These Highlights Do Not Include All The Information Needed To Use Invega Hafyera Safely And Effectively. See Full Prescribing Information For Invega Hafyera.
6cd61892-d2cb-434d-83ed-5c1b2c4e7a0b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA HAFYERA is not approved for use in patients with dementia-related psychosis [see Warnings and Precautions (5.1) ] .
Indications and Usage
INVEGA HAFYERA, an every-six-month injection, is indicated for the treatment of schizophrenia in adults after they have been adequately treated with: A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA) for at least four months, or An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA) for at least one three-month cycle.
Dosage and Administration
Administer INVEGA HAFYERA by gluteal injection once every 6 months by a healthcare professional. Do not administer by any other route. ( 2.1 ) See Full Prescribing Information for complete dosing information. ( 2.2 ) Initiate INVEGA HAFYERA when the next once-a-month or every three-month paliperidone palmitate extended-release injectable suspension dose is scheduled. Dose is based on the previous once-a-month or every-three-month product. ( 2.2 ): INVEGA HAFYERA Doses for Adults Adequately Treated with Once-a-month paliperidone palmitate extended-release injectable suspension (PP1M) Switching from the PP1M 39 mg, 78 mg and 117 mg doses was not studied. If the Last Dose of PP1M is: Initiate INVEGA HAFYERA at the Following Dose: 156 mg 1,092 mg 234 mg 1,560 mg INVEGA HAFYERA Doses for Adults Adequately Treated with Every-three-month paliperidone palmitate injectable suspension (PP3M) Switching from the PP3M 273 mg and 410 mg doses was not studied. If the Last Dose of PP3M is: Initiate INVEGA HAFYERA at the Following Dose: 546 mg 1,092 mg 819 mg 1,560 mg Missed Doses: Refer to the Full Prescribing Information. ( 2.3 ) See Full Prescribing Information for important preparation and administration information. ( 2.5 ) Moderate to severe renal impairment (creatinine clearance <50 mL/min): INVEGA HAFYERA is not recommended. ( 2.4 ) Mild renal impairment (creatinine clearance ≥ 50 mL/min to < 80 mL/min): Adjust dosage and stabilize the patient using PP1M before transitioning to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA. See appropriate table above. ( 2.4 )
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis : Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2 ) Neuroleptic Malignant Syndrome : Manage with immediate discontinuation of drug and close monitoring. ( 5.3 ) QT Prolongation : Avoid use with drugs that also increase QT interval and in patients with risk factors for prolonged QT interval. ( 5.4 ) Tardive Dyskinesia : Discontinue treatment if clinically appropriate ( 5.5 ) Metabolic Changes : Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6 ) Orthostatic Hypotension and Syncope : Use with caution in patients with known cardiovascular or cerebrovascular disease and patients predisposed to hypotension. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or a history of leukopenia or neutropenia. Consider discontinuing INVEGA HAFYERA if a clinically significant decline in WBC occurs in the absence of other causative factors. ( 5.9 ) Hyperprolactinemia : Prolactin elevations occur and persist during chronic administration. ( 5.10 ) Potential for Cognitive and Motor Impairment : Use caution when operating machinery. ( 5.11 ) Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.12 )
Contraindications
INVEGA HAFYERA is contraindicated in patients with a known hypersensitivity to either paliperidone or risperidone, or to any of the excipients in the INVEGA HAFYERA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone palmitate is converted to paliperidone, which is a metabolite of risperidone.
Adverse Reactions
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. No studies have been conducted with oral paliperidone, the 1-month paliperidone palmitate extended-release injectable suspension, the 3-month paliperidone extended-release injectable suspension or INVEGA HAFYERA in elderly patients with dementia. These medications are not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1) ] .
Drug Interactions
Strong CYP3A4/P-glycoprotein (P-gp) inducers : Avoid using strong CYP3A4 and/or P-gp inducers during a dosing interval for INVEGA HAFYERA. If administering a strong inducer is necessary, consider managing the patient using paliperidone extended-release tablets. ( 7.1 , 12.3 )
Storage and Handling
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle. 1,092 mg paliperidone palmitate kit (NDC 50458-611-01) 1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
How Supplied
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle. 1,092 mg paliperidone palmitate kit (NDC 50458-611-01) 1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
Medication Information
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis : Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2 ) Neuroleptic Malignant Syndrome : Manage with immediate discontinuation of drug and close monitoring. ( 5.3 ) QT Prolongation : Avoid use with drugs that also increase QT interval and in patients with risk factors for prolonged QT interval. ( 5.4 ) Tardive Dyskinesia : Discontinue treatment if clinically appropriate ( 5.5 ) Metabolic Changes : Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6 ) Orthostatic Hypotension and Syncope : Use with caution in patients with known cardiovascular or cerebrovascular disease and patients predisposed to hypotension. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis : Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or a history of leukopenia or neutropenia. Consider discontinuing INVEGA HAFYERA if a clinically significant decline in WBC occurs in the absence of other causative factors. ( 5.9 ) Hyperprolactinemia : Prolactin elevations occur and persist during chronic administration. ( 5.10 ) Potential for Cognitive and Motor Impairment : Use caution when operating machinery. ( 5.11 ) Seizures : Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.12 )
Indications and Usage
INVEGA HAFYERA, an every-six-month injection, is indicated for the treatment of schizophrenia in adults after they have been adequately treated with: A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA) for at least four months, or An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA) for at least one three-month cycle.
Dosage and Administration
Administer INVEGA HAFYERA by gluteal injection once every 6 months by a healthcare professional. Do not administer by any other route. ( 2.1 ) See Full Prescribing Information for complete dosing information. ( 2.2 ) Initiate INVEGA HAFYERA when the next once-a-month or every three-month paliperidone palmitate extended-release injectable suspension dose is scheduled. Dose is based on the previous once-a-month or every-three-month product. ( 2.2 ): INVEGA HAFYERA Doses for Adults Adequately Treated with Once-a-month paliperidone palmitate extended-release injectable suspension (PP1M) Switching from the PP1M 39 mg, 78 mg and 117 mg doses was not studied. If the Last Dose of PP1M is: Initiate INVEGA HAFYERA at the Following Dose: 156 mg 1,092 mg 234 mg 1,560 mg INVEGA HAFYERA Doses for Adults Adequately Treated with Every-three-month paliperidone palmitate injectable suspension (PP3M) Switching from the PP3M 273 mg and 410 mg doses was not studied. If the Last Dose of PP3M is: Initiate INVEGA HAFYERA at the Following Dose: 546 mg 1,092 mg 819 mg 1,560 mg Missed Doses: Refer to the Full Prescribing Information. ( 2.3 ) See Full Prescribing Information for important preparation and administration information. ( 2.5 ) Moderate to severe renal impairment (creatinine clearance <50 mL/min): INVEGA HAFYERA is not recommended. ( 2.4 ) Mild renal impairment (creatinine clearance ≥ 50 mL/min to < 80 mL/min): Adjust dosage and stabilize the patient using PP1M before transitioning to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA. See appropriate table above. ( 2.4 )
Contraindications
INVEGA HAFYERA is contraindicated in patients with a known hypersensitivity to either paliperidone or risperidone, or to any of the excipients in the INVEGA HAFYERA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone palmitate is converted to paliperidone, which is a metabolite of risperidone.
Adverse Reactions
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. No studies have been conducted with oral paliperidone, the 1-month paliperidone palmitate extended-release injectable suspension, the 3-month paliperidone extended-release injectable suspension or INVEGA HAFYERA in elderly patients with dementia. These medications are not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1) ] .
Drug Interactions
Strong CYP3A4/P-glycoprotein (P-gp) inducers : Avoid using strong CYP3A4 and/or P-gp inducers during a dosing interval for INVEGA HAFYERA. If administering a strong inducer is necessary, consider managing the patient using paliperidone extended-release tablets. ( 7.1 , 12.3 )
Storage and Handling
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle. 1,092 mg paliperidone palmitate kit (NDC 50458-611-01) 1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
How Supplied
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle. 1,092 mg paliperidone palmitate kit (NDC 50458-611-01) 1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA HAFYERA is not approved for use in patients with dementia-related psychosis [see Warnings and Precautions (5.1) ] .
Section 42229-5
Switching to INVEGA HAFYERA from a PP1M Product
The recommended initial INVEGA HAFYERA dose is based on the previous PP1M dose (see Table 1). Initiate INVEGA HAFYERA when the next PP1M dose is scheduled. INVEGA HAFYERA may be administered up to 1 week before or 1 week after the next scheduled PP1M dose. When switching from PP1M to INVEGA HAFYERA, the two injection cycles immediately preceding the switch should be the same dosage strength before starting INVEGA HAFYERA.
| Last Dose of PP1M
There are no equivalent doses of INVEGA HAFYERA for 39 mg, 78 mg, or 117 mg doses of a PP1M product, which were not studied
[see
Clinical Studies (14)]
.
|
Initial Dose of INVEGA HAFYERA |
|---|---|
| 156 mg | 1,092 mg |
| 234 mg | 1,560 mg |
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: January 2025 | |||
|
PATIENT INFORMATION
INVEGA HAFYERA ® (in-VAY-guh HAF-ye-RA) (paliperidone palmitate) extended-release injectable suspension |
||||
|
What is the most important information I should know about INVEGA HAFYERA?
INVEGA HAFYERA may cause serious side effects, including:
|
||||
|
What is INVEGA HAFYERA?
INVEGA HAFYERA is a prescription medicine given by injection by a healthcare provider 1 time every 6 months and used for the treatment of schizophrenia in adults who have been adequately treated with either:
|
||||
| Do not receive INVEGA HAFYERA if you are allergic to paliperidone palmitate, risperidone, or any of the ingredients in INVEGA HAFYERA. See the end of this Patient Information leaflet for a complete list of ingredients in INVEGA HAFYERA. | ||||
Before receiving INVEGA HAFYERA, tell your healthcare provider about all your medical conditions, including if you:
INVEGA HAFYERA and other medicines may affect each other causing possible serious side effects. INVEGA HAFYERA may affect the way other medicines work, and other medicines may affect how INVEGA HAFYERA works. Your healthcare provider can tell you if it is safe to receive INVEGA HAFYERA with your other medicines. Do not start or stop any medicines during treatment with INVEGA HAFYERA without talking to your healthcare provider first. Know the medicines you take. Keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine. |
||||
How will I receive INVEGA HAFYERA?
|
||||
What should I avoid while receiving INVEGA HAFYERA?
|
||||
|
What are the possible side effects of INVEGA HAFYERA?
INVEGA HAFYERA may cause serious side effects, including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
||||
|
|
|||
| These are not all the possible side effects of INVEGA HAFYERA.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
|
General information about INVEGA HAFYERA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about INVEGA HAFYERA that is written for health professionals. |
||||
|
What are the ingredients in INVEGA HAFYERA?
Active ingredient: paliperidone palmitate Inactive ingredients: polysorbate 20, polyethylene glycol 4000, citric acid monohydrate, sodium dihydrogen phosphate monohydrate, sodium hydroxide, and water for injection Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, NJ 08560, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2021 For more information, go to www.invegahafyerahcp.com or call 1-800-526-7736. |
Section 43683-2
Section 44425-7
Storage and Handling
Store at room temperature 20 °C to 25 °C (68 °F to 77 °F); excursions between 15 °C and 30 °C (59 °F and 86 °F) are permitted. Do not mix with any other product or diluent.
Ship and store in a horizontal position. See arrows on product carton for proper orientation.
5.8 Falls
Somnolence, postural hypotension, motor and sensory instability have been reported with the use of antipsychotics, including paliperidone palmitate, which may lead to falls and, consequently, fractures or other fall-related injuries. For patients, particularly the elderly, with diseases, conditions, or medications that could exacerbate these effects, assess the risk of falls when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
9.2 Abuse
Paliperidone has not been systematically studied in animals or humans for its potential for abuse.
5.12 Seizures
In the 6-month paliperidone palmitate extended-release injectable suspension double-blind active controlled trial there were no reports of seizures or convulsions, nor were any reports made in the long-term maintenance trial of PP3M. In the pivotal clinical studies with PP1M which included four fixed-dose, double-blind, placebo-controlled studies in subjects with schizophrenia, <1% (1/1293) of subjects treated with the PP1M experienced an adverse event of convulsion compared with <1% (1/510) of placebo-treated subjects who experienced an adverse event of grand mal convulsion.
Like other antipsychotic drugs, INVEGA HAFYERA should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older.
5.14 Priapism
A case (0.2%) of priapism was reported in the clinical trial with INVEGA HAFYERA. Priapism has been reported with oral paliperidone during postmarketing surveillance. Drugs with alpha-adrenergic blocking effects have been reported to induce priapism. Severe priapism may require surgical intervention.
11 Description
INVEGA HAFYERA ® contains a racemic mixture of (+)- and (-)- paliperidone palmitate. Paliperidone palmitate is an atypical antipsychotic belonging to the chemical class of benzisoxazole derivatives. The chemical name is (9 RS)-3-[2-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)piperidin-1-yl]ethyl]-2-methyl-4-oxo-6,7,8,9-tetrahydro-4 H-pyrido[1,2- a]pyrimadin-9-yl hexadecanoate. Its molecular formula is C 39H 57FN 4O 4 and its molecular weight is 664.89. The structural formula is:
Paliperidone palmitate is very slightly soluble in ethanol and methanol, practically insoluble in polyethylene glycol 400 and propylene glycol, and slightly soluble in ethyl acetate.
INVEGA HAFYERA is available as a white to off-white sterile aqueous extended-release suspension for intramuscular injection in dose strengths of 1,092 mg and 1,560 mg paliperidone palmitate. The drug product hydrolyzes to the active moiety, paliperidone, resulting in dose strengths of 700 mg, and 1,000 mg of paliperidone, respectively. The inactive ingredients are polysorbate 20 (10 mg/mL), polyethylene glycol 4000 (75 mg/mL), citric acid monohydrate (7.5 mg/mL), sodium dihydrogen phosphate monohydrate (6 mg/mL), sodium hydroxide (5.4 mg/mL), and water for injection.
INVEGA HAFYERA is provided in a single-dose prefilled syringe (cyclic-olefin-copolymer) prefilled with either 700 mg (3.5 mL), or 1,000 mg (5.0 mL) paliperidone (as 1,092 mg, or 1,560 mg paliperidone palmitate) suspension with a tip cap, plunger rod, backstop and a thin walled 20G, 1½-inch safety needle.
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. INVEGA HAFYERA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
9.3 Dependence
Paliperidone has not been systematically studied in animals or humans for its potential for tolerance or physical dependence.
8.4 Pediatric Use
Safety and effectiveness of INVEGA HAFYERA in patients less than 18 years of age have not been established. Use of INVEGA HAFYERA is not recommended in pediatric patients because of the potential longer duration of an adverse event. In clinical trials of oral paliperidone, there were notably higher incidences of dystonia, hyperkinesia, tremor, and parkinsonism in the adolescent population as compared to the adult studies.
8.5 Geriatric Use
The clinical study of INVEGA HAFYERA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
This drug is substantially excreted by the kidney and clearance is decreased in patients with renal impairment [see Clinical Pharmacology (12.3)] . Because elderly patients are more likely to have decreased renal function, INVEGA HAFYERA is not recommended to be used in elderly patients with mild, moderate or severe renal impairment [see Use in Specific Populations (8.6)] .
14 Clinical Studies
The efficacy of INVEGA HAFYERA for the treatment of schizophrenia in patients who had previously been stably treated with either PP1M for at least 4 months or PP3M for at least one 3-month injection cycle was evaluated in a randomized, double-blind, active-controlled, interventional, parallel-group, multicenter, non-inferiority study designed to evaluate time to relapse in adults with a DSM-5 diagnosis of schizophrenia.
Patients could enter the study if previously treated with PP1M at dosages of 156 or 234 mg, PP3M at dosages of 546 or 819 mg, injectable risperidone at dosages of 50 mg, or any oral antipsychotic with a reason to change (e.g., efficacy, safety, tolerability, or a preference for a long-acting injectable medication) and with a PANSS total score of <70 points.
After establishing tolerability with PP1M (at dosages of 156 or 234 mg) or PP3M (at dosages of 546 or 819 mg) and clinical stability, defined by having a PANSS total score of <70 points for the previous 2 assessments prior to the double-blind phase, patients were randomized in a 2:1 ratio to receive INVEGA HAFYERA (478 patients) or PP3M (224 patients).
The primary efficacy variable was time to first relapse in the double-blind phase. The primary efficacy analysis was based on the difference in Kaplan-Meier 12-month estimates of percentage of subjects remaining relapse-free between INVEGA HAFYERA and 3-month paliperidone palmitate extended-release injectable suspension. Relapse was pre-defined as emergence of one or more of the following: psychiatric hospitalization, ≥25% increase (if the baseline score was >40) or a 10-point increase (if the baseline score was ≤40) in total PANSS score on two consecutive assessments, deliberate self-injury, violent behavior, suicidal/homicidal ideation: a score of ≥5 (if the maximum baseline score was ≤3) or ≥6 (if the maximum baseline score was 4) on two consecutive assessments of the specific PANSS items.
A relapse event was experienced by 7.5% and 4.9% of patients in the INVEGA HAFYERA and PP3M treatment groups, respectively, with the Kaplan-Meier estimated difference (INVEGA HAFYERA – PP3M) of 2.9% (95% CI: -1.1 to 6.8). The upper bound of the 95% CI (6.8%) was less than 10%, the prespecified non-inferiority margin. The study demonstrated non-inferiority of INVEGA HAFYERA to PP3M. A Kaplan-Meier plot of time to relapse by treatment group is shown in Figure 4.
Figure 4: Kaplan-Meier Plot of Cumulative Proportion of Patients with Relapse Over Time
An evaluation of population subgroups did not reveal any clinically significant differences in responsiveness on the basis of gender, age, or race.
4 Contraindications
INVEGA HAFYERA is contraindicated in patients with a known hypersensitivity to either paliperidone or risperidone, or to any of the excipients in the INVEGA HAFYERA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone palmitate is converted to paliperidone, which is a metabolite of risperidone.
5.4 Qt Prolongation
Paliperidone causes a modest increase in the corrected QT (QTc) interval. The use of paliperidone should be avoided in combination with other drugs that are known to prolong QTc including Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications, antipsychotic medications (e.g., chlorpromazine, thioridazine), antibiotics (e.g., gatifloxacin, moxifloxacin), or any other class of medications known to prolong the QTc interval. Paliperidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Certain circumstances may increase the risk of the occurrence of Torsades de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including (1) bradycardia; (2) hypokalemia or hypomagnesemia; (3) concomitant use of other drugs that prolong the QTc interval; and (4) presence of congenital prolongation of the QT interval.
The effects of paliperidone on the QT interval were evaluated in a double-blind, active-controlled (moxifloxacin 400 mg single dose), multicenter Thorough QT study with oral paliperidone in adult patients, and in four fixed-dose efficacy studies and one maintenance study of the 1-month paliperidone palmitate injectable product.
In the Thorough QT study (n=141), the 8 mg dose of immediate-release oral paliperidone (n=50) showed a mean placebo-subtracted increase from baseline in QTcLD (QT interval corrected for heart rate using the population specified linear derived method) of 12.3 msec (90% CI: 8.9; 15.6) on day 8 at 1.5 hours post-dose. The mean steady-state peak plasma concentration for this 8 mg dose of paliperidone immediate release (C max ss=113 ng/mL) was approximately 1.3-fold the exposure with the maximum recommended 1,560 mg dose of INVEGA HAFYERA administered in the gluteal muscle (mean C max md=89.3 ng/mL). In this same study, a 4 mg dose of the immediate-release oral formulation of paliperidone, for which C max ss=35 ng/mL, showed an increased placebo-subtracted QTcLD of 6.8 msec (90% CI: 3.6; 10.1) on day 2 at 1.5 hours post-dose.
In the four fixed-dose efficacy studies of the 1-month paliperidone palmitate injectable product, no subject had a change in QTcLD exceeding 60 msec and no subject had a QTcLD value of >500 msec at any time point. In the maintenance study, no subject had a QTcLD change >60 msec, and one subject had a QTcLD value of 507 msec (Bazett's QT corrected interval [QTcB] value of 483 msec); this latter subject also had a heart rate of 45 beats per minute.
In the INVEGA HAFYERA randomized double-blind active controlled study in subjects with schizophrenia, during the double-blind Phase, QTcLD exceeding 60 msec was observed in 2 subjects (0.4%) in the INVEGA HAFYERA treatment group and in 2 subjects (0.9%) in the PP3M treatment group. No subject had a QTcLD value of >480 msec at any point in the study.
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1)]
- Cerebrovascular adverse reactions, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2)]
- Neuroleptic malignant syndrome [see Warnings and Precautions (5.3)]
- QT prolongation [see Warnings and Precautions (5.4)]
- Tardive dyskinesia [see Warnings and Precautions (5.5)]
- Metabolic changes [see Warnings and Precautions (5.6)]
- Orthostatic hypotension and syncope [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia, neutropenia, and agranulocytosis [see Warnings and Precautions (5.9)]
- Hyperprolactinemia [see Warnings and Precautions (5.10)]
- Potential for cognitive and motor impairment [see Warnings and Precautions (5.11)]
- Seizures [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
- Priapism [see Warnings and Precautions (5.14)]
- Disruption of body temperature regulation [see Warnings and Precautions (5.15)]
7 Drug Interactions
8.6 Renal Impairment
Use of INVEGA HAFYERA is not recommended for use in patients with moderate or severe renal impairment (creatinine clearance <50 mL/min). Use of INVEGA HAFYERA in patients with mild renal impairment (creatinine clearance ≥50 mL/min to <80 mL/min) is based on the patient’s previous dose of PP1M or PP3M before transitioning to INVEGA HAFYERA [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)] .
Instructions for Use
INVEGA HAFYERA
®
in-VAY-guh HAF-ye-RA
(paliperidone palmitate)
extended-release injectable suspension
For Gluteal Intramuscular
Injection Only
Administer every 6 months
| Shake syringe with the syringe tip cap pointing up VERY FAST for at least 15 seconds, rest briefly, then shake again for 15 seconds |
Important
| Shipping and storing the carton in a horizontal orientation improves the ability to resuspend this highly concentrated product. |
INVEGA HAFYERA must be administered by a healthcare professional as a single injection.
Do not divide dose into multiple injections.
INVEGA HAFYERA is intended for gluteal intramuscular use only. Inject slowly, deep into the muscle taking care to avoid injection into a blood vessel.
Dosing
Administer INVEGA HAFYERA once every 6 months.
Thin Wall Safety Needle
Thin wall safety needle is designed to be used with INVEGA HAFYERA. Therefore, it is important to only use the needle provided in the INVEGA HAFYERA suspension kit.
Dose pack contents
- Prepare for the injection: this highly concentrated product requires specific steps to resuspend
Hold syringe with the tip cap pointing up
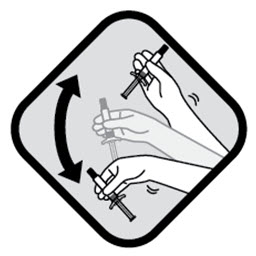
Shake syringe VERY FAST for at least 15 seconds, rest briefly, then shake again for 15 seconds
To ensure complete resuspension shake syringe with:
- Short, VERY FAST up and down motion
- Loose wrist
Proceed to the next step immediately after shaking.
Check suspension for solid product
|
|
|
|
Product may clog. Shake syringe with the syringe tip cap pointing up VERY FAST for at least 15 seconds, rest, then shake again for 15 seconds. |
Open needle pouch
Peel off the pouch cover.
Place pouch with the needle inside on a clean surface.
Remove syringe tip cap and attach needle
Hold the syringe with the tip cap pointing up.
Twist and pull off the cap.
Attach the safety needle to the syringe using a gentle twisting motion to avoid needle hub cracks or damage. Always check for signs of damage or leakage prior to administration.
Pull back plunger
Hold the syringe upright.
Gently pull back the plunger to clear the syringe tip of any solid product. This will make pressing the plunger easier during the injection.
Remove air bubbles
Press the plunger carefully until a drop of liquid comes out of the needle tip.
- Slowly inject entire content and confirm
|
Select and clean a gluteal injection site
Wipe the gluteal site with an alcohol swab and allow it to dry. Do not touch, fan or blow on the injection site after you have cleaned it. |
|
|
Remove needle sheath
Pull the needle sheath away from the needle in a straight motion. Do not twist the sheath, as this may loosen the needle from the syringe. |
|
|
Slowly inject and confirm
Use slow, firm, consistent pressure to press the plunger completely. This should take approximately 30 seconds. Continue to press the plunger if you feel resistance. This is normal. While the needle is in the gluteal muscle, confirm that the entire content of the syringe has been injected. Remove needle from the muscle. |
- After the injection
|
Secure needle
After the injection is complete, use your thumb or a flat surface to secure the needle in the safety device. The needle is secure when you hear a "click" sound. |
|
|
Dispose of properly and check injection site
Dispose of the syringe in an approved sharps container. There may be a small amount of blood or liquid at the injection site. Hold pressure over the skin with a cotton ball or gauze pad until any bleeding stops. Do not rub the injection site. If needed, cover injection site with a bandage. |
Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560, USA
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2021
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Revised: 2/2025
12.2 Pharmacodynamics
In vitro, paliperidone acts as an antagonist at the central dopamine D 2 and serotonin 5HT 2A receptors with binding affinities (Ki values) of 1.6–2.8 nM and 0.8–1.2 nM, respectively. Paliperidone also acts as an antagonist at histamine H 1 and α 1 and α 2 adrenergic receptors with binding affinities of 32 nM, 4 nM, and 17 nM, respectively. Paliperidone has no appreciable affinity for cholinergic muscarinic or β 1- and β 2-adrenergic receptors. The pharmacological activity of the (+)- and (-)- paliperidone enantiomers is qualitatively and quantitatively similar.
12.3 Pharmacokinetics
The pharmacokinetics for INVEGA HAFYERA presented below are based on gluteal administration only.
INVEGA HAFYERA delivers paliperidone over a 6-month period, compared to the 1-month or 3-month products which are administered every month or every three months, respectively. INVEGA HAFYERA doses of 1,092 mg and 1,560 mg result in paliperidone total exposure ranges that are encompassed within the exposure range for corresponding doses of 1-month paliperidone palmitate injections (PP1M) (156 mg and 234 mg) or corresponding doses of 3-month paliperidone palmitate (PP3M) injections (546 mg and 819 mg, respectively) or to corresponding once daily doses of paliperidone extended-release tablets. However, mean trough concentrations (C trough) at the end of the dosing interval were approximately 20 – 25% lower for INVEGA HAFYERA as compared to corresponding doses of 3-month paliperidone palmitate. The mean peak concentration (C max) was higher (1.4 to 1.5-fold) for INVEGA HAFYERA as compared to corresponding doses of 3-month paliperidone palmitate.
Inter-subject variability in paliperidone PK parameters for INVEGA HAFYERA ranged from 42 to 48% for AUC 6months and ranged from 56 to 103% for C max. Because of the difference in pharmacokinetic profiles among the four paliperidone products, caution should be exercised when making a direct comparison of their pharmacokinetic properties.
5.6 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.5 Tardive Dyskinesia
Tardive dyskinesia, a syndrome of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose. The syndrome can develop after relatively brief treatment periods, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, INVEGA HAFYERA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with INVEGA HAFYERA, drug discontinuation should be considered. Consideration should be given to the long-acting nature of INVEGA HAFYERA. However, some patients may require treatment with INVEGA HAFYERA despite the presence of the syndrome.
8.7 Hepatic Impairment
INVEGA HAFYERA has not been studied in patients with hepatic impairment. Based on a study with oral paliperidone, no dose adjustment is required in patients with mild or moderate hepatic impairment. Paliperidone has not been studied in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
INVEGA HAFYERA, an every-six-month injection, is indicated for the treatment of schizophrenia in adults after they have been adequately treated with:
- A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA) for at least four months, or
- An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA) for at least one three-month cycle.
5.10 Hyperprolactinemia
Like other drugs that antagonize dopamine D 2receptors, paliperidone elevates prolactin levels and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to that seen with risperidone, a drug that is associated with higher levels of prolactin than other antipsychotic drugs.
Hyperprolactinemia, regardless of etiology, may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously detected breast cancer. An increase in the incidence of pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)] . Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
Median prolactin levels remained relatively stable throughout the open-label and double-blind phases in male subjects, whereas in female subjects, median prolactin levels increased. During the double-blind phase, median prolactin levels continued to increase after dosing in both the INVEGA HAFYERA and PP3M groups, returning to baseline level at Month 6 and at Month 12 (end of double-blind phase).
During the double-blind phase, prolactin levels relative to reference range (>13.13 ng/mL in males and >26.72 ng/mL in females) from maintenance baseline were noted in a similar percentage of subjects in the INVEGA HAFYERA and PP3M groups in both males (35% vs 36%) and females (29% vs. 30%). In the INVEGA HAFYERA group, 14 females (2.9%) and 4 males (0.8%) experienced potentially prolactin-related adverse reactions, while 6 females (2.7%) and 1 male (0.4%) in the PP3M experienced potentially prolactin-related adverse reactions.
12.1 Mechanism of Action
Paliperidone palmitate is hydrolyzed to paliperidone [see Clinical Pharmacology (12.3)] . Paliperidone is the major active metabolite of risperidone. The mechanism of action of paliperidone is unclear. However, its efficacy in the treatment of schizophrenia could be mediated through a combination of central dopamine D 2 and serotonin 5HT 2A receptor antagonism.
9.1 Controlled Substance
INVEGA HAFYERA contains paliperidone, which is not a controlled substance.
5 Warnings and Precautions
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation of drug and close monitoring. ( 5.3)
- QT Prolongation: Avoid use with drugs that also increase QT interval and in patients with risk factors for prolonged QT interval. ( 5.4)
- Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.5)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6)
- Orthostatic Hypotension and Syncope: Use with caution in patients with known cardiovascular or cerebrovascular disease and patients predisposed to hypotension. ( 5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or a history of leukopenia or neutropenia. Consider discontinuing INVEGA HAFYERA if a clinically significant decline in WBC occurs in the absence of other causative factors. ( 5.9)
- Hyperprolactinemia: Prolactin elevations occur and persist during chronic administration. ( 5.10)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. ( 5.11)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.12)
2 Dosage and Administration
- Administer INVEGA HAFYERA by gluteal injection once every 6 months by a healthcare professional. Do not administer by any other route. ( 2.1)
- See Full Prescribing Information for complete dosing information. ( 2.2)
- Initiate INVEGA HAFYERA when the next once-a-month or every three-month paliperidone palmitate extended-release injectable suspension dose is scheduled. Dose is based on the previous once-a-month or every-three-month product. (
2.2):
INVEGA HAFYERA Doses for Adults Adequately Treated with Once-a-month paliperidone palmitate extended-release injectable suspension (PP1M)Switching from the PP1M 39 mg, 78 mg and 117 mg doses was not studied.
If the Last Dose of PP1M is: Initiate INVEGA HAFYERA at the Following Dose: 156 mg 1,092 mg 234 mg 1,560 mg INVEGA HAFYERA Doses for Adults Adequately Treated with Every-three-month paliperidone palmitate injectable suspension (PP3M)Switching from the PP3M 273 mg and 410 mg doses was not studied.If the Last Dose of PP3M is: Initiate INVEGA HAFYERA at the Following Dose: 546 mg 1,092 mg 819 mg 1,560 mg - Missed Doses: Refer to the Full Prescribing Information. ( 2.3)
- See Full Prescribing Information for important preparation and administration information. ( 2.5)
- Moderate to severe renal impairment (creatinine clearance <50 mL/min): INVEGA HAFYERA is not recommended. ( 2.4)
- Mild renal impairment (creatinine clearance ≥ 50 mL/min to < 80 mL/min): Adjust dosage and stabilize the patient using PP1M before transitioning to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA. See appropriate table above. ( 2.4)
3 Dosage Forms and Strengths
INVEGA HAFYERA is a white to off-white aqueous extended-release injectable suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate in single-dose prefilled syringes.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of paliperidone; because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: angioedema, catatonia, ileus, somnambulism, swollen tongue, thrombotic thrombocytopenic purpura, urinary incontinence, and urinary retention.
Cases of anaphylactic reaction after injection with the 1-month paliperidone palmitate extended-release suspension have been reported during postmarketing experience in patients who have previously tolerated oral risperidone or oral paliperidone.
Paliperidone is the major active metabolite of risperidone. Adverse reactions reported with oral risperidone and risperidone long-acting injection can be found in the Adverse Reactions (6) section of the Prescribing Information for those products.
8 Use in Specific Populations
- Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.3 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with antipsychotic drugs, including paliperidone.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, including delirium, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, discontinue INVEGA HAFYERA and provide symptomatic treatment and monitoring.
16 How Supplied/storage and Handling
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle.
1,092 mg paliperidone palmitate kit (NDC 50458-611-01)
1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
5.7 Orthostatic Hypotension and Syncope
Paliperidone can induce orthostatic hypotension and syncope in some patients because of its alpha-adrenergic blocking activity.
Use INVEGA HAFYERA with caution in patients with known cardiovascular disease (e.g., heart failure, history of myocardial infarction or ischemia, conduction abnormalities), cerebrovascular disease, or conditions that predispose the patient to hypotension (e.g., dehydration, hypovolemia, and treatment with antihypertensive medications). Monitoring of orthostatic vital signs should be considered in patients who are vulnerable to hypotension.
13.2 Animal Toxicology And/or Pharmacology
Injection site toxicity was assessed in minipigs injected intramuscularly with the 6-month paliperidone palmitate extended-release injectable suspension at doses up to 2,115 mg, which is slightly above the MRHD. Injection site inflammatory reactions were greater and more advanced than reactions to the 1-month paliperidone palmitate extended-release injectable suspension. Reversibility of these findings was not examined.
Principal Display Panel 5 Ml Syringe Carton
NDC 50458-612-01
Single-dose prefilled syringe. Use entire contents of syringe.
INVEGA HAFYERA™
(paliperidone palmitate)
extended-release injectable suspension
FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
Each injection must be administered only by
a healthcare professional.
Shake before using.
CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle).
Recommended Dosage: See Prescribing Information.
Rx only
Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted.
SHIP AND STORE
THIS SIDE UP
LAY
FLAT
1,560 mg/5 mL
Each single-dose prefilled syringe contains
1,560 mg paliperidone palmitate.
6
MONTHS
Administer
every 6 months
Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds
5.15 Disruption of Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing INVEGA HAFYERA to patients who will be experiencing conditions which may contribute to an elevation in core body temperature, e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration.
Principal Display Panel 3.5 Ml Syringe Carton
NDC 50458-611-01
Single-dose prefilled syringe. Use entire contents of syringe.
INVEGA HAFYERA™
(paliperidone palmitate)
extended-release injectable suspension
FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
Each injection must be administered only by
a healthcare professional.
Shake before using.
CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle).
Recommended Dosage: See Prescribing Information.
Rx only
Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted.
SHIP AND STORE
THIS SIDE UP
LAY
FLAT
1,092 mg/3.5 mL
Each single-dose prefilled syringe contains
1,092 mg paliperidone palmitate.
6
MONTHS
Administer
every 6 months
Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds
5.9 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trial and/or postmarketing experience, events of leukopenia and neutropenia have been reported temporally related to antipsychotic agents, including INVEGA HAFYERA. Agranulocytosis has also been reported.
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or a drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of INVEGA HAFYERA at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Discontinue INVEGA HAFYERA in patients with severe neutropenia (absolute neutrophil count <1000/mm 3) and follow their WBC until recovery.
5.11 Potential for Cognitive and Motor Impairment
Somnolence and sedation were reported as adverse reactions in patients treated with INVEGA HAFYERA [see Adverse Reactions (6.1)] . Antipsychotics, including INVEGA HAFYERA, have the potential to impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that paliperidone therapy does not adversely affect them.
2.1 Important Dosage and Administration Information
- INVEGA HAFYERA must be administered as a gluteal intramuscular injection by a healthcare professional once every 6 months. Do not administer by any other route [see Dosage and Administration (2.5)] .
- Initiate INVEGA HAFYERA only after adequate treatment has been established with either:
- A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA), referred to as PP1M, once monthly for at least four months; or
- An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA), referred to as PP3M, once every three months for at least one three-month injection cycle.
- See Prescribing Information of the PP1M and PP3M products for the recommended dosage of these products.
2.5 Instructions for Preparation and Administration
- To be prepared and administered by a healthcare provider only.
- Read the instructions for preparation and administration below and consider referring to the separate Healthcare Provider "Instructions for Use" for preparation and administration considerations.
- For gluteal intramuscular injection only. Do not inject by any other route. As a universal precaution, always wear gloves.
- Inspect INVEGA HAFYERA for particulate matter and discoloration prior to administration.
- Do not mix with any other product or diluent.
- After shaking, INVEGA HAFYERA should appear uniform, thick and milky white .
- Do not use needles from the PP1M or PP3M products or other commercially-available needles to reduce the risk of blockage.
- Avoid inadvertent injection into a blood vessel. Administer the dose in a single injection; do not administer the dose in divided injections. Inject slowly, deep into the upper-outer quadrant of the gluteal muscle. Future injections should be alternated between the two gluteal muscles.
8.8 Patients With Parkinson's Disease Or Lewy Body Dementia
Patients with Parkinson's Disease or Dementia with Lewy Bodies can experience increased sensitivity to INVEGA HAFYERA. Manifestations can include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with neuroleptic malignant syndrome.
2.4 Dosage Recommendations in Patients With Renal Impairment
INVEGA HAFYERA has not been systematically studied in patients with renal impairment [see Clinical Pharmacology (12.3)] . For patients with mild renal impairment (creatinine clearance ≥50 mL/min to < 80 mL/min [Cockcroft-Gault Formula]), adjust dosage and stabilize the patient using PP1M before transitioning from PP1M to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA (see Table 1) [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)]. Refer to the Prescribing Information of PP1M or PP3M product for the recommended PP1M or PP3M dosage in patients with mild renal impairment.
INVEGA HAFYERA is not recommended in patients with moderate or severe renal impairment (creatinine clearance <50 mL/min) [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
7.1 Drugs Having Clinically Important Interactions With Invega Hafyera
Because paliperidone palmitate is hydrolyzed to paliperidone, results from studies with oral paliperidone should be taken into consideration when assessing drug-drug interaction potential. In addition, consider the 6-month dosing interval and the half-life of INVEGA HAFYERA [see Clinical Pharmacology (12.3)] .
Table 10 presents clinically significant drug interactions with INVEGA HAFYERA.
| Centrally acting Drugs and Alcohol | |
| Clinical Rationale | Given the primary CNS effects of paliperidone, concomitant use of centrally acting drugs and alcohol may modulate the CNS effects of INVEGA HAFYERA. |
| Clinical Recommendation | INVEGA HAFYERA should be used with caution with other centrally acting drugs and alcohol. |
| Drugs with Potential for Inducing Orthostatic Hypotension | |
| Clinical Rationale | Because INVEGA HAFYERA has the potential for inducing orthostatic hypotension, an additive effect may occur when INVEGA HAFYERA is administered with other therapeutic agents that have this potential [see Warnings and Precautions (5.7)] . |
| Clinical Recommendation | Monitor orthostatic vital signs in patients who are vulnerable to hypotension [see Warnings and Precautions (5.7)] . |
| Strong Inducers of CYP3A4 and P-gp | |
| Clinical Rationale | The concomitant use of INVEGA HAFYERA and strong inducers of CYP3A4 and P-gp may decrease the exposure of paliperidone [see Clinical Pharmacology (12.3)] . |
| Clinical Recommendation | Avoid using CYP3A4 and/or P-gp inducers with INVEGA HAFYERA during the 6-month dosing interval, if possible. If administering a strong inducer is necessary, consider managing the patient using paliperidone extended-release tablets [see Dosage and Administration (2.1)] . |
| Examples | carbamazepine, rifampin, or St. John's Wort |
| Levodopa and Other Dopamine Agonists | |
| Clinical Rationale | Paliperidone may antagonize the effect of levodopa and other dopamine agonists. |
| Clinical Recommendation | Monitor and manage patient as clinically appropriate. |
7.2 Drugs Having No Clinically Important Interactions With Invega Hafyera
Based on pharmacokinetic studies with oral paliperidone, no dosage adjustment of INVEGA HAFYERA is required when administered concomitantly with valproate [see Clinical Pharmacology (12.3)] . Additionally, no dosage adjustment is necessary for valproate when co-administered with INVEGA HAFYERA [see Clinical Pharmacology (12.3)] .
Pharmacokinetic interaction between lithium and INVEGA HAFYERA is unlikely.
Paliperidone is not expected to cause clinically important pharmacokinetic interactions with drugs that are metabolized by cytochrome P450 isozymes. In vitro studies indicate that CYP2D6 and CYP3A4 may be involved in paliperidone metabolism; however, there is no evidence in vivo that inhibitors of these enzymes significantly affect the metabolism of paliperidone. Paliperidone is not a substrate of CYP1A2, CYP2A6, CYP2C9, and CYP2C19; an interaction with inhibitors or inducers of these isozymes is unlikely [see Clinical Pharmacology (12.3)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. INVEGA HAFYERA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.2)] .
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA HAFYERA is not approved for use in patients with dementia-related psychosis [see Warnings and Precautions (5.1)] .
5.2 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients With Dementia Related Psychosis
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. No studies have been conducted with oral paliperidone, the 1-month paliperidone palmitate extended-release injectable suspension, the 3-month paliperidone extended-release injectable suspension or INVEGA HAFYERA in elderly patients with dementia. These medications are not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1)] .
Structured Label Content
Section 42229-5 (42229-5)
Switching to INVEGA HAFYERA from a PP1M Product
The recommended initial INVEGA HAFYERA dose is based on the previous PP1M dose (see Table 1). Initiate INVEGA HAFYERA when the next PP1M dose is scheduled. INVEGA HAFYERA may be administered up to 1 week before or 1 week after the next scheduled PP1M dose. When switching from PP1M to INVEGA HAFYERA, the two injection cycles immediately preceding the switch should be the same dosage strength before starting INVEGA HAFYERA.
| Last Dose of PP1M
There are no equivalent doses of INVEGA HAFYERA for 39 mg, 78 mg, or 117 mg doses of a PP1M product, which were not studied
[see
Clinical Studies (14)]
.
|
Initial Dose of INVEGA HAFYERA |
|---|---|
| 156 mg | 1,092 mg |
| 234 mg | 1,560 mg |
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: January 2025 | |||
|
PATIENT INFORMATION
INVEGA HAFYERA ® (in-VAY-guh HAF-ye-RA) (paliperidone palmitate) extended-release injectable suspension |
||||
|
What is the most important information I should know about INVEGA HAFYERA?
INVEGA HAFYERA may cause serious side effects, including:
|
||||
|
What is INVEGA HAFYERA?
INVEGA HAFYERA is a prescription medicine given by injection by a healthcare provider 1 time every 6 months and used for the treatment of schizophrenia in adults who have been adequately treated with either:
|
||||
| Do not receive INVEGA HAFYERA if you are allergic to paliperidone palmitate, risperidone, or any of the ingredients in INVEGA HAFYERA. See the end of this Patient Information leaflet for a complete list of ingredients in INVEGA HAFYERA. | ||||
Before receiving INVEGA HAFYERA, tell your healthcare provider about all your medical conditions, including if you:
INVEGA HAFYERA and other medicines may affect each other causing possible serious side effects. INVEGA HAFYERA may affect the way other medicines work, and other medicines may affect how INVEGA HAFYERA works. Your healthcare provider can tell you if it is safe to receive INVEGA HAFYERA with your other medicines. Do not start or stop any medicines during treatment with INVEGA HAFYERA without talking to your healthcare provider first. Know the medicines you take. Keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine. |
||||
How will I receive INVEGA HAFYERA?
|
||||
What should I avoid while receiving INVEGA HAFYERA?
|
||||
|
What are the possible side effects of INVEGA HAFYERA?
INVEGA HAFYERA may cause serious side effects, including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
||||
|
|
|||
| These are not all the possible side effects of INVEGA HAFYERA.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
|
General information about INVEGA HAFYERA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about INVEGA HAFYERA that is written for health professionals. |
||||
|
What are the ingredients in INVEGA HAFYERA?
Active ingredient: paliperidone palmitate Inactive ingredients: polysorbate 20, polyethylene glycol 4000, citric acid monohydrate, sodium dihydrogen phosphate monohydrate, sodium hydroxide, and water for injection Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, NJ 08560, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2021 For more information, go to www.invegahafyerahcp.com or call 1-800-526-7736. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Store at room temperature 20 °C to 25 °C (68 °F to 77 °F); excursions between 15 °C and 30 °C (59 °F and 86 °F) are permitted. Do not mix with any other product or diluent.
Ship and store in a horizontal position. See arrows on product carton for proper orientation.
5.8 Falls
Somnolence, postural hypotension, motor and sensory instability have been reported with the use of antipsychotics, including paliperidone palmitate, which may lead to falls and, consequently, fractures or other fall-related injuries. For patients, particularly the elderly, with diseases, conditions, or medications that could exacerbate these effects, assess the risk of falls when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
9.2 Abuse
Paliperidone has not been systematically studied in animals or humans for its potential for abuse.
5.12 Seizures
In the 6-month paliperidone palmitate extended-release injectable suspension double-blind active controlled trial there were no reports of seizures or convulsions, nor were any reports made in the long-term maintenance trial of PP3M. In the pivotal clinical studies with PP1M which included four fixed-dose, double-blind, placebo-controlled studies in subjects with schizophrenia, <1% (1/1293) of subjects treated with the PP1M experienced an adverse event of convulsion compared with <1% (1/510) of placebo-treated subjects who experienced an adverse event of grand mal convulsion.
Like other antipsychotic drugs, INVEGA HAFYERA should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older.
5.14 Priapism
A case (0.2%) of priapism was reported in the clinical trial with INVEGA HAFYERA. Priapism has been reported with oral paliperidone during postmarketing surveillance. Drugs with alpha-adrenergic blocking effects have been reported to induce priapism. Severe priapism may require surgical intervention.
11 Description (11 DESCRIPTION)
INVEGA HAFYERA ® contains a racemic mixture of (+)- and (-)- paliperidone palmitate. Paliperidone palmitate is an atypical antipsychotic belonging to the chemical class of benzisoxazole derivatives. The chemical name is (9 RS)-3-[2-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)piperidin-1-yl]ethyl]-2-methyl-4-oxo-6,7,8,9-tetrahydro-4 H-pyrido[1,2- a]pyrimadin-9-yl hexadecanoate. Its molecular formula is C 39H 57FN 4O 4 and its molecular weight is 664.89. The structural formula is:
Paliperidone palmitate is very slightly soluble in ethanol and methanol, practically insoluble in polyethylene glycol 400 and propylene glycol, and slightly soluble in ethyl acetate.
INVEGA HAFYERA is available as a white to off-white sterile aqueous extended-release suspension for intramuscular injection in dose strengths of 1,092 mg and 1,560 mg paliperidone palmitate. The drug product hydrolyzes to the active moiety, paliperidone, resulting in dose strengths of 700 mg, and 1,000 mg of paliperidone, respectively. The inactive ingredients are polysorbate 20 (10 mg/mL), polyethylene glycol 4000 (75 mg/mL), citric acid monohydrate (7.5 mg/mL), sodium dihydrogen phosphate monohydrate (6 mg/mL), sodium hydroxide (5.4 mg/mL), and water for injection.
INVEGA HAFYERA is provided in a single-dose prefilled syringe (cyclic-olefin-copolymer) prefilled with either 700 mg (3.5 mL), or 1,000 mg (5.0 mL) paliperidone (as 1,092 mg, or 1,560 mg paliperidone palmitate) suspension with a tip cap, plunger rod, backstop and a thin walled 20G, 1½-inch safety needle.
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. INVEGA HAFYERA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
9.3 Dependence
Paliperidone has not been systematically studied in animals or humans for its potential for tolerance or physical dependence.
8.4 Pediatric Use
Safety and effectiveness of INVEGA HAFYERA in patients less than 18 years of age have not been established. Use of INVEGA HAFYERA is not recommended in pediatric patients because of the potential longer duration of an adverse event. In clinical trials of oral paliperidone, there were notably higher incidences of dystonia, hyperkinesia, tremor, and parkinsonism in the adolescent population as compared to the adult studies.
8.5 Geriatric Use
The clinical study of INVEGA HAFYERA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
This drug is substantially excreted by the kidney and clearance is decreased in patients with renal impairment [see Clinical Pharmacology (12.3)] . Because elderly patients are more likely to have decreased renal function, INVEGA HAFYERA is not recommended to be used in elderly patients with mild, moderate or severe renal impairment [see Use in Specific Populations (8.6)] .
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of INVEGA HAFYERA for the treatment of schizophrenia in patients who had previously been stably treated with either PP1M for at least 4 months or PP3M for at least one 3-month injection cycle was evaluated in a randomized, double-blind, active-controlled, interventional, parallel-group, multicenter, non-inferiority study designed to evaluate time to relapse in adults with a DSM-5 diagnosis of schizophrenia.
Patients could enter the study if previously treated with PP1M at dosages of 156 or 234 mg, PP3M at dosages of 546 or 819 mg, injectable risperidone at dosages of 50 mg, or any oral antipsychotic with a reason to change (e.g., efficacy, safety, tolerability, or a preference for a long-acting injectable medication) and with a PANSS total score of <70 points.
After establishing tolerability with PP1M (at dosages of 156 or 234 mg) or PP3M (at dosages of 546 or 819 mg) and clinical stability, defined by having a PANSS total score of <70 points for the previous 2 assessments prior to the double-blind phase, patients were randomized in a 2:1 ratio to receive INVEGA HAFYERA (478 patients) or PP3M (224 patients).
The primary efficacy variable was time to first relapse in the double-blind phase. The primary efficacy analysis was based on the difference in Kaplan-Meier 12-month estimates of percentage of subjects remaining relapse-free between INVEGA HAFYERA and 3-month paliperidone palmitate extended-release injectable suspension. Relapse was pre-defined as emergence of one or more of the following: psychiatric hospitalization, ≥25% increase (if the baseline score was >40) or a 10-point increase (if the baseline score was ≤40) in total PANSS score on two consecutive assessments, deliberate self-injury, violent behavior, suicidal/homicidal ideation: a score of ≥5 (if the maximum baseline score was ≤3) or ≥6 (if the maximum baseline score was 4) on two consecutive assessments of the specific PANSS items.
A relapse event was experienced by 7.5% and 4.9% of patients in the INVEGA HAFYERA and PP3M treatment groups, respectively, with the Kaplan-Meier estimated difference (INVEGA HAFYERA – PP3M) of 2.9% (95% CI: -1.1 to 6.8). The upper bound of the 95% CI (6.8%) was less than 10%, the prespecified non-inferiority margin. The study demonstrated non-inferiority of INVEGA HAFYERA to PP3M. A Kaplan-Meier plot of time to relapse by treatment group is shown in Figure 4.
Figure 4: Kaplan-Meier Plot of Cumulative Proportion of Patients with Relapse Over Time
An evaluation of population subgroups did not reveal any clinically significant differences in responsiveness on the basis of gender, age, or race.
4 Contraindications (4 CONTRAINDICATIONS)
INVEGA HAFYERA is contraindicated in patients with a known hypersensitivity to either paliperidone or risperidone, or to any of the excipients in the INVEGA HAFYERA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone palmitate is converted to paliperidone, which is a metabolite of risperidone.
5.4 Qt Prolongation (5.4 QT Prolongation)
Paliperidone causes a modest increase in the corrected QT (QTc) interval. The use of paliperidone should be avoided in combination with other drugs that are known to prolong QTc including Class 1A (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications, antipsychotic medications (e.g., chlorpromazine, thioridazine), antibiotics (e.g., gatifloxacin, moxifloxacin), or any other class of medications known to prolong the QTc interval. Paliperidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Certain circumstances may increase the risk of the occurrence of Torsades de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including (1) bradycardia; (2) hypokalemia or hypomagnesemia; (3) concomitant use of other drugs that prolong the QTc interval; and (4) presence of congenital prolongation of the QT interval.
The effects of paliperidone on the QT interval were evaluated in a double-blind, active-controlled (moxifloxacin 400 mg single dose), multicenter Thorough QT study with oral paliperidone in adult patients, and in four fixed-dose efficacy studies and one maintenance study of the 1-month paliperidone palmitate injectable product.
In the Thorough QT study (n=141), the 8 mg dose of immediate-release oral paliperidone (n=50) showed a mean placebo-subtracted increase from baseline in QTcLD (QT interval corrected for heart rate using the population specified linear derived method) of 12.3 msec (90% CI: 8.9; 15.6) on day 8 at 1.5 hours post-dose. The mean steady-state peak plasma concentration for this 8 mg dose of paliperidone immediate release (C max ss=113 ng/mL) was approximately 1.3-fold the exposure with the maximum recommended 1,560 mg dose of INVEGA HAFYERA administered in the gluteal muscle (mean C max md=89.3 ng/mL). In this same study, a 4 mg dose of the immediate-release oral formulation of paliperidone, for which C max ss=35 ng/mL, showed an increased placebo-subtracted QTcLD of 6.8 msec (90% CI: 3.6; 10.1) on day 2 at 1.5 hours post-dose.
In the four fixed-dose efficacy studies of the 1-month paliperidone palmitate injectable product, no subject had a change in QTcLD exceeding 60 msec and no subject had a QTcLD value of >500 msec at any time point. In the maintenance study, no subject had a QTcLD change >60 msec, and one subject had a QTcLD value of 507 msec (Bazett's QT corrected interval [QTcB] value of 483 msec); this latter subject also had a heart rate of 45 beats per minute.
In the INVEGA HAFYERA randomized double-blind active controlled study in subjects with schizophrenia, during the double-blind Phase, QTcLD exceeding 60 msec was observed in 2 subjects (0.4%) in the INVEGA HAFYERA treatment group and in 2 subjects (0.9%) in the PP3M treatment group. No subject had a QTcLD value of >480 msec at any point in the study.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1)]
- Cerebrovascular adverse reactions, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2)]
- Neuroleptic malignant syndrome [see Warnings and Precautions (5.3)]
- QT prolongation [see Warnings and Precautions (5.4)]
- Tardive dyskinesia [see Warnings and Precautions (5.5)]
- Metabolic changes [see Warnings and Precautions (5.6)]
- Orthostatic hypotension and syncope [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia, neutropenia, and agranulocytosis [see Warnings and Precautions (5.9)]
- Hyperprolactinemia [see Warnings and Precautions (5.10)]
- Potential for cognitive and motor impairment [see Warnings and Precautions (5.11)]
- Seizures [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
- Priapism [see Warnings and Precautions (5.14)]
- Disruption of body temperature regulation [see Warnings and Precautions (5.15)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
Use of INVEGA HAFYERA is not recommended for use in patients with moderate or severe renal impairment (creatinine clearance <50 mL/min). Use of INVEGA HAFYERA in patients with mild renal impairment (creatinine clearance ≥50 mL/min to <80 mL/min) is based on the patient’s previous dose of PP1M or PP3M before transitioning to INVEGA HAFYERA [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)] .
Instructions for Use (INSTRUCTIONS FOR USE)
INVEGA HAFYERA
®
in-VAY-guh HAF-ye-RA
(paliperidone palmitate)
extended-release injectable suspension
For Gluteal Intramuscular
Injection Only
Administer every 6 months
| Shake syringe with the syringe tip cap pointing up VERY FAST for at least 15 seconds, rest briefly, then shake again for 15 seconds |
Important
| Shipping and storing the carton in a horizontal orientation improves the ability to resuspend this highly concentrated product. |
INVEGA HAFYERA must be administered by a healthcare professional as a single injection.
Do not divide dose into multiple injections.
INVEGA HAFYERA is intended for gluteal intramuscular use only. Inject slowly, deep into the muscle taking care to avoid injection into a blood vessel.
Dosing
Administer INVEGA HAFYERA once every 6 months.
Thin Wall Safety Needle
Thin wall safety needle is designed to be used with INVEGA HAFYERA. Therefore, it is important to only use the needle provided in the INVEGA HAFYERA suspension kit.
Dose pack contents
- Prepare for the injection: this highly concentrated product requires specific steps to resuspend
Hold syringe with the tip cap pointing up
Shake syringe VERY FAST for at least 15 seconds, rest briefly, then shake again for 15 seconds
To ensure complete resuspension shake syringe with:
- Short, VERY FAST up and down motion
- Loose wrist
Proceed to the next step immediately after shaking.
Check suspension for solid product
|
|
|
|
Product may clog. Shake syringe with the syringe tip cap pointing up VERY FAST for at least 15 seconds, rest, then shake again for 15 seconds. |
Open needle pouch
Peel off the pouch cover.
Place pouch with the needle inside on a clean surface.
Remove syringe tip cap and attach needle
Hold the syringe with the tip cap pointing up.
Twist and pull off the cap.
Attach the safety needle to the syringe using a gentle twisting motion to avoid needle hub cracks or damage. Always check for signs of damage or leakage prior to administration.
Pull back plunger
Hold the syringe upright.
Gently pull back the plunger to clear the syringe tip of any solid product. This will make pressing the plunger easier during the injection.
Remove air bubbles
Press the plunger carefully until a drop of liquid comes out of the needle tip.
- Slowly inject entire content and confirm
|
Select and clean a gluteal injection site
Wipe the gluteal site with an alcohol swab and allow it to dry. Do not touch, fan or blow on the injection site after you have cleaned it. |
|
|
Remove needle sheath
Pull the needle sheath away from the needle in a straight motion. Do not twist the sheath, as this may loosen the needle from the syringe. |
|
|
Slowly inject and confirm
Use slow, firm, consistent pressure to press the plunger completely. This should take approximately 30 seconds. Continue to press the plunger if you feel resistance. This is normal. While the needle is in the gluteal muscle, confirm that the entire content of the syringe has been injected. Remove needle from the muscle. |
- After the injection
|
Secure needle
After the injection is complete, use your thumb or a flat surface to secure the needle in the safety device. The needle is secure when you hear a "click" sound. |
|
|
Dispose of properly and check injection site
Dispose of the syringe in an approved sharps container. There may be a small amount of blood or liquid at the injection site. Hold pressure over the skin with a cotton ball or gauze pad until any bleeding stops. Do not rub the injection site. If needed, cover injection site with a bandage. |
Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560, USA
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2021
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Revised: 2/2025
12.2 Pharmacodynamics
In vitro, paliperidone acts as an antagonist at the central dopamine D 2 and serotonin 5HT 2A receptors with binding affinities (Ki values) of 1.6–2.8 nM and 0.8–1.2 nM, respectively. Paliperidone also acts as an antagonist at histamine H 1 and α 1 and α 2 adrenergic receptors with binding affinities of 32 nM, 4 nM, and 17 nM, respectively. Paliperidone has no appreciable affinity for cholinergic muscarinic or β 1- and β 2-adrenergic receptors. The pharmacological activity of the (+)- and (-)- paliperidone enantiomers is qualitatively and quantitatively similar.
12.3 Pharmacokinetics
The pharmacokinetics for INVEGA HAFYERA presented below are based on gluteal administration only.
INVEGA HAFYERA delivers paliperidone over a 6-month period, compared to the 1-month or 3-month products which are administered every month or every three months, respectively. INVEGA HAFYERA doses of 1,092 mg and 1,560 mg result in paliperidone total exposure ranges that are encompassed within the exposure range for corresponding doses of 1-month paliperidone palmitate injections (PP1M) (156 mg and 234 mg) or corresponding doses of 3-month paliperidone palmitate (PP3M) injections (546 mg and 819 mg, respectively) or to corresponding once daily doses of paliperidone extended-release tablets. However, mean trough concentrations (C trough) at the end of the dosing interval were approximately 20 – 25% lower for INVEGA HAFYERA as compared to corresponding doses of 3-month paliperidone palmitate. The mean peak concentration (C max) was higher (1.4 to 1.5-fold) for INVEGA HAFYERA as compared to corresponding doses of 3-month paliperidone palmitate.
Inter-subject variability in paliperidone PK parameters for INVEGA HAFYERA ranged from 42 to 48% for AUC 6months and ranged from 56 to 103% for C max. Because of the difference in pharmacokinetic profiles among the four paliperidone products, caution should be exercised when making a direct comparison of their pharmacokinetic properties.
5.6 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.5 Tardive Dyskinesia
Tardive dyskinesia, a syndrome of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible appear to increase as the duration of treatment and the total cumulative dose. The syndrome can develop after relatively brief treatment periods, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment itself may suppress (or partially suppress) the signs and symptoms of the syndrome and may thus mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, INVEGA HAFYERA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that is known to respond to antipsychotic drugs. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with INVEGA HAFYERA, drug discontinuation should be considered. Consideration should be given to the long-acting nature of INVEGA HAFYERA. However, some patients may require treatment with INVEGA HAFYERA despite the presence of the syndrome.
8.7 Hepatic Impairment
INVEGA HAFYERA has not been studied in patients with hepatic impairment. Based on a study with oral paliperidone, no dose adjustment is required in patients with mild or moderate hepatic impairment. Paliperidone has not been studied in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
INVEGA HAFYERA, an every-six-month injection, is indicated for the treatment of schizophrenia in adults after they have been adequately treated with:
- A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA) for at least four months, or
- An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA) for at least one three-month cycle.
5.10 Hyperprolactinemia
Like other drugs that antagonize dopamine D 2receptors, paliperidone elevates prolactin levels and the elevation persists during chronic administration. Paliperidone has a prolactin-elevating effect similar to that seen with risperidone, a drug that is associated with higher levels of prolactin than other antipsychotic drugs.
Hyperprolactinemia, regardless of etiology, may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously detected breast cancer. An increase in the incidence of pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)] . Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
Median prolactin levels remained relatively stable throughout the open-label and double-blind phases in male subjects, whereas in female subjects, median prolactin levels increased. During the double-blind phase, median prolactin levels continued to increase after dosing in both the INVEGA HAFYERA and PP3M groups, returning to baseline level at Month 6 and at Month 12 (end of double-blind phase).
During the double-blind phase, prolactin levels relative to reference range (>13.13 ng/mL in males and >26.72 ng/mL in females) from maintenance baseline were noted in a similar percentage of subjects in the INVEGA HAFYERA and PP3M groups in both males (35% vs 36%) and females (29% vs. 30%). In the INVEGA HAFYERA group, 14 females (2.9%) and 4 males (0.8%) experienced potentially prolactin-related adverse reactions, while 6 females (2.7%) and 1 male (0.4%) in the PP3M experienced potentially prolactin-related adverse reactions.
12.1 Mechanism of Action
Paliperidone palmitate is hydrolyzed to paliperidone [see Clinical Pharmacology (12.3)] . Paliperidone is the major active metabolite of risperidone. The mechanism of action of paliperidone is unclear. However, its efficacy in the treatment of schizophrenia could be mediated through a combination of central dopamine D 2 and serotonin 5HT 2A receptor antagonism.
9.1 Controlled Substance
INVEGA HAFYERA contains paliperidone, which is not a controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities). ( 5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation of drug and close monitoring. ( 5.3)
- QT Prolongation: Avoid use with drugs that also increase QT interval and in patients with risk factors for prolonged QT interval. ( 5.4)
- Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.5)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. ( 5.6)
- Orthostatic Hypotension and Syncope: Use with caution in patients with known cardiovascular or cerebrovascular disease and patients predisposed to hypotension. ( 5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with pre-existing low white blood cell count (WBC) or a history of leukopenia or neutropenia. Consider discontinuing INVEGA HAFYERA if a clinically significant decline in WBC occurs in the absence of other causative factors. ( 5.9)
- Hyperprolactinemia: Prolactin elevations occur and persist during chronic administration. ( 5.10)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery. ( 5.11)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. ( 5.12)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer INVEGA HAFYERA by gluteal injection once every 6 months by a healthcare professional. Do not administer by any other route. ( 2.1)
- See Full Prescribing Information for complete dosing information. ( 2.2)
- Initiate INVEGA HAFYERA when the next once-a-month or every three-month paliperidone palmitate extended-release injectable suspension dose is scheduled. Dose is based on the previous once-a-month or every-three-month product. (
2.2):
INVEGA HAFYERA Doses for Adults Adequately Treated with Once-a-month paliperidone palmitate extended-release injectable suspension (PP1M)Switching from the PP1M 39 mg, 78 mg and 117 mg doses was not studied.
If the Last Dose of PP1M is: Initiate INVEGA HAFYERA at the Following Dose: 156 mg 1,092 mg 234 mg 1,560 mg INVEGA HAFYERA Doses for Adults Adequately Treated with Every-three-month paliperidone palmitate injectable suspension (PP3M)Switching from the PP3M 273 mg and 410 mg doses was not studied.If the Last Dose of PP3M is: Initiate INVEGA HAFYERA at the Following Dose: 546 mg 1,092 mg 819 mg 1,560 mg - Missed Doses: Refer to the Full Prescribing Information. ( 2.3)
- See Full Prescribing Information for important preparation and administration information. ( 2.5)
- Moderate to severe renal impairment (creatinine clearance <50 mL/min): INVEGA HAFYERA is not recommended. ( 2.4)
- Mild renal impairment (creatinine clearance ≥ 50 mL/min to < 80 mL/min): Adjust dosage and stabilize the patient using PP1M before transitioning to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA. See appropriate table above. ( 2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
INVEGA HAFYERA is a white to off-white aqueous extended-release injectable suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate in single-dose prefilled syringes.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of paliperidone; because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: angioedema, catatonia, ileus, somnambulism, swollen tongue, thrombotic thrombocytopenic purpura, urinary incontinence, and urinary retention.
Cases of anaphylactic reaction after injection with the 1-month paliperidone palmitate extended-release suspension have been reported during postmarketing experience in patients who have previously tolerated oral risperidone or oral paliperidone.
Paliperidone is the major active metabolite of risperidone. Adverse reactions reported with oral risperidone and risperidone long-acting injection can be found in the Adverse Reactions (6) section of the Prescribing Information for those products.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. ( 8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.3 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with antipsychotic drugs, including paliperidone.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, including delirium, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, discontinue INVEGA HAFYERA and provide symptomatic treatment and monitoring.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
INVEGA HAFYERA ® is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle.
1,092 mg paliperidone palmitate kit (NDC 50458-611-01)
1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
5.7 Orthostatic Hypotension and Syncope
Paliperidone can induce orthostatic hypotension and syncope in some patients because of its alpha-adrenergic blocking activity.
Use INVEGA HAFYERA with caution in patients with known cardiovascular disease (e.g., heart failure, history of myocardial infarction or ischemia, conduction abnormalities), cerebrovascular disease, or conditions that predispose the patient to hypotension (e.g., dehydration, hypovolemia, and treatment with antihypertensive medications). Monitoring of orthostatic vital signs should be considered in patients who are vulnerable to hypotension.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Injection site toxicity was assessed in minipigs injected intramuscularly with the 6-month paliperidone palmitate extended-release injectable suspension at doses up to 2,115 mg, which is slightly above the MRHD. Injection site inflammatory reactions were greater and more advanced than reactions to the 1-month paliperidone palmitate extended-release injectable suspension. Reversibility of these findings was not examined.
Principal Display Panel 5 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 5 mL Syringe Carton)
NDC 50458-612-01
Single-dose prefilled syringe. Use entire contents of syringe.
INVEGA HAFYERA™
(paliperidone palmitate)
extended-release injectable suspension
FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
Each injection must be administered only by
a healthcare professional.
Shake before using.
CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle).
Recommended Dosage: See Prescribing Information.
Rx only
Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted.
SHIP AND STORE
THIS SIDE UP
LAY
FLAT
1,560 mg/5 mL
Each single-dose prefilled syringe contains
1,560 mg paliperidone palmitate.
6
MONTHS
Administer
every 6 months
Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds
5.15 Disruption of Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing INVEGA HAFYERA to patients who will be experiencing conditions which may contribute to an elevation in core body temperature, e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration.
Principal Display Panel 3.5 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 3.5 mL Syringe Carton)
NDC 50458-611-01
Single-dose prefilled syringe. Use entire contents of syringe.
INVEGA HAFYERA™
(paliperidone palmitate)
extended-release injectable suspension
FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
Each injection must be administered only by
a healthcare professional.
Shake before using.
CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle).
Recommended Dosage: See Prescribing Information.
Rx only
Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted.
SHIP AND STORE
THIS SIDE UP
LAY
FLAT
1,092 mg/3.5 mL
Each single-dose prefilled syringe contains
1,092 mg paliperidone palmitate.
6
MONTHS
Administer
every 6 months
Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds
5.9 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trial and/or postmarketing experience, events of leukopenia and neutropenia have been reported temporally related to antipsychotic agents, including INVEGA HAFYERA. Agranulocytosis has also been reported.
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or a drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of INVEGA HAFYERA at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Discontinue INVEGA HAFYERA in patients with severe neutropenia (absolute neutrophil count <1000/mm 3) and follow their WBC until recovery.
5.11 Potential for Cognitive and Motor Impairment
Somnolence and sedation were reported as adverse reactions in patients treated with INVEGA HAFYERA [see Adverse Reactions (6.1)] . Antipsychotics, including INVEGA HAFYERA, have the potential to impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that paliperidone therapy does not adversely affect them.
2.1 Important Dosage and Administration Information
- INVEGA HAFYERA must be administered as a gluteal intramuscular injection by a healthcare professional once every 6 months. Do not administer by any other route [see Dosage and Administration (2.5)] .
- Initiate INVEGA HAFYERA only after adequate treatment has been established with either:
- A once-a-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA SUSTENNA), referred to as PP1M, once monthly for at least four months; or
- An every-three-month paliperidone palmitate extended-release injectable suspension (e.g., INVEGA TRINZA), referred to as PP3M, once every three months for at least one three-month injection cycle.
- See Prescribing Information of the PP1M and PP3M products for the recommended dosage of these products.
2.5 Instructions for Preparation and Administration
- To be prepared and administered by a healthcare provider only.
- Read the instructions for preparation and administration below and consider referring to the separate Healthcare Provider "Instructions for Use" for preparation and administration considerations.
- For gluteal intramuscular injection only. Do not inject by any other route. As a universal precaution, always wear gloves.
- Inspect INVEGA HAFYERA for particulate matter and discoloration prior to administration.
- Do not mix with any other product or diluent.
- After shaking, INVEGA HAFYERA should appear uniform, thick and milky white .
- Do not use needles from the PP1M or PP3M products or other commercially-available needles to reduce the risk of blockage.
- Avoid inadvertent injection into a blood vessel. Administer the dose in a single injection; do not administer the dose in divided injections. Inject slowly, deep into the upper-outer quadrant of the gluteal muscle. Future injections should be alternated between the two gluteal muscles.
8.8 Patients With Parkinson's Disease Or Lewy Body Dementia (8.8 Patients with Parkinson's Disease or Lewy Body Dementia)
Patients with Parkinson's Disease or Dementia with Lewy Bodies can experience increased sensitivity to INVEGA HAFYERA. Manifestations can include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with neuroleptic malignant syndrome.
2.4 Dosage Recommendations in Patients With Renal Impairment (2.4 Dosage Recommendations in Patients with Renal Impairment)
INVEGA HAFYERA has not been systematically studied in patients with renal impairment [see Clinical Pharmacology (12.3)] . For patients with mild renal impairment (creatinine clearance ≥50 mL/min to < 80 mL/min [Cockcroft-Gault Formula]), adjust dosage and stabilize the patient using PP1M before transitioning from PP1M to INVEGA HAFYERA, or from PP1M to PP3M before transitioning to INVEGA HAFYERA (see Table 1) [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)]. Refer to the Prescribing Information of PP1M or PP3M product for the recommended PP1M or PP3M dosage in patients with mild renal impairment.
INVEGA HAFYERA is not recommended in patients with moderate or severe renal impairment (creatinine clearance <50 mL/min) [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
7.1 Drugs Having Clinically Important Interactions With Invega Hafyera (7.1 Drugs Having Clinically Important Interactions with INVEGA HAFYERA)
Because paliperidone palmitate is hydrolyzed to paliperidone, results from studies with oral paliperidone should be taken into consideration when assessing drug-drug interaction potential. In addition, consider the 6-month dosing interval and the half-life of INVEGA HAFYERA [see Clinical Pharmacology (12.3)] .
Table 10 presents clinically significant drug interactions with INVEGA HAFYERA.
| Centrally acting Drugs and Alcohol | |
| Clinical Rationale | Given the primary CNS effects of paliperidone, concomitant use of centrally acting drugs and alcohol may modulate the CNS effects of INVEGA HAFYERA. |
| Clinical Recommendation | INVEGA HAFYERA should be used with caution with other centrally acting drugs and alcohol. |
| Drugs with Potential for Inducing Orthostatic Hypotension | |
| Clinical Rationale | Because INVEGA HAFYERA has the potential for inducing orthostatic hypotension, an additive effect may occur when INVEGA HAFYERA is administered with other therapeutic agents that have this potential [see Warnings and Precautions (5.7)] . |
| Clinical Recommendation | Monitor orthostatic vital signs in patients who are vulnerable to hypotension [see Warnings and Precautions (5.7)] . |
| Strong Inducers of CYP3A4 and P-gp | |
| Clinical Rationale | The concomitant use of INVEGA HAFYERA and strong inducers of CYP3A4 and P-gp may decrease the exposure of paliperidone [see Clinical Pharmacology (12.3)] . |
| Clinical Recommendation | Avoid using CYP3A4 and/or P-gp inducers with INVEGA HAFYERA during the 6-month dosing interval, if possible. If administering a strong inducer is necessary, consider managing the patient using paliperidone extended-release tablets [see Dosage and Administration (2.1)] . |
| Examples | carbamazepine, rifampin, or St. John's Wort |
| Levodopa and Other Dopamine Agonists | |
| Clinical Rationale | Paliperidone may antagonize the effect of levodopa and other dopamine agonists. |
| Clinical Recommendation | Monitor and manage patient as clinically appropriate. |
7.2 Drugs Having No Clinically Important Interactions With Invega Hafyera (7.2 Drugs Having No Clinically Important Interactions with INVEGA HAFYERA)
Based on pharmacokinetic studies with oral paliperidone, no dosage adjustment of INVEGA HAFYERA is required when administered concomitantly with valproate [see Clinical Pharmacology (12.3)] . Additionally, no dosage adjustment is necessary for valproate when co-administered with INVEGA HAFYERA [see Clinical Pharmacology (12.3)] .
Pharmacokinetic interaction between lithium and INVEGA HAFYERA is unlikely.
Paliperidone is not expected to cause clinically important pharmacokinetic interactions with drugs that are metabolized by cytochrome P450 isozymes. In vitro studies indicate that CYP2D6 and CYP3A4 may be involved in paliperidone metabolism; however, there is no evidence in vivo that inhibitors of these enzymes significantly affect the metabolism of paliperidone. Paliperidone is not a substrate of CYP1A2, CYP2A6, CYP2C9, and CYP2C19; an interaction with inhibitors or inducers of these isozymes is unlikely [see Clinical Pharmacology (12.3)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. INVEGA HAFYERA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.2)] .
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. INVEGA HAFYERA is not approved for use in patients with dementia-related psychosis [see Warnings and Precautions (5.1)] .
5.2 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients With Dementia Related Psychosis (5.2 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis)
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. No studies have been conducted with oral paliperidone, the 1-month paliperidone palmitate extended-release injectable suspension, the 3-month paliperidone extended-release injectable suspension or INVEGA HAFYERA in elderly patients with dementia. These medications are not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:47.165984 · Updated: 2026-03-14T22:23:15.770287