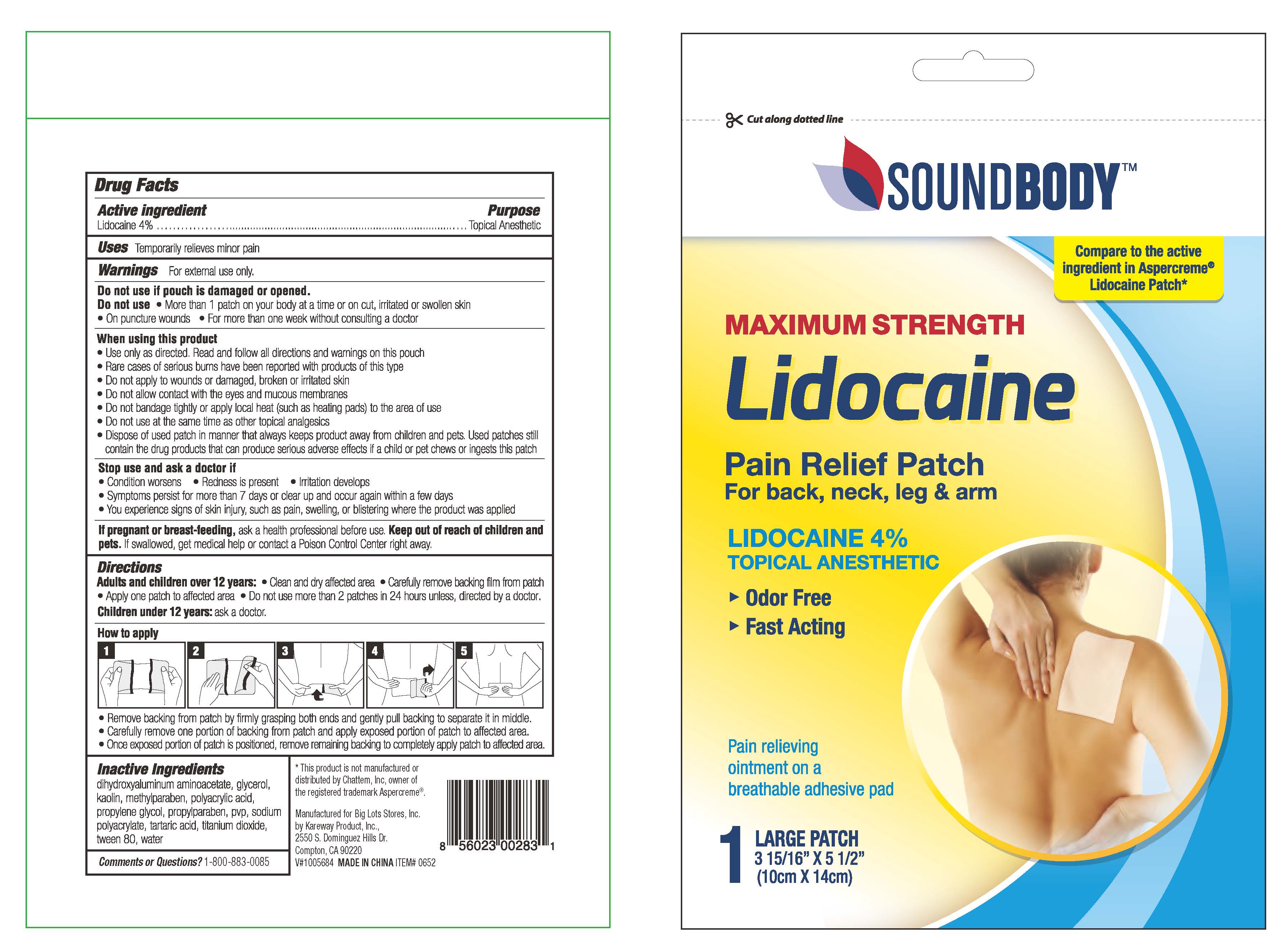
Sound Body Lidocaine Patch
6cbb6d67-ded2-3b73-e053-2a91aa0a363b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Purpose
Topical Anesthetic
Medication Information
Purpose
Topical Anesthetic
Description
Lidocaine 4%
Uses
Temporarily relieves minor pain
Purposes
Topical Anesthetic
Warnings
For external use only
Directions
adults and children over 12 years:
- clean and dry affected area
- remove backing from patch by firmly grasping both ends and gently pulling until backing separates in middle
- carefully remove smaller portion of backing from patch and apply exposed portion of patch to affected area
- once exposed portion of patch is positioned, carefully remove remaining backing to completely apply patch to affected area
- use 2 patch for up to 24 hours
children 12 years or younger: ask a doctor
Do Not Use
- more than 1 patch on your body at a time or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredient
Lidocaine 4%
Inactive Ingredients
dihydroxyaluminum aminoacetate, glycerol, kaolin, methylparaben, polyacrylic acid,
propylene glycol, propylparaben, pvp, sodium polyacrylate, tartaric acid, titanium dioxide, tween 80, water
When Using This Product
- use only as directed. Read and follow all directions and warnings on this label.
- rare cases of serious burns have been reported with products of this type
- do not apply to wounds or damaged, broken or irritated skin
- do not allow contact with the eyes and mucous membranes
- do not bandage tightly or apply local heat (such as heating pads) to the area of use
- do not use at the same time as other topical analgesics
- dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Stop Use and Ask A Doctor If
- condition worsens
- redness is present
- irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
- you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding
ask a health professional before use.
Package/label Principal Display Panel
Soundbody
MAXIMUM STRENGTH
LIDOCAINE PATCH
Label
Keep Out of Reach of Children and Pets
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
Temporarily relieves minor pain
Purposes
Topical Anesthetic
Warnings
For external use only
Directions
adults and children over 12 years:
- clean and dry affected area
- remove backing from patch by firmly grasping both ends and gently pulling until backing separates in middle
- carefully remove smaller portion of backing from patch and apply exposed portion of patch to affected area
- once exposed portion of patch is positioned, carefully remove remaining backing to completely apply patch to affected area
- use 2 patch for up to 24 hours
children 12 years or younger: ask a doctor
Do Not Use
- more than 1 patch on your body at a time or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredient (Active ingredient)
Lidocaine 4%
Inactive Ingredients
dihydroxyaluminum aminoacetate, glycerol, kaolin, methylparaben, polyacrylic acid,
propylene glycol, propylparaben, pvp, sodium polyacrylate, tartaric acid, titanium dioxide, tween 80, water
When Using This Product
- use only as directed. Read and follow all directions and warnings on this label.
- rare cases of serious burns have been reported with products of this type
- do not apply to wounds or damaged, broken or irritated skin
- do not allow contact with the eyes and mucous membranes
- do not bandage tightly or apply local heat (such as heating pads) to the area of use
- do not use at the same time as other topical analgesics
- dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Stop Use and Ask A Doctor If (Stop Use and Ask a Doctor if)
- condition worsens
- redness is present
- irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
- you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding (If Pregnant or Breast-Feeding)
ask a health professional before use.
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Soundbody
MAXIMUM STRENGTH
LIDOCAINE PATCH
Label
Keep Out of Reach of Children and Pets
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:34.613030 · Updated: 2026-03-14T23:01:03.935839