Lidopro Patch
6c9a076e-6316-427b-9ce6-c8fe2d388837
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Purpose
Topical Anesthetic
Medication Information
Purpose
Topical Anesthetic
Description
Lidocaine 4%
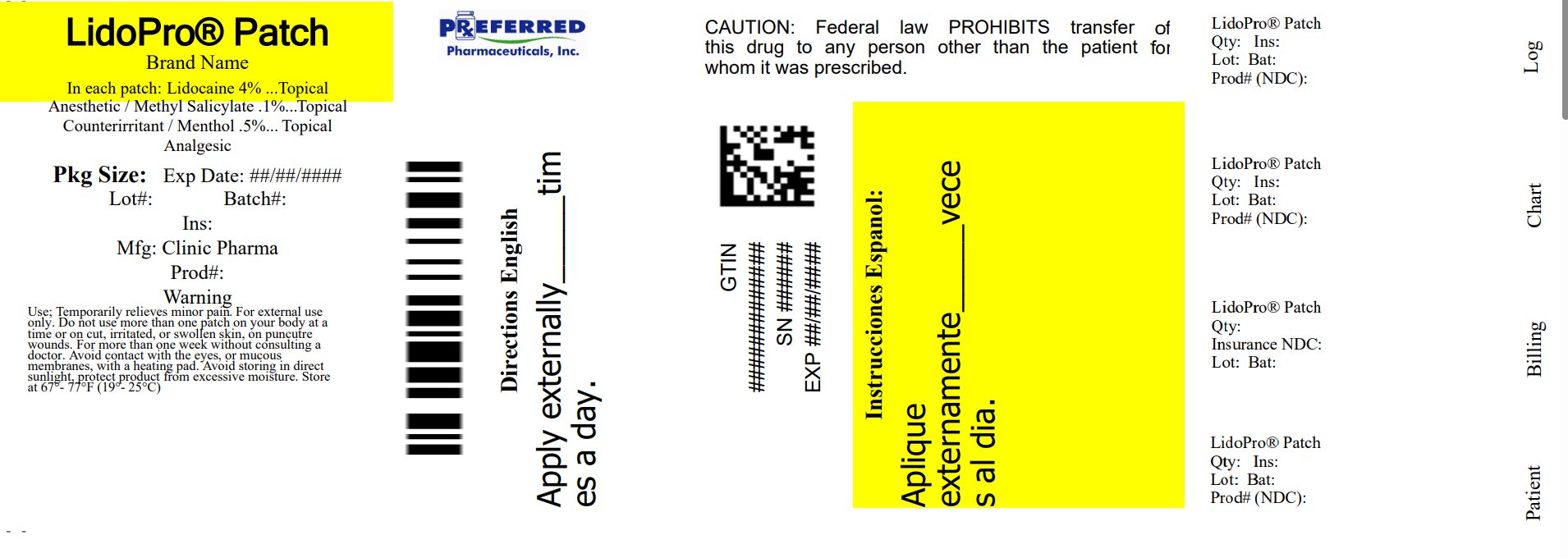
Uses
Temporarily relieves minor pain
Warnings
For external use only
Directions
adults and children over 12 years:
-
•clean and dry the affected area
-
•remove patch from plastic liner and place on the affected area
-
•use 1 patch for up to 12 hours
-
•place used patch on the liner when not in use
-
•re-use the patch up to 2 times.
children 12 years or younger: ask a doctor
Do Not Use
• more than one patch on your bidy at a time or on cuts, irritated, or swollen skin
• on puncture wounds
• for more than one week without consulting a doctor.
Active Ingredient
Lidocaine 4%
Other Ingredients:
Vegan Glycerol, Polyacrylate, Aqua. Polysorbate 80
Principal Display Panel
LidoPro patch
Relabeled By: Preferred Pharmaceuticals Inc.
NDC 68788-8594-3
When Using This Product
-
•Use only as directed. Read and follow directions and warning on this packaging.
-
•Do not apply to wounds or damaged, broken, or irritated skin
-
•Avoid contact with the eyes or mucous membranes
-
•Do not bandage tightly or apply local heat (such as heating pads) to area of use
-
•Do not use at the same time as other topical analgesics
-
•Dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Stop Use and Ask A Doctor If
-
•conditions worsens
-
•redness is present
-
•irritation develops
-
•symptoms persist for more than 7 days or clear up and occur again within a few days
-
•you experience signs of injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding,
ask health professional before use.
Keep Out of Reach of Children and Pets.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
Temporarily relieves minor pain
Purpose
Topical Anesthetic
Warnings
For external use only
Directions (DIRECTIONS)
adults and children over 12 years:
-
•clean and dry the affected area
-
•remove patch from plastic liner and place on the affected area
-
•use 1 patch for up to 12 hours
-
•place used patch on the liner when not in use
-
•re-use the patch up to 2 times.
children 12 years or younger: ask a doctor
Do Not Use (Do not use)
• more than one patch on your bidy at a time or on cuts, irritated, or swollen skin
• on puncture wounds
• for more than one week without consulting a doctor.
Active Ingredient
Lidocaine 4%
Other Ingredients: (OTHER INGREDIENTS:)
Vegan Glycerol, Polyacrylate, Aqua. Polysorbate 80
Principal Display Panel
LidoPro patch
Relabeled By: Preferred Pharmaceuticals Inc.
NDC 68788-8594-3
When Using This Product (When using this product)
-
•Use only as directed. Read and follow directions and warning on this packaging.
-
•Do not apply to wounds or damaged, broken, or irritated skin
-
•Avoid contact with the eyes or mucous membranes
-
•Do not bandage tightly or apply local heat (such as heating pads) to area of use
-
•Do not use at the same time as other topical analgesics
-
•Dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
-
•conditions worsens
-
•redness is present
-
•irritation develops
-
•symptoms persist for more than 7 days or clear up and occur again within a few days
-
•you experience signs of injury, such as pain, swelling, or blistering where the product was applied
If Pregnant Or Breast Feeding, (If pregnant or breast feeding,)
ask health professional before use.
Keep Out of Reach of Children and Pets. (Keep out of reach of children and pets.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:40.695514 · Updated: 2026-03-14T23:09:34.496634