aurum iodatum 6c
6c40ea02-4076-3880-e053-2991aa0ad6c1
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
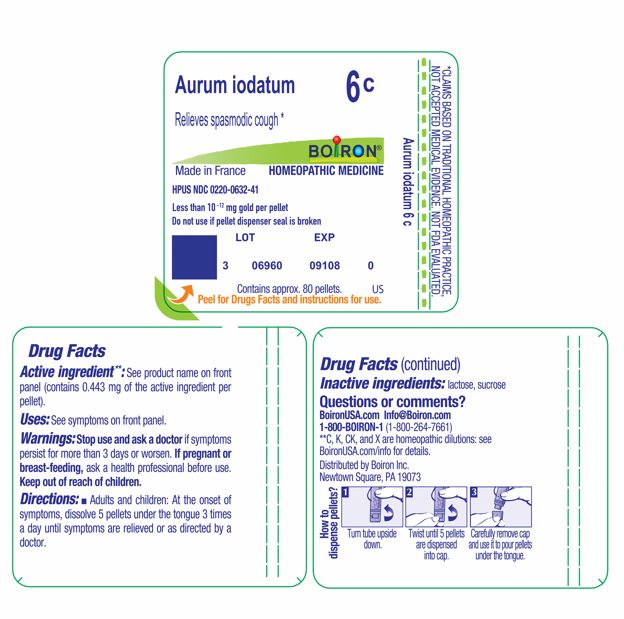
Aurum iodatum 6C HPUS Less than 10 -12 mg gold per pellet Active ingredient**: See product name on front panel (contains 0.443 mg of the active ingredient per pellet).
Medication Information
Active Ingredient
Aurum iodatum 6C HPUS
Less than 10 -12 mg gold per pellet
Active ingredient**: See product name on front panel (contains 0.443 mg of the active ingredient per pellet).
Indications and Usage
Relieves spasmodic cough *
Uses: See symptoms on front panel.
Dosage and Administration
Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
Description
Aurum iodatum 6C HPUS Less than 10 -12 mg gold per pellet Active ingredient**: See product name on front panel (contains 0.443 mg of the active ingredient per pellet).
Section 42229-5
Do not use if pellet dispenser seal is broken.
Contains approx 80 pellets.
How to dispense pellets? Turn tube upside down. Twist until 5 pellets are dispensed into cap. Carefully remove the cap and use it to pour pellets under the tongue.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen.
Section 51727-6
lactose, sucrose
Section 51945-4
Section 53413-1
1-800-BOIRON-1 (1-800-264-7661),
BoironUSA.com [email protected]
Distributed by Boiron, Inc. Newtown Square, PA 19073
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Structured Label Content
Active Ingredient (55106-9)
Aurum iodatum 6C HPUS
Less than 10 -12 mg gold per pellet
Active ingredient**: See product name on front panel (contains 0.443 mg of the active ingredient per pellet).
Indications and Usage (34067-9)
Relieves spasmodic cough *
Uses: See symptoms on front panel.
Dosage and Administration (34068-7)
Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
Section 42229-5 (42229-5)
Do not use if pellet dispenser seal is broken.
Contains approx 80 pellets.
How to dispense pellets? Turn tube upside down. Twist until 5 pellets are dispensed into cap. Carefully remove the cap and use it to pour pellets under the tongue.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen.
Section 51727-6 (51727-6)
lactose, sucrose
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
1-800-BOIRON-1 (1-800-264-7661),
BoironUSA.com [email protected]
Distributed by Boiron, Inc. Newtown Square, PA 19073
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:05.509061 · Updated: 2026-03-14T23:17:27.453851