Hevert®
6c07bbb5-7770-4ab1-b336-00703e949fc9
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
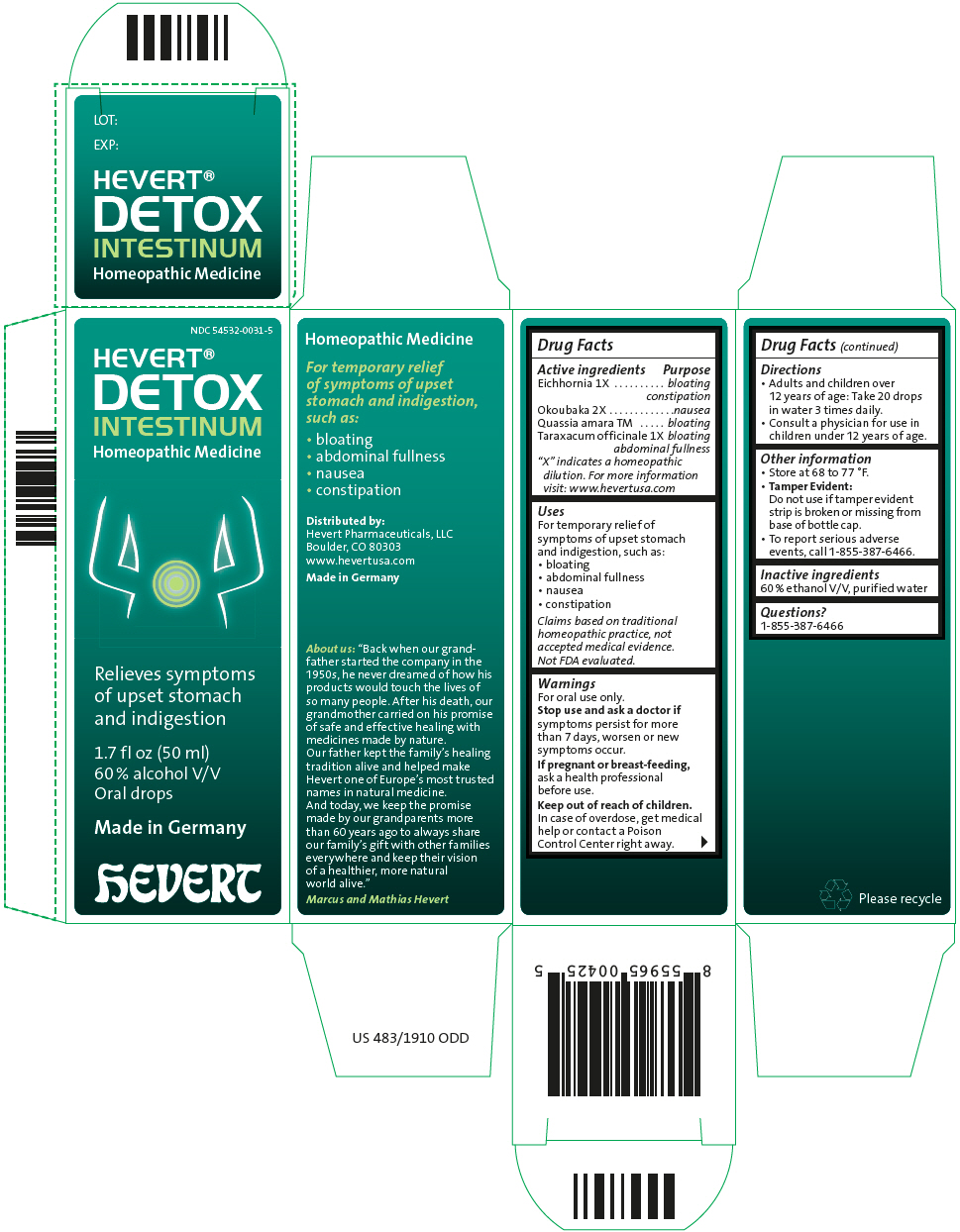
Uses
For temporary relief of symptoms of upset stomach and indigestion, such as:
- bloating
- abdominal fullness
- nausea
- constipation
Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if symptoms persist for more than 7 days, worsen or new symptoms occur.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| "X" indicates a homeopathic dilution. For more information visit: www.hevertusa.com | |
| Eichhornia 1X |
bloating constipation |
| Okoubaka 2X | nausea |
| Quassia amara TM | bloating |
| Taraxacum officinale 1X |
bloating abdominal fullness |
Warnings
For oral use only.
Directions
- Adults and children over 12 years of age: Take 20 drops in water 3 times daily.
- Consult a physician for use in children under 12 years of age.
Questions?
1-855-387-6466
Other Information
- Store at 68 to 77 °F.
-
Tamper Evident:
Do not use if tamper evident strip is broken or missing from base of bottle cap. - To report serious adverse events, call 1-855-387-6466.
Inactive Ingredients
60 % ethanol V/V, purified water
Principal Display Panel 50 Ml Bottle Carton
NDC 54532-0031-5
HEVERT®
DETOX
INTESTINUM
Homeopathic Medicine
Relieves symptoms
of upset stomach
and indigestion
1.7 fl oz (50 ml)
60 % alcohol V/V
Oral drops
Made in Germany
hEVERT
Structured Label Content
Uses
For temporary relief of symptoms of upset stomach and indigestion, such as:
- bloating
- abdominal fullness
- nausea
- constipation
Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if symptoms persist for more than 7 days, worsen or new symptoms occur.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| "X" indicates a homeopathic dilution. For more information visit: www.hevertusa.com | |
| Eichhornia 1X |
bloating constipation |
| Okoubaka 2X | nausea |
| Quassia amara TM | bloating |
| Taraxacum officinale 1X |
bloating abdominal fullness |
Warnings
For oral use only.
Directions
- Adults and children over 12 years of age: Take 20 drops in water 3 times daily.
- Consult a physician for use in children under 12 years of age.
Questions?
1-855-387-6466
Other Information (Other information)
- Store at 68 to 77 °F.
-
Tamper Evident:
Do not use if tamper evident strip is broken or missing from base of bottle cap. - To report serious adverse events, call 1-855-387-6466.
Inactive Ingredients (Inactive ingredients)
60 % ethanol V/V, purified water
Principal Display Panel 50 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 50 ml Bottle Carton)
NDC 54532-0031-5
HEVERT®
DETOX
INTESTINUM
Homeopathic Medicine
Relieves symptoms
of upset stomach
and indigestion
1.7 fl oz (50 ml)
60 % alcohol V/V
Oral drops
Made in Germany
hEVERT
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:10:53.619368 · Updated: 2026-03-14T23:15:51.329649