Meclizine Hydrochloride Chewable Tablets 25 Mg
6ab6cf0e-a6c9-4b5a-95c1-37c279bf18dc
34390-5
HUMAN OTC DRUG LABEL
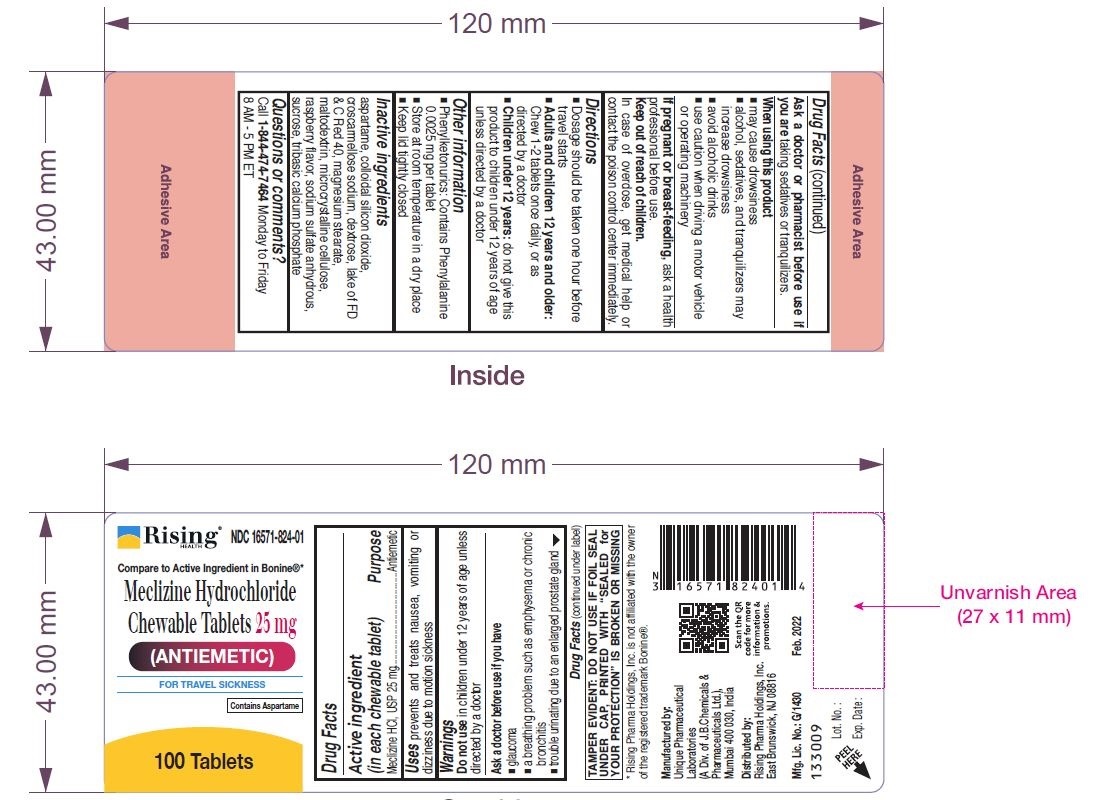
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Meclizine HCl, USP 25 mg
Purpose
Antiemetic
Medication Information
Purpose
Antiemetic
Description
Meclizine HCl, USP 25 mg
Uses
prevents and treats nausea, vomiting or dizziness due to motion sickness.
Section 50568-5
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
Do not use in children under 12 years of age unless directed by a doctor
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Directions
- Dosage should be taken one hour before travel starts
- Adults and children 12 years and older: Chew 1-2 tablets once daily, or as directed by a doctor
- Children under 12 years: do not give this product to children under 12 years of age unless directed by a doctor
Drug Facts
Meclizine HCl, USP 25 mg
Other Information
- Phenylketonurics: Contains Phenylalanine 0.0025 mg per tablet
- Store at room temperature in a dry place
- Keep lid tightly closed
Inactive Ingredients
aspartame, colloidal silicon dioxide, croscarmellose sodium, dextrose, lake of FD & C Red 40, magnesium stearate, maltodextrin, microcrystalline cellulose, raspberry flavor, sodium sulfate anhydrous, sucrose, tribasic calcium phosphate
Questions Or Comments?
Call 1-844-474-7464 Monday to Friday 8 AM - 5 PM ET
When Using This Product
- may cause drowsiness
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
Keep Out of Reach of Children.
In case of overdose, get medical help or contact the Poison Control Center immediately.
Principal Display Panel 25 Mg Chewable Tablet Label
Meclizine Hydrochloride Chewable Tablets
25 mg
100 Tablets / 1000 tablets
Tamper Evident: Do Not Use If Foil Seal Under Cap, Printed With"sealed for Your Protection" Is Broken Or Missing.
Structured Label Content
Uses
prevents and treats nausea, vomiting or dizziness due to motion sickness.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Antiemetic
Warnings
Do not use in children under 12 years of age unless directed by a doctor
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- trouble urinating due to an enlarged prostate gland
Directions
- Dosage should be taken one hour before travel starts
- Adults and children 12 years and older: Chew 1-2 tablets once daily, or as directed by a doctor
- Children under 12 years: do not give this product to children under 12 years of age unless directed by a doctor
Drug Facts
Meclizine HCl, USP 25 mg
Other Information
- Phenylketonurics: Contains Phenylalanine 0.0025 mg per tablet
- Store at room temperature in a dry place
- Keep lid tightly closed
Inactive Ingredients (Inactive ingredients)
aspartame, colloidal silicon dioxide, croscarmellose sodium, dextrose, lake of FD & C Red 40, magnesium stearate, maltodextrin, microcrystalline cellulose, raspberry flavor, sodium sulfate anhydrous, sucrose, tribasic calcium phosphate
Questions Or Comments? (Questions or comments?)
Call 1-844-474-7464 Monday to Friday 8 AM - 5 PM ET
When Using This Product (When using this product)
- may cause drowsiness
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact the Poison Control Center immediately.
Principal Display Panel 25 Mg Chewable Tablet Label (PRINCIPAL DISPLAY PANEL - 25 mg Chewable Tablet Label)
Meclizine Hydrochloride Chewable Tablets
25 mg
100 Tablets / 1000 tablets
Tamper Evident: Do Not Use If Foil Seal Under Cap, Printed With"sealed for Your Protection" Is Broken Or Missing. (TAMPER EVIDENT: DO NOT USE IF FOIL SEAL UNDER CAP, PRINTED WITH"SEALED for YOUR PROTECTION" IS BROKEN OR MISSING.)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:25.976597 · Updated: 2026-03-14T23:05:09.820545