These Highlights Do Not Include All The Information Needed To Use Trospium Chloride Tablets
6a711666-9e80-4728-b745-e7dc86f57510
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
Indications and Usage
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
Dosage and Administration
The recommended dose is 20 mg twice daily. Trospium chloride tablets should be dosed at least one hour before meals or given on an empty stomach. Dosage modification is recommended in the following patient populations: • For patients with severe renal impairment (creatinine clearance less than 30 mL/min), the recommended dose is 20 mg once daily at bedtime [ see Warnings and Precautions ( 5.5 ), Use in Specific Populations ( 8.6 ), and Clinical Pharmacology ( 12.3 ) ]. • In geriatric patients greater than or equal to 75 years of age, dose may be titrated down to 20 mg once daily based upon tolerability [ see Use in Specific Populations ( 8.5 ) ].
Warnings and Precautions
• Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction or gastrointestinal obstructive disorders due to risk of urinary or gastric retention. ( 5.1 , 5.3 ) • Angioedema of the face, lips, tongue and/or larynx has been reported with trospium chloride. ( 5.2 ) • In patients with controlled narrow angle glaucoma trospium chloride tablets should be used only with careful monitoring. ( 5.4 ) • Central Nervous System Effects: Somnolence has been reported with trospium chloride tablets Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them ( 5.5 ). • Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, the risk of anticholinergic adverse reactions is expected to be in patients with moderate renal impairment. ( 5.6 )
Contraindications
Trospium chloride tablets are contraindicated in patients with: • urinary retention • gastric retention • uncontrolled narrow-angle glaucoma. • known hypersensitivity to the drug or its ingredients. Angioedema, rash and anaphylactic reaction have been reported.
Adverse Reactions
The most common adverse reactions (greater than or equal to 1%) with trospium chloride tablets are dry mouth (20.1%), constipation (9.6%), and headache (4.2%). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Cipla Ltd. at 1-866-604-3268 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
• Concomitant use with digoxin did not affect the pharmacokinetics of either drug. ( 7.1 ) • Some drugs which are actively secreted by the kidney may interact with trospium chloride tablets by competing for renal tubular secretion. ( 7.2 ) • Concomitant use with metformin immediate release tablets reduced exposure and peak concentration of trospium. ( 7.4 )
Storage and Handling
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows: Bottles of 30s count (NDC 69097-912-02), Bottles of 60s count (NDC 69097-912-03) and Bottles of 1000s count (NDC 69097-912-15)
How Supplied
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows: Bottles of 30s count (NDC 69097-912-02), Bottles of 60s count (NDC 69097-912-03) and Bottles of 1000s count (NDC 69097-912-15)
Patient Information
Manufactured by: InvaGen Pharmaceuticals, Inc. (a subsidiary of Cipla Ltd.) Hauppauge, NY 11788 Manufactured for: Cipla USA, Inc. 10 Independence Boulevard, Suite 300 Warren, NJ 07059
Medication Information
Warnings and Precautions
• Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction or gastrointestinal obstructive disorders due to risk of urinary or gastric retention. ( 5.1 , 5.3 ) • Angioedema of the face, lips, tongue and/or larynx has been reported with trospium chloride. ( 5.2 ) • In patients with controlled narrow angle glaucoma trospium chloride tablets should be used only with careful monitoring. ( 5.4 ) • Central Nervous System Effects: Somnolence has been reported with trospium chloride tablets Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them ( 5.5 ). • Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, the risk of anticholinergic adverse reactions is expected to be in patients with moderate renal impairment. ( 5.6 )
Indications and Usage
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
Dosage and Administration
The recommended dose is 20 mg twice daily. Trospium chloride tablets should be dosed at least one hour before meals or given on an empty stomach. Dosage modification is recommended in the following patient populations: • For patients with severe renal impairment (creatinine clearance less than 30 mL/min), the recommended dose is 20 mg once daily at bedtime [ see Warnings and Precautions ( 5.5 ), Use in Specific Populations ( 8.6 ), and Clinical Pharmacology ( 12.3 ) ]. • In geriatric patients greater than or equal to 75 years of age, dose may be titrated down to 20 mg once daily based upon tolerability [ see Use in Specific Populations ( 8.5 ) ].
Contraindications
Trospium chloride tablets are contraindicated in patients with: • urinary retention • gastric retention • uncontrolled narrow-angle glaucoma. • known hypersensitivity to the drug or its ingredients. Angioedema, rash and anaphylactic reaction have been reported.
Adverse Reactions
The most common adverse reactions (greater than or equal to 1%) with trospium chloride tablets are dry mouth (20.1%), constipation (9.6%), and headache (4.2%). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Cipla Ltd. at 1-866-604-3268 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
• Concomitant use with digoxin did not affect the pharmacokinetics of either drug. ( 7.1 ) • Some drugs which are actively secreted by the kidney may interact with trospium chloride tablets by competing for renal tubular secretion. ( 7.2 ) • Concomitant use with metformin immediate release tablets reduced exposure and peak concentration of trospium. ( 7.4 )
Storage and Handling
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows: Bottles of 30s count (NDC 69097-912-02), Bottles of 60s count (NDC 69097-912-03) and Bottles of 1000s count (NDC 69097-912-15)
How Supplied
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows: Bottles of 30s count (NDC 69097-912-02), Bottles of 60s count (NDC 69097-912-03) and Bottles of 1000s count (NDC 69097-912-15)
Patient Information
Manufactured by: InvaGen Pharmaceuticals, Inc. (a subsidiary of Cipla Ltd.) Hauppauge, NY 11788 Manufactured for: Cipla USA, Inc. 10 Independence Boulevard, Suite 300 Warren, NJ 07059
Description
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
Section 42229-5
Teratogenic Effects
Pregnancy Category C: There are no adequate and well-controlled studies of trospium chloride tablets in pregnant women. Trospium chloride tablets
should be used during pregnancy only if the potential benefit to the patient outweighs the risk to the patient and fetus. Women who become pregnant during trospium chloride tablets treatment are encouraged to contact their physician.
Section 44425-7
Store at 20° to 25°C (68° to 77°F) (see USP Controlled Room Temperature).
7.1 Digoxin
Concomitant use of trospium chloride tablets and digoxin did not affect the pharmacokinetics of either drug [see Clinical Pharmacology (12.3)].
10 Overdosage
Overdosage with antimuscarinic agents, including trospium chloride tablets, can result in severe antimuscarinic effects. Supportive treatment should be provided according to symptoms. In the event of overdosage, electrocardiographic monitoring is recommended.
A 7-month-old baby experienced tachycardia and mydriasis after administration of a single dose of trospium 10 mg given by a sibling. The baby’s weight was reported as 5 kg. Following admission into the hospital and about 1 hour after ingestion of the trospium, medicinal charcoal was administered for detoxification. While hospitalized, the baby experienced mydriasis and tachycardia up to 230 beats per minute. Therapeutic intervention was not deemed necessary. The baby was discharged as completely recovered the following day.
7.4 Metformin
Co-administration of 500 mg metformin immediate release tablets twice daily with trospium chloride extended release tablets, 60 mg reduced the steady-state systemic exposure of trospium by approximately 29% for mean AUC0-24 and by 34% for mean Cmax[see Clinical Pharmacology (12.3)].
11 Description
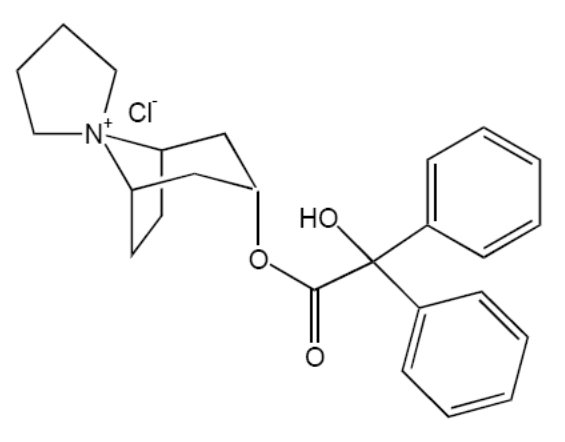
Trospium chloride, USP is a quaternary ammonium compound with the chemical name of Spiro[8azoniabicyclo[3.2.1]octane-8,1'-pyrrolidinium], 3-[(hydroxydiphenylacetyl)oxy]-, chloride, (1α, 3β, 5α). The empirical formula of trospium chloride, USP is C25H30ClNO3 and its molecular weight is 427.97. The structural formula of trospium chloride is represented below:
Trospium chloride, USP is a fine, colorless to slightly yellow, crystalline solid. The compound’s solubility in water is approximately 1 g per 2 mL.
Each trospium chloride tablet contains 20 mg of trospium chloride, USP a muscarinic antagonist, for oral administration. Each tablet also contains the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, hypromellose 2910, titanium dioxide, polyethylene glycol 400, polysorbate 80, xylitol, sucralose, red iron oxide and yellow iron oxide.
Meets USP Dissolution Test 2.
5.2 Angioedema
Angioedema of the face, lips, tongue, and/or larynx has been reported with trospium chloride, the active ingredient in trospium chloride tablets. In one case, angioedema occurred after the first dose of trospium chloride. Angioedema associated with upper airway swelling may be life threatening. If involvement of the tongue, hypopharynx, or larynx occurs, trospium chloride tablets should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided.
17.1 Angioedema
Patients should be informed that trospium chloride, the active ingredient in trospium chloride tablets, may produce angioedema which could result in life-threatening airway obstruction. Patients should be advised to promptly discontinue trospium chloride tablets and seek immediate medical attention if they experience edema of the tongue, edema of the laryngopharynx, or difficulty breathing.
8.4 Pediatric Use
The safety and effectiveness of trospium chloride tablets in pediatric patients have not been established.
8.5 Geriatric Use
Of the 591 patients with overactive bladder who received treatment with trospium chloride tablets in the two U.S., placebo-controlled, efficacy and safety studies, 249 patients (42%) were 65 years of age and older. Eighty-eight trospium chloride tablets treated patients (15%) were greater than or equal to 75 years of age.
In these 2 studies, the incidence of commonly reported anticholinergic adverse reactions in patients treated with trospium chloride tablets (including dry mouth, constipation, dyspepsia, urinary tract infection, and urinary retention) was higher in patients 75 years of age and older as compared to younger patients. This effect may be related to an enhanced sensitivity to anticholinergic agents in this patient population [see Clinical Pharmacology (12.3)]. Therefore, based upon tolerability, the dose frequency of trospium chloride tablets may be reduced to 20 mg once daily in patients 75 years of age and older.
14 Clinical Studies
Trospium chloride tablets was evaluated for the treatment of patients with overactive bladder who had symptoms of urinary frequency, urgency, and urge incontinence in two U.S. 12-week, placebo-controlled studies and one 9-month open label extension.
Study 1 was a randomized, double-blind, placebo-controlled, parallel-group study in 523 patients. A total of 262 patients received trospium chloride tablets 20 mg twice daily and 261 patients received placebo. The majority of patients were Caucasian (85%) and female (74%) with a mean age of 61 years (range: 21 to 90 years). Entry criteria required that patients have urge or mixed incontinence (with a predominance of urge), urge incontinence episodes of at least 7 per week, and greater than 70 micturitions per week. The patient’s medical history and urinary diary during the treatment-free baseline confirmed the diagnosis. Reductions in urinary frequency, urge incontinence episodes and urinary void volume for placebo and trospium chloride tablets treatment groups are summarized in Table 3 and Figures 2 and 3.
Table 3. Mean (SE) change from baseline to end of treatment (Week 12 or last observation carried forward) for urinary frequency, urge incontinence episodes, and void volume in Study 1
|
Efficacy endpoint |
Placebo N=256 |
Trospium chloride N=253 |
P-value |
|
Urinary frequency/24 hours a,* |
|||
|
Mean baseline |
12.9 |
12.7 |
|
|
Mean change from baseline |
-1.3 (0.2) |
-2.4 (0.2) |
<0.001 |
|
Urge incontinence episodes/week b,* |
|||
|
Mean baseline |
30.1 |
27.3 |
|
|
Mean change from baseline |
-13.9 (1.2) |
-15.4 (1.1) |
0.012 |
|
Urinary void volume/toilet void (mL)a ,c |
|||
|
Mean baseline |
156.6 |
155.1 |
|
|
Mean change from baseline |
7.7 (3.1) |
32.1 (3.1) |
<0.001 |
|
a Treatment differences assessed by analysis of variance for ITT:LOCF data set. b Treatment differences assessed by ranked analysis of variance for ITT:LOCF data set. c Placebo N=253, Trospium chloride N=248. * Denotes co-primary endpoint ITT=intent-to-treat, LOCF=last observation carried forward. |
Figure 2 – Mean Change from Baseline in Urinary Frequency/24 Hours, by Visit: Study 1
Figure 3 – Mean Change from Baseline in Urge Incontinence/Week, by Visit: Study 1
Study 2 was nearly identical in design to Study 1. A total of 329 patients received trospium chloride tablets 20 mg twice daily and 329 patients received placebo. The majority of patients were Caucasian (88%) and female (82%) with a mean age of 61 years (range: 19 to 94 years). Entry criteria were identical to Study 1. Reductions in urinary frequency, urge incontinence episodes, and urinary void volume for placebo and trospium chloride tablets treatment groups are summarized in Table 4 and Figures 4 and 5.
Table 4. Mean (SE) change from baseline to end of treatment\(Week 12 or last observation carried forward) for urinary frequency, urge incontinence episodes, and void volume in Study 2.
|
Efficacy endpoint |
Placebo N=325 |
Trospium N=323 |
P-value |
|
Urinary frequency/24 hours a,* |
|||
|
Mean baseline |
13.2 |
12.9 |
|
|
Mean change from baseline |
-1.8 (0.2) |
-2.7 (0.2) |
<0.001 |
|
Urge incontinence episodes/week b |
|||
|
Mean baseline |
27.3 |
26.9 |
|
|
Mean change from baseline |
-12.1 (1.0) |
-16.1 (1.0) |
<0.001 |
|
Urinary void volume/toilet void (mL)a,c |
|||
|
Mean baseline |
154.6 |
154.8 |
|
|
Mean change from baseline |
9.4 (2.8) |
35.6 (2.8) |
<0.001 |
|
a Treatment differences assessed by analysis of variance for ITT:LOCF data set. b Treatment differences assessed by ranked analysis of variance for ITT:LOCF data set. c Placebo N=320, Trospium chloride N=319. * Denotes co-primary endpoint ITT=intent-to-treat, LOCF=last observation carried forward. |
Figure 4 – Mean Change from Baseline in Urinary Frequency/24 Hours, by Visit: Study 2
Figure 5 – Mean Change from Baseline in Urge Incontinence/Week, by Visit: Study 2
17.3 Administration
Patients should be instructed regarding the recommended dosing and administration of trospium chloride tablets:
-
•Take one trospium chloride tablet twice daily with water.
-
•Take trospium chloride tablets on an empty stomach or at least 1 hour before a meal.
4 Contraindications
Trospium chloride tablets are contraindicated in patients with:
-
•urinary retention
-
•gastric retention
-
•uncontrolled narrow-angle glaucoma.
-
•known hypersensitivity to the drug or its ingredients. Angioedema, rash and anaphylactic reaction have been reported.
6 Adverse Reactions
The most common adverse reactions (greater than or equal to 1%) with trospium chloride tablets are dry mouth (20.1%), constipation (9.6%), and headache (4.2%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Cipla Ltd. at 1-866-604-3268 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
-
•Concomitant use with digoxin did not affect the pharmacokinetics of either drug. (7.1)
-
•Some drugs which are actively secreted by the kidney may interact with trospium chloride tablets by competing for renal tubular secretion. (7.2)
-
•Concomitant use with metformin immediate release tablets reduced exposure and peak concentration of trospium. (7.4)
8.3 Nursing Mothers
Trospium chloride (2 mg/kg orally and 50 mcg/kg intravenously) was excreted, to a limited extent (less than 1%), into the milk of lactating rats (primarily as parent compound). It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, trospium chloride tablets should be used during lactation only if the potential benefit justifies the potential risk to the newborn.
17.2 When Not to Use
Prior to treatment, patients should fully understand the risks and benefits of trospium chloride tablets. In particular, patients should be informed not to take trospium chloride tablets if they:
-
•have urinary retention;
-
•gastric retention;
-
•uncontrolled narrow-angle glaucoma;
-
•are allergic to any component of trospium chloride tablets.
8.6 Renal Impairment
Severe renal impairment (creatinine clearance less than 30 mL/minute) significantly altered the disposition of trospium chloride tablets A 4.2-fold and 1.8-fold increase in mean AUC(0-∞) and Cmax, respectively, and the appearance of an additional elimination phase with a long half-life (~33 hr) were detected in patients with severe renal impairment compared with nearly age-matched subjects with creatinine clearance equal to or higher than 80 mL/min. The different pharmacokinetic behavior of trospium chloride tablets in patients with severe renal impairment necessitates adjustment of dosage frequency [see Dosage and Administration (2)]. The pharmacokinetics of trospium have not been studied in patients with creatinine clearance ranging from 30 to 80 mL/min.
Trospium is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function.
12.2 Pharmacodynamics
Placebo-controlled studies employing urodynamic variables were conducted in patients with conditions characterized by involuntary detrusor contractions. The results demonstrate that trospium chloride tablet increases maximum cystometric bladder capacity and volume at first detrusor contraction.
17.4 Adverse Reactions
Patients should be informed that the most common side effects with trospium chloride tablets are dry mouth and constipation and that other less common side effects include trouble emptying the bladder, blurred vision, and heat prostration. Because anticholinergics, such as trospium chloride tablets, may produce dizziness or blurred vision, patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until the drug’s effects have been determined. Patients should be informed that alcohol may enhance the drowsiness caused by anticholinergic agents.
Revised: 06/2016
8.2 Labor and Delivery
The effect of trospium chloride tablets on labor and delivery is unknown.
8.7 Hepatic Impairment
There is no information regarding the effect of severe hepatic impairment on exposure to trospium chloride tablets. In a study of patients with mild and with moderate hepatic impairment, given 40 mg of immediate-release trospium chloride, mean Cmax increased 12% and 63%, respectively, and mean AUC(0-∞) decreased 5% and 15%, respectively, compared to healthy subjects. The clinical significance of these findings is unknown. Caution should be used when administering trospium chloride tablets to patients with moderate and severe hepatic impairment.
1 Indications and Usage
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
12.1 Mechanism of Action
Trospium chloride tablet is a muscarinic antagonist.
Trospium chloride antagonizes the effect of acetylcholine on muscarinic receptors in cholinergically innervated organs including the bladder. Its parasympatholytic action reduces the tonus of smooth muscle in the bladder.
Receptor assays showed that trospium chloride has negligible affinity for nicotinic receptors as compared to muscarinic receptors at concentrations obtained from therapeutic doses.
7.3 Antimuscarinic Agents
The concomitant use of trospium chloride tablets with other antimuscarinic agents that produce dry mouth, constipation, and other anticholinergic pharmacological effects may increase the frequency and/or severity of such effects. Trospium chloride tablets may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility.
5 Warnings and Precautions
-
•Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction or gastrointestinal obstructive disorders due to risk of urinary or gastric retention. (5.1, 5.3)
-
•Angioedema of the face, lips, tongue and/or larynx has been reported with trospium chloride. (5.2)
-
•In patients with controlled narrow angle glaucoma trospium chloride tablets should be used only with careful monitoring. (5.4)
-
•Central Nervous System Effects: Somnolence has been reported with trospium chloride tablets Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them (5.5).
-
•Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, the risk of anticholinergic adverse reactions is expected to be in patients with moderate renal impairment. (5.6)
2 Dosage and Administration
The recommended dose is 20 mg twice daily. Trospium chloride tablets should be dosed at least one hour before meals or given on an empty stomach.
Dosage modification is recommended in the following patient populations:
-
•For patients with severe renal impairment (creatinine clearance less than 30 mL/min), the recommended dose is 20 mg once daily at bedtime [see Warnings and Precautions (5.5), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
-
•In geriatric patients greater than or equal to 75 years of age, dose may be titrated down to 20 mg once daily based upon tolerability [see Use in Specific Populations (8.5)].
3 Dosage Forms and Strengths
Trospium chloride tablets, USP 20 mg tablets are light yellow, film coated, round biconvex, debossed with ‘IG’ on one side and ‘336’ on other.
5.1 Risk of Urinary Retention
Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention [see Contraindications (4)].
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of trospium chloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal – gastritis; Cardiovascular – palpitations, supraventricular tachycardia, chest pain, syncope, “hypertensive crisis”; Immunological – Stevens-Johnson syndrome, anaphylactic reaction, angioedema; Nervous System – dizziness, confusion, vision abnormal, hallucinations, somnolence and delirium; Musculoskeletal – rhabdomyolysis; General – rash.
8 Use in Specific Populations
-
•The safety and effectiveness of trospium chloride tablets in pediatric patients have not been established. (8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of trospium chloride tablets was evaluated in controlled clinical trials in a total of 2975 patients, who were treated with trospium chloride tablets (N=1673), placebo (N=1056) or active control medications (N=246). Of this total, 1181 patients participated in two, 12-week, U.S., efficacy and safety studies and a 9-month open-label extension. Of this total, 591 patients received trospium chloride tablets 20 mg twice daily. In all controlled trials combined, 232 and 208 patients received treatment with trospium chloride tablets for at least 24 and 52 weeks, respectively.
In all placebo-controlled trials combined, the incidence of serious adverse events was 2.9% among patients receiving trospium chloride tablets 20 mg twice daily and 1.5% among patients receiving placebo. Table 1 lists adverse reactions from the combined 12-week U.S. safety and efficacy trials were reported by at least 1% of patients, and were reported more frequently in the trospium chloride tablets group than in the placebo group.
The two most common adverse reactions reported by patients receiving trospium chloride tablets 20 mg twice daily were dry mouth and constipation. The single most frequently reported adverse reaction for trospium chloride tablets, dry mouth, occurred in 20.1% of trospium chloride tablets treated patients and 5.8% of patients receiving placebo. In the two U.S. studies, dry mouth led to discontinuation in 1.9% of patients treated with trospium chloride tablets 20 mg twice daily. For the patients who reported dry mouth, most had their first occurrence of the event within the first month of treatment.
Table 1. Incidence (%) of adverse reactions with trospium chloride tablets, reported in greater than or equal to 1% of all patients treated with trospium chloride tablets and more frequent with trospium chloride tablets (20 mg twice daily) than placebo in Studies 1 and 2 combined
|
Adverse Reaction |
Placebo (N=590) |
Trospium Chloride Tablets 20 mg Twice daily (N=591) |
|
Gastrointestinal Disorders |
||
|
Dry mouth |
34 ( 5.8) |
119 (20.1) |
|
Constipation |
27 (4.6) |
57 (9.6) |
|
Abdominal pain upper |
7 (1.2) |
9 (1.5) |
|
Constipation aggravated |
5 (0.8) |
8 (1.4) |
|
Dyspepsia |
2 (0.3) |
7 (1.2) |
|
Flatulence |
5 (0.8) |
7 (1.2) |
|
Nervous System Disorders |
||
|
Headache |
12 (2.0) |
25 (4.2) |
|
General Disorders |
||
|
Fatigue |
8 (1.4) |
11 (1.9) |
|
Renal and Urinary Disorders |
||
|
Urinary retention |
2 (0.3) |
7 (1.2) |
|
Eye Disorders |
||
|
Dry eyes |
2 (0.3) |
7 (1.2) |
Other adverse reactions from the U.S., placebo-controlled trials , occurring in greater than or equal to 0.5% and less than 1.0% of trospium chloride tablets treated patients, and more common with trospium chloride tablets than placebo are: tachycardia , vision blurred, abdominal distension, vomiting, dysgeusia, dry throat, and dry skin.
During controlled clinical studies, one adverse reaction of angioneurotic edema was reported.
17 Patient Counseling Information
“See FDA-approved Patient Labeling (Patient Information)”
5.5 Central Nervous System Effects
Trospium chloride tablets are associated with anticholinergic central nervous system (CNS) effects [see Adverse Reactions (6.2)]. A variety of CNS anticholinergic effects have been reported, including dizziness, confusion, hallucinations and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
16 How Supplied/storage and Handling
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows:
Bottles of 30s count (NDC 69097-912-02),
Bottles of 60s count (NDC 69097-912-03) and
Bottles of 1000s count (NDC 69097-912-15)
5.4 Controlled Narrow Angle Glaucoma
In patients being treated for narrow-angle glaucoma, trospium chloride tablets should only be used if the potential benefits outweigh the risks and in that circumstance only with careful monitoring [see Contraindications (4)].
Package/label Principal Display Panel
NDC 69097-912-03 Rx Only
Trospium Chloride
Tablets, USP
20 mg
Pharmacist: Please dispense with Patient Information Leaflet Provided Separately to Each Patient.
60 Tablets
Cipla
5.3 Decreased Gastrointestinal Motility
Trospium chloride tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)]. Trospium chloride tablets, like other antimuscarinic agents, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis, intestinal atony and myasthenia gravis.
7.2 Drugs Eliminated By Active Tubular Secretion
Although demonstrated in a drug-drug interaction study not to affect the pharmacokinetics of digoxin, trospium chloride tablets has the potential for pharmacokinetic interactions with other drugs that are eliminated by active tubular secretion (e.g., procainamide, pancuronium, morphine, vancomycin, and tenofovir). Coadministration of trospium chloride tablets with these drugs may increase the serum concentration of trospium chloride tablets and/or the coadministered drug due to competition for this elimination pathway. Careful patient monitoring is recommended in patients receiving such drugs [see Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: Carcinogenicity studies with trospium chloride were conducted in mice and rats for 78 weeks and 104 weeks, respectively, at maximally tolerated doses. No evidence of a carcinogenic effect was found in either mice or rats administered up to 200 mg/kg/day, approximately 9 times the expected clinical exposure levels at the maximum recommended human dose (MRHD) of 40 mg.
Mutagenesis: Trospium chloride was not mutagenic nor genotoxic in tests in vitro in bacteria (Ames test) and mammalian cells (L5178Y mouse lymphoma and CHO cells) or in vivo in the rat micronucleus test.
Impairment of Fertility: No evidence of impaired fertility was observed in rats administered doses up to 200 mg/kg/day (about 16 times the expected clinical exposure at the MRHD, based on AUC).
5.6 Anticholinergic Adverse Reactions in Patients With Moderate Renal Impairment
Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, anticholinergic adverse reactions (including dry mouth, constipation, dyspepsia, urinary tract infection, and urinary retention) are expected to be greater in patients with moderate renal impairment [see Dosage and Administration (2), and Use in Specific Populations (8.6)].
patient Information
Manufactured by:
InvaGen Pharmaceuticals, Inc.
(a subsidiary of Cipla Ltd.)
Hauppauge, NY 11788
Manufactured for:
Cipla USA, Inc.
10 Independence Boulevard, Suite 300
Warren, NJ 07059
Structured Label Content
Section 42229-5 (42229-5)
Teratogenic Effects
Pregnancy Category C: There are no adequate and well-controlled studies of trospium chloride tablets in pregnant women. Trospium chloride tablets
should be used during pregnancy only if the potential benefit to the patient outweighs the risk to the patient and fetus. Women who become pregnant during trospium chloride tablets treatment are encouraged to contact their physician.
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F) (see USP Controlled Room Temperature).
7.1 Digoxin
Concomitant use of trospium chloride tablets and digoxin did not affect the pharmacokinetics of either drug [see Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
Overdosage with antimuscarinic agents, including trospium chloride tablets, can result in severe antimuscarinic effects. Supportive treatment should be provided according to symptoms. In the event of overdosage, electrocardiographic monitoring is recommended.
A 7-month-old baby experienced tachycardia and mydriasis after administration of a single dose of trospium 10 mg given by a sibling. The baby’s weight was reported as 5 kg. Following admission into the hospital and about 1 hour after ingestion of the trospium, medicinal charcoal was administered for detoxification. While hospitalized, the baby experienced mydriasis and tachycardia up to 230 beats per minute. Therapeutic intervention was not deemed necessary. The baby was discharged as completely recovered the following day.
7.4 Metformin
Co-administration of 500 mg metformin immediate release tablets twice daily with trospium chloride extended release tablets, 60 mg reduced the steady-state systemic exposure of trospium by approximately 29% for mean AUC0-24 and by 34% for mean Cmax[see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Trospium chloride, USP is a quaternary ammonium compound with the chemical name of Spiro[8azoniabicyclo[3.2.1]octane-8,1'-pyrrolidinium], 3-[(hydroxydiphenylacetyl)oxy]-, chloride, (1α, 3β, 5α). The empirical formula of trospium chloride, USP is C25H30ClNO3 and its molecular weight is 427.97. The structural formula of trospium chloride is represented below:
Trospium chloride, USP is a fine, colorless to slightly yellow, crystalline solid. The compound’s solubility in water is approximately 1 g per 2 mL.
Each trospium chloride tablet contains 20 mg of trospium chloride, USP a muscarinic antagonist, for oral administration. Each tablet also contains the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, hypromellose 2910, titanium dioxide, polyethylene glycol 400, polysorbate 80, xylitol, sucralose, red iron oxide and yellow iron oxide.
Meets USP Dissolution Test 2.
5.2 Angioedema
Angioedema of the face, lips, tongue, and/or larynx has been reported with trospium chloride, the active ingredient in trospium chloride tablets. In one case, angioedema occurred after the first dose of trospium chloride. Angioedema associated with upper airway swelling may be life threatening. If involvement of the tongue, hypopharynx, or larynx occurs, trospium chloride tablets should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided.
17.1 Angioedema
Patients should be informed that trospium chloride, the active ingredient in trospium chloride tablets, may produce angioedema which could result in life-threatening airway obstruction. Patients should be advised to promptly discontinue trospium chloride tablets and seek immediate medical attention if they experience edema of the tongue, edema of the laryngopharynx, or difficulty breathing.
8.4 Pediatric Use
The safety and effectiveness of trospium chloride tablets in pediatric patients have not been established.
8.5 Geriatric Use
Of the 591 patients with overactive bladder who received treatment with trospium chloride tablets in the two U.S., placebo-controlled, efficacy and safety studies, 249 patients (42%) were 65 years of age and older. Eighty-eight trospium chloride tablets treated patients (15%) were greater than or equal to 75 years of age.
In these 2 studies, the incidence of commonly reported anticholinergic adverse reactions in patients treated with trospium chloride tablets (including dry mouth, constipation, dyspepsia, urinary tract infection, and urinary retention) was higher in patients 75 years of age and older as compared to younger patients. This effect may be related to an enhanced sensitivity to anticholinergic agents in this patient population [see Clinical Pharmacology (12.3)]. Therefore, based upon tolerability, the dose frequency of trospium chloride tablets may be reduced to 20 mg once daily in patients 75 years of age and older.
14 Clinical Studies (14 CLINICAL STUDIES)
Trospium chloride tablets was evaluated for the treatment of patients with overactive bladder who had symptoms of urinary frequency, urgency, and urge incontinence in two U.S. 12-week, placebo-controlled studies and one 9-month open label extension.
Study 1 was a randomized, double-blind, placebo-controlled, parallel-group study in 523 patients. A total of 262 patients received trospium chloride tablets 20 mg twice daily and 261 patients received placebo. The majority of patients were Caucasian (85%) and female (74%) with a mean age of 61 years (range: 21 to 90 years). Entry criteria required that patients have urge or mixed incontinence (with a predominance of urge), urge incontinence episodes of at least 7 per week, and greater than 70 micturitions per week. The patient’s medical history and urinary diary during the treatment-free baseline confirmed the diagnosis. Reductions in urinary frequency, urge incontinence episodes and urinary void volume for placebo and trospium chloride tablets treatment groups are summarized in Table 3 and Figures 2 and 3.
Table 3. Mean (SE) change from baseline to end of treatment (Week 12 or last observation carried forward) for urinary frequency, urge incontinence episodes, and void volume in Study 1
|
Efficacy endpoint |
Placebo N=256 |
Trospium chloride N=253 |
P-value |
|
Urinary frequency/24 hours a,* |
|||
|
Mean baseline |
12.9 |
12.7 |
|
|
Mean change from baseline |
-1.3 (0.2) |
-2.4 (0.2) |
<0.001 |
|
Urge incontinence episodes/week b,* |
|||
|
Mean baseline |
30.1 |
27.3 |
|
|
Mean change from baseline |
-13.9 (1.2) |
-15.4 (1.1) |
0.012 |
|
Urinary void volume/toilet void (mL)a ,c |
|||
|
Mean baseline |
156.6 |
155.1 |
|
|
Mean change from baseline |
7.7 (3.1) |
32.1 (3.1) |
<0.001 |
|
a Treatment differences assessed by analysis of variance for ITT:LOCF data set. b Treatment differences assessed by ranked analysis of variance for ITT:LOCF data set. c Placebo N=253, Trospium chloride N=248. * Denotes co-primary endpoint ITT=intent-to-treat, LOCF=last observation carried forward. |
Figure 2 – Mean Change from Baseline in Urinary Frequency/24 Hours, by Visit: Study 1
Figure 3 – Mean Change from Baseline in Urge Incontinence/Week, by Visit: Study 1
Study 2 was nearly identical in design to Study 1. A total of 329 patients received trospium chloride tablets 20 mg twice daily and 329 patients received placebo. The majority of patients were Caucasian (88%) and female (82%) with a mean age of 61 years (range: 19 to 94 years). Entry criteria were identical to Study 1. Reductions in urinary frequency, urge incontinence episodes, and urinary void volume for placebo and trospium chloride tablets treatment groups are summarized in Table 4 and Figures 4 and 5.
Table 4. Mean (SE) change from baseline to end of treatment\(Week 12 or last observation carried forward) for urinary frequency, urge incontinence episodes, and void volume in Study 2.
|
Efficacy endpoint |
Placebo N=325 |
Trospium N=323 |
P-value |
|
Urinary frequency/24 hours a,* |
|||
|
Mean baseline |
13.2 |
12.9 |
|
|
Mean change from baseline |
-1.8 (0.2) |
-2.7 (0.2) |
<0.001 |
|
Urge incontinence episodes/week b |
|||
|
Mean baseline |
27.3 |
26.9 |
|
|
Mean change from baseline |
-12.1 (1.0) |
-16.1 (1.0) |
<0.001 |
|
Urinary void volume/toilet void (mL)a,c |
|||
|
Mean baseline |
154.6 |
154.8 |
|
|
Mean change from baseline |
9.4 (2.8) |
35.6 (2.8) |
<0.001 |
|
a Treatment differences assessed by analysis of variance for ITT:LOCF data set. b Treatment differences assessed by ranked analysis of variance for ITT:LOCF data set. c Placebo N=320, Trospium chloride N=319. * Denotes co-primary endpoint ITT=intent-to-treat, LOCF=last observation carried forward. |
Figure 4 – Mean Change from Baseline in Urinary Frequency/24 Hours, by Visit: Study 2
Figure 5 – Mean Change from Baseline in Urge Incontinence/Week, by Visit: Study 2
17.3 Administration
Patients should be instructed regarding the recommended dosing and administration of trospium chloride tablets:
-
•Take one trospium chloride tablet twice daily with water.
-
•Take trospium chloride tablets on an empty stomach or at least 1 hour before a meal.
4 Contraindications (4 CONTRAINDICATIONS)
Trospium chloride tablets are contraindicated in patients with:
-
•urinary retention
-
•gastric retention
-
•uncontrolled narrow-angle glaucoma.
-
•known hypersensitivity to the drug or its ingredients. Angioedema, rash and anaphylactic reaction have been reported.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (greater than or equal to 1%) with trospium chloride tablets are dry mouth (20.1%), constipation (9.6%), and headache (4.2%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Cipla Ltd. at 1-866-604-3268 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•Concomitant use with digoxin did not affect the pharmacokinetics of either drug. (7.1)
-
•Some drugs which are actively secreted by the kidney may interact with trospium chloride tablets by competing for renal tubular secretion. (7.2)
-
•Concomitant use with metformin immediate release tablets reduced exposure and peak concentration of trospium. (7.4)
8.3 Nursing Mothers
Trospium chloride (2 mg/kg orally and 50 mcg/kg intravenously) was excreted, to a limited extent (less than 1%), into the milk of lactating rats (primarily as parent compound). It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, trospium chloride tablets should be used during lactation only if the potential benefit justifies the potential risk to the newborn.
17.2 When Not to Use
Prior to treatment, patients should fully understand the risks and benefits of trospium chloride tablets. In particular, patients should be informed not to take trospium chloride tablets if they:
-
•have urinary retention;
-
•gastric retention;
-
•uncontrolled narrow-angle glaucoma;
-
•are allergic to any component of trospium chloride tablets.
8.6 Renal Impairment
Severe renal impairment (creatinine clearance less than 30 mL/minute) significantly altered the disposition of trospium chloride tablets A 4.2-fold and 1.8-fold increase in mean AUC(0-∞) and Cmax, respectively, and the appearance of an additional elimination phase with a long half-life (~33 hr) were detected in patients with severe renal impairment compared with nearly age-matched subjects with creatinine clearance equal to or higher than 80 mL/min. The different pharmacokinetic behavior of trospium chloride tablets in patients with severe renal impairment necessitates adjustment of dosage frequency [see Dosage and Administration (2)]. The pharmacokinetics of trospium have not been studied in patients with creatinine clearance ranging from 30 to 80 mL/min.
Trospium is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function.
12.2 Pharmacodynamics
Placebo-controlled studies employing urodynamic variables were conducted in patients with conditions characterized by involuntary detrusor contractions. The results demonstrate that trospium chloride tablet increases maximum cystometric bladder capacity and volume at first detrusor contraction.
17.4 Adverse Reactions
Patients should be informed that the most common side effects with trospium chloride tablets are dry mouth and constipation and that other less common side effects include trouble emptying the bladder, blurred vision, and heat prostration. Because anticholinergics, such as trospium chloride tablets, may produce dizziness or blurred vision, patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until the drug’s effects have been determined. Patients should be informed that alcohol may enhance the drowsiness caused by anticholinergic agents.
Revised: 06/2016
8.2 Labor and Delivery
The effect of trospium chloride tablets on labor and delivery is unknown.
8.7 Hepatic Impairment
There is no information regarding the effect of severe hepatic impairment on exposure to trospium chloride tablets. In a study of patients with mild and with moderate hepatic impairment, given 40 mg of immediate-release trospium chloride, mean Cmax increased 12% and 63%, respectively, and mean AUC(0-∞) decreased 5% and 15%, respectively, compared to healthy subjects. The clinical significance of these findings is unknown. Caution should be used when administering trospium chloride tablets to patients with moderate and severe hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Trospium chloride tablets are a muscarinic antagonist indicated for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency.
12.1 Mechanism of Action
Trospium chloride tablet is a muscarinic antagonist.
Trospium chloride antagonizes the effect of acetylcholine on muscarinic receptors in cholinergically innervated organs including the bladder. Its parasympatholytic action reduces the tonus of smooth muscle in the bladder.
Receptor assays showed that trospium chloride has negligible affinity for nicotinic receptors as compared to muscarinic receptors at concentrations obtained from therapeutic doses.
7.3 Antimuscarinic Agents
The concomitant use of trospium chloride tablets with other antimuscarinic agents that produce dry mouth, constipation, and other anticholinergic pharmacological effects may increase the frequency and/or severity of such effects. Trospium chloride tablets may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction or gastrointestinal obstructive disorders due to risk of urinary or gastric retention. (5.1, 5.3)
-
•Angioedema of the face, lips, tongue and/or larynx has been reported with trospium chloride. (5.2)
-
•In patients with controlled narrow angle glaucoma trospium chloride tablets should be used only with careful monitoring. (5.4)
-
•Central Nervous System Effects: Somnolence has been reported with trospium chloride tablets Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them (5.5).
-
•Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, the risk of anticholinergic adverse reactions is expected to be in patients with moderate renal impairment. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dose is 20 mg twice daily. Trospium chloride tablets should be dosed at least one hour before meals or given on an empty stomach.
Dosage modification is recommended in the following patient populations:
-
•For patients with severe renal impairment (creatinine clearance less than 30 mL/min), the recommended dose is 20 mg once daily at bedtime [see Warnings and Precautions (5.5), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
-
•In geriatric patients greater than or equal to 75 years of age, dose may be titrated down to 20 mg once daily based upon tolerability [see Use in Specific Populations (8.5)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Trospium chloride tablets, USP 20 mg tablets are light yellow, film coated, round biconvex, debossed with ‘IG’ on one side and ‘336’ on other.
5.1 Risk of Urinary Retention
Trospium chloride tablets should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention [see Contraindications (4)].
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following adverse reactions have been identified during post-approval use of trospium chloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal – gastritis; Cardiovascular – palpitations, supraventricular tachycardia, chest pain, syncope, “hypertensive crisis”; Immunological – Stevens-Johnson syndrome, anaphylactic reaction, angioedema; Nervous System – dizziness, confusion, vision abnormal, hallucinations, somnolence and delirium; Musculoskeletal – rhabdomyolysis; General – rash.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•The safety and effectiveness of trospium chloride tablets in pediatric patients have not been established. (8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of trospium chloride tablets was evaluated in controlled clinical trials in a total of 2975 patients, who were treated with trospium chloride tablets (N=1673), placebo (N=1056) or active control medications (N=246). Of this total, 1181 patients participated in two, 12-week, U.S., efficacy and safety studies and a 9-month open-label extension. Of this total, 591 patients received trospium chloride tablets 20 mg twice daily. In all controlled trials combined, 232 and 208 patients received treatment with trospium chloride tablets for at least 24 and 52 weeks, respectively.
In all placebo-controlled trials combined, the incidence of serious adverse events was 2.9% among patients receiving trospium chloride tablets 20 mg twice daily and 1.5% among patients receiving placebo. Table 1 lists adverse reactions from the combined 12-week U.S. safety and efficacy trials were reported by at least 1% of patients, and were reported more frequently in the trospium chloride tablets group than in the placebo group.
The two most common adverse reactions reported by patients receiving trospium chloride tablets 20 mg twice daily were dry mouth and constipation. The single most frequently reported adverse reaction for trospium chloride tablets, dry mouth, occurred in 20.1% of trospium chloride tablets treated patients and 5.8% of patients receiving placebo. In the two U.S. studies, dry mouth led to discontinuation in 1.9% of patients treated with trospium chloride tablets 20 mg twice daily. For the patients who reported dry mouth, most had their first occurrence of the event within the first month of treatment.
Table 1. Incidence (%) of adverse reactions with trospium chloride tablets, reported in greater than or equal to 1% of all patients treated with trospium chloride tablets and more frequent with trospium chloride tablets (20 mg twice daily) than placebo in Studies 1 and 2 combined
|
Adverse Reaction |
Placebo (N=590) |
Trospium Chloride Tablets 20 mg Twice daily (N=591) |
|
Gastrointestinal Disorders |
||
|
Dry mouth |
34 ( 5.8) |
119 (20.1) |
|
Constipation |
27 (4.6) |
57 (9.6) |
|
Abdominal pain upper |
7 (1.2) |
9 (1.5) |
|
Constipation aggravated |
5 (0.8) |
8 (1.4) |
|
Dyspepsia |
2 (0.3) |
7 (1.2) |
|
Flatulence |
5 (0.8) |
7 (1.2) |
|
Nervous System Disorders |
||
|
Headache |
12 (2.0) |
25 (4.2) |
|
General Disorders |
||
|
Fatigue |
8 (1.4) |
11 (1.9) |
|
Renal and Urinary Disorders |
||
|
Urinary retention |
2 (0.3) |
7 (1.2) |
|
Eye Disorders |
||
|
Dry eyes |
2 (0.3) |
7 (1.2) |
Other adverse reactions from the U.S., placebo-controlled trials , occurring in greater than or equal to 0.5% and less than 1.0% of trospium chloride tablets treated patients, and more common with trospium chloride tablets than placebo are: tachycardia , vision blurred, abdominal distension, vomiting, dysgeusia, dry throat, and dry skin.
During controlled clinical studies, one adverse reaction of angioneurotic edema was reported.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
“See FDA-approved Patient Labeling (Patient Information)”
5.5 Central Nervous System Effects
Trospium chloride tablets are associated with anticholinergic central nervous system (CNS) effects [see Adverse Reactions (6.2)]. A variety of CNS anticholinergic effects have been reported, including dizziness, confusion, hallucinations and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how trospium chloride tablets affect them. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Trospium chloride tablets, USP 20 mg (light yellow film coated, round biconvex tablets de-bossed with ‘IG’ on one side and ‘336’ on other) are supplied as follows:
Bottles of 30s count (NDC 69097-912-02),
Bottles of 60s count (NDC 69097-912-03) and
Bottles of 1000s count (NDC 69097-912-15)
5.4 Controlled Narrow Angle Glaucoma (5.4 Controlled Narrow-angle Glaucoma)
In patients being treated for narrow-angle glaucoma, trospium chloride tablets should only be used if the potential benefits outweigh the risks and in that circumstance only with careful monitoring [see Contraindications (4)].
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
NDC 69097-912-03 Rx Only
Trospium Chloride
Tablets, USP
20 mg
Pharmacist: Please dispense with Patient Information Leaflet Provided Separately to Each Patient.
60 Tablets
Cipla
5.3 Decreased Gastrointestinal Motility
Trospium chloride tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)]. Trospium chloride tablets, like other antimuscarinic agents, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis, intestinal atony and myasthenia gravis.
7.2 Drugs Eliminated By Active Tubular Secretion (7.2 Drugs Eliminated by Active Tubular Secretion)
Although demonstrated in a drug-drug interaction study not to affect the pharmacokinetics of digoxin, trospium chloride tablets has the potential for pharmacokinetic interactions with other drugs that are eliminated by active tubular secretion (e.g., procainamide, pancuronium, morphine, vancomycin, and tenofovir). Coadministration of trospium chloride tablets with these drugs may increase the serum concentration of trospium chloride tablets and/or the coadministered drug due to competition for this elimination pathway. Careful patient monitoring is recommended in patients receiving such drugs [see Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: Carcinogenicity studies with trospium chloride were conducted in mice and rats for 78 weeks and 104 weeks, respectively, at maximally tolerated doses. No evidence of a carcinogenic effect was found in either mice or rats administered up to 200 mg/kg/day, approximately 9 times the expected clinical exposure levels at the maximum recommended human dose (MRHD) of 40 mg.
Mutagenesis: Trospium chloride was not mutagenic nor genotoxic in tests in vitro in bacteria (Ames test) and mammalian cells (L5178Y mouse lymphoma and CHO cells) or in vivo in the rat micronucleus test.
Impairment of Fertility: No evidence of impaired fertility was observed in rats administered doses up to 200 mg/kg/day (about 16 times the expected clinical exposure at the MRHD, based on AUC).
5.6 Anticholinergic Adverse Reactions in Patients With Moderate Renal Impairment (5.6 Anticholinergic Adverse Reactions in Patients with Moderate Renal Impairment)
Trospium is substantially excreted by the kidney. The effects of moderate renal impairment on systemic exposure are not known but systemic exposure is likely increased. Therefore, anticholinergic adverse reactions (including dry mouth, constipation, dyspepsia, urinary tract infection, and urinary retention) are expected to be greater in patients with moderate renal impairment [see Dosage and Administration (2), and Use in Specific Populations (8.6)].
patient Information ( Patient Information)
Manufactured by:
InvaGen Pharmaceuticals, Inc.
(a subsidiary of Cipla Ltd.)
Hauppauge, NY 11788
Manufactured for:
Cipla USA, Inc.
10 Independence Boulevard, Suite 300
Warren, NJ 07059
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:30.485824 · Updated: 2026-03-14T21:43:40.690874