1001-fst-2023-0609
6a5d43c8-f4b7-4447-b2e1-53eb12208ef0
34390-5
HUMAN OTC DRUG LABEL
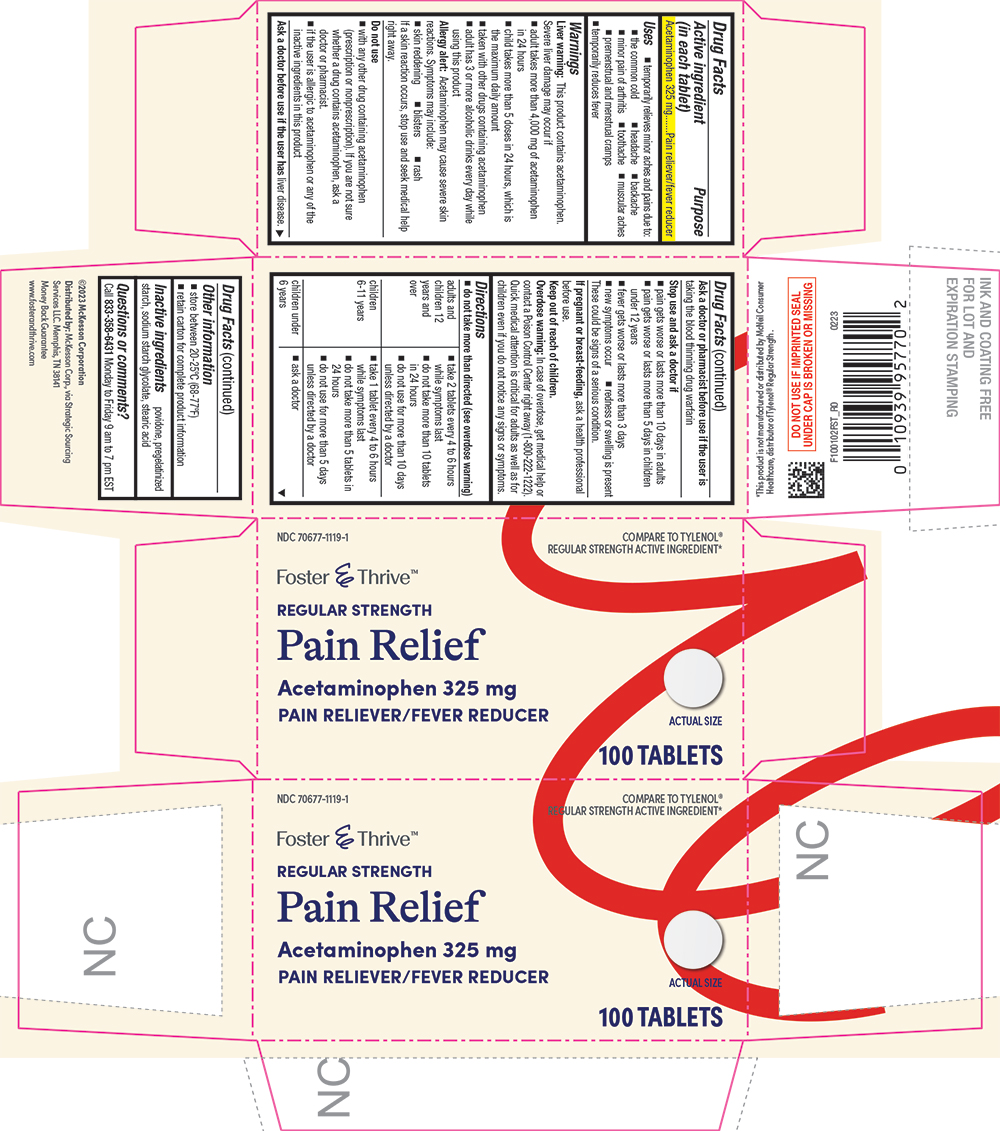
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Pain reliever/fever reducer
Description
Drug Facts
Medication Information
Active Ingredient
Acetaminophen 325 mg
Uses
- temporarily relieves minor aches and pains due to:
- the common cold
- headache
- backache
- minor pain of arthritis
- toothache
- muscular aches
- premenstrual and menstrual cramps
- temporarily reduces fever
Purpose
Pain reliever/fever reducer
Directions
- do not take more than directed (see overdose warning)
| adults and children 12 years and over |
|
| children 6-11 years |
|
| children under 6 years |
|
Other Information
- store between 20-25°C (68-77°F)
- retain carton for complete product information
Inactive Ingredients
povidone, pregelatinized starch, sodium starch glycolate, stearic acid
Description
Drug Facts
Principal Display Panel
NDC 70677-1119-1
COMPARE TO TYLENOL® REGULAR STRENGTH ACTIVE INGREDIENT*
Foster & Thrive ™
REGULAR STRENGTH
Pain Relief
Acetaminophen 325 mg
PAIN RELIEVER/FEVER REDUCER
ACTUAL SIZE
100 TABLETS
Section 42229-5
Drug Facts
Section 50568-5
Ask a doctor or pharmacist before use if the user is taking the blood thinning drug warfarin
Section 50569-3
Ask a doctor before use if the user has liver disease.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Do Not Use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if the user is allergic to acetaminophen or any of the inactive ingredients in this product
Allergy Alert
acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Liver Warning
This product contains acetaminophen. Severe liver damage may occur if
- adult takes more than 4,000 mg of acetaminophen in 24 hours
- child takes more than 5 doses in 24 hours, which is the maximum daily amount
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Overdose Warning
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Questions Or Comments?
Call 833-358-6431 Monday to Friday 9 am to 7 pm EST
Stop Use and Ask A Doctor If
- pain gets worse or lasts more than 10 days in adults
- pain gets worse or lasts more than 5 days in children under 12 years
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
Structured Label Content
Active Ingredient (Active ingredient (in each tablet))
Acetaminophen 325 mg
Uses
- temporarily relieves minor aches and pains due to:
- the common cold
- headache
- backache
- minor pain of arthritis
- toothache
- muscular aches
- premenstrual and menstrual cramps
- temporarily reduces fever
Purpose
Pain reliever/fever reducer
Directions
- do not take more than directed (see overdose warning)
| adults and children 12 years and over |
|
| children 6-11 years |
|
| children under 6 years |
|
Other Information (Other information)
- store between 20-25°C (68-77°F)
- retain carton for complete product information
Inactive Ingredients (Inactive ingredients)
povidone, pregelatinized starch, sodium starch glycolate, stearic acid
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 70677-1119-1
COMPARE TO TYLENOL® REGULAR STRENGTH ACTIVE INGREDIENT*
Foster & Thrive ™
REGULAR STRENGTH
Pain Relief
Acetaminophen 325 mg
PAIN RELIEVER/FEVER REDUCER
ACTUAL SIZE
100 TABLETS
Section 42229-5 (42229-5)
Drug Facts
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if the user is taking the blood thinning drug warfarin
Section 50569-3 (50569-3)
Ask a doctor before use if the user has liver disease.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Do Not Use (Do not use)
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if the user is allergic to acetaminophen or any of the inactive ingredients in this product
Allergy Alert (Allergy alert)
acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Liver Warning (Liver warning)
This product contains acetaminophen. Severe liver damage may occur if
- adult takes more than 4,000 mg of acetaminophen in 24 hours
- child takes more than 5 doses in 24 hours, which is the maximum daily amount
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Overdose Warning (Overdose warning)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Questions Or Comments? (Questions or comments?)
Call 833-358-6431 Monday to Friday 9 am to 7 pm EST
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- pain gets worse or lasts more than 10 days in adults
- pain gets worse or lasts more than 5 days in children under 12 years
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:52.617619 · Updated: 2026-03-14T23:04:12.212732