69f31121-79b9-420d-947b-90228100543b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
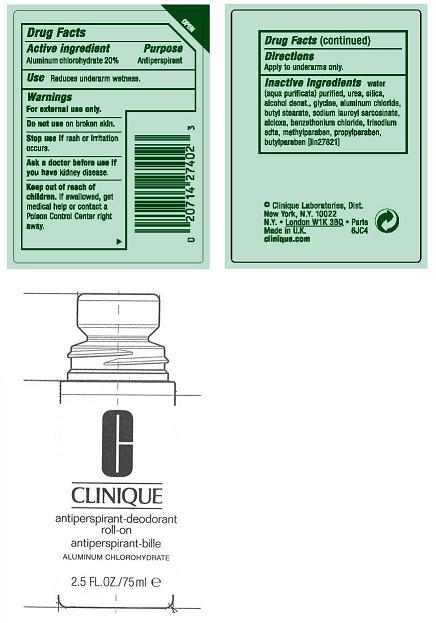
ACTIVE INGREDIENT : ALUMINUM CHLOROHYDRATE 20.00%
Medication Information
Warnings and Precautions
WARNINGS:
- FOR EXTERNAL USE ONLY
- DO NOT USE ON BROKEN SKIN
- STOP USE IF RASH OR IRRITATION OCCURS
- ASK A DOCTOR BEFORE USE IF YOU HAVE KIDNEY DISEASE
Indications and Usage
DIRECTIONS: APPLY TO UNDERARMS ONLY.
Description
ACTIVE INGREDIENT : ALUMINUM CHLOROHYDRATE 20.00%
Section 50565-1
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 51727-6
inactive ingredients: water[] urea [] silica [] alcohol denat. [] glycine [] aluminum chloride [] butyl stearate [] sodium lauroyl sarcosinate [] alcloxa [] benzethonium chloride [] trisodium edta [] methylparaben [] propylparaben [] butylparaben iln27821
Section 51945-4
PRINCIPAL DISPLAY PANEL:
CLINIQUE
anti-perspirant deodorant roll-on
ALUMINUM CHLOROHYDRATE
2.5FL OZ./ 70ML
CLINIQUE LABORATORIES, DIST.
NEW YORK, NY 10022
6183
CLINIQUE.COM
Section 55105-1
USES: DECREASES UNDERARM PERSPIRATION
Section 55106-9
ACTIVE INGREDIENT : ALUMINUM CHLOROHYDRATE 20.00%
Structured Label Content
Indications and Usage (34067-9)
DIRECTIONS: APPLY TO UNDERARMS ONLY.
Warnings and Precautions (34071-1)
WARNINGS:
- FOR EXTERNAL USE ONLY
- DO NOT USE ON BROKEN SKIN
- STOP USE IF RASH OR IRRITATION OCCURS
- ASK A DOCTOR BEFORE USE IF YOU HAVE KIDNEY DISEASE
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 51727-6 (51727-6)
inactive ingredients: water[] urea [] silica [] alcohol denat. [] glycine [] aluminum chloride [] butyl stearate [] sodium lauroyl sarcosinate [] alcloxa [] benzethonium chloride [] trisodium edta [] methylparaben [] propylparaben [] butylparaben iln27821
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL:
CLINIQUE
anti-perspirant deodorant roll-on
ALUMINUM CHLOROHYDRATE
2.5FL OZ./ 70ML
CLINIQUE LABORATORIES, DIST.
NEW YORK, NY 10022
6183
CLINIQUE.COM
Section 55105-1 (55105-1)
USES: DECREASES UNDERARM PERSPIRATION
Section 55106-9 (55106-9)
ACTIVE INGREDIENT : ALUMINUM CHLOROHYDRATE 20.00%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:02.329983 · Updated: 2026-03-14T22:52:21.082332