Exemestane
69abd0ff-b200-4d1a-b14a-80e980a8e781
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
EXEMESTANE is an aromatase inhibitor indicated for: • adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy ( 14.1 ). • treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy ( 14.2 ).
Dosage and Administration
Recommended Dose: One 25 mg tablet once daily after a meal ( 2.1 ).
Contraindications
EXEMESTANE is contraindicated in patients with a known hypersensitivity to the drug or to any of the excipients.
Warnings and Precautions
• Reductions in bone mineral density (BMD) over time are seen with exemestane use ( 5.1 ). • Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed ( 5.2 ). • Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception ( 5.6 , 8.1 , 8.3 ).
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Reductions in Bone Mineral Density (BMD) [see Warnings and Precautions (5.1) ]
Drug Interactions
Strong CYP 3A4 inducers: Concomitant use of strong CYP 3A4 inducers decreases exemestane exposure. Increase the EXEMESTANE dose to 50 mg ( 2.2 , 7 ).
How Supplied
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets. 30-tablet HDPE bottle NDC 59762-2858-1
Storage and Handling
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets. 30-tablet HDPE bottle NDC 59762-2858-1
Description
EXEMESTANE is an aromatase inhibitor indicated for: • adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy ( 14.1 ). • treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy ( 14.2 ).
Medication Information
Warnings and Precautions
• Reductions in bone mineral density (BMD) over time are seen with exemestane use ( 5.1 ). • Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed ( 5.2 ). • Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception ( 5.6 , 8.1 , 8.3 ).
Indications and Usage
EXEMESTANE is an aromatase inhibitor indicated for: • adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy ( 14.1 ). • treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy ( 14.2 ).
Dosage and Administration
Recommended Dose: One 25 mg tablet once daily after a meal ( 2.1 ).
Contraindications
EXEMESTANE is contraindicated in patients with a known hypersensitivity to the drug or to any of the excipients.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Reductions in Bone Mineral Density (BMD) [see Warnings and Precautions (5.1) ]
Drug Interactions
Strong CYP 3A4 inducers: Concomitant use of strong CYP 3A4 inducers decreases exemestane exposure. Increase the EXEMESTANE dose to 50 mg ( 2.2 , 7 ).
Storage and Handling
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets. 30-tablet HDPE bottle NDC 59762-2858-1
How Supplied
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets. 30-tablet HDPE bottle NDC 59762-2858-1
Description
EXEMESTANE is an aromatase inhibitor indicated for: • adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy ( 14.1 ). • treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy ( 14.2 ).
Section 42229-5
Adjuvant Therapy
The data described below reflect exposure to EXEMESTANE in 2325 postmenopausal women with early breast cancer. EXEMESTANE tolerability in postmenopausal women with early breast cancer was evaluated in two well-controlled trials: the IES study [see Clinical Studies (14.1)] and the 027 study (a randomized, placebo-controlled, double-blind, parallel group study specifically designed to assess the effects of exemestane on bone metabolism, hormones, lipids, and coagulation factors over 2 years of treatment).
The median duration of adjuvant treatment was 27.4 months and 27.3 months for patients receiving EXEMESTANE or tamoxifen, respectively, within the IES study and 23.9 months for patients receiving EXEMESTANE or placebo within the 027 study. Median duration of observation after randomization for EXEMESTANE was 34.5 months and for tamoxifen was 34.6 months. Median duration of observation was 30 months for both groups in the 027 study.
Certain adverse reactions, which were expected based on the known pharmacological properties and side effect profiles of test drugs, were actively sought through a positive checklist. Signs and symptoms were graded for severity using CTC in both studies. Within the IES study, the presence of some illnesses/conditions was monitored through a positive checklist without assessment of severity. These included myocardial infarction, other cardiovascular disorders, gynecological disorders, osteoporosis, osteoporotic fractures, other primary cancer, and hospitalizations.
Within the IES study, discontinuations due to adverse reactions occurred in 6% and 5% of patients receiving EXEMESTANE and tamoxifen, respectively, and in 12% and 4.1% of patients receiving EXEMESTANE or placebo respectively within study 027.
Deaths due to any cause were reported for 1.3% of the exemestane treated patients and 1.4% of the tamoxifen treated patients within the IES study. There were 6 deaths due to stroke on the exemestane arm compared to 2 on tamoxifen. There were 5 deaths due to cardiac failure on the exemestane arm compared to 2 on tamoxifen.
The incidence of cardiac ischemic events (myocardial infarction, angina, and myocardial ischemia) was 1.6% in exemestane treated patients and 0.6% in tamoxifen treated patients in the IES study. Cardiac failure was observed in 0.4% of exemestane treated patients and 0.3% of tamoxifen treated patients.
In the adjuvant treatment of early breast cancer, the most common adverse reactions occurring in ≥10% of patients in any treatment group (EXEMESTANE vs. tamoxifen) were hot flushes (21% vs. 20%), fatigue (16% vs. 15%), arthralgia (15% vs. 9%), headache (13% vs. 11%), insomnia (12% vs. 9%), and increased sweating (12% vs. 10%). Discontinuation rates due to AEs were similar between EXEMESTANE and tamoxifen (6% vs. 5%). Incidences of cardiac ischemic events (myocardial infarction, angina, and myocardial ischemia) were EXEMESTANE 1.6%, tamoxifen 0.6%. Incidence of cardiac failure: EXEMESTANE 0.4%, tamoxifen 0.3%.
Treatment-emergent adverse reactions and illnesses including all causalities and occurring with an incidence of ≥5% in either treatment group of the IES study during or within one month of the end of treatment are shown in Table 2.
| % of patients | ||
|---|---|---|
| Body system and Adverse Reaction by MedDRA dictionary |
EXEMESTANE
25 mg daily (N=2252) |
Tamoxifen
20 mg daily 75 patients received tamoxifen 30 mg daily;
(N=2280) |
|
Eye |
||
|
Visual disturbances Event actively sought.
|
5 |
3.8 |
|
Gastrointestinal |
||
|
Nausea |
9 |
9 |
|
General Disorders |
||
|
Fatigue |
16 |
15 |
|
Musculoskeletal |
||
|
Arthralgia |
15 |
9 |
|
Pain in limb |
9 |
6 |
|
Back pain |
9 |
7 |
|
Osteoarthritis |
6 |
4.5 |
|
Nervous System |
||
|
Headache |
13 |
11 |
|
Dizziness |
10 |
8 |
|
Psychiatric |
||
|
Insomnia |
12 |
9 |
|
Depression |
6 |
6 |
|
Skin & Subcutaneous Tissue |
||
|
Increased sweating |
12 |
10 |
|
Vascular |
||
|
Hot flushes |
21 |
20 |
|
Hypertension |
10 |
8 |
In the IES study, as compared to tamoxifen, EXEMESTANE was associated with a higher incidence of events in musculoskeletal disorders and in nervous system disorders, including the following events occurring with frequency lower than 5% (osteoporosis [4.6% vs. 2.8%], osteochondrosis [0.3% vs. 0%] and stenosing tenosynovitis (trigger finger) [0.3% vs. 0%], paresthesia [2.6% vs. 0.9%], carpal tunnel syndrome [2.4% vs. 0.2%], and neuropathy [0.6% vs. 0.1%]). Diarrhea was also more frequent in the exemestane group (4.2% vs. 2.2%). Clinical fractures were reported in 94 patients receiving exemestane (4.2%) and 71 patients receiving tamoxifen (3.1%). After a median duration of therapy of about 30 months and a median follow-up of about 52 months, gastric ulcer was observed at a slightly higher frequency in the EXEMESTANE group compared to tamoxifen (0.7% vs. <0.1%). The majority of patients on EXEMESTANE with gastric ulcer received concomitant treatment with non-steroidal anti-inflammatory agents and/or had a prior history.
Tamoxifen was associated with a higher incidence of muscle cramps [3.1% vs. 1.5%], thromboembolism [2.0% vs. 0.9%], endometrial hyperplasia [1.7% vs. 0.6%], and uterine polyps [2.4% vs. 0.4%].
Common adverse reactions occurring in study 027 are described in Table 3.
|
Adverse Reaction |
Exemestane
|
Placebo
|
|
Hot flushes |
33 |
25 |
|
Arthralgia |
29 |
29 |
|
Increased sweating |
18 |
21 |
|
Alopecia |
15 |
4.1 |
|
Hypertension |
15 |
7 |
|
Insomnia |
14 |
15 |
|
Nausea |
12 |
16 |
|
Fatigue |
11 |
19 |
|
Abdominal pain |
11 |
14 |
|
Depression |
10 |
7 |
|
Diarrhea |
10 |
1.4 |
|
Dizziness |
10 |
10 |
|
Dermatitis |
8 |
1.4 |
|
Headache |
7 |
4.1 |
|
Myalgia |
6 |
4.1 |
|
Edema |
6 |
7 |
Section 42230-3
|
Patient Information
EXEMESTANE (exemestane) tablets |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration Revised: 12/2024 | |||
|
What is EXEMESTANE? EXEMESTANE is used in women who are past menopause for the treatment of: |
|||
|
|||
|
It is not known if EXEMESTANE is safe and effective in children. |
|||
|
Do not take EXEMESTANE if you are allergic to EXEMESTANE or any of the ingredients in EXEMESTANE. See the end of this leaflet for a complete list of ingredients in EXEMESTANE. |
|||
|
Before you take EXEMESTANE, tell your doctor about all your medical conditions, including if you: |
|||
|
|||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Especially tell your doctor if you take medicines that contain estrogen, including other hormone replacement therapy or birth control pills or patches. EXEMESTANE should not be taken with medicines that contain estrogen as they could affect how well EXEMESTANE works. |
|||
|
How should I take EXEMESTANE?
|
|||
|
What are the possible side effects of EXEMESTANE? EXEMESTANE may cause serious side effects, including: |
|||
|
|||
|
The most common side effects of EXEMESTANE in women with early breast cancer include: |
|||
|
|
|
|
|
The most common side effects of EXEMESTANE in women with advanced breast cancer include: |
|||
|
|
|
|
|
Your doctor will do blood tests to check your vitamin D level before starting treatment with EXEMESTANE. EXEMESTANE may cause decreased fertility in males and females. Talk to your doctor if you have concerns about fertility. These are not all the possible side effects of EXEMESTANE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store EXEMESTANE?
|
|||
|
General information about the safe and effective use of EXEMESTANE. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use EXEMESTANE for a condition for which it was not prescribed. Do not give EXEMESTANE to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or doctor for information about EXEMESTANE that is written for health professionals. |
|||
|
What is in EXEMESTANE? Active ingredient: exemestane Inactive ingredients: mannitol, crospovidone, polysorbate 80, hypromellose, colloidal silicon dioxide, microcrystalline cellulose, sodium starch glycolate, magnesium stearate, simethicone, polyethylene glycol 6000, sucrose, magnesium carbonate, titanium dioxide, methylparaben, and polyvinyl alcohol.
|
Section 44425-7
Store at 25°C (77ºF); excursions permitted to 15°–30°C (59°–86°F) [see USP Controlled Room Temperature].
10 Overdosage
Clinical trials have been conducted with exemestane given as a single dose to healthy female volunteers at doses as high as 800 mg and daily for 12 weeks to postmenopausal women with advanced breast cancer at doses as high as 600 mg. These dosages were well tolerated. There is no specific antidote to overdosage and treatment must be symptomatic. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is indicated.
A male child (age unknown) accidentally ingested a 25-mg tablet of exemestane. The initial physical examination was normal, but blood tests performed 1 hour after ingestion indicated leucocytosis (WBC 25000/mm3 with 90% neutrophils). Blood tests were repeated 4 days after the incident and were normal. No treatment was given.
In mice, mortality was observed after a single oral dose of exemestane of 3200 mg/kg, the lowest dose tested (about 640 times the recommended human dose on a mg/m2 basis). In rats and dogs, mortality was observed after single oral doses of exemestane of 5000 mg/kg (about 2000 times the recommended human dose on a mg/m2 basis) and of 3000 mg/kg (about 4000 times the recommended human dose on a mg/m2 basis), respectively.
Convulsions were observed after single doses of exemestane of 400 mg/kg and 3000 mg/kg in mice and dogs (approximately 80 and 4000 times the recommended human dose on a mg/m2 basis), respectively.
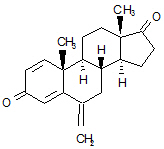
11 Description
EXEMESTANE tablets for oral administration contain 25 mg of exemestane, an irreversible, steroidal aromatase inactivator. Exemestane is chemically described as 6-methylenandrosta-1,4-diene-3,17-dione. Its molecular formula is C20H24O2 and its structural formula is as follows:
The active ingredient is a white to slightly yellow crystalline powder with a molecular weight of 296.41. Exemestane is freely soluble in N, N-dimethylformamide, soluble in methanol, and practically insoluble in water.
Each EXEMESTANE tablet contains the following inactive ingredients: mannitol, crospovidone, polysorbate 80, hypromellose, colloidal silicon dioxide, microcrystalline cellulose, sodium starch glycolate, magnesium stearate, simethicone, polyethylene glycol 6000, sucrose, magnesium carbonate, titanium dioxide, methylparaben, and polyvinyl alcohol.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
4 Contraindications
EXEMESTANE is contraindicated in patients with a known hypersensitivity to the drug or to any of the excipients.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Reductions in Bone Mineral Density (BMD) [see Warnings and Precautions (5.1)]
7 Drug Interactions
2.1 Recommended Dose
The recommended dose of EXEMESTANE in early and advanced breast cancer is one 25 mg tablet once daily after a meal.
-
•adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy.
-
•the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy.
8.7 Renal Impairment
The AUC of exemestane was increased in subjects with moderate or severe renal impairment (creatinine clearance <35 mL/min/1.73 m2) [see Clinical Pharmacology (12.3)]. However, based on experience with exemestane at repeated doses up to 200 mg daily that demonstrated a moderate increase in non life-threatening adverse reactions, dosage adjustment does not appear to be necessary.
12.3 Pharmacokinetics
Following oral administration to healthy postmenopausal women, plasma concentrations of exemestane decline polyexponentially with a mean terminal half-life of about 24 hours. The pharmacokinetics of exemestane are dose proportional after single (10 to 200 mg) or repeated oral doses (0.5 to 50 mg). Following repeated daily doses of exemestane 25 mg, plasma concentrations of unchanged drug are similar to levels measured after a single dose.
Pharmacokinetic parameters in postmenopausal women with advanced breast cancer following single or repeated doses have been compared with those in healthy, postmenopausal women. After repeated dosing, the average oral clearance in women with advanced breast cancer was 45% lower than the oral clearance in healthy postmenopausal women, with corresponding higher systemic exposure. Mean AUC values following repeated doses in women with breast cancer (75.4 ng∙h/mL) were about twice those in healthy women (41.4 ng∙h/mL).
2.2 Dose Modifications
Concomitant use of strong CYP 3A4 inducers decreases exemestane exposure, For patients receiving EXEMESTANE with a strong CYP 3A4 inducer such as rifampicin or phenytoin, the recommended dose of EXEMESTANE is 50 mg once daily after a meal. [see Drug Interactions (7) and Clinical Pharmacology (12.3)]
8.6 Hepatic Impairment
The AUC of exemestane was increased in subjects with moderate or severe hepatic impairment (Childs-Pugh B or C) [see Clinical Pharmacology (12.3)]. However, based on experience with exemestane at repeated doses up to 200 mg daily that demonstrated a moderate increase in non life-threatening adverse reactions, dosage adjustment does not appear to be necessary.
1 Indications and Usage
EXEMESTANE is an aromatase inhibitor indicated for:
-
•adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy (14.1).
-
•treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy (14.2).
12.1 Mechanism of Action
Breast cancer cell growth may be estrogen-dependent. Aromatase is the principal enzyme that converts androgens to estrogens both in pre- and postmenopausal women. While the main source of estrogen (primarily estradiol) is the ovary in premenopausal women, the principal source of circulating estrogens in postmenopausal women is from conversion of adrenal and ovarian androgens (androstenedione and testosterone) to estrogens (estrone and estradiol) by the aromatase enzyme in peripheral tissues.
Exemestane is an irreversible, steroidal aromatase inactivator, structurally related to the natural substrate androstenedione. It acts as a false substrate for the aromatase enzyme, and is processed to an intermediate that binds irreversibly to the active site of the enzyme, causing its inactivation, an effect also known as "suicide inhibition". Exemestane significantly lowers circulating estrogen concentrations in postmenopausal women, but has no detectable effect on adrenal biosynthesis of corticosteroids or aldosterone. Exemestane has no effect on other enzymes involved in the steroidogenic pathway up to a concentration at least 600 times higher than that inhibiting the aromatase enzyme.
5.2 Vitamin D Assessment
Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed, due to the high prevalence of vitamin D deficiency in women with early breast cancer (EBC). Women with vitamin D deficiency should receive supplementation with vitamin D.
5.6 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, EXEMESTANE can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of exemestane to pregnant rats and rabbits caused increased incidence of abortions and embryo-fetal toxicity. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with EXEMESTANE and for 1 month after the last dose [see Use in Specific Populations (8.1), (8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
-
•Reductions in bone mineral density (BMD) over time are seen with exemestane use (5.1).
-
•Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed (5.2).
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception (5.6, 8.1, 8.3).
2 Dosage and Administration
Recommended Dose: One 25 mg tablet once daily after a meal (2.1).
3 Dosage Forms and Strengths
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane.
5.4 Laboratory Abnormalities
In patients with early breast cancer, the incidence of hematological abnormalities of Common Toxicity Criteria (CTC) grade ≥1 was lower in the exemestane treatment group, compared with tamoxifen. Incidence of CTC grade 3 or 4 abnormalities was low (approximately 0.1%) in both treatment groups. Approximately 20% of patients receiving exemestane in clinical studies in advanced breast cancer experienced CTC grade 3 or 4 lymphocytopenia. Of these patients, 89% had a pre-existing lower grade lymphopenia. Forty percent of patients either recovered or improved to a lesser severity while on treatment. Patients did not have a significant increase in viral infections, and no opportunistic infections were observed. Elevations of serum levels of AST, ALT, alkaline phosphatase, and gamma glutamyl transferase >5 times the upper value of the normal range (i.e., ≥ CTC grade 3) have been rarely reported in patients treated for advanced breast cancer but appear mostly attributable to the underlying presence of liver and/or bone metastases. In the comparative study in advanced breast cancer patients, CTC grade 3 or 4 elevation of gamma glutamyl transferase without documented evidence of liver metastasis was reported in 2.7% of patients treated with EXEMESTANE and in 1.8% of patients treated with megestrol acetate.
In patients with early breast cancer, elevations in bilirubin, alkaline phosphatase, and creatinine were more common in those receiving exemestane than either tamoxifen or placebo. Treatment-emergent bilirubin elevations (any CTC grade) occurred in 5% of exemestane patients and 0.8% of tamoxifen patients on the Intergroup Exemestane Study (IES), and in 7% of exemestane treated patients vs. 0% of placebo treated patients in the 027 study. CTC grade 3–4 increases in bilirubin occurred in 0.9% of exemestane treated patients compared to 0.1% of tamoxifen treated patients. Alkaline phosphatase elevations of any CTC grade occurred in 15% of exemestane treated patients on the IES compared to 2.6% of tamoxifen treated patients, and in 14% of exemestane treated patients compared to 7% of placebo treated patients in study 027. Creatinine elevations occurred in 6% of exemestane treated patients and 4.3% of tamoxifen treated patients on the IES and in 6% of exemestane treated patients and 0% of placebo treated patients in study 027.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post approval use of EXEMESTANE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders- hypersensitivity
Hepatobiliary disorders- hepatitis including cholestatic hepatitis
Nervous system disorders- paresthesia
Musculoskeletal and connective tissue disorder- tendon disorders including tendon rupture, tendonitis, and tenosynovitis
Skin and subcutaneous tissue disorders- acute generalized exanthematous pustulosis, urticaria, pruritus
8 Use in Specific Populations
Lactation: Advise not to breastfeed (8.2).
5.5 Use in Premenopausal Women
EXEMESTANE is not indicated for the treatment of breast cancer in premenopausal women.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets.
30-tablet HDPE bottle NDC 59762-2858-1
14.2 Treatment of Advanced Breast Cancer
Exemestane 25 mg administered once daily was evaluated in a randomized double-blind, multicenter, multinational comparative study and in two multicenter single-arm studies of postmenopausal women with advanced breast cancer who had disease progression after treatment with tamoxifen for metastatic disease or as adjuvant therapy. Some patients also have received prior cytotoxic therapy, either as adjuvant treatment or for metastatic disease.
The primary purpose of the three studies was evaluation of objective response rate (complete response [CR] and partial response [PR]). Time to tumor progression and overall survival were also assessed in the comparative trial. Response rates were assessed based on World Health Organization (WHO) criteria, and in the comparative study, were submitted to an external review committee that was blinded to patient treatment. In the comparative study, 769 patients were randomized to receive EXEMESTANE tablets 25 mg once daily (N = 366) or megestrol acetate 40 mg four times daily (N = 403). Demographics and baseline characteristics are presented in Table 9.
| Parameter |
EXEMESTANE
(N = 366) |
Megestrol Acetate
(N = 403) |
|---|---|---|
|
Median Age (range) |
65 (35–89) |
65 (30–91) |
|
ECOG Performance Status |
||
|
0 |
167 (46%) |
187 (46%) |
|
1 |
162 (44%) |
172 (43%) |
|
2 |
34 (9%) |
42 (10%) |
|
Receptor Status |
||
|
ER and/or PgR + |
246 (67%) |
274 (68%) |
|
ER and PgR unknown |
116 (32%) |
128 (32%) |
|
Responders to prior tamoxifen |
68 (19%) |
85 (21%) |
|
NE for response to prior tamoxifen |
46 (13%) |
41 (10%) |
|
Site of Metastasis |
||
|
Visceral ± other sites |
207 (57%) |
239 (59%) |
|
Bone only |
61 (17%) |
73 (18%) |
|
Soft tissue only |
54 (15%) |
51 (13%) |
|
Bone & soft tissue |
43 (12%) |
38 (9%) |
|
Measurable Disease |
287 (78%) |
314 (78%) |
|
Prior Tamoxifen Therapy |
||
|
Adjuvant or Neoadjuvant |
145 (40%) |
152 (38%) |
|
Advanced Disease, Outcome |
||
|
CR, PR, or SD ≥ 6 months |
179 (49%) |
210 (52%) |
|
SD < 6 months, PD or NE |
42 (12%) |
41 (10%) |
|
Prior Chemotherapy |
||
|
For advanced disease ± adjuvant |
58 (16%) |
67 (17%) |
|
Adjuvant only |
104 (28%) |
108 (27%) |
|
No chemotherapy |
203 (56%) |
226 (56%) |
The efficacy results from the comparative study are shown in Table 10. The objective response rates observed in the two treatment arms showed that EXEMESTANE was not different from megestrol acetate. Response rates for EXEMESTANE from the two single-arm trials were 23.4% and 28.1%.
| Response Characteristics |
EXEMESTANE
(N=366) |
Megestrol Acetate
(N=403) |
|---|---|---|
| Abbreviations: CR = complete response, PR = partial response, SD = stable disease (no change), TTP = time to tumor progression, C.I. = confidence interval, MA = megestrol acetate, AR = EXEMESTANE | ||
|
Objective Response Rate = CR + PR (%) |
15.0 |
12.4 |
|
Difference in Response Rate (AR-MA) |
2.6 |
|
|
95% C.I. |
7.5, -2.3 |
|
|
CR (%) |
2.2 |
1.2 |
|
PR (%) |
12.8 |
11.2 |
|
SD ≥ 24 Weeks (%) |
21.3 |
21.1 |
|
Median Duration of Response (weeks) |
76.1 |
71.0 |
|
Median TTP (weeks) |
20.3 |
16.6 |
|
Hazard Ratio (AR-MA) |
0.84 |
There were too few deaths occurring across treatment groups to draw conclusions on overall survival differences. The Kaplan-Meier curve for time to tumor progression in the comparative study is shown in Figure 2.
| Figure 2. Time to Tumor Progression in the Comparative Study of Postmenopausal Women With Advanced Breast Cancer Whose Disease Had Progressed After Tamoxifen Therapy |
|---|
5.1 Reductions in Bone Mineral Density (bmd)
Reductions in bone mineral density (BMD) over time are seen with exemestane use. Table 1 describes changes in BMD from baseline to 24 months in patients receiving exemestane compared to patients receiving tamoxifen (IES) or placebo (027). Concomitant use of bisphosphonates, vitamin D supplementation, and calcium was not allowed.
|
IES |
027 |
|||
|
BMD |
Exemestane
|
Tamoxifen1
|
Exemestane
|
Placebo1
|
|
Lumbar spine (%) |
-3.1 |
-0.2 |
-3.5 |
-2.4 |
|
Femoral neck (%) |
-4.2 |
-0.3 |
-4.6 |
-2.6 |
During adjuvant treatment with exemestane, women with osteoporosis or at risk of osteoporosis should have their bone mineral density formally assessed by bone densitometry at the commencement of treatment. Monitor patients for bone mineral density loss and treat as appropriate.
1.1 Adjuvant Treatment of Postmenopausal Women
EXEMESTANE is indicated for adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy [see Clinical Studies (14.1)].
14.1 Adjuvant Treatment in Early Breast Cancer
The Intergroup Exemestane Study 031 (IES) was a randomized, double-blind, multicenter, multinational study comparing exemestane (25 mg/day) vs. tamoxifen (20 or 30 mg/day) in postmenopausal women with early breast cancer. Patients who remained disease-free after receiving adjuvant tamoxifen therapy for 2 to 3 years were randomized to receive an additional 3 or 2 years of EXEMESTANE or tamoxifen to complete a total of 5 years of hormonal therapy.
The primary objective of the study was to determine whether, in terms of disease-free survival, it was more effective to switch to EXEMESTANE rather than continuing tamoxifen therapy for the remainder of five years. Disease-free survival was defined as the time from randomization to time of local or distant recurrence of breast cancer, contralateral invasive breast cancer, or death from any cause.
The secondary objectives were to compare the two regimens in terms of overall survival and long-term tolerability. Time to contralateral invasive breast cancer and distant recurrence-free survival were also evaluated.
A total of 4724 patients in the intent-to-treat (ITT) analysis were randomized to EXEMESTANE (exemestane tablets) 25 mg once daily (N = 2352) or to continue to receive tamoxifen once daily at the same dose received before randomization (N = 2372). Demographics and baseline tumor characteristics are presented in Table 5. Prior breast cancer therapy is summarized in Table 6.
| Parameter |
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|---|---|---|
|
Age (years): |
||
|
Median age (range) |
63.0 (38.0 – 96.0) |
63.0 (31.0 – 90.0) |
|
Race, n (%): |
||
|
Caucasian |
2315 (98.4) |
2333 (98.4) |
|
Hispanic |
13 (0.6) |
13 (0.5) |
|
Asian |
10 (0.4) |
9 (0.4) |
|
Black |
7 (0.3) |
10 (0.4) |
|
Other/not reported |
7 (0.3) |
7 (0.3) |
|
Nodal status, n (%): |
||
|
Negative |
1217 (51.7) |
1228 (51.8) |
|
Positive |
1051 (44.7) |
1044 (44.0) |
|
1–3 Positive nodes |
721 (30.7) |
708 (29.8) |
|
4–9 Positive nodes |
239 (10.2) |
244 (10.3) |
|
>9 Positive nodes |
88 (3.7) |
86 (3.6) |
|
Not reported |
3 (0.1) |
6 (0.3) |
|
Unknown or missing |
84 (3.6) |
100 (4.2) |
|
Histologic type, n (%): |
||
|
Infiltrating ductal |
1777 (75.6) |
1830 (77.2) |
|
Infiltrating lobular |
341 (14.5) |
321 (13.5) |
|
Other |
231 (9.8) |
213 (9.0) |
|
Unknown or missing |
3 (0.1) |
8 (0.3) |
|
Receptor status Results for receptor status include the results of the post-randomization testing of specimens from subjects for whom receptor status was unknown at randomization.
, n (%):
|
||
|
ER and PgR Positive |
1331 (56.6) |
1319 (55.6) |
|
ER Positive and PgR Negative/Unknown |
677 (28.8) |
692 (29.2) |
|
ER Unknown and PgR Positive Only one subject in the exemestane group had unknown ER status and positive PgR status. /Unknown
|
288 (12.2) |
291 (12.3) |
|
ER Negative and PgR Positive |
6 (0.3) |
7 (0.3) |
|
ER Negative and PgR Negative/Unknown (none positive) |
48 (2.0) |
58 (2.4) |
|
Missing |
2 (0.1) |
5 (0.2) |
|
Tumor Size, n (%): |
||
|
≤ 0.5 cm |
58 (2.5) |
46 (1.9) |
|
> 0.5 – 1.0 cm |
315 (13.4) |
302 (12.7) |
|
> 1.0 – 2 cm |
1031 (43.8) |
1033 (43.5) |
|
> 2.0 – 5.0 cm |
833 (35.4) |
883 (37.2) |
|
> 5.0 cm |
62 (2.6) |
59 (2.5) |
|
Not reported |
53 (2.3) |
49 (2.1) |
|
Tumor Grade, n (%): |
||
|
G1 |
397 (16.9) |
393 (16.6) |
|
G2 |
977 (41.5) |
1007 (42.5) |
|
G3 |
454 (19.3) |
428 (18.0) |
|
G4 |
23 (1.0) |
19 (0.8) |
|
Unknown/Not Assessed/Not reported |
501 (21.3) |
525 (22.1) |
| Parameter |
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|---|---|---|
|
Type of surgery, n (%): |
||
|
Mastectomy |
1232 (52.4) |
1242 (52.4) |
|
Breast-conserving |
1116 (47.4) |
1123 (47.3) |
|
Unknown or missing |
4 (0.2) |
7 (0.3) |
|
Radiotherapy to the breast, n (%): |
||
|
Yes |
1524 (64.8) |
1523 (64.2) |
|
No |
824 (35.5) |
843 (35.5) |
|
Not reported |
4 (0.2) |
6 (0.3) |
|
Prior therapy, n (%): |
||
|
Chemotherapy |
774 (32.9) |
769 (32.4) |
|
Hormone replacement therapy |
567 (24.1) |
561 (23.7) |
|
Bisphosphonates |
43 (1.8) |
34 (1.4) |
|
Duration of tamoxifen therapy at randomization (months): |
||
|
Median (range) |
28.5 (15.8 – 52.2) |
28.4 (15.6 – 63.0) |
|
Tamoxifen dose, n (%): |
||
|
20 mg |
2270 (96.5) |
2287 (96.4) |
|
30 mg The 30 mg dose was used only in Denmark, where this dose was the standard of care.
|
78 (3.3) |
75 (3.2) |
|
Not reported |
4 (0.2) |
10 (0.4) |
After a median duration of therapy of 27 months and with a median follow-up of 34.5 months, 520 events were reported, 213 in the EXEMESTANE group and 307 in the tamoxifen group (Table 7).
| Event |
First Events
N (%) |
|
|---|---|---|
|
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|
|
Loco-regional recurrence |
34 (1.45) |
45 (1.90) |
|
Distant recurrence |
126 (5.36) |
183 (7.72) |
|
Second primary – contralateral breast cancer |
7 (0.30) |
25 (1.05) |
|
Death – breast cancer |
1 (0.04) |
6 (0.25) |
|
Death – other reason |
41 (1.74) |
43 (1.81) |
|
Death – missing/unknown |
3 (0.13) |
5 (0.21) |
|
Ipsilateral breast cancer |
1 (0.04) |
0 |
|
Total number of events |
213 (9.06) |
307 (12.94) |
Disease-free survival in the intent-to-treat population was statistically significantly improved [Hazard Ratio (HR) = 0.69, 95% CI: 0.58, 0.82, P = 0.00003, Table 8, Figure 1] in the EXEMESTANE arm compared to the tamoxifen arm. In the hormone receptor-positive subpopulation representing about 85% of the trial patients, disease-free survival was also statistically significantly improved (HR = 0.65, 95% CI: 0.53, 0.79, P = 0.00001) in the EXEMESTANE arm compared to the tamoxifen arm. Consistent results were observed in the subgroups of patients with node negative or positive disease, and patients who had or had not received prior chemotherapy.
An overall survival update at 119 months median follow-up showed no significant difference between the two groups, with 467 deaths (19.9%) occurring in the EXEMESTANE group and 510 deaths (21.5%) in the tamoxifen group.
|
|
Hazard Ratio
|
p-value
|
|
Disease-free survival |
0.69 (0.58–0.82) |
0.00003 |
|
Time to contralateral breast cancer |
0.32 (0.15–0.72) |
0.00340 |
|
Distant recurrence-free survival |
0.74 (0.62–0.90) |
0.00207 |
|
Overall survival |
0.91 (0.81–1.04) |
0.16 Not adjusted for multiple testing.
|
|
ER and/or PgR positive |
||
|
Disease-free survival |
0.65 (0.53–0.79) |
0.00001 |
|
Time to contralateral breast cancer |
0.22 (0.08–0.57) |
0.00069 |
|
Distant recurrence-free survival |
0.73 (0.59–0.90) |
0.00367 |
|
Overall survival |
0.89 (0.78–1.02) |
0.09065 |
Figure 1. Disease-Free Survival in the IES Study of Postmenopausal Women with Early Breast Cancer (ITT Population)
1.2 Advanced Breast Cancer in Postmenopausal Women
EXEMESTANE is indicated for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy [see Clinical Studies (14.2)].
5.3 Administration With Estrogen Containing Agents
EXEMESTANE should not be coadministered with systemic estrogen-containing agents as these could interfere with its pharmacologic action.
Principal Display Panel 25 Mg Tablet Bottle Label
NDC 59762-2858-1
30 Tablets
GREENSTONE® BRAND
exemestane
tablets
25 mg
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A 2-year carcinogenicity study in mice at doses of 50, 150, and 450 mg/kg/day exemestane (gavage), resulted in an increased incidence of hepatocellular adenomas and/or carcinomas in both genders at the high dose level. Plasma AUC (0–24hr) at the high dose were 2575 ± 386 and 5667 ± 1833 ng.hr/mL in males and females (approx. 34 and 75 fold the AUC in postmenopausal patients at the recommended clinical dose). An increased incidence of renal tubular adenomas was observed in male mice at the high dose of 450 mg/kg/day. Since the doses tested in mice did not achieve an MTD, neoplastic findings in organs other than liver and kidneys remain unknown.
A separate carcinogenicity study was conducted in rats at the doses of 30, 100, and 315 mg/kg/day exemestane (gavage) for 92 weeks in males and 2 years in females. No evidence of carcinogenic activity up to the highest dose tested of 315 mg/kg/day was observed in females. The male rat study was inconclusive since it was terminated prematurely at Week 92. At the highest dose, plasma AUC(0–24hr) levels in male (1418 ± 287 ng.hr/mL) and female (2318 ± 1067 ng.hr/mL) rats were 19 and 31 fold higher than those measured in postmenopausal cancer patients receiving the recommended clinical dose.
Exemestane was not mutagenic in vitro in bacteria (Ames test) or mammalian cells (V79 Chinese hamster lung cells). Exemestane was clastogenic in human lymphocytes in vitro without metabolic activation but was not clastogenic in vivo (micronucleus assay in mouse bone marrow). Exemestane did not increase unscheduled DNA synthesis in rat hepatocytes when tested in vitro.
In a pilot reproductive study in rats, male rats were treated with doses of 125–1000 mg/kg/day exemestane, beginning 63 days prior to and during cohabitation. Untreated female rats showed reduced fertility when mated to males treated with ≥500 mg/kg/day exemestane (≥200 times the recommended human dose on a mg/m2 basis). In a separate study, exemestane was given to female rats at 4–100 mg/kg/day beginning 14 days prior to mating and through day 15 or 20 of gestation. Exemestane increased the placental weights at ≥4 mg/kg/day (≥1.5 times the human dose on a mg/m2 basis). Exemestane showed no effects on ovarian function, mating behavior, and conception rate in rats given doses up to 20 mg/kg/day (approximately 8 times the recommended human dose on a mg/m2 basis); however, decreases in mean litter size and fetal body weight, along with delayed ossification were evidenced at ≥20 mg/kg/day. In general toxicology studies, changes in the ovary, including hyperplasia, an increase in the incidence of ovarian cysts, and a decrease in corpora lutea were observed with variable frequency in mice, rats, and dogs at doses that ranged from 3–20 times the human dose on a mg/m2 basis.
Structured Label Content
Section 42229-5 (42229-5)
Adjuvant Therapy
The data described below reflect exposure to EXEMESTANE in 2325 postmenopausal women with early breast cancer. EXEMESTANE tolerability in postmenopausal women with early breast cancer was evaluated in two well-controlled trials: the IES study [see Clinical Studies (14.1)] and the 027 study (a randomized, placebo-controlled, double-blind, parallel group study specifically designed to assess the effects of exemestane on bone metabolism, hormones, lipids, and coagulation factors over 2 years of treatment).
The median duration of adjuvant treatment was 27.4 months and 27.3 months for patients receiving EXEMESTANE or tamoxifen, respectively, within the IES study and 23.9 months for patients receiving EXEMESTANE or placebo within the 027 study. Median duration of observation after randomization for EXEMESTANE was 34.5 months and for tamoxifen was 34.6 months. Median duration of observation was 30 months for both groups in the 027 study.
Certain adverse reactions, which were expected based on the known pharmacological properties and side effect profiles of test drugs, were actively sought through a positive checklist. Signs and symptoms were graded for severity using CTC in both studies. Within the IES study, the presence of some illnesses/conditions was monitored through a positive checklist without assessment of severity. These included myocardial infarction, other cardiovascular disorders, gynecological disorders, osteoporosis, osteoporotic fractures, other primary cancer, and hospitalizations.
Within the IES study, discontinuations due to adverse reactions occurred in 6% and 5% of patients receiving EXEMESTANE and tamoxifen, respectively, and in 12% and 4.1% of patients receiving EXEMESTANE or placebo respectively within study 027.
Deaths due to any cause were reported for 1.3% of the exemestane treated patients and 1.4% of the tamoxifen treated patients within the IES study. There were 6 deaths due to stroke on the exemestane arm compared to 2 on tamoxifen. There were 5 deaths due to cardiac failure on the exemestane arm compared to 2 on tamoxifen.
The incidence of cardiac ischemic events (myocardial infarction, angina, and myocardial ischemia) was 1.6% in exemestane treated patients and 0.6% in tamoxifen treated patients in the IES study. Cardiac failure was observed in 0.4% of exemestane treated patients and 0.3% of tamoxifen treated patients.
In the adjuvant treatment of early breast cancer, the most common adverse reactions occurring in ≥10% of patients in any treatment group (EXEMESTANE vs. tamoxifen) were hot flushes (21% vs. 20%), fatigue (16% vs. 15%), arthralgia (15% vs. 9%), headache (13% vs. 11%), insomnia (12% vs. 9%), and increased sweating (12% vs. 10%). Discontinuation rates due to AEs were similar between EXEMESTANE and tamoxifen (6% vs. 5%). Incidences of cardiac ischemic events (myocardial infarction, angina, and myocardial ischemia) were EXEMESTANE 1.6%, tamoxifen 0.6%. Incidence of cardiac failure: EXEMESTANE 0.4%, tamoxifen 0.3%.
Treatment-emergent adverse reactions and illnesses including all causalities and occurring with an incidence of ≥5% in either treatment group of the IES study during or within one month of the end of treatment are shown in Table 2.
| % of patients | ||
|---|---|---|
| Body system and Adverse Reaction by MedDRA dictionary |
EXEMESTANE
25 mg daily (N=2252) |
Tamoxifen
20 mg daily 75 patients received tamoxifen 30 mg daily;
(N=2280) |
|
Eye |
||
|
Visual disturbances Event actively sought.
|
5 |
3.8 |
|
Gastrointestinal |
||
|
Nausea |
9 |
9 |
|
General Disorders |
||
|
Fatigue |
16 |
15 |
|
Musculoskeletal |
||
|
Arthralgia |
15 |
9 |
|
Pain in limb |
9 |
6 |
|
Back pain |
9 |
7 |
|
Osteoarthritis |
6 |
4.5 |
|
Nervous System |
||
|
Headache |
13 |
11 |
|
Dizziness |
10 |
8 |
|
Psychiatric |
||
|
Insomnia |
12 |
9 |
|
Depression |
6 |
6 |
|
Skin & Subcutaneous Tissue |
||
|
Increased sweating |
12 |
10 |
|
Vascular |
||
|
Hot flushes |
21 |
20 |
|
Hypertension |
10 |
8 |
In the IES study, as compared to tamoxifen, EXEMESTANE was associated with a higher incidence of events in musculoskeletal disorders and in nervous system disorders, including the following events occurring with frequency lower than 5% (osteoporosis [4.6% vs. 2.8%], osteochondrosis [0.3% vs. 0%] and stenosing tenosynovitis (trigger finger) [0.3% vs. 0%], paresthesia [2.6% vs. 0.9%], carpal tunnel syndrome [2.4% vs. 0.2%], and neuropathy [0.6% vs. 0.1%]). Diarrhea was also more frequent in the exemestane group (4.2% vs. 2.2%). Clinical fractures were reported in 94 patients receiving exemestane (4.2%) and 71 patients receiving tamoxifen (3.1%). After a median duration of therapy of about 30 months and a median follow-up of about 52 months, gastric ulcer was observed at a slightly higher frequency in the EXEMESTANE group compared to tamoxifen (0.7% vs. <0.1%). The majority of patients on EXEMESTANE with gastric ulcer received concomitant treatment with non-steroidal anti-inflammatory agents and/or had a prior history.
Tamoxifen was associated with a higher incidence of muscle cramps [3.1% vs. 1.5%], thromboembolism [2.0% vs. 0.9%], endometrial hyperplasia [1.7% vs. 0.6%], and uterine polyps [2.4% vs. 0.4%].
Common adverse reactions occurring in study 027 are described in Table 3.
|
Adverse Reaction |
Exemestane
|
Placebo
|
|
Hot flushes |
33 |
25 |
|
Arthralgia |
29 |
29 |
|
Increased sweating |
18 |
21 |
|
Alopecia |
15 |
4.1 |
|
Hypertension |
15 |
7 |
|
Insomnia |
14 |
15 |
|
Nausea |
12 |
16 |
|
Fatigue |
11 |
19 |
|
Abdominal pain |
11 |
14 |
|
Depression |
10 |
7 |
|
Diarrhea |
10 |
1.4 |
|
Dizziness |
10 |
10 |
|
Dermatitis |
8 |
1.4 |
|
Headache |
7 |
4.1 |
|
Myalgia |
6 |
4.1 |
|
Edema |
6 |
7 |
Section 42230-3 (42230-3)
|
Patient Information
EXEMESTANE (exemestane) tablets |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration Revised: 12/2024 | |||
|
What is EXEMESTANE? EXEMESTANE is used in women who are past menopause for the treatment of: |
|||
|
|||
|
It is not known if EXEMESTANE is safe and effective in children. |
|||
|
Do not take EXEMESTANE if you are allergic to EXEMESTANE or any of the ingredients in EXEMESTANE. See the end of this leaflet for a complete list of ingredients in EXEMESTANE. |
|||
|
Before you take EXEMESTANE, tell your doctor about all your medical conditions, including if you: |
|||
|
|||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Especially tell your doctor if you take medicines that contain estrogen, including other hormone replacement therapy or birth control pills or patches. EXEMESTANE should not be taken with medicines that contain estrogen as they could affect how well EXEMESTANE works. |
|||
|
How should I take EXEMESTANE?
|
|||
|
What are the possible side effects of EXEMESTANE? EXEMESTANE may cause serious side effects, including: |
|||
|
|||
|
The most common side effects of EXEMESTANE in women with early breast cancer include: |
|||
|
|
|
|
|
The most common side effects of EXEMESTANE in women with advanced breast cancer include: |
|||
|
|
|
|
|
Your doctor will do blood tests to check your vitamin D level before starting treatment with EXEMESTANE. EXEMESTANE may cause decreased fertility in males and females. Talk to your doctor if you have concerns about fertility. These are not all the possible side effects of EXEMESTANE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store EXEMESTANE?
|
|||
|
General information about the safe and effective use of EXEMESTANE. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use EXEMESTANE for a condition for which it was not prescribed. Do not give EXEMESTANE to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or doctor for information about EXEMESTANE that is written for health professionals. |
|||
|
What is in EXEMESTANE? Active ingredient: exemestane Inactive ingredients: mannitol, crospovidone, polysorbate 80, hypromellose, colloidal silicon dioxide, microcrystalline cellulose, sodium starch glycolate, magnesium stearate, simethicone, polyethylene glycol 6000, sucrose, magnesium carbonate, titanium dioxide, methylparaben, and polyvinyl alcohol.
|
Section 44425-7 (44425-7)
Store at 25°C (77ºF); excursions permitted to 15°–30°C (59°–86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Clinical trials have been conducted with exemestane given as a single dose to healthy female volunteers at doses as high as 800 mg and daily for 12 weeks to postmenopausal women with advanced breast cancer at doses as high as 600 mg. These dosages were well tolerated. There is no specific antidote to overdosage and treatment must be symptomatic. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is indicated.
A male child (age unknown) accidentally ingested a 25-mg tablet of exemestane. The initial physical examination was normal, but blood tests performed 1 hour after ingestion indicated leucocytosis (WBC 25000/mm3 with 90% neutrophils). Blood tests were repeated 4 days after the incident and were normal. No treatment was given.
In mice, mortality was observed after a single oral dose of exemestane of 3200 mg/kg, the lowest dose tested (about 640 times the recommended human dose on a mg/m2 basis). In rats and dogs, mortality was observed after single oral doses of exemestane of 5000 mg/kg (about 2000 times the recommended human dose on a mg/m2 basis) and of 3000 mg/kg (about 4000 times the recommended human dose on a mg/m2 basis), respectively.
Convulsions were observed after single doses of exemestane of 400 mg/kg and 3000 mg/kg in mice and dogs (approximately 80 and 4000 times the recommended human dose on a mg/m2 basis), respectively.
11 Description (11 DESCRIPTION)
EXEMESTANE tablets for oral administration contain 25 mg of exemestane, an irreversible, steroidal aromatase inactivator. Exemestane is chemically described as 6-methylenandrosta-1,4-diene-3,17-dione. Its molecular formula is C20H24O2 and its structural formula is as follows:
The active ingredient is a white to slightly yellow crystalline powder with a molecular weight of 296.41. Exemestane is freely soluble in N, N-dimethylformamide, soluble in methanol, and practically insoluble in water.
Each EXEMESTANE tablet contains the following inactive ingredients: mannitol, crospovidone, polysorbate 80, hypromellose, colloidal silicon dioxide, microcrystalline cellulose, sodium starch glycolate, magnesium stearate, simethicone, polyethylene glycol 6000, sucrose, magnesium carbonate, titanium dioxide, methylparaben, and polyvinyl alcohol.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
4 Contraindications (4 CONTRAINDICATIONS)
EXEMESTANE is contraindicated in patients with a known hypersensitivity to the drug or to any of the excipients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Reductions in Bone Mineral Density (BMD) [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
2.1 Recommended Dose
The recommended dose of EXEMESTANE in early and advanced breast cancer is one 25 mg tablet once daily after a meal.
-
•adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy.
-
•the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy.
8.7 Renal Impairment
The AUC of exemestane was increased in subjects with moderate or severe renal impairment (creatinine clearance <35 mL/min/1.73 m2) [see Clinical Pharmacology (12.3)]. However, based on experience with exemestane at repeated doses up to 200 mg daily that demonstrated a moderate increase in non life-threatening adverse reactions, dosage adjustment does not appear to be necessary.
12.3 Pharmacokinetics
Following oral administration to healthy postmenopausal women, plasma concentrations of exemestane decline polyexponentially with a mean terminal half-life of about 24 hours. The pharmacokinetics of exemestane are dose proportional after single (10 to 200 mg) or repeated oral doses (0.5 to 50 mg). Following repeated daily doses of exemestane 25 mg, plasma concentrations of unchanged drug are similar to levels measured after a single dose.
Pharmacokinetic parameters in postmenopausal women with advanced breast cancer following single or repeated doses have been compared with those in healthy, postmenopausal women. After repeated dosing, the average oral clearance in women with advanced breast cancer was 45% lower than the oral clearance in healthy postmenopausal women, with corresponding higher systemic exposure. Mean AUC values following repeated doses in women with breast cancer (75.4 ng∙h/mL) were about twice those in healthy women (41.4 ng∙h/mL).
2.2 Dose Modifications
Concomitant use of strong CYP 3A4 inducers decreases exemestane exposure, For patients receiving EXEMESTANE with a strong CYP 3A4 inducer such as rifampicin or phenytoin, the recommended dose of EXEMESTANE is 50 mg once daily after a meal. [see Drug Interactions (7) and Clinical Pharmacology (12.3)]
8.6 Hepatic Impairment
The AUC of exemestane was increased in subjects with moderate or severe hepatic impairment (Childs-Pugh B or C) [see Clinical Pharmacology (12.3)]. However, based on experience with exemestane at repeated doses up to 200 mg daily that demonstrated a moderate increase in non life-threatening adverse reactions, dosage adjustment does not appear to be necessary.
1 Indications and Usage (1 INDICATIONS AND USAGE)
EXEMESTANE is an aromatase inhibitor indicated for:
-
•adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy (14.1).
-
•treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy (14.2).
12.1 Mechanism of Action
Breast cancer cell growth may be estrogen-dependent. Aromatase is the principal enzyme that converts androgens to estrogens both in pre- and postmenopausal women. While the main source of estrogen (primarily estradiol) is the ovary in premenopausal women, the principal source of circulating estrogens in postmenopausal women is from conversion of adrenal and ovarian androgens (androstenedione and testosterone) to estrogens (estrone and estradiol) by the aromatase enzyme in peripheral tissues.
Exemestane is an irreversible, steroidal aromatase inactivator, structurally related to the natural substrate androstenedione. It acts as a false substrate for the aromatase enzyme, and is processed to an intermediate that binds irreversibly to the active site of the enzyme, causing its inactivation, an effect also known as "suicide inhibition". Exemestane significantly lowers circulating estrogen concentrations in postmenopausal women, but has no detectable effect on adrenal biosynthesis of corticosteroids or aldosterone. Exemestane has no effect on other enzymes involved in the steroidogenic pathway up to a concentration at least 600 times higher than that inhibiting the aromatase enzyme.
5.2 Vitamin D Assessment
Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed, due to the high prevalence of vitamin D deficiency in women with early breast cancer (EBC). Women with vitamin D deficiency should receive supplementation with vitamin D.
5.6 Embryo Fetal Toxicity (5.6 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, EXEMESTANE can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of exemestane to pregnant rats and rabbits caused increased incidence of abortions and embryo-fetal toxicity. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with EXEMESTANE and for 1 month after the last dose [see Use in Specific Populations (8.1), (8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Reductions in bone mineral density (BMD) over time are seen with exemestane use (5.1).
-
•Routine assessment of 25-hydroxy vitamin D levels prior to the start of aromatase inhibitor treatment should be performed (5.2).
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception (5.6, 8.1, 8.3).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Recommended Dose: One 25 mg tablet once daily after a meal (2.1).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane.
5.4 Laboratory Abnormalities
In patients with early breast cancer, the incidence of hematological abnormalities of Common Toxicity Criteria (CTC) grade ≥1 was lower in the exemestane treatment group, compared with tamoxifen. Incidence of CTC grade 3 or 4 abnormalities was low (approximately 0.1%) in both treatment groups. Approximately 20% of patients receiving exemestane in clinical studies in advanced breast cancer experienced CTC grade 3 or 4 lymphocytopenia. Of these patients, 89% had a pre-existing lower grade lymphopenia. Forty percent of patients either recovered or improved to a lesser severity while on treatment. Patients did not have a significant increase in viral infections, and no opportunistic infections were observed. Elevations of serum levels of AST, ALT, alkaline phosphatase, and gamma glutamyl transferase >5 times the upper value of the normal range (i.e., ≥ CTC grade 3) have been rarely reported in patients treated for advanced breast cancer but appear mostly attributable to the underlying presence of liver and/or bone metastases. In the comparative study in advanced breast cancer patients, CTC grade 3 or 4 elevation of gamma glutamyl transferase without documented evidence of liver metastasis was reported in 2.7% of patients treated with EXEMESTANE and in 1.8% of patients treated with megestrol acetate.
In patients with early breast cancer, elevations in bilirubin, alkaline phosphatase, and creatinine were more common in those receiving exemestane than either tamoxifen or placebo. Treatment-emergent bilirubin elevations (any CTC grade) occurred in 5% of exemestane patients and 0.8% of tamoxifen patients on the Intergroup Exemestane Study (IES), and in 7% of exemestane treated patients vs. 0% of placebo treated patients in the 027 study. CTC grade 3–4 increases in bilirubin occurred in 0.9% of exemestane treated patients compared to 0.1% of tamoxifen treated patients. Alkaline phosphatase elevations of any CTC grade occurred in 15% of exemestane treated patients on the IES compared to 2.6% of tamoxifen treated patients, and in 14% of exemestane treated patients compared to 7% of placebo treated patients in study 027. Creatinine elevations occurred in 6% of exemestane treated patients and 4.3% of tamoxifen treated patients on the IES and in 6% of exemestane treated patients and 0% of placebo treated patients in study 027.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post approval use of EXEMESTANE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders- hypersensitivity
Hepatobiliary disorders- hepatitis including cholestatic hepatitis
Nervous system disorders- paresthesia
Musculoskeletal and connective tissue disorder- tendon disorders including tendon rupture, tendonitis, and tenosynovitis
Skin and subcutaneous tissue disorders- acute generalized exanthematous pustulosis, urticaria, pruritus
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed (8.2).
5.5 Use in Premenopausal Women
EXEMESTANE is not indicated for the treatment of breast cancer in premenopausal women.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
EXEMESTANE tablets are round, biconvex, off-white to slightly greyish sugar-coated tablets, about 6 mm diameter, printed with numbers 2858 on one side in black ink. Each tablet contains 25 mg of exemestane. EXEMESTANE is packaged in HDPE bottles with a child-resistant screw cap, supplied in packs of 30 tablets.
30-tablet HDPE bottle NDC 59762-2858-1
14.2 Treatment of Advanced Breast Cancer
Exemestane 25 mg administered once daily was evaluated in a randomized double-blind, multicenter, multinational comparative study and in two multicenter single-arm studies of postmenopausal women with advanced breast cancer who had disease progression after treatment with tamoxifen for metastatic disease or as adjuvant therapy. Some patients also have received prior cytotoxic therapy, either as adjuvant treatment or for metastatic disease.
The primary purpose of the three studies was evaluation of objective response rate (complete response [CR] and partial response [PR]). Time to tumor progression and overall survival were also assessed in the comparative trial. Response rates were assessed based on World Health Organization (WHO) criteria, and in the comparative study, were submitted to an external review committee that was blinded to patient treatment. In the comparative study, 769 patients were randomized to receive EXEMESTANE tablets 25 mg once daily (N = 366) or megestrol acetate 40 mg four times daily (N = 403). Demographics and baseline characteristics are presented in Table 9.
| Parameter |
EXEMESTANE
(N = 366) |
Megestrol Acetate
(N = 403) |
|---|---|---|
|
Median Age (range) |
65 (35–89) |
65 (30–91) |
|
ECOG Performance Status |
||
|
0 |
167 (46%) |
187 (46%) |
|
1 |
162 (44%) |
172 (43%) |
|
2 |
34 (9%) |
42 (10%) |
|
Receptor Status |
||
|
ER and/or PgR + |
246 (67%) |
274 (68%) |
|
ER and PgR unknown |
116 (32%) |
128 (32%) |
|
Responders to prior tamoxifen |
68 (19%) |
85 (21%) |
|
NE for response to prior tamoxifen |
46 (13%) |
41 (10%) |
|
Site of Metastasis |
||
|
Visceral ± other sites |
207 (57%) |
239 (59%) |
|
Bone only |
61 (17%) |
73 (18%) |
|
Soft tissue only |
54 (15%) |
51 (13%) |
|
Bone & soft tissue |
43 (12%) |
38 (9%) |
|
Measurable Disease |
287 (78%) |
314 (78%) |
|
Prior Tamoxifen Therapy |
||
|
Adjuvant or Neoadjuvant |
145 (40%) |
152 (38%) |
|
Advanced Disease, Outcome |
||
|
CR, PR, or SD ≥ 6 months |
179 (49%) |
210 (52%) |
|
SD < 6 months, PD or NE |
42 (12%) |
41 (10%) |
|
Prior Chemotherapy |
||
|
For advanced disease ± adjuvant |
58 (16%) |
67 (17%) |
|
Adjuvant only |
104 (28%) |
108 (27%) |
|
No chemotherapy |
203 (56%) |
226 (56%) |
The efficacy results from the comparative study are shown in Table 10. The objective response rates observed in the two treatment arms showed that EXEMESTANE was not different from megestrol acetate. Response rates for EXEMESTANE from the two single-arm trials were 23.4% and 28.1%.
| Response Characteristics |
EXEMESTANE
(N=366) |
Megestrol Acetate
(N=403) |
|---|---|---|
| Abbreviations: CR = complete response, PR = partial response, SD = stable disease (no change), TTP = time to tumor progression, C.I. = confidence interval, MA = megestrol acetate, AR = EXEMESTANE | ||
|
Objective Response Rate = CR + PR (%) |
15.0 |
12.4 |
|
Difference in Response Rate (AR-MA) |
2.6 |
|
|
95% C.I. |
7.5, -2.3 |
|
|
CR (%) |
2.2 |
1.2 |
|
PR (%) |
12.8 |
11.2 |
|
SD ≥ 24 Weeks (%) |
21.3 |
21.1 |
|
Median Duration of Response (weeks) |
76.1 |
71.0 |
|
Median TTP (weeks) |
20.3 |
16.6 |
|
Hazard Ratio (AR-MA) |
0.84 |
There were too few deaths occurring across treatment groups to draw conclusions on overall survival differences. The Kaplan-Meier curve for time to tumor progression in the comparative study is shown in Figure 2.
| Figure 2. Time to Tumor Progression in the Comparative Study of Postmenopausal Women With Advanced Breast Cancer Whose Disease Had Progressed After Tamoxifen Therapy |
|---|
5.1 Reductions in Bone Mineral Density (bmd) (5.1 Reductions in Bone Mineral Density (BMD))
Reductions in bone mineral density (BMD) over time are seen with exemestane use. Table 1 describes changes in BMD from baseline to 24 months in patients receiving exemestane compared to patients receiving tamoxifen (IES) or placebo (027). Concomitant use of bisphosphonates, vitamin D supplementation, and calcium was not allowed.
|
IES |
027 |
|||
|
BMD |
Exemestane
|
Tamoxifen1
|
Exemestane
|
Placebo1
|
|
Lumbar spine (%) |
-3.1 |
-0.2 |
-3.5 |
-2.4 |
|
Femoral neck (%) |
-4.2 |
-0.3 |
-4.6 |
-2.6 |
During adjuvant treatment with exemestane, women with osteoporosis or at risk of osteoporosis should have their bone mineral density formally assessed by bone densitometry at the commencement of treatment. Monitor patients for bone mineral density loss and treat as appropriate.
1.1 Adjuvant Treatment of Postmenopausal Women
EXEMESTANE is indicated for adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer who have received two to three years of tamoxifen and are switched to EXEMESTANE for completion of a total of five consecutive years of adjuvant hormonal therapy [see Clinical Studies (14.1)].
14.1 Adjuvant Treatment in Early Breast Cancer
The Intergroup Exemestane Study 031 (IES) was a randomized, double-blind, multicenter, multinational study comparing exemestane (25 mg/day) vs. tamoxifen (20 or 30 mg/day) in postmenopausal women with early breast cancer. Patients who remained disease-free after receiving adjuvant tamoxifen therapy for 2 to 3 years were randomized to receive an additional 3 or 2 years of EXEMESTANE or tamoxifen to complete a total of 5 years of hormonal therapy.
The primary objective of the study was to determine whether, in terms of disease-free survival, it was more effective to switch to EXEMESTANE rather than continuing tamoxifen therapy for the remainder of five years. Disease-free survival was defined as the time from randomization to time of local or distant recurrence of breast cancer, contralateral invasive breast cancer, or death from any cause.
The secondary objectives were to compare the two regimens in terms of overall survival and long-term tolerability. Time to contralateral invasive breast cancer and distant recurrence-free survival were also evaluated.
A total of 4724 patients in the intent-to-treat (ITT) analysis were randomized to EXEMESTANE (exemestane tablets) 25 mg once daily (N = 2352) or to continue to receive tamoxifen once daily at the same dose received before randomization (N = 2372). Demographics and baseline tumor characteristics are presented in Table 5. Prior breast cancer therapy is summarized in Table 6.
| Parameter |
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|---|---|---|
|
Age (years): |
||
|
Median age (range) |
63.0 (38.0 – 96.0) |
63.0 (31.0 – 90.0) |
|
Race, n (%): |
||
|
Caucasian |
2315 (98.4) |
2333 (98.4) |
|
Hispanic |
13 (0.6) |
13 (0.5) |
|
Asian |
10 (0.4) |
9 (0.4) |
|
Black |
7 (0.3) |
10 (0.4) |
|
Other/not reported |
7 (0.3) |
7 (0.3) |
|
Nodal status, n (%): |
||
|
Negative |
1217 (51.7) |
1228 (51.8) |
|
Positive |
1051 (44.7) |
1044 (44.0) |
|
1–3 Positive nodes |
721 (30.7) |
708 (29.8) |
|
4–9 Positive nodes |
239 (10.2) |
244 (10.3) |
|
>9 Positive nodes |
88 (3.7) |
86 (3.6) |
|
Not reported |
3 (0.1) |
6 (0.3) |
|
Unknown or missing |
84 (3.6) |
100 (4.2) |
|
Histologic type, n (%): |
||
|
Infiltrating ductal |
1777 (75.6) |
1830 (77.2) |
|
Infiltrating lobular |
341 (14.5) |
321 (13.5) |
|
Other |
231 (9.8) |
213 (9.0) |
|
Unknown or missing |
3 (0.1) |
8 (0.3) |
|
Receptor status Results for receptor status include the results of the post-randomization testing of specimens from subjects for whom receptor status was unknown at randomization.
, n (%):
|
||
|
ER and PgR Positive |
1331 (56.6) |
1319 (55.6) |
|
ER Positive and PgR Negative/Unknown |
677 (28.8) |
692 (29.2) |
|
ER Unknown and PgR Positive Only one subject in the exemestane group had unknown ER status and positive PgR status. /Unknown
|
288 (12.2) |
291 (12.3) |
|
ER Negative and PgR Positive |
6 (0.3) |
7 (0.3) |
|
ER Negative and PgR Negative/Unknown (none positive) |
48 (2.0) |
58 (2.4) |
|
Missing |
2 (0.1) |
5 (0.2) |
|
Tumor Size, n (%): |
||
|
≤ 0.5 cm |
58 (2.5) |
46 (1.9) |
|
> 0.5 – 1.0 cm |
315 (13.4) |
302 (12.7) |
|
> 1.0 – 2 cm |
1031 (43.8) |
1033 (43.5) |
|
> 2.0 – 5.0 cm |
833 (35.4) |
883 (37.2) |
|
> 5.0 cm |
62 (2.6) |
59 (2.5) |
|
Not reported |
53 (2.3) |
49 (2.1) |
|
Tumor Grade, n (%): |
||
|
G1 |
397 (16.9) |
393 (16.6) |
|
G2 |
977 (41.5) |
1007 (42.5) |
|
G3 |
454 (19.3) |
428 (18.0) |
|
G4 |
23 (1.0) |
19 (0.8) |
|
Unknown/Not Assessed/Not reported |
501 (21.3) |
525 (22.1) |
| Parameter |
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|---|---|---|
|
Type of surgery, n (%): |
||
|
Mastectomy |
1232 (52.4) |
1242 (52.4) |
|
Breast-conserving |
1116 (47.4) |
1123 (47.3) |
|
Unknown or missing |
4 (0.2) |
7 (0.3) |
|
Radiotherapy to the breast, n (%): |
||
|
Yes |
1524 (64.8) |
1523 (64.2) |
|
No |
824 (35.5) |
843 (35.5) |
|
Not reported |
4 (0.2) |
6 (0.3) |
|
Prior therapy, n (%): |
||
|
Chemotherapy |
774 (32.9) |
769 (32.4) |
|
Hormone replacement therapy |
567 (24.1) |
561 (23.7) |
|
Bisphosphonates |
43 (1.8) |
34 (1.4) |
|
Duration of tamoxifen therapy at randomization (months): |
||
|
Median (range) |
28.5 (15.8 – 52.2) |
28.4 (15.6 – 63.0) |
|
Tamoxifen dose, n (%): |
||
|
20 mg |
2270 (96.5) |
2287 (96.4) |
|
30 mg The 30 mg dose was used only in Denmark, where this dose was the standard of care.
|
78 (3.3) |
75 (3.2) |
|
Not reported |
4 (0.2) |
10 (0.4) |
After a median duration of therapy of 27 months and with a median follow-up of 34.5 months, 520 events were reported, 213 in the EXEMESTANE group and 307 in the tamoxifen group (Table 7).
| Event |
First Events
N (%) |
|
|---|---|---|
|
Exemestane
(N = 2352) |
Tamoxifen
(N = 2372) |
|
|
Loco-regional recurrence |
34 (1.45) |
45 (1.90) |
|
Distant recurrence |
126 (5.36) |
183 (7.72) |
|
Second primary – contralateral breast cancer |
7 (0.30) |
25 (1.05) |
|
Death – breast cancer |
1 (0.04) |
6 (0.25) |
|
Death – other reason |
41 (1.74) |
43 (1.81) |
|
Death – missing/unknown |
3 (0.13) |
5 (0.21) |
|
Ipsilateral breast cancer |
1 (0.04) |
0 |
|
Total number of events |
213 (9.06) |
307 (12.94) |
Disease-free survival in the intent-to-treat population was statistically significantly improved [Hazard Ratio (HR) = 0.69, 95% CI: 0.58, 0.82, P = 0.00003, Table 8, Figure 1] in the EXEMESTANE arm compared to the tamoxifen arm. In the hormone receptor-positive subpopulation representing about 85% of the trial patients, disease-free survival was also statistically significantly improved (HR = 0.65, 95% CI: 0.53, 0.79, P = 0.00001) in the EXEMESTANE arm compared to the tamoxifen arm. Consistent results were observed in the subgroups of patients with node negative or positive disease, and patients who had or had not received prior chemotherapy.
An overall survival update at 119 months median follow-up showed no significant difference between the two groups, with 467 deaths (19.9%) occurring in the EXEMESTANE group and 510 deaths (21.5%) in the tamoxifen group.
|
|
Hazard Ratio
|
p-value
|
|
Disease-free survival |
0.69 (0.58–0.82) |
0.00003 |
|
Time to contralateral breast cancer |
0.32 (0.15–0.72) |
0.00340 |
|
Distant recurrence-free survival |
0.74 (0.62–0.90) |
0.00207 |
|
Overall survival |
0.91 (0.81–1.04) |
0.16 Not adjusted for multiple testing.
|
|
ER and/or PgR positive |
||
|
Disease-free survival |
0.65 (0.53–0.79) |
0.00001 |
|
Time to contralateral breast cancer |
0.22 (0.08–0.57) |
0.00069 |
|
Distant recurrence-free survival |
0.73 (0.59–0.90) |
0.00367 |
|
Overall survival |
0.89 (0.78–1.02) |
0.09065 |
Figure 1. Disease-Free Survival in the IES Study of Postmenopausal Women with Early Breast Cancer (ITT Population)
1.2 Advanced Breast Cancer in Postmenopausal Women
EXEMESTANE is indicated for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen therapy [see Clinical Studies (14.2)].
5.3 Administration With Estrogen Containing Agents (5.3 Administration with Estrogen-Containing Agents)
EXEMESTANE should not be coadministered with systemic estrogen-containing agents as these could interfere with its pharmacologic action.
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
NDC 59762-2858-1
30 Tablets
GREENSTONE® BRAND
exemestane
tablets
25 mg
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A 2-year carcinogenicity study in mice at doses of 50, 150, and 450 mg/kg/day exemestane (gavage), resulted in an increased incidence of hepatocellular adenomas and/or carcinomas in both genders at the high dose level. Plasma AUC (0–24hr) at the high dose were 2575 ± 386 and 5667 ± 1833 ng.hr/mL in males and females (approx. 34 and 75 fold the AUC in postmenopausal patients at the recommended clinical dose). An increased incidence of renal tubular adenomas was observed in male mice at the high dose of 450 mg/kg/day. Since the doses tested in mice did not achieve an MTD, neoplastic findings in organs other than liver and kidneys remain unknown.
A separate carcinogenicity study was conducted in rats at the doses of 30, 100, and 315 mg/kg/day exemestane (gavage) for 92 weeks in males and 2 years in females. No evidence of carcinogenic activity up to the highest dose tested of 315 mg/kg/day was observed in females. The male rat study was inconclusive since it was terminated prematurely at Week 92. At the highest dose, plasma AUC(0–24hr) levels in male (1418 ± 287 ng.hr/mL) and female (2318 ± 1067 ng.hr/mL) rats were 19 and 31 fold higher than those measured in postmenopausal cancer patients receiving the recommended clinical dose.
Exemestane was not mutagenic in vitro in bacteria (Ames test) or mammalian cells (V79 Chinese hamster lung cells). Exemestane was clastogenic in human lymphocytes in vitro without metabolic activation but was not clastogenic in vivo (micronucleus assay in mouse bone marrow). Exemestane did not increase unscheduled DNA synthesis in rat hepatocytes when tested in vitro.
In a pilot reproductive study in rats, male rats were treated with doses of 125–1000 mg/kg/day exemestane, beginning 63 days prior to and during cohabitation. Untreated female rats showed reduced fertility when mated to males treated with ≥500 mg/kg/day exemestane (≥200 times the recommended human dose on a mg/m2 basis). In a separate study, exemestane was given to female rats at 4–100 mg/kg/day beginning 14 days prior to mating and through day 15 or 20 of gestation. Exemestane increased the placental weights at ≥4 mg/kg/day (≥1.5 times the human dose on a mg/m2 basis). Exemestane showed no effects on ovarian function, mating behavior, and conception rate in rats given doses up to 20 mg/kg/day (approximately 8 times the recommended human dose on a mg/m2 basis); however, decreases in mean litter size and fetal body weight, along with delayed ossification were evidenced at ≥20 mg/kg/day. In general toxicology studies, changes in the ovary, including hyperplasia, an increase in the incidence of ovarian cysts, and a decrease in corpora lutea were observed with variable frequency in mice, rats, and dogs at doses that ranged from 3–20 times the human dose on a mg/m2 basis.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:36.901168 · Updated: 2026-03-14T22:41:00.111917