Live Better
6915940c-2eae-445c-ba58-e39d221e9d3d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
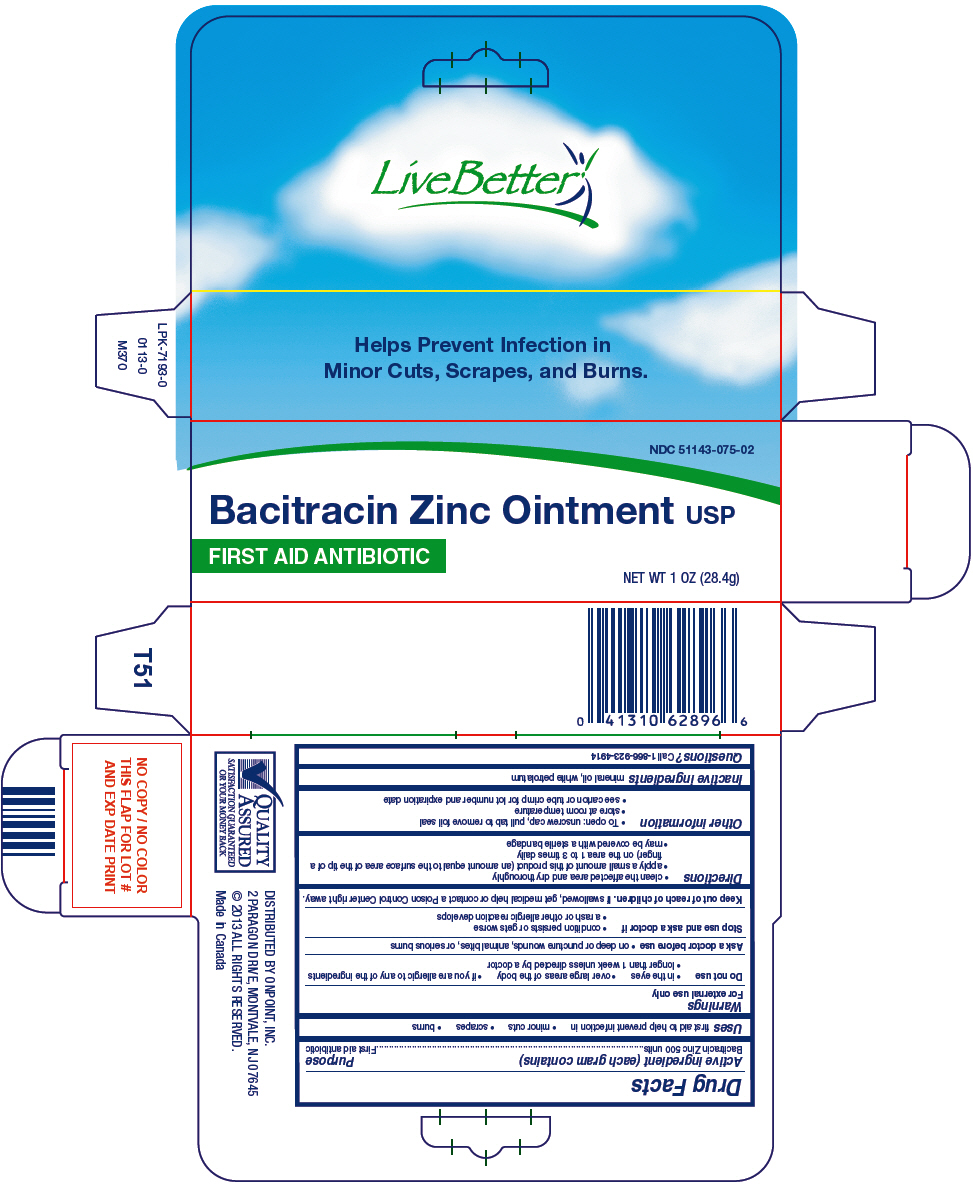
Bacitracin Zinc 500 units
Purpose
First aid antibiotic
Medication Information
Purpose
First aid antibiotic
Description
Bacitracin Zinc 500 units
Uses
first aid to help prevent infection in
- minor cuts
- scrapes
- burns
Section 42229-5
DISTRIBUTED BY ONPOINT, INC.
2 PARAGON DRIVE, MONTVALE, NJ 07645
Section 50565-1
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only
Directions
- clean the affected area and dry thoroughly
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Do Not Use
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
- longer than 1 week unless directed by a doctor
Questions?
Call 1-866-923-4914
Other Information
- To open: unscrew cap, pull tab to remove foil seal
- store at room temperature
- see carton or tube crimp for lot number and expiration date
Inactive Ingredients
mineral oil, white petrolatum
Ask A Doctor Before Use
- on deep or puncture wounds, animal bites, or serious burns
Stop Use and Ask A Doctor If
- condition persists or gets worse
- a rash or other allergic reaction develops
Active Ingredient (each Gram Contains)
Bacitracin Zinc 500 units
Principal Display Panel 28.4 G Tube Carton
NDC 51143-075-02
Bacitracin Zinc Ointment USP
FIRST AID ANTIBIOTIC
NET WT 1 OZ (28.4g)
Structured Label Content
Uses
first aid to help prevent infection in
- minor cuts
- scrapes
- burns
Section 42229-5 (42229-5)
DISTRIBUTED BY ONPOINT, INC.
2 PARAGON DRIVE, MONTVALE, NJ 07645
Section 50565-1 (50565-1)
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
First aid antibiotic
Warnings
For external use only
Directions
- clean the affected area and dry thoroughly
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Do Not Use (Do not use)
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
- longer than 1 week unless directed by a doctor
Questions?
Call 1-866-923-4914
Other Information (Other information)
- To open: unscrew cap, pull tab to remove foil seal
- store at room temperature
- see carton or tube crimp for lot number and expiration date
Inactive Ingredients (Inactive ingredients)
mineral oil, white petrolatum
Ask A Doctor Before Use (Ask a doctor before use)
- on deep or puncture wounds, animal bites, or serious burns
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition persists or gets worse
- a rash or other allergic reaction develops
Active Ingredient (each Gram Contains) (Active ingredient (each gram contains))
Bacitracin Zinc 500 units
Principal Display Panel 28.4 G Tube Carton (PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton)
NDC 51143-075-02
Bacitracin Zinc Ointment USP
FIRST AID ANTIBIOTIC
NET WT 1 OZ (28.4g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:30.598820 · Updated: 2026-03-14T23:09:19.585027