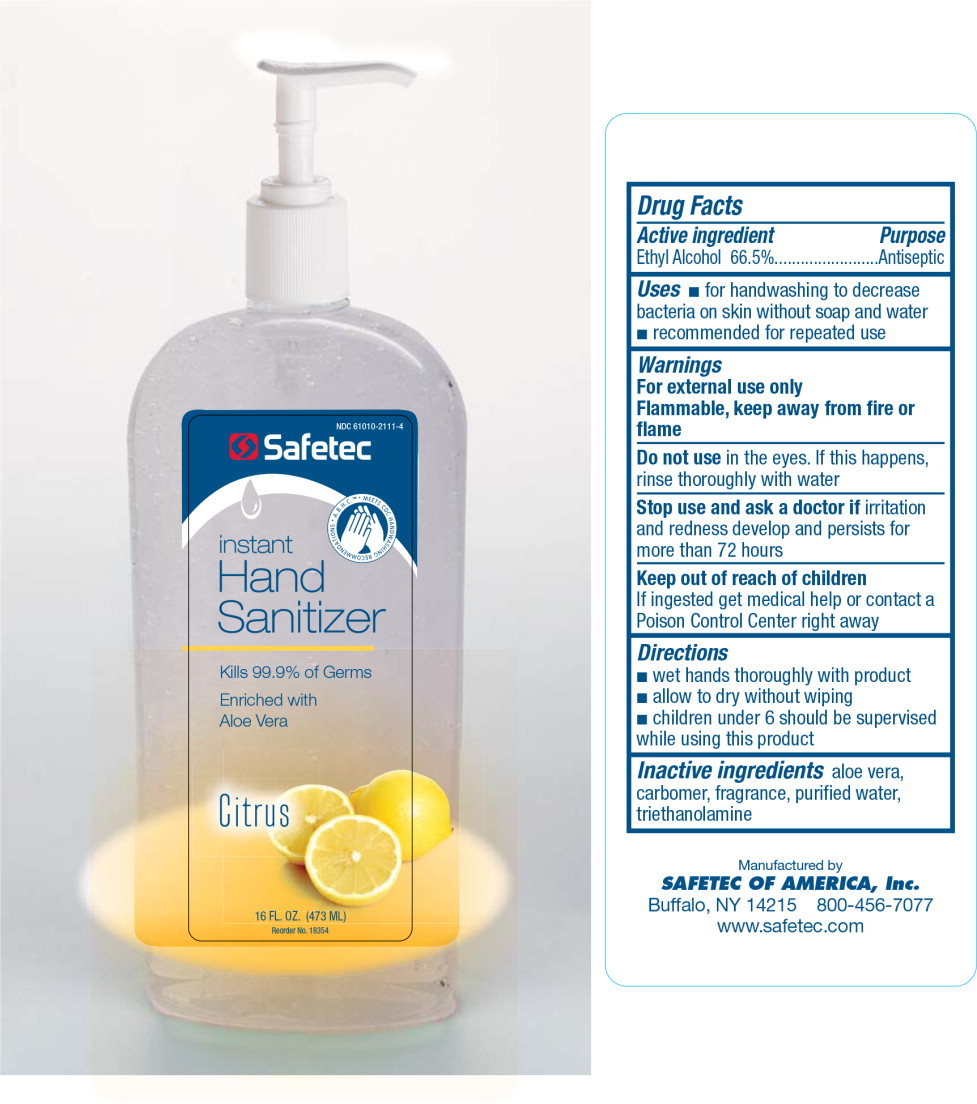
A.b.h.c. Instant Hand Sanitizer Citrus Scent (alcohol) Gel
691462b7-d118-4018-8985-5e32f38146d9
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Drug Facts
Uses
- for handwashing to decrease bacteria on skin without soap and water
- recommended for repeated use
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children If ingested get medical help or contact a Poison Control Center right away
Section 50566-9
Stop use and ask doctor if irritation and redness develop and persists for more than 72 hours
Section 50570-1
Do not use in the eyes. If this happens, rinse thoroughly with water
Warnings
For external use only.
Flammable, keep away from fire or flame
Directions
- wet hands and wrists thoroughly with product
- allow to dry without wiping
- children under 6 should be supervised while using this product
Active Ingredient
Ethyl Alcohol 66.5%
Inactive Ingredients
aloe vera, carbomer, fragrance, purified water, triethanolamine
Principal Display Panel – 16 Fl. Oz. Bottle Label
NDC 61010-2111-4
Safetec
Instant
Hand
Sanitizer
A.B.H.C. ™ MEETS CDC HANDWASHING RECOMMENDATIONS
Kills 99.9% of Germs
Enriched with
Aloe Vera
Citrus
16 FL. OZ. (473 ML)
Reorder No. 18354
Structured Label Content
Uses
- for handwashing to decrease bacteria on skin without soap and water
- recommended for repeated use
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children If ingested get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
Stop use and ask doctor if irritation and redness develop and persists for more than 72 hours
Section 50570-1 (50570-1)
Do not use in the eyes. If this happens, rinse thoroughly with water
Purpose
Antiseptic
Warnings
For external use only.
Flammable, keep away from fire or flame
Directions
- wet hands and wrists thoroughly with product
- allow to dry without wiping
- children under 6 should be supervised while using this product
Active Ingredient (Active ingredient)
Ethyl Alcohol 66.5%
Inactive Ingredients (Inactive ingredients)
aloe vera, carbomer, fragrance, purified water, triethanolamine
Principal Display Panel – 16 Fl. Oz. Bottle Label (Principal Display Panel – 16 Fl. OZ. Bottle Label)
NDC 61010-2111-4
Safetec
Instant
Hand
Sanitizer
A.B.H.C. ™ MEETS CDC HANDWASHING RECOMMENDATIONS
Kills 99.9% of Germs
Enriched with
Aloe Vera
Citrus
16 FL. OZ. (473 ML)
Reorder No. 18354
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:26.101853 · Updated: 2026-03-14T22:57:58.648528