These Highlights Do Not Include All The Information Needed To Use Xeljanz/xeljanz Xr Safely And Effectively. See Full Prescribing Information For Xeljanz/xeljanz Xr.
68e3d6b2-7838-4d2d-a417-09d919b43e13
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Boxed Warning 10/2025 Indications and Usage, Psoriatic Arthritis ( 1.2 ) 10/2025 Dosage and Administration, Recommended Dosage in Pediatric Patients 2 Years of Age and Older with Psoriatic Arthritis or Polyarticular Course Juvenile Idiopathic Arthritis ( 2.4 ) 10/2025
Indications and Usage
XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) are Janus kinase (JAK) inhibitors. XELJANZ tablets and XELJANZ XR are indicated for the treatment of adult patients with: • Moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers. • Active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers. • Active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers. • Moderately to severely active ulcerative colitis (UC), who have had an inadequate response or intolerance to one or more TNF blockers. XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with: • Active PsA, who have had an inadequate response or intolerance to one or more TNF blockers. • Active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers. Limitations of Use : • Use of XELJANZ/XELJANZ XR for RA, AS, PsA, or pcJIA in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended. ( 1.1 , 1.2 , 1.3 , 1.4 ) • Use of XELJANZ tablets and XELJANZ XR for UC in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended. ( 1.5 )
Dosage and Administration
Recommended Evaluations and Immunization Prior to Treatment Initiation • Prior to initiating XELJANZ/XELJANZ XR, consider performing an active and latent TB evaluation, viral hepatitis screening, a complete blood count, and updating immunizations. Avoid XELJANZ or XELJANZ XR initiation if absolute lymphocyte count <500 cells/mm 3 , an absolute neutrophil count (ANC) <1000 cells/mm 3 or hemoglobin <9 g/dL. ( 2.1 ) Important Administration Instructions • XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). ( 2.2) • Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider. ( 2.2 ) Recommended Dosage Adult Patients with RA, PsA or AS • XELJANZ tablets 5 mg twice daily or XELJANZ XR (extended-release tablets) 11 mg once daily. ( 2.3 ) Pediatric Patients 2 Years of Age and Older with PsA or pcJIA Who Weigh At Least 10 kg • XELJANZ (tablets or oral solution) 5 mg twice daily for those ≥40 kg or weight-based equivalent twice daily for those <40 kg. ( 2.4 ) Adult Patients with UC • Induction: XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed, continue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for a maximum of 16 weeks. Discontinue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. ( 2.5 ) • Maintenance: XELJANZ tablets 5 mg twice daily or XELJANZ XR 11 mg once daily. For patients with loss of response during maintenance treatment, XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily may be considered and limited to the shortest duration, with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. ( 2.5 ) Dosage in Patients with Renal Impairment or Hepatic Impairment • Use of XELJANZ (tablets and oral solution) or XELJANZ XR in patients with severe HI is not recommended. ( 2.3 , 2.4 , 2.5 , 8.7 ) • See full prescribing information (FPI) for recommended dosage in patients with moderate or severe RI or moderate HI. ( 2.3 , 2.4 , 2.5 , 8.6 , 8.7 ) Dosage Modification See the full prescribing information for dosage modification by indication for patients who concomitantly use CYP2C19 and/or CYP3A4 inhibitors and patients with lymphopenia, neutropenia, or anemia. ( 2.3 , 2.4 , 2.5 , 7 )
Warnings and Precautions
• Serious Infections : Avoid use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) during an active serious infection, including localized infections. ( 5.1 ) • Gastrointestinal Perforations : Promptly evaluate patients at increased risk for gastrointestinal perforation who present with new onset abdominal symptoms. ( 5.6 ) • Laboratory Monitoring : Recommended due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes and lipids. ( 5.8 ) • Vaccinations : Avoid use of live vaccines concurrently with XELJANZ or XELJANZ XR. ( 5.9 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Serious Infections [see Warnings and Precautions (5.1) ] • Increased Risk of Mortality [see Warnings and Precautions (5.2) ] • Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3) ] • Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4) ] • Thrombosis [see Warnings and Precautions (5.5) ] • Gastrointestinal Perforations [see Warnings and Precautions (5.6) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] • Laboratory Abnormalities [see Warnings and Precautions (5.8) ]
Drug Interactions
Table 7 includes drugs with clinically significant drug interactions when concomitantly used with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) and instructions for preventing or managing them. Table 7: Clinically Significant Interactions Affecting XELJANZ/XELJANZ XR When Concomitantly Used with Other Drugs Strong CYP3A4 Inhibitors (e.g., ketoconazole) Clinical Impact Increased exposure to tofacitinib Intervention Dosage modification of XELJANZ/XELJANZ XR is recommended [see Dosage and Administration (2) , Clinical Pharmacology, Figure 3 (12.3) ] Moderate CYP3A4 Inhibitors Concomitantly Used with Strong CYP2C19 Inhibitors (e.g., fluconazole) Clinical Impact Increased exposure to tofacitinib Intervention Dosage modification of XELJANZ/XELJANZ XR is recommended [see Dosage and Administration (2) , Clinical Pharmacology, Figure 3 (12.3) ] Strong CYP3A4 Inducers (e.g., rifampin) Clinical Impact Decreased exposure to tofacitinib and may result in loss of or reduced clinical response Intervention Concomitant use with XELJANZ/XELJANZ XR is not recommended [see Clinical Pharmacology, Figure 3 (12.3) ] Immunosuppressive Drugs (e.g., azathioprine, tacrolimus, cyclosporine) Clinical Impact Risk of added immunosuppression; concomitant use of XELJANZ/XELJANZ XR with biologic DMARDs or potent immunosuppressants has not been studied in patients with RA, PsA, AS, UC, or pcJIA. Intervention Concomitant use with XELJANZ/XELJANZ XR is not recommended [see Indications and Usage (1) , Clinical Pharmacology, Figure 3 (12.3) ]
Storage and Handling
How Supplied Information for XELJANZ Tablets and XELJANZ XR How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23. Table 23: How Supplied Information for XELJANZ Tablets and XELJANZ XR Dosage Form, Strength, and Description Bottle Size (number of tablets) NDC Number XELJANZ (tofacitinib) tablets, 5 mg White, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 5” on the other side 60 NDC 0069-1001-01 XELJANZ (tofacitinib) tablets, 10 mg Blue, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 10” on the other side 60 NDC 0069-1002-01 XELJANZ XR (tofacitinib) extended-release tablets, 11 mg Pink, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 11” printed on one side of the tablet 30 NDC 0069-0501-30 XELJANZ XR (tofacitinib) extended-release tablets, 22 mg Beige, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 22” printed on one side of the tablet 30 NDC 0069-0502-30 How Supplied Information for XELJANZ Oral Solution XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex. Storage and Handling for XELJANZ Tablets/XELJANZ XR Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature]. Do not repackage. Storage and Handling for XELJANZ Oral Solution Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature]. Store in the original bottle and carton to protect from light. Use contents of bottle within 60 days of opening. Discard unused oral solution after 60 days.
How Supplied
How Supplied Information for XELJANZ Tablets and XELJANZ XR How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23. Table 23: How Supplied Information for XELJANZ Tablets and XELJANZ XR Dosage Form, Strength, and Description Bottle Size (number of tablets) NDC Number XELJANZ (tofacitinib) tablets, 5 mg White, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 5” on the other side 60 NDC 0069-1001-01 XELJANZ (tofacitinib) tablets, 10 mg Blue, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 10” on the other side 60 NDC 0069-1002-01 XELJANZ XR (tofacitinib) extended-release tablets, 11 mg Pink, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 11” printed on one side of the tablet 30 NDC 0069-0501-30 XELJANZ XR (tofacitinib) extended-release tablets, 22 mg Beige, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 22” printed on one side of the tablet 30 NDC 0069-0502-30 How Supplied Information for XELJANZ Oral Solution XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex. Storage and Handling for XELJANZ Tablets/XELJANZ XR Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature]. Do not repackage. Storage and Handling for XELJANZ Oral Solution Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature]. Store in the original bottle and carton to protect from light. Use contents of bottle within 60 days of opening. Discard unused oral solution after 60 days.
Medication Information
Warnings and Precautions
• Serious Infections : Avoid use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) during an active serious infection, including localized infections. ( 5.1 ) • Gastrointestinal Perforations : Promptly evaluate patients at increased risk for gastrointestinal perforation who present with new onset abdominal symptoms. ( 5.6 ) • Laboratory Monitoring : Recommended due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes and lipids. ( 5.8 ) • Vaccinations : Avoid use of live vaccines concurrently with XELJANZ or XELJANZ XR. ( 5.9 )
Indications and Usage
XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) are Janus kinase (JAK) inhibitors. XELJANZ tablets and XELJANZ XR are indicated for the treatment of adult patients with: • Moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers. • Active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers. • Active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers. • Moderately to severely active ulcerative colitis (UC), who have had an inadequate response or intolerance to one or more TNF blockers. XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with: • Active PsA, who have had an inadequate response or intolerance to one or more TNF blockers. • Active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers. Limitations of Use : • Use of XELJANZ/XELJANZ XR for RA, AS, PsA, or pcJIA in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended. ( 1.1 , 1.2 , 1.3 , 1.4 ) • Use of XELJANZ tablets and XELJANZ XR for UC in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended. ( 1.5 )
Dosage and Administration
Recommended Evaluations and Immunization Prior to Treatment Initiation • Prior to initiating XELJANZ/XELJANZ XR, consider performing an active and latent TB evaluation, viral hepatitis screening, a complete blood count, and updating immunizations. Avoid XELJANZ or XELJANZ XR initiation if absolute lymphocyte count <500 cells/mm 3 , an absolute neutrophil count (ANC) <1000 cells/mm 3 or hemoglobin <9 g/dL. ( 2.1 ) Important Administration Instructions • XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). ( 2.2) • Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider. ( 2.2 ) Recommended Dosage Adult Patients with RA, PsA or AS • XELJANZ tablets 5 mg twice daily or XELJANZ XR (extended-release tablets) 11 mg once daily. ( 2.3 ) Pediatric Patients 2 Years of Age and Older with PsA or pcJIA Who Weigh At Least 10 kg • XELJANZ (tablets or oral solution) 5 mg twice daily for those ≥40 kg or weight-based equivalent twice daily for those <40 kg. ( 2.4 ) Adult Patients with UC • Induction: XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed, continue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for a maximum of 16 weeks. Discontinue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. ( 2.5 ) • Maintenance: XELJANZ tablets 5 mg twice daily or XELJANZ XR 11 mg once daily. For patients with loss of response during maintenance treatment, XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily may be considered and limited to the shortest duration, with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. ( 2.5 ) Dosage in Patients with Renal Impairment or Hepatic Impairment • Use of XELJANZ (tablets and oral solution) or XELJANZ XR in patients with severe HI is not recommended. ( 2.3 , 2.4 , 2.5 , 8.7 ) • See full prescribing information (FPI) for recommended dosage in patients with moderate or severe RI or moderate HI. ( 2.3 , 2.4 , 2.5 , 8.6 , 8.7 ) Dosage Modification See the full prescribing information for dosage modification by indication for patients who concomitantly use CYP2C19 and/or CYP3A4 inhibitors and patients with lymphopenia, neutropenia, or anemia. ( 2.3 , 2.4 , 2.5 , 7 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Serious Infections [see Warnings and Precautions (5.1) ] • Increased Risk of Mortality [see Warnings and Precautions (5.2) ] • Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3) ] • Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4) ] • Thrombosis [see Warnings and Precautions (5.5) ] • Gastrointestinal Perforations [see Warnings and Precautions (5.6) ] • Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] • Laboratory Abnormalities [see Warnings and Precautions (5.8) ]
Drug Interactions
Table 7 includes drugs with clinically significant drug interactions when concomitantly used with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) and instructions for preventing or managing them. Table 7: Clinically Significant Interactions Affecting XELJANZ/XELJANZ XR When Concomitantly Used with Other Drugs Strong CYP3A4 Inhibitors (e.g., ketoconazole) Clinical Impact Increased exposure to tofacitinib Intervention Dosage modification of XELJANZ/XELJANZ XR is recommended [see Dosage and Administration (2) , Clinical Pharmacology, Figure 3 (12.3) ] Moderate CYP3A4 Inhibitors Concomitantly Used with Strong CYP2C19 Inhibitors (e.g., fluconazole) Clinical Impact Increased exposure to tofacitinib Intervention Dosage modification of XELJANZ/XELJANZ XR is recommended [see Dosage and Administration (2) , Clinical Pharmacology, Figure 3 (12.3) ] Strong CYP3A4 Inducers (e.g., rifampin) Clinical Impact Decreased exposure to tofacitinib and may result in loss of or reduced clinical response Intervention Concomitant use with XELJANZ/XELJANZ XR is not recommended [see Clinical Pharmacology, Figure 3 (12.3) ] Immunosuppressive Drugs (e.g., azathioprine, tacrolimus, cyclosporine) Clinical Impact Risk of added immunosuppression; concomitant use of XELJANZ/XELJANZ XR with biologic DMARDs or potent immunosuppressants has not been studied in patients with RA, PsA, AS, UC, or pcJIA. Intervention Concomitant use with XELJANZ/XELJANZ XR is not recommended [see Indications and Usage (1) , Clinical Pharmacology, Figure 3 (12.3) ]
Storage and Handling
How Supplied Information for XELJANZ Tablets and XELJANZ XR How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23. Table 23: How Supplied Information for XELJANZ Tablets and XELJANZ XR Dosage Form, Strength, and Description Bottle Size (number of tablets) NDC Number XELJANZ (tofacitinib) tablets, 5 mg White, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 5” on the other side 60 NDC 0069-1001-01 XELJANZ (tofacitinib) tablets, 10 mg Blue, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 10” on the other side 60 NDC 0069-1002-01 XELJANZ XR (tofacitinib) extended-release tablets, 11 mg Pink, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 11” printed on one side of the tablet 30 NDC 0069-0501-30 XELJANZ XR (tofacitinib) extended-release tablets, 22 mg Beige, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 22” printed on one side of the tablet 30 NDC 0069-0502-30 How Supplied Information for XELJANZ Oral Solution XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex. Storage and Handling for XELJANZ Tablets/XELJANZ XR Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature]. Do not repackage. Storage and Handling for XELJANZ Oral Solution Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature]. Store in the original bottle and carton to protect from light. Use contents of bottle within 60 days of opening. Discard unused oral solution after 60 days.
How Supplied
How Supplied Information for XELJANZ Tablets and XELJANZ XR How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23. Table 23: How Supplied Information for XELJANZ Tablets and XELJANZ XR Dosage Form, Strength, and Description Bottle Size (number of tablets) NDC Number XELJANZ (tofacitinib) tablets, 5 mg White, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 5” on the other side 60 NDC 0069-1001-01 XELJANZ (tofacitinib) tablets, 10 mg Blue, round, immediate-release film-coated tablets, debossed with “Pfizer” on one side, and “JKI 10” on the other side 60 NDC 0069-1002-01 XELJANZ XR (tofacitinib) extended-release tablets, 11 mg Pink, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 11” printed on one side of the tablet 30 NDC 0069-0501-30 XELJANZ XR (tofacitinib) extended-release tablets, 22 mg Beige, oval, extended-release film-coated tablets with a drilled hole at one end of the tablet band and “JKI 22” printed on one side of the tablet 30 NDC 0069-0502-30 How Supplied Information for XELJANZ Oral Solution XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex. Storage and Handling for XELJANZ Tablets/XELJANZ XR Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature]. Do not repackage. Storage and Handling for XELJANZ Oral Solution Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature]. Store in the original bottle and carton to protect from light. Use contents of bottle within 60 days of opening. Discard unused oral solution after 60 days.
Description
Boxed Warning 10/2025 Indications and Usage, Psoriatic Arthritis ( 1.2 ) 10/2025 Dosage and Administration, Recommended Dosage in Pediatric Patients 2 Years of Age and Older with Psoriatic Arthritis or Polyarticular Course Juvenile Idiopathic Arthritis ( 2.4 ) 10/2025
Section 42229-5
SERIOUS INFECTIONS
Patients treated with XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets) are at increased risk for developing serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), that may lead to hospitalization or death [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids. Reported infections included:
-
•Active TB, which may present with pulmonary or extrapulmonary disease. Patients should be tested for latent TB before XELJANZ/XELJANZ XR use and during therapy. Treatment for latent infection should be initiated prior to XELJANZ/XELJANZ XR use.
-
•Invasive fungal infections, including cryptococcosis and pneumocystosis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral, including herpes zoster, and other infections due to opportunistic pathogens.
The risks and benefits of XELJANZ/XELJANZ XR treatment should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after XELJANZ/XELJANZ XR treatment, including the possible development of TB in patients who tested negative for latent TB infection prior to initiating therapy. If a serious infection develops, interrupt XELJANZ/XELJANZ XR until the infection is controlled [see Warnings and Precautions (5.1)].
Section 42231-1
|
MEDICATION GUIDE |
||
|
XELJANZ (ZEL' JANS')
|
XELJANZ XR (ZEL' JANS' EKS-AHR)
|
XELJANZ (ZEL' JANS')
|
|
What is the most important information I should know about XELJANZ/XELJANZ XR/XELJANZ oral solution?
|
||
|
1. |
Serious infections. XELJANZ/XELJANZ XR/XELJANZ oral solution are medicines that affect your immune system. XELJANZ/XELJANZ XR/XELJANZ oral solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR/XELJANZ oral solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
You should not start taking XELJANZ/XELJANZ XR/XELJANZ oral solution if you have any kind of infection unless your healthcare provider tells you it is okay. You may be at a higher risk of developing shingles (herpes zoster).
|
|
|
|
|
After starting XELJANZ/XELJANZ XR/XELJANZ oral solution, call your healthcare provider right away if you have any symptoms of an infection. XELJANZ/XELJANZ XR/XELJANZ oral solution can make you more likely to get infections or make worse any infection that you have. |
||
|
2. |
Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg or 10 mg twice daily. |
|
|
3. |
Cancer and immune system problems. XELJANZ/XELJANZ XR/XELJANZ oral solution may increase your risk of certain cancers by changing the way your immune system works.
|
|
|
4. |
Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg or 10 mg twice daily, especially if you are a current or past smoker.
|
|
|
5. |
Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking XELJANZ 5 mg or 10 mg twice daily. Blood clots in the lungs have also happened in people with ulcerative colitis. Some people have died from these blood clots.
|
|
|
6. |
Tears (perforation) in the stomach or intestines.
|
|
|
7. |
Allergic reactions.
|
|
|
8. |
Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking XELJANZ/XELJANZ XR/XELJANZ oral solution and while you take XELJANZ/XELJANZ XR/XELJANZ oral solution to check for the following side effects:
Your healthcare provider should routinely check certain liver tests. |
|
|
See " What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution? " for more information about side effects. |
||
|
What is XELJANZ/XELJANZ XR/XELJANZ oral solution?
It is not known if XELJANZ/XELJANZ XR/XELJANZ oral solution is safe and effective in people with Hepatitis B or C. |
||
|
What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR/XELJANZ oral solution?
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. XELJANZ/XELJANZ XR/XELJANZ oral solution and other medicines may affect each other causing side effects.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||
|
How should I take XELJANZ/XELJANZ XR/XELJANZ oral solution?
|
||
|
What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution? XELJANZ/XELJANZ XR/XELJANZ oral solution may cause serious side effects, including:
|
||
|
|
|
|
Common side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
Common side effects of XELJANZ/XELJANZ XR in people with ulcerative colitis include:
Common side effects of XELJANZ/XELJANZ oral solution in children with polyarticular course juvenile arthritis and psoriatic arthritis include:
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. |
||
|
How should I store XELJANZ/XELJANZ XR/XELJANZ oral solution?
Keep XELJANZ/XELJANZ XR/XELJANZ oral solution and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of XELJANZ/XELJANZ XR/XELJANZ oral solution.
|
||
|
What are the ingredients in XELJANZ 5 mg?
This Medication Guide may have been updated. For the most recent Medication Guide, please visit www.pfizer.com. LAB-0535-16.0 |
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: October 2025
Section 43683-2
10 Overdosage
There is no specific antidote for overdose with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). In case of an overdose, it is recommended that the patient be monitored for signs and symptoms of adverse reactions.
In a study in patients with end-stage renal disease (ESRD) undergoing hemodialysis, plasma tofacitinib concentrations declined more rapidly during the period of hemodialysis and dialyzer efficiency, calculated as dialyzer clearance/blood flow entering the dialyzer, was high [mean (SD) = 0.73 (0.15)]. However, due to the significant non-renal clearance of tofacitinib, the fraction of total elimination occurring by hemodialysis was small, and thus, limits the value of hemodialysis for treatment of overdose with XELJANZ/XELJANZ XR.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
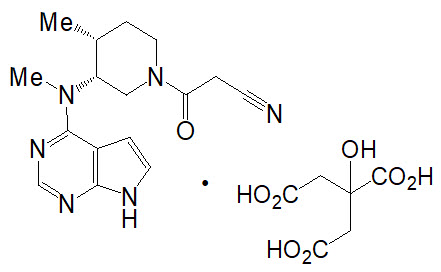
11 Description
XELJANZ (tofacitinib) tablets, XELJANZ XR (tofacitinib) extended-release tablets and XELJANZ (tofacitinib) oral solution are formulated with the citrate salt of tofacitinib, a JAK inhibitor.
Tofacitinib citrate is a white to off-white powder with the following chemical name: (3R,4R)-4-methyl-3-(methyl-7H-pyrrolo [2,3-d]pyrimidin-4-ylamino)-ß-oxo-1-piperidinepropanenitrile, 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
The solubility of tofacitinib citrate in water is 2.9 mg/mL.
Tofacitinib citrate has a molecular weight of 504.5 Daltons (or 312.4 Daltons as the tofacitinib free base) and a molecular formula of C16H20N6O•C6H8O7. The chemical structure of tofacitinib citrate is:
XELJANZ tablets is supplied for oral administration as a:
-
•5 mg white round, immediate-release film-coated tablet. Each tablet contains 5 mg of tofacitinib (equivalent to 8.08 mg of tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
-
•10 mg blue round, immediate-release film-coated tablet. Each tablet contains 10 mg of tofacitinib (equivalent to 16.16 mg of tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
XELJANZ XR is supplied for oral administration as a:
-
•11 mg pink, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each tablet contains 11 mg of tofacitinib (equivalent to 17.77 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide and triacetin. Printing ink contains, ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
-
•22 mg beige, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each tablet contains 22 mg of tofacitinib (equivalent to 35.54 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
XELJANZ oral solution is supplied for oral administration as a 1 mg/mL clear, colorless solution. Each 1 mL contains 1 mg of tofacitinib (equivalent to 1.62 mg of tofacitinib citrate) and the following inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
5.5 Thrombosis
Thrombosis may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Thrombosis, including pulmonary embolism (PE), deep venous thrombosis (DVT), and arterial thrombosis, have occurred in patients treated with XELJANZ and other Janus kinase (JAK) inhibitors used to treat inflammatory conditions. Many of these events were serious and some resulted in death [see Warnings and Precautions (5.2)].
Patients with RA 50 years of age and older with at least one cardiovascular risk factor treated with XELJANZ tablets 5 mg or 10 mg twice daily compared to TNF blockers in RA Safety Study 1 had an observed increase in incidence of these thrombotic events. The incidence rate of DVT per 100 patient-years was 0.28 for XELJANZ tablets 10 mg twice a day, 0.22 for XELJANZ tablets 5 mg twice a day, and 0.16 for TNF blockers. The incidence rate of PE per 100 patient-years was 0.49 for XELJANZ tablets 10 mg twice a day, 0.18 for XELJANZ tablets 5 mg twice a day, and 0.05 for TNF blockers [see Clinical Studies (14.6)].
XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
In a long-term extension study in patients with UC, five cases of pulmonary embolism were reported in patients taking XELJANZ 10 mg twice daily, including one death in a patient with advanced cancer.
Promptly evaluate patients with symptoms of thrombosis and discontinue XELJANZ/XELJANZ XR in patients with symptoms of thrombosis.
Avoid XELJANZ/XELJANZ XR in patients that may be at increased risk of thrombosis. For the treatment of UC, use XELJANZ tablets or XELJANZ XR at the lowest effective dose and for the shortest duration needed to achieve/maintain therapeutic response [see Dosage and Administration (2.5) ].
5.9 Vaccinations
Avoid use of live vaccines concurrently with XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets). Prior to initiating XELJANZ/XELJANZ XR therapy, update immunizations in agreement with current immunization guidelines. The interval between live vaccinations and initiation of XELJANZ/XELJANZ XR therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
8.4 Pediatric Use
The safety and effectiveness of XELJANZ (tablets and oral solution) in pediatric patients for indications, other than in patients with active pcJIA and PsA, have not been established.
The safety and effectiveness of XELJANZ have not been established in pediatric patients less than 2 years of age.
The safety and effectiveness of XELJANZ XR (extended-release tablets) in pediatric patients have not been established.
Polyarticular Course Juvenile Idiopathic Arthritis (pcJIA)
The safety and effectiveness of XELJANZ (tablets and oral solution) for the treatment of active pcJIA have been established in pediatric patients 2 years of age and older who have had an inadequate response or intolerance to one or more TNF blockers.
Use of XELJANZ for this indication is supported by evidence from adequate and well-controlled studies of XELJANZ tablets in adults with RA, pharmacokinetic (PK) data from adult patients with RA, and with additional safety, efficacy, and PK data from a clinical trial of XELJANZ in pediatric patients 2 years and older with active pcJIA (Study pcJIA-I) [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1, 14.4)].
Adverse reactions observed in pediatric patients with pcJIA who received XELJANZ were consistent with those reported in adults with RA [see Adverse Reactions (6.1)].
Psoriatic Arthritis
The safety and effectiveness of XELJANZ (tablets and oral solution) for the treatment of active PsA have been established in pediatric patients 2 years of age and older who have had an inadequate response or intolerance to one or more TNF blockers.
Use of XELJANZ for this indication is supported by evidence from well-controlled studies of XELJANZ tablets in adults with PsA, PK data from adults with PsA, and PK data from a clinical trial of XELJANZ in 225 pediatric patients with JIA, and safety data from 280 pediatric patients 2 years of age and older with JIA [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)]. Following administration of the recommended XELJANZ dosage in pediatric patients 2 years of age and older with PsA, tofacitinib plasma exposures are predicted to be comparable to those observed in adults with PsA based on population PK modeling and simulation [see Clinical Pharmacology (12.3)].
Systemic Juvenile Idiopathic Arthritis
The safety and effectiveness of XELJANZ for the treatment of pediatric patients with systemic juvenile idiopathic arthritis (sJIA) have not been established.
The results from a two-part study (an open-label, run-in phase, followed by a double-blind, placebo-controlled, randomized event-driven withdrawal phase) in 100 patients 2 years to 17 years of age with sJIA with active systemic features did not demonstrate that XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) was efficacious in the treatment of sJIA with active systemic features.
Of the 100 patients enrolled in the open-label run-in phase, 59 (59%) patients achieved a clinical response and were eligible for the double-blind withdrawal phase. There were 28 patients randomized to XELJANZ and 31 patients to placebo. The study data were insufficient to demonstrate efficacy and, therefore, XELJANZ is not recommended for the treatment of sJIA.
Adverse reactions observed in pediatric patients with sJIA receiving XELJANZ/XELJANZ oral solution were consistent with those reported in pcJIA and RA patients [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the 3315 adults who were enrolled in clinical trials with RA (Studies RA-I to V), a total of 505 patients were 65 years of age and older, including 71 patients 75 years and older. The frequency of serious infection among XELJANZ tablets-treated patients 65 years of age and older was higher than among those adults under the age of 65.
Of the 1156 XELJANZ tablet-treated patients in clinical trials of patients with UC, a total of 77 patients (7%) were 65 years of age or older. Clinical studies of XELJANZ in patients with UC did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger adult patients.
Of the 783 XELJANZ tablet-treated patients in clinical trials of patients with PsA, a total of 72 (9.2%) patients were 65 years of age and older, including 2 (0.3%) patients 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with PsA to determine if they respond differently from younger adult patients.
Of the 420 XELJANZ tablet-treated patients in clinical trials of patients with AS, a total of 12 (2.9%) patients were 65 years of age and older, including 1 (0.2%) patient 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with AS to determine if they respond differently from younger adult patients.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Increased Risk of Mortality [see Warnings and Precautions (5.2)]
-
•Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3)]
-
•Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4)]
-
•Thrombosis [see Warnings and Precautions (5.5)]
-
•Gastrointestinal Perforations [see Warnings and Precautions (5.6)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
-
•Laboratory Abnormalities [see Warnings and Precautions (5.8)]
7 Drug Interactions
Table 7 includes drugs with clinically significant drug interactions when concomitantly used with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) and instructions for preventing or managing them.
|
Strong CYP3A4 Inhibitors (e.g., ketoconazole) |
|
|
Clinical Impact |
Increased exposure to tofacitinib |
|
Intervention |
Dosage modification of XELJANZ/XELJANZ XR is recommended |
|
Moderate CYP3A4 Inhibitors Concomitantly Used with Strong CYP2C19 Inhibitors (e.g., fluconazole) |
|
|
Clinical Impact |
Increased exposure to tofacitinib |
|
Intervention |
Dosage modification of XELJANZ/XELJANZ XR is recommended |
|
Strong CYP3A4 Inducers (e.g., rifampin) |
|
|
Clinical Impact |
Decreased exposure to tofacitinib and may result in loss of or reduced clinical response |
|
Intervention |
Concomitant use with XELJANZ/XELJANZ XR is not recommended |
|
Immunosuppressive Drugs (e.g., azathioprine, tacrolimus, cyclosporine) |
|
|
Clinical Impact |
Risk of added immunosuppression; concomitant use of XELJANZ/XELJANZ XR with biologic DMARDs or potent immunosuppressants has not been studied in patients with RA, PsA, AS, UC, or pcJIA. |
|
Intervention |
Concomitant use with XELJANZ/XELJANZ XR is not recommended |
8.6 Renal Impairment
Moderate and Severe Renal Impairment
XELJANZ-treated patients with moderate renal impairment (RI) (CLcr ≥30 and ≤50 mL/minute) or severe RI (<30 mL/minute) had greater tofacitinib blood concentrations than XELJANZ-treated patients with normal renal function (CLcr >80 mL/minute). The recommended dosage of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) in patients with moderate or severe RI (including those with severe RI who are undergoing hemodialysis) is lower than the recommended dosage in patients with normal renal function [see Dosage and Administration (2.3, 2.4, 2.5)].
Mild Renal Impairment
The recommended dosage in patients with mild RI (CLcr >50 and ≤80 mL/minute) is the same as patients with normal renal function.
Instructions for Use
XELJANZ
®
(ZEL' JANS')
(tofacitinib)
oral solution
Read this Instructions for Use before you start taking XELJANZ oral solution and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring XELJANZ oral solution:
Always use the oral dosing syringe that comes with XELJANZ oral solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store XELJANZ?
-
•Store XELJANZ oral solution at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Always store XELJANZ oral solution in the original bottle and carton to protect from light.
Keep XELJANZ and all medicines out of the reach of children.
Use XELJANZ oral solution within 60 days of opening the bottle. Throw away (discard) remaining XELJANZ oral solution after 60 days.
To help you remember when to throw away your bottle of XELJANZ oral solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
Each carton of XELJANZ oral solution contains:
-
•1 press-in bottle adapter
-
•1 bottle of XELJANZ oral solution
-
•1 oral dosing syringe
Step 1. Remove bottle from carton
Open the carton and remove the bottle of XELJANZ oral solution.
Step 2. Open bottle
Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)
Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe
Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe
Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle
With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe
Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose
Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of XELJANZ
Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle
Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect XELJANZ oral solution from light.
Step 11. Clean oral dosing syringe
Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the XELJANZ oral solution.
Do not throw away the oral dosing syringe.
LAB-1422-3.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: October 2025
This Instructions for Use may have been updated. For the most recent Instructions for Use, please visit www.pfizer.com.
12.2 Pharmacodynamics
Treatment with XELJANZ was associated with dose-dependent reductions of circulating CD16/56+ natural killer cells, with estimated maximum reductions occurring at approximately 8-10 weeks after initiation of therapy. These changes generally resolved within 2-6 weeks after discontinuation of treatment. Treatment with XELJANZ was associated with dose-dependent increases in B cell counts. Changes in circulating T-lymphocyte counts and T-lymphocyte subsets (CD3+, CD4+ and CD8+) were small and inconsistent. The clinical significance of these changes is unknown.
Total serum IgG, IgM, and IgA levels after 6-month dosing in patients with rheumatoid arthritis (RA) were lower than in patients who received placebo; however, changes were small and not dose-dependent.
After treatment with XELJANZ in patients with RA, rapid decreases in serum C-reactive protein (CRP) were observed and maintained throughout dosing. Changes in CRP observed with XELJANZ treatment do not reverse fully within 2 weeks after discontinuation, indicating a longer duration of pharmacodynamic activity compared to the pharmacokinetic half-life.
Similar changes in T cells, B cells, and serum CRP have been observed in patients with active psoriatic arthritis (PsA) although reversibility was not assessed. Total serum immunoglobulins were not assessed in patients with active PsA.
1.5 Ulcerative Colitis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with moderately to severely active ulcerative colitis (UC), who have an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
5.1 Serious Infections
Serious and sometimes fatal infections may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, or other opportunistic pathogens have been reported in patients receiving XELJANZ. The most common serious infections reported with XELJANZ included pneumonia, cellulitis, herpes zoster, urinary tract infection, diverticulitis, and appendicitis. Among opportunistic infections, tuberculosis and other mycobacterial infections, cryptococcosis, histoplasmosis, esophageal candidiasis, pneumocystosis, multi-dermatomal herpes zoster, cytomegalovirus infections, BK virus infection, and listeriosis were reported with XELJANZ. Some patients have presented with disseminated rather than localized disease, and were often taking concomitant immunomodulating agents such as methotrexate or corticosteroids.
In the UC population, treatment with XELJANZ tablets 10 mg twice daily was associated with greater risk of serious infections compared to 5 mg twice daily. Additionally, opportunistic herpes zoster infections (including meningoencephalitis, ophthalmologic, and disseminated cutaneous) were seen in patients who were treated with XELJANZ tablets 10 mg twice daily.
Other serious infections that were not reported in clinical studies may also occur (e.g., coccidioidomycosis).
Avoid use of XELJANZ/XELJANZ XR in patients with an active, serious infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating XELJANZ/XELJANZ XR in patients:
-
•with chronic or recurrent infection
-
•who have been exposed to tuberculosis
-
•with a history of a serious or an opportunistic infection
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
-
•with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with XELJANZ/XELJANZ XR. Interrupt XELJANZ/XELJANZ XR if a patient develops a serious infection, an opportunistic infection, or sepsis. In patients who develop a new infection during treatment with XELJANZ/XELJANZ XR, promptly complete diagnostic testing appropriate for an immunocompromised patient; initiate appropriate antimicrobial therapy, and monitor the patients closely.
Caution is also recommended in patients with a history of chronic lung disease, or in those who develop interstitial lung disease, as they may be more prone to infections.
Risk of infection may be higher with increasing degrees of lymphopenia and consideration should be given to lymphocyte counts when assessing individual patient risk of infection. Discontinuation and monitoring criteria for lymphopenia are recommended [see Dosage and Administration (2.3 , 2.4 , 2.5) ].
1 Indications and Usage
XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) are Janus kinase (JAK) inhibitors.
XELJANZ tablets and XELJANZ XR are indicated for the treatment of adult patients with:
-
•Moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Moderately to severely active ulcerative colitis (UC), who have had an inadequate response or intolerance to one or more TNF blockers.
XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with:
-
•Active PsA, who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use:
-
•Use of XELJANZ/XELJANZ XR for RA, AS, PsA, or pcJIA in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended. (1.1, 1.2, 1.3, 1.4)
-
•Use of XELJANZ tablets and XELJANZ XR for UC in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended. (1.5)
1.2 Psoriatic Arthritis
XELJANZ (tablets and oral solution) is indicated for the treatment of adult and pediatric patients 2 years of age and older with active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers.
XELJANZ XR (extended-release tablets) is indicated for the treatment of adults with active PsA who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ or XELJANZ XR in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
1.1 Rheumatoid Arthritis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biologic disease-modifying antirheumatic drugs (DMARDs) or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
12.1 Mechanism of Action
Tofacitinib is a Janus kinase (JAK) inhibitor. JAKs are intracellular enzymes which transmit signals arising from cytokine or growth factor-receptor interactions on the cellular membrane to influence cellular processes of hematopoiesis and immune cell function. Within the signaling pathway, JAKs phosphorylate and activate Signal Transducers and Activators of Transcription (STATs) which modulate intracellular activity including gene expression. Tofacitinib modulates the signaling pathway at the point of JAKs, preventing the phosphorylation and activation of STATs. JAK enzymes transmit cytokine signaling through pairing of JAKs (e.g., JAK1/JAK3, JAK1/JAK2, JAK1/TyK2, JAK2/JAK2). Tofacitinib inhibited the in vitro activities of JAK1/JAK2, JAK1/JAK3, and JAK2/JAK2 combinations with IC50 of 406, 56, and 1377 nM, respectively. However, the relevance of specific JAK combinations to therapeutic effectiveness is not known.
1.3 Ankylosing Spondylitis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
5 Warnings and Precautions
-
•Serious Infections: Avoid use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) during an active serious infection, including localized infections. (5.1)
-
•Gastrointestinal Perforations: Promptly evaluate patients at increased risk for gastrointestinal perforation who present with new onset abdominal symptoms. (5.6)
-
•Laboratory Monitoring: Recommended due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes and lipids. (5.8)
-
•Vaccinations: Avoid use of live vaccines concurrently with XELJANZ or XELJANZ XR. (5.9)
2 Dosage and Administration
Recommended Evaluations and Immunization Prior to Treatment Initiation
-
•Prior to initiating XELJANZ/XELJANZ XR, consider performing an active and latent TB evaluation, viral hepatitis screening, a complete blood count, and updating immunizations. Avoid XELJANZ or XELJANZ XR initiation if absolute lymphocyte count <500 cells/mm3, an absolute neutrophil count (ANC) <1000 cells/mm3 or hemoglobin <9 g/dL. (2.1)
Important Administration Instructions
-
•XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). (2.2)
-
•Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider. (2.2)
Recommended Dosage
Adult Patients with RA, PsA or AS
-
•XELJANZ tablets 5 mg twice daily or XELJANZ XR (extended-release tablets) 11 mg once daily. (2.3)
Pediatric Patients 2 Years of Age and Older with PsA or pcJIA Who Weigh At Least 10 kg
-
•XELJANZ (tablets or oral solution) 5 mg twice daily for those ≥40 kg or weight-based equivalent twice daily for those <40 kg. (2.4)
Adult Patients with UC
-
•Induction: XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed, continue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for a maximum of 16 weeks. Discontinue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. (2.5)
-
•Maintenance: XELJANZ tablets 5 mg twice daily or XELJANZ XR 11 mg once daily. For patients with loss of response during maintenance treatment, XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily may be considered and limited to the shortest duration, with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. (2.5)
Dosage in Patients with Renal Impairment or Hepatic Impairment
-
•Use of XELJANZ (tablets and oral solution) or XELJANZ XR in patients with severe HI is not recommended. (2.3, 2.4, 2.5, 8.7)
-
•See full prescribing information (FPI) for recommended dosage in patients with moderate or severe RI or moderate HI. (2.3, 2.4, 2.5, 8.6, 8.7)
Dosage Modification
See the full prescribing information for dosage modification by indication for patients who concomitantly use CYP2C19 and/or CYP3A4 inhibitors and patients with lymphopenia, neutropenia, or anemia. (2.3, 2.4, 2.5, 7)
3 Dosage Forms and Strengths
5.8 Laboratory Abnormalities
Laboratory abnormalities may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Drug hypersensitivity (events such as angioedema and urticaria have been observed)
Skin and subcutaneous tissue disorders: Acne
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
5.7 Hypersensitivity Reactions
Hypersensitivity reactions may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Reactions such as angioedema and urticaria that may reflect drug hypersensitivity have been observed in patients receiving XELJANZ/XELJANZ XR. Some events were serious. If a serious hypersensitivity reaction occurs, promptly discontinue XELJANZ/XELJANZ XR while evaluating the potential cause or causes of the reaction [see Adverse Reactions (6.2)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
The clinical studies described in this subsection were conducted using XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) and/or XELJANZ oral solution.
5.2 Increased Risk of Mortality
Increased risk of mortality may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Adult patients with rheumatoid arthritis (RA), 50 years of age and older, with at least one cardiovascular risk factor treated with XELJANZ tablets 5 mg or 10 mg twice a day had a higher observed rate of all-cause mortality, including sudden cardiovascular death, compared to those treated with TNF blockers in a large, randomized, postmarketing safety study (RA Safety Study 1). The incidence rate of all-cause mortality per 100 patient-years was 1.23 for XELJANZ tablets 10 mg twice a day, 0.88 for XELJANZ tablets 5 mg twice a day, and 0.69 for TNF blockers [see Clinical Studies (14.6)]. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR.
XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
For the treatment of UC, use XELJANZ/XELJANZ XR at the lowest effective dose and for the shortest duration needed to achieve/maintain therapeutic response [see Dosage and Administration (2.5) ].
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
5.6 Gastrointestinal Perforations
Gastrointestinal perforations may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Events of gastrointestinal perforation have been reported in clinical studies with XELJANZ tablets, although the role of JAK inhibition in these events is not known. In these studies, many patients with RA received background therapy with nonsteroidal anti-inflammatory drugs (NSAIDs).
There was no discernable difference in frequency of gastrointestinal perforation between the placebo and the XELJANZ tablets treatment groups in clinical trials of patients with UC, and many of them were receiving background corticosteroids.
Promptly evaluate patients treated with XELJANZ/XELJANZ XR who may be at increased risk for gastrointestinal perforation (e.g., patients with a history of diverticulitis or taking NSAIDs) and who present with new onset abdominal symptoms for early identification of gastrointestinal perforation [see Adverse Reactions (6.1)].
16 How Supplied/storage and Handling
How Supplied Information for XELJANZ Tablets and XELJANZ XR
How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23.
| Dosage Form, Strength, and Description |
Bottle Size
(number of tablets) |
NDC Number |
|---|---|---|
|
XELJANZ (tofacitinib) tablets, |
60 |
NDC 0069-1001-01 |
|
XELJANZ (tofacitinib) tablets, 10 mg
|
60 |
NDC 0069-1002-01 |
|
XELJANZ XR (tofacitinib) extended-release tablets, 11 mg
|
30 |
NDC 0069-0501-30 |
|
XELJANZ XR (tofacitinib) extended-release tablets, 22 mg
|
30 |
NDC 0069-0502-30 |
How Supplied Information for XELJANZ Oral Solution
XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex.
Storage and Handling for XELJANZ Tablets/XELJANZ XR
Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature].
Do not repackage.
Storage and Handling for XELJANZ Oral Solution
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature].
Store in the original bottle and carton to protect from light.
Use contents of bottle within 60 days of opening.
Discard unused oral solution after 60 days.
5.4 Major Adverse Cardiovascular Events
Major adverse cardiovascular events may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). In RA Safety Study 1, patients with RA who were 50 years of age and older with at least one cardiovascular risk factor and treated with XELJANZ tablets 5 mg or 10 mg twice daily had a higher rate of major adverse cardiovascular events (MACE) defined as cardiovascular death, non-fatal myocardial infarction (MI), and non-fatal stroke, compared to those treated with TNF blockers. The incidence rate of MACE per 100 patient-years was 1.11 for XELJANZ tablets 10 mg twice a day, 0.91 for XELJANZ tablets 5 mg twice a day, and 0.79 for TNF blockers. The incidence rate of fatal or non-fatal myocardial infarction per 100 patient-years was 0.39 for XELJANZ tablets 10 mg twice a day, 0.36 for XELJANZ tablets 5 mg twice a day, and 0.2 for TNF blockers [see Clinical Studies (14.6)]. Patients who are current or past smokers are at additional increased risk.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Inform patients about the symptoms of serious cardiovascular events and the steps to take if they occur. Discontinue XELJANZ/XELJANZ XR in patients that have experienced a MI or stroke. XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
2.2 Important Administration Instructions
-
•XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider.
-
•Dose interruption is recommended for management of lymphopenia, neutropenia, and anemia [see Warnings and Precautions (5.8) and Adverse Reactions (6.1)].
-
•Interrupt use of XELJANZ/XELJANZ XR if a patient develops a serious infection until the infection is controlled [see Warnings and Precautions (5.1)].
-
•Take XELJANZ/XELJANZ XR with or without food [see Clinical Pharmacology (12.3)].
-
•Swallow XELJANZ XR whole and intact. Do not crush, split, or chew the extended-release tablets [see Clinical Pharmacology (12.3)].
14.2 Clinical Studies in Psoriatic Arthritis
The psoriatic arthritis (PsA) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included 2 multicenter, randomized, double-blind, placebo-controlled trials in 816 adults with active PsA (Studies PsA-I and PsA-II).
Trial Designs and Population
All patients had active PsA for at least 6 months based upon the Classification Criteria for Psoriatic Arthritis (CASPAR), at least 3 tender/painful joints and at least 3 swollen joints, and active plaque psoriasis. Patients randomized and treated across the 2 clinical trials represented different PsA subtypes at screening, including <5 joints or asymmetric involvement (21%), ≥5 joints involved (90%), distal interphalangeal (DIP) joint involvement (61%), arthritis mutilans (8%), and spondylitis (19%). Patients in these clinical trials had a diagnosis of PsA for a mean (SD) of 7.7 (7.2) years. At baseline, 80% and 53% of patients had enthesitis and dactylitis, respectively. At baseline, all patients were required to receive treatment with a stable dose of a nonbiologic DMARD (79% received methotrexate, 13% received sulfasalazine, 7% received leflunomide, 1% received other nonbiologic DMARDs). In both clinical trials, the primary endpoints were the ACR20 response and the change from baseline in HAQ-DI at Month 3.
-
•Study PsA-I was a 12-month clinical trial in 422 patients who had an inadequate response to a nonbiologic DMARD (67% and 33% were inadequate responders to 1 nonbiologic DMARD and ≥2 nonbiologic DMARDs, respectively) and who were naïve to treatment with a TNF blocker. Although Study PsA-1 included patients who are TNF blocker-naïve, XELJANZ and XELJANZ XR are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)]. Patients were randomized in a 2:2:2:1:1 ratio to receive XELJANZ 5 mg twice daily, XELJANZ 10 mg twice daily, adalimumab 40 mg subcutaneously once every 2 weeks, placebo to XELJANZ 5 mg twice daily treatment sequence, or placebo to XELJANZ 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a predetermined XELJANZ dosage of 5 mg or 10 mg twice daily. Study PsA-I was not designed to demonstrate noninferiority or superiority to adalimumab.
-
•Study PsA-II was a 6-month clinical trial in 394 patients who had an inadequate response to at least 1 approved TNF blocker (66%, 19%, and 15% were inadequate responders to 1 TNF blocker, 2 TNF blockers and ≥3 TNF blockers, respectively). Patients were randomized in a 2:2:1:1 ratio to receive XELJANZ 5 mg twice daily, XELJANZ 10 mg twice daily, placebo to XELJANZ 5 mg twice daily treatment sequence, or placebo to XELJANZ 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, placebo patients were switched in a blinded fashion to a predetermined XELJANZ dosage of 5 mg or 10 mg twice daily as in Study PsA-I.
Although other dosages have been studied, the recommended dosage of XELJANZ is 5 mg twice daily. XELJANZ 10 mg twice daily is not recommended for treatment of PsA [see Dosage and Administration (2.3)].
14.1 Clinical Studies in Rheumatoid Arthritis
The rheumatoid arthritis (RA) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included six randomized controlled trials in adults with moderate to severe active RA.
Trial Design
-
•Study RA-I (NCT00814307) was a 6-month monotherapy trial in which 610 patients with moderate to severe active RA who had an inadequate response to a DMARD (nonbiologic or biologic) received XELJANZ 5 mg or 10 mg twice daily or placebo added to their background DMARD. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 mg or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, changes in Health Assessment Questionnaire – Disability Index (HAQ-DI), and rates of Disease Activity Score DAS28-4(ESR) less than 2.6.
-
•Study RA-II (NCT00856544) was a 12-month trial in which 792 patients with moderate to severe active RA who had an inadequate response to a nonbiologic DMARD received XELJANZ 5 mg or 10 mg twice daily or placebo added to background DMARD treatment (excluding potent immunosuppressive treatments such as azathioprine or cyclosporine). At the Month 3 visit, nonresponding patients were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 mg or 10 mg twice daily. At the end of Month 6, all patients treated with placebo were switched to their second predetermined XELJANZ treatment in a blinded fashion. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, changes in HAQ-DI at Month 3, and rates of DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-III (NCT00853385) was a 12-month trial in 717 patients with moderate to severe active RA who had an inadequate response to methotrexate (MTX). Patients received XELJANZ 5 mg or 10 mg orally twice daily, adalimumab 40 mg subcutaneously every other week, or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-IV (NCT00847613) was a 2-year trial with a planned analysis at 1 year in which 797 patients with moderate to severe active RA who had an inadequate response to MTX received XELJANZ 5 mg or 10 mg twice daily or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, mean change from baseline in van der Heijde-modified total Sharp Score (mTSS) at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-V (NCT00960440) was a 6-month trial in which 399 patients with moderate to severe active RA who had an inadequate response to at least one approved TNF blocking biological product received XELJANZ 5 mg or 10 mg twice daily or placebo added to background MTX. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, HAQ-DI, and DAS28-4(ESR) less than 2.6.
-
•Study RA-VI (NCT01039688) was a 2-year monotherapy trial with a planned analysis at 1 year in which 952 MTX-naïve patients with moderate to severe active RA received XELJANZ 5 or 10 mg twice daily or MTX dose-titrated over 8 weeks to 20 mg weekly. The primary endpoints were mean change from baseline in van der Heijde-modified Total Sharp Score (mTSS) at Month 6 and the proportion of patients who achieved an ACR70 response at Month 6.
Although other dosages have been studied, the recommended dosage of XELJANZ is 5 mg twice daily. XELJANZ 10 mg twice daily is not recommended for the treatment of RA [see Dosage and Administration (2.3)].
Clinical Response
The percentages of XELJANZ-treated patients who achieved ACR20, ACR50, and ACR70 responses in Studies RA-I, IV, and V are shown in Table 10. Similar results were observed with Studies RA-II and III. In trials RA-I through V, patients treated with 5 mg twice daily XELJANZ had higher ACR20, ACR50, and ACR70 response rates versus patients treated with placebo, with or without background DMARD treatment, at Month 3 and Month 6. Higher ACR20 response rates were observed within 2 weeks compared to placebo. In the 12-month trials, ACR response rates in XELJANZ-treated patients were consistent at 6 and 12 months.
|
Monotherapy in Nonbiologic or Biologic DMARD Inadequate Responders Inadequate response to at least one DMARD (biologic or nonbiologic) due to lack of efficacy or toxicity.
|
MTX Inadequate Responders Inadequate response to MTX defined as the presence of sufficient residual disease activity to meet the entry criteria.
|
TNF Blocker Inadequate Responders Inadequate response to a least one TNF blocker due to lack of efficacy and/or intolerance.
|
||||
|
Study RA-I |
Study RA-IV |
Study RA-V |
||||
|
N N is number of randomized and treated patients.
|
Placebo + background DMARD |
XELJANZ 5 mg Twice Daily + background DMARD |
Placebo + background MTX |
XELJANZ 5 mg Twice Daily + background MTX |
Placebo + background MTX |
XELJANZ 5 mg Twice Daily + background MTX |
|
122 |
243 |
160 |
321 |
132 |
133 |
|
|
ACR20 Month 3 Month 6 |
26% NA NA (not applicable), as data for placebo treatment is not available beyond 3 months in Studies RA-I and RA-V due to placebo advancement.
|
59% 69% |
27% 25% |
55% 50% |
24% NA |
41% 51% |
|
ACR50 Month 3 Month 6 |
12% NA |
31% 42% |
8% 9% |
29% 32% |
8% NA |
26% 37% |
|
ACR70 Month 3 Month 6 |
6% NA |
15% 22% |
3% 1% |
11% 14% |
2% NA |
14% 16% |
In Study RA-IV, a greater proportion of patients treated with XELJANZ 5 mg twice daily plus MTX achieved a low level of disease activity as measured by a DAS28-4(ESR) less than 2.6 at 6 months compared to those treated with MTX alone (Table 11).
|
Study RA-IV |
||
|
DAS28-4(ESR) Less Than 2.6 |
Placebo + MTX 160 |
XELJANZ 5 mg Twice Daily + MTX 321 |
|
Proportion of responders at Month 6 (n) |
1% (2) |
6% (19) |
|
Of responders, proportion with 0 active joints (n) |
50% (1) |
42% (8) |
|
Of responders, proportion with 1 active joint (n) |
0 |
5% (1) |
|
Of responders, proportion with 2 active joints (n) |
0 |
32% (6) |
|
Of responders, proportion with 3 or more active joints (n) |
50% (1) |
21% (4) |
The results of the components of the ACR response criteria for Study RA-IV are shown in Table 12. Similar results were observed for XELJANZ in Studies RA-I, II, III, V, and VI.
|
Study RA-IV |
||||
|
Placebo + MTX N=160 |
XELJANZ 5 mg Twice Daily + MTX N=321 |
|||
|
Component (mean) Data shown is mean (Standard Deviation) at Month 3.
|
Baseline |
Month 3 |
Baseline |
Month 3 |
|
Number of tender joints (0-68) |
23 (13) |
18 (14) |
24 (14) |
13 (14) |
|
Number of swollen joints (0-66) |
14 (9) |
10 (9) |
14 (8) |
6 (8) |
|
Pain Visual analog scale: 0 = best, 100 = worst.
|
55 (24) |
47 (24) |
58 (23) |
34 (23) |
|
Patient global assessment |
54 (23) |
47 (24) |
58 (24) |
35 (23) |
|
Disability index (HAQ-DI) Health Assessment Questionnaire Disability Index: 0 = best, 3 = worst; 20 questions; categories: dressing and grooming, arising, eating, walking, hygiene, reach, grip, and activities.
|
1.32 (0.67) |
1.19 (0.68) |
1.41 (0.68) |
0.99 (0.65) |
|
Physician global assessment |
56 (18) |
43 (22) |
59 (16) |
30 (19) |
|
CRP (mg/L) |
13.7 (14.9) |
14.6 (18.7) |
15.3 (19.0) |
7.1 (19.1) |
The percent of ACR20 responders by visit for Study RA-IV is shown in Figure 4. Similar responses were observed for XELJANZ in Studies RA-I, II, III, V, and VI.
Figure 4: Percentage of ACR20 Responders by Visit Through Month 6 in Study RA-IV
Principal Display Panel 240 Ml Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
NDC 63539-029-01
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
Xeljanz®
(tofacitinib) oral solution
1 mg/mL
240 mL
Rx only
Principal Display Panel 240 Ml Bottle Carton
PROFESSIONAL SAMPLE – NOT FOR SALE
NDC 63539-029-02
ALWAYS DISPENSE WITH MEDICATION GUIDE
Pfizer
Xeljanz®
(tofacitinib) oral solution
1 mg/mL
Contents:
-
•Oral solution bottle
-
•1 Oral dosing syringe
-
•1 Press-in bottle adapter
-
•Prescribing Information
-
•Medication Guide
-
•Instructions for Use
240 mL
Rx only
14.3 Clinical Studies in Ankylosing Spondylitis
The ankylosing spondylitis (AS) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included one placebo-controlled trial (Study AS-I) in adults with active AS. Patients had active disease as defined by both Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and back pain score (BASDAI question 2) of greater or equal to 4 despite non-steroidal anti-inflammatory drug (NSAID), corticosteroid or disease modifying anti-rheumatic drug (DMARD) therapy.
5.3 Malignancy and Lymphoproliferative Disorders
Malignancies and lymphoproliferative disorders may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Malignancies, including lymphomas and solid cancers, were observed in clinical studies of XELJANZ [see Adverse Reactions (6.1)].
Other malignancies were observed in XELJANZ clinical studies and the postmarketing setting, including, but not limited to, lung cancer, breast cancer, melanoma, prostate cancer, and pancreatic cancer.
In RA Safety Study 1, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in patients treated with XELJANZ tablets 5 mg or 10 mg twice a day compared with TNF blockers. The incidence rate of malignancies (excluding NMSC) per 100 patient-years was 1.13 for XELJANZ tablets 10 mg twice a day, 1.13 for XELJANZ tablets 5 mg twice a day, and 0.77 for TNF blockers. Patients who are current or past smokers are at additional increased risk [see Clinical Studies (14.6)].
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with XELJANZ tablets 5 mg twice a day and XELJANZ tablets 10 mg twice a day compared to those treated with TNF blockers. The incidence rate of lymphomas per 100 patient-years was 0.11 for XELJANZ tablets 10 mg twice a day, 0.07 for XELJANZ tablets 5 mg twice a day, and 0.02 for TNF blockers. The incidence rate of lung cancers per 100 patient-years among current and past smokers was 0.59 for XELJANZ tablets 10 mg twice a day, 0.48 for XELJANZ tablets 5 mg twice a day, and 0.27 for TNF blockers [see Clinical Studies (14.6)].
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR, particularly in patients with a known malignancy (other than a successfully treated NMSC), patients who develop a malignancy while on treatment, and patients who are current or past smokers. XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
Principal Display Panel 5 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-012-02
Pfizer
Xeljanz
™
(tofacitinib tablets)
5 mg*
60 Tablets
Rx only
Principal Display Panel 10 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-016-02
Pfizer
Xeljanz
®
(tofacitinib) tablets
10 mg*
10mg Is Recommended Only In
Ulcerative Colitis
60 Tablets
Rx only
Principal Display Panel 11 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-501-30
Pfizer
Xeljanz
®
XR
(tofacitinib) tablets
Extended Release Tablets
11 mg*
30 Tablets
Rx only
Principal Display Panel 22 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
Pfizer
NDC 63539-502-30
Xeljanz® XR
(tofacitinib) tablets
Extended Release Tablets
22 mg*
22mg Is Recommended Only In
Ulcerative Colitis
30 Tablets
Rx only
1.4 Polyarticular Course Juvenile Idiopathic Arthritis
XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with of active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
2.5 Recommended Dosage in Adults With Ulcerative Colitis
Table 3 displays the recommended dosage of XELJANZ tablets and XELJANZ XR (extended-release tablets) in adult patients with ulcerative colitis (UC) [see Indications and Usage (1.5)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. Table 4 displays the recommended dosage modification for patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and patients with lymphopenia, neutropenia, or anemia.
| Adults |
XELJANZ
tablets |
XELJANZ XR
(extended-release tablets) |
|---|---|---|
|
Patients with Normal Renal and Hepatic Function |
Induction: 10 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 10 mg twice daily for a maximum of 16 weeks. Discontinue 10 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 22 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 22 mg once daily for a maximum of 16 weeks. Discontinue 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Maintenance: 5 mg twice daily. For patients with loss of response during maintenance treatment, may consider a dosage of 10 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: 11 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 22 mg once daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. |
|
|
Recommended Dosage in Patients with Renal Impairment (RI) Tofacitinib PK was evaluated in subjects with varying degrees of renal impairment, where the severity of renal impairment was defined based on creatinine clearance (CLcr) estimated using the Cockcroft‑Gault equation: CLcr >80 mL/min (normal renal function); CLcr >50 and ≤80 mL/min (mild renal impairment); ≥30 and ≤50 mL/min (moderate renal impairment); <30 mL/min (severe renal impairment).
|
||
|
Mild RI |
Same as patients with normal renal function. |
|
|
Moderate RI |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Severe RI |
Maintenance: 5 mg once daily.
|
Maintenance: XELJANZ XR is not recommended. See Maintenance Dosage for XELJANZ tablets for Moderate or Severe RI.
|
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
||
|
Mild HI |
Same as patients with normal hepatic function. |
|
|
Moderate HI |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Maintenance: 5 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 5 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: XELJANZ XR is not recommended. See Maintenance Dosage for XELJANZ tablets for Moderate HI. |
|
|
Severe HI |
Use of XELJANZ tablets/XELJANZ XR is not recommended. |
| Adults |
XELJANZ
Tablets |
XELJANZ XR
(extended-release tablets) |
|---|---|---|
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
||
|
Strong CYP2C19 inhibitor(s) |
No dosage modification is recommended. |
|
|
Moderate CYP2C19 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Strong CYP3A4 inhibitor(s) |
||
|
Maintenance: 5 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 5 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: XELJANZ XR is not recommended. see Maintenance Dosage for XELJANZ tablets for Strong CYP3A4 inhibitors. |
|
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
||
|
Lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
|
ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
|
ANC 500 to 1000 cells/mm3 |
If taking:
|
If taking:
|
|
Hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 39-week toxicology study in monkeys, tofacitinib at exposure levels approximately 6 times the recommended dose of 5 mg twice daily, and approximately 3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 5 mg/kg twice daily) produced lymphomas. No lymphomas were observed in this study at exposure levels 1 times the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg twice daily).
The carcinogenic potential of tofacitinib was assessed in 6-month rasH2 transgenic mouse carcinogenicity and 2-year rat carcinogenicity studies. Tofacitinib, at exposure levels approximately 34 times the recommended dose of 5 mg twice daily, and approximately 17 times the 10 mg twice daily dose (on an AUC basis at oral doses of 200 mg/kg/day) was not carcinogenic in mice.
In the 24-month oral carcinogenicity study in Sprague-Dawley rats, tofacitinib caused benign Leydig cell tumors, hibernomas (malignancy of brown adipose tissue), and benign thymomas at doses greater than or equal to 30 mg/kg/day (approximately 42 times the exposure levels at the recommended dose of 5 mg twice daily, and approximately 21 times the 10 mg twice daily dose on an AUC basis). The relevance of benign Leydig cell tumors to human risk is not known.
Tofacitinib was not mutagenic in the bacterial reverse mutation assay. It was positive for clastogenicity in the in vitro chromosome aberration assay with human lymphocytes in the presence of metabolic enzymes, but negative in the absence of metabolic enzymes. Tofacitinib was negative in the in vivo rat micronucleus assay and in the in vitro CHO-HGPRT assay and the in vivo rat hepatocyte unscheduled DNA synthesis assay.
In rats, tofacitinib at exposure levels approximately 17 times the recommended dose of 5 mg twice daily, and approximately 8.3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 10 mg/kg/day) reduced female fertility due to increased post-implantation loss. There was no impairment of female rat fertility at exposure levels of tofacitinib equal to the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg/day). Tofacitinib exposure levels at approximately 133 times the recommended dose of 5 mg twice daily, and approximately 67 times the 10 mg twice daily dose (on an AUC basis at oral doses of 100 mg/kg/day) had no effect on male fertility, sperm motility, or sperm concentration.
2.1 Recommended Evaluations and Immunization Prior to Treatment Initiation
Prior to initiating XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets), consider performing the following:
-
•Active and latent tuberculosis (TB) infection evaluation: If the patient has latent TB, treat for TB prior to XELJANZ/XELJANZ XR treatment [see Warnings and Precautions (5.1)].
-
•Viral hepatitis screening in accordance with clinical guidelines [see Warnings and Precautions (5.1)].
-
•A complete blood count: Avoid initiation of XELJANZ/XELJANZ XR treatment in patients with a lymphocyte count less than 500 cells/mm3, absolute neutrophil count less than 1000 cells/mm3, or hemoglobin level less than 9 g/dL [see Warnings and Precautions (5.8)].
-
•Baseline hepatic function evaluation: XELJANZ/XELJANZ XR is not recommended for patients with severe hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
-
•Update immunizations according to current immunization guidelines. The interval between live vaccinations and initiation of XELJANZ/XELJANZ XR should be in accordance with current vaccination guidelines regarding immunosuppressive agents [see Warnings and Precautions (5.9)].
14.4 Clinical Studies in Polyarticular Course Juvenile Idiopathic Arthritis
The efficacy of XELJANZ (tablets and oral solution) for pcJIA was assessed in Study pcJIA-I (NCT02592434). This was a 44-week, two-part study (that consisted of an 18-week, open-label, run-in phase, followed by a 26-week double-blind, placebo-controlled, randomized withdrawal phase) in pediatric patients 2 years to 17 years of age with active rheumatoid factor (RF) negative polyarthritis, RF positive polyarthritis, extended oligoarthritis, and systemic JIA without systemic manifestations who had an inadequate response or intolerance to at least one DMARD which could have included MTX or biologic agents. This study also included patients ages 2 years to 17 years of age with active juvenile psoriatic arthritis (jPsA) and enthesitis-related arthritis (ERA) who had an inadequate response to NSAIDs. Although the clinical studies included some patients who are TNF blocker-naïve, XELJANZ is not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.4)].
Patients received XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) for 18 weeks (run-in phase) followed by randomization to either XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) or placebo for 26 weeks (double-blind phase). Only patients who achieved at least a JIA ACR30 response at the end of the run-in phase were randomized (1:1) to the double-blind phase. Treatment with a stable dose of MTX was permitted but was not required during the study. Concurrent use of biologics or DMARDs other than MTX was not permitted in the study.
Baseline Disease Characteristics
A total of 225 pediatric patients with JIA (56 male and 169 female) with active polyarthritis were enrolled in the run-in phase including RF negative (104), RF positive (39), extended oligoarthritis (28), systemic JIA without systemic manifestations (13), jPsA (20), and ERA (21). Patients had a mean (SD) disease duration of 3.8 ± 3.5 years, and a mean (SD) number of active joints of 12.2 ± 8.1.
Efficacy Results
Of the 225 patients, 173 (77%) patients achieved JIA ACR30 response at Week 18 and were randomized into the double-blind phase to either XELJANZ (n=88) or placebo (n=85). At the conclusion of the 18-week, open-label, run-in phase, pediatric ACR 30/50/70 responses were 77%, 70%, and 49%, respectively.
In both the run-in and double-blind phases, approximately one-third of the patients were taking concomitant oral corticosteroids, and approximately two-thirds were taking concomitant MTX.
The primary endpoint was the occurrence of disease flare at Week 44 relative to the double-blind phase baseline at Week 18. Disease flare was defined (according to Pediatric Rheumatology Collaborative Study Group (PRCSG)/Pediatric Rheumatology International Trials Organization (PRINTO) Disease Flare criteria) as worsening of ≥30% in 3 or more of the 6 JIA core response variables with no more than 1 of the remaining JIA core response variables improving by ≥30%.
XELJANZ-treated patients experienced significantly fewer disease flares at Week 44 compared to placebo-treated patients (31% [27/88] vs. 55% [47/85]; difference in proportions -25% [95% CI: -39%, -10%]; p=0.0007). The occurrence of disease flare by visit in Study pcJIA-I is shown in Figure 7.
| BID = twice daily; SE = standard error; N = total number of patients. |
| The 26-week double-blind phase is from Week 18 through Week 44 on and after randomization day. |
14.6 Safety Study in Adults With Rheumatoid Arthritis (xeljanz Versus Tnf Blocker)
A randomized open-label trial (RA Safety Study 1; NCT02092467) was conducted to evaluate safety with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) at two doses, 5 mg twice daily (N=1455) and 10 mg twice daily (N=1456), versus the TNF-blocker control (N=1451) in RA patients 50 years of age and older with at least one cardiovascular risk factor. The co-primary endpoints were adjudicated MACE (defined as cardiovascular death, non-fatal MI, and non-fatal stroke) and adjudicated malignancy (excluding non-melanoma skin cancer). The study was designed to exclude a prespecified risk margin of 1.8 for the hazard ratio of combined XELJANZ regimens versus the TNF-blocker control for each co-primary endpoint. An independent committee conducted a blinded evaluation of the co-primary endpoints according to predefined criteria (adjudication). The study was event-driven and patients were followed until a sufficient number of primary outcome events accrued. Other endpoints included mortality, serious infections, and thromboembolic events. The median on-study follow-up time was 4 years.
The mean age of the population was 61 years (range: 50 to 88 years). Most patients were female (78%) and Caucasian (77%). Patients had a diagnosis of RA for a mean of 10 years, and a median swollen and tender joint count of 11 and 15 respectively. Cardiovascular risk factors included cigarette smoking (current or past) (48%), hypertension (66%), high density lipoprotein <40 mg/dL (12%), diabetes mellitus (17%), family history of premature coronary heart disease (15%), extra-articular disease associated with RA (37%), and history of coronary artery disease (11%).
The non-inferiority criterion was not met for the primary comparison of the combined XELJANZ dosages to TNF blockers since the upper limit of the 95% CI exceeded the pre-specified non-inferiority criterion of 1.8 (for MACE, the upper limit of the 95% CI was 1.94; for malignancies excluding NMSC, the upper limit of the 95% CI was 2.09).
Table 22 shows the study results for each of the co-primary endpoints, and other endpoints. There was an increased risk of death, MACE, malignancies, serious infections, and thromboembolic events associated with both dosages of XELJANZ.
| Endpoint |
TNF Blocker
N=1451 PY=5468 |
XELJANZ
5 mg Twice Daily N=1455 PY=5490 |
XELJANZ
10 mg Twice Daily N=1456 PY=5298 |
|---|---|---|---|
| Note: XELJANZ 10 mg twice daily was discontinued by the Data Monitoring Committee due to safety concerns, and ongoing patients switched from XELJANZ 10 mg to XELJANZ 5 mg. The column “XELJANZ 10 mg Twice Daily” includes all events and follow-up for patients randomized to XELJANZ 10 mg twice daily. A XELJANZ (refers to tablets and oral solution) 10 mg twice daily (or a XELJANZ XR 22 mg once daily) dosage is not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3)]. N indicates number of patients; n indicates number of patients with events. IR indicates incidence rate per 100 person-year (PY). NMSC: Non-melanoma Skin Cancer; MACE: Major Adverse Cardiac Events; HR: Hazard Ratio; DVT: Deep Vein Thrombosis; PE: Pulmonary Embolism; VTE: Venous Thromboembolism, first occurrence of a VTE, defined as the composite of adjudicated DVT and adjudicated PE; ATE: Arterial Thromboembolism; TE: Thromboembolism, first occurrence of a TE, defined as the composite of adjudicated VTE and unadjudicated ATE. |
|||
|
MACE, n [IR] |
43 [0.79] |
50 [0.91] |
59 [1.11] |
|
HR (95% CI) HR (95%) CI for XELJANZ vs. TNF Blocker (Univariate Cox Proportional Hazard Model).
|
1.16 (0.77, 1.74) |
1.41 (0.95, 2.10) |
|
|
MI, MI and Stroke include fatal and non-fatal events. n [IR]
|
11 [0.20] |
20 [0.36] |
21 [0.39] |
|
HR (95% CI) |
1.81 (0.87, 3.79) |
1.97 (0.95, 4.09) |
|
|
Stroke, n [IR] |
20 [0.36] |
18 [0.33] |
21 [0.39] |
|
HR (95% CI) |
0.89 (0.47, 1.69) |
1.08 (0.59, 2.00) |
|
|
Cardiovascular Death, n [IR] |
15 [0.27] |
18 [0.32] |
25 [0.47] |
|
HR (95% CI) |
1.20 (0.60, 2.37) |
1.71 (0.90, 3.24) |
|
|
Malignancies Excl. NMSC, n [IR] |
42 [0.77] |
62 [1.13] |
60 [1.13] |
|
HR (95% CI) |
1.47 (1.00, 2.18) |
1.48 (1.00, 2.19) |
|
|
Malignancies Excl. NMSC
Data and analyses for Malignancies excluding NMSC for current and ex-smokers are included. There were 720 current and ex-smokers randomized to XELJANZ 5 mg, 704 to XELJANZ 10 mg, and 679 to TNF blockers.
|
25 [0.99] |
41 [1.53] |
48 [1.91] |
|
HR (95% CI) |
1.55 (0.94, 2.55) |
1.94 (1.19, 3.14) |
|
|
All Death |
38 [0.69] |
49 [0.88] |
66 [1.23] |
|
HR (95% CI) |
1.29 (0.84, 1.96) |
1.79 (1.20, 2.66) |
|
|
Serious Infections |
133 [2.52] |
155 [2.95] |
184 [3.65] |
|
HR (95% CI) |
1.17 (0.93, 1.47) |
1.44 (1.15, 1.80) |
|
|
DVT |
9 [0.16] |
12 [0.22] |
15 [0.28] |
|
HR (95% CI) |
1.33 (0.56, 3.15) |
1.72 (0.75, 3.92) |
|
|
PE |
3 [0.05] |
10 [0.18] |
26 [0.49] |
|
HR (95% CI) |
3.32 (0.91, 12.08) |
8.95 (2.71, 29.56) |
|
|
VTE |
12 [0.22] |
18 [0.33] |
36 [0.68] |
|
HR (95% CI) |
1.50 (0.72, 3.10) |
3.10 (1.61, 5.96) |
|
|
ATE |
45 [0.83] |
51 [0.93] |
55 [1.04] |
|
HR (95% CI) |
1.13 (0.76, 1.69) |
1.26 (0.85, 1.87) |
|
|
TE |
56 [1.03] |
67 [1.23] |
86 [1.65] |
|
HR (95% CI) |
1.19 (0.84, 1.70) |
1.60 (1.14, 2.23) |
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with XELJANZ 5 mg twice a day and XELJANZ 10 mg twice a day compared to those treated with TNF blockers. Lymphoma was reported for 4 patients who received XELJANZ 5 mg twice a day, 6 patients who received XELJANZ 10 mg twice a day, and 1 patient who received TNF blockers (Incidence Rate [IR] of 0.07, 0.11, and 0.02 per 100 patient-years, respectively). Among current and past smokers, lung cancer was reported for 13 patients who received XELJANZ 5 mg twice a day, 15 patients who received XELJANZ 10 mg twice a day, and 7 patients who received TNF blockers (IR of 0.48, 0.59, and 0.27 per 100 patient-years, respectively).
Given these increased risks, XELJANZ (tablets and oral solution) 10 mg twice daily (or XELJANZ XR (extended-release tablets) 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS or pcJIA [see Dosage and Administration (2.3, 2.4)].
Warning: Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS
See full prescribing information for complete boxed warning.
-
•Increased risk of serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), leading to hospitalization or death. Interrupt XELJANZ/XELJANZ XR treatment if serious infection occurs until the infection is controlled. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test. (5.1)
-
•Higher rate of all-cause mortality, including sudden cardiovascular (CV) death with XELJANZ vs. TNF blockers in rheumatoid arthritis (RA) patients. (5.2)
-
•Malignancies have occurred in patients treated with XELJANZ. Higher rate of lymphomas and lung cancers with XELJANZ vs. TNF blockers in RA patients. (5.3)
-
•Higher rate of major adverse CV events (defined as CV death, myocardial infarction, and stroke) with XELJANZ vs. TNF blockers in RA patients. (5.4)
-
•Thrombosis has occurred in patients treated with XELJANZ. Increased incidence of pulmonary embolism, venous and arterial thrombosis with XELJANZ vs. TNF blockers in RA patients. (5.5)
5.10 Risk of Gastrointestinal Obstruction With Xeljanz Xr A Non Deformable Extended Release Formulation
Gastrointestinal obstruction may occur with XELJANZ XR (extended-release tablets). Avoid use of XELJANZ XR in patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs utilizing a non-deformable extended-release formulation.
2.3 Recommended Dosage in Adults With Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis
Table 1 displays the recommended dosage of XELJANZ tablets and XELJANZ XR (extended-release tablets) for adults with RA, PsA, and AS [see Indication and Usage (1.1, 1.2, 1.3)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. The table also displays the recommended dosage modifications for patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and patients with lymphopenia, neutropenia, or anemia.
|
Adults |
XELJANZ Tablets |
XELJANZ XR (extended-release tablets) |
|
Patients with Normal Renal and Hepatic Function Excludes patients who concomitantly use XELJANZ tablets/XELJANZ XR with strong CYP3A4 inhibitor(s) or moderate CYP3A4 inhibitor(s) and strong CYP2C19 inhibitor(s), as well as patients with lymphocyte count less than 500 cells/mm3, ANC <1000 cells/mm3, or hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL.
|
5 mg twice daily |
11 mg once daily |
|
Recommended Dosage in Patients with Renal Impairment (RI) Tofacitinib PK was evaluated in subjects with varying degrees of renal impairment, where the severity of renal impairment was defined based on creatinine clearance (CLcr) estimated using the Cockcroft‑Gault equation: CLcr >80 mL/min (normal renal function); >50 and ≤80 mL/min (mild renal impairment); ≥30 and ≤50 mL/min (moderate renal impairment); <30 mL/min (severe renal impairment).
|
||
|
Mild RI (CLcr >50 and ≤80 mL/min) |
5 mg twice daily |
11 mg once daily |
|
Moderate RI (CLcr ≥30 and ≤50 mL/min) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Severe RI (CLcr <30 mL/min) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
For patients undergoing hemodialysis, administer the dose after the dialysis session on dialysis days. If a dose was taken before the dialysis procedure, supplemental doses are not recommended after dialysis. |
||
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
||
|
Mild HI (Child-Pugh A) |
5 mg twice daily |
11 mg once daily |
|
Moderate HI (Child-Pugh B) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Severe HI (Child-Pugh C) |
Use of XELJANZ tablets/XELJANZ XR is not recommended. |
|
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
||
|
Strong CYP2C19 inhibitor(s) |
5 mg twice daily |
11 mg once daily |
|
Moderate CYP2C19 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Strong CYP3A4 inhibitor(s) |
||
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
||
|
Patients with lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
|
Patients with ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
|
Patients with ANC 500 to 1000 cells/mm3 |
Interrupt dosing. When ANC is greater than 1000, resume 5 mg twice daily. |
Interrupt dosing. When ANC is greater than 1000, resume 11 mg once daily. |
|
Patients with hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
2.4 Recommended Dosage in Pediatric Patients 2 Years of Age and Older With Psoriatic Arthritis Or Polyarticular Course Juvenile Idiopathic Arthritis
Table 2 displays the recommended body weight-based dosages for XELJANZ tablets and XELJANZ oral solution in pediatric patients 2 years of age and older with PsA or pcJIA [see Indication and Usage (1.2, 1.4)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. The table also includes recommended dosage modification for pediatric patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and pediatric patients with lymphopenia, neutropenia, or anemia.
Administer XELJANZ oral solution using the included press-in bottle adapter and oral dosing syringe [see Instructions for Use].
| Pediatric Patients 2 Years of Age and Older | XELJANZ tablets and XELJANZ oral solution |
|---|---|
|
Patients with Normal Renal and Hepatic Function
|
|
|
Recommended Dosage in Patients with Renal Impairment (RI) |
|
|
Mild RI |
Same as patients with normal renal function. |
|
Moderate RI |
|
|
Severe RI |
|
|
For patients undergoing hemodialysis, administer the dose after the dialysis session on dialysis days. If a dose was taken before the dialysis procedure, supplemental doses are not recommended after dialysis. |
|
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
|
|
Mild HI |
Same as patients with normal hepatic function. |
|
Moderate HI |
|
|
Severe HI |
Use of XELJANZ tablets/XELJANZ oral solution is not recommended. |
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
|
|
Strong CYP2C19 inhibitor(s) |
No dosage modification is recommended. |
|
Moderate CYP2C19 inhibitor(s) |
|
|
Moderate CYP3A4 inhibitor(s) |
|
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
|
|
Strong CYP3A4 inhibitor(s) |
|
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
|
|
Patients with lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
Patients with ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
Patients with ANC 500 to 1000 cells/mm3 |
Interrupt dosing until ANC is greater than 1000 cells/mm3. |
|
Patients with hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
Structured Label Content
Section 42229-5 (42229-5)
SERIOUS INFECTIONS
Patients treated with XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets) are at increased risk for developing serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), that may lead to hospitalization or death [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids. Reported infections included:
-
•Active TB, which may present with pulmonary or extrapulmonary disease. Patients should be tested for latent TB before XELJANZ/XELJANZ XR use and during therapy. Treatment for latent infection should be initiated prior to XELJANZ/XELJANZ XR use.
-
•Invasive fungal infections, including cryptococcosis and pneumocystosis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral, including herpes zoster, and other infections due to opportunistic pathogens.
The risks and benefits of XELJANZ/XELJANZ XR treatment should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after XELJANZ/XELJANZ XR treatment, including the possible development of TB in patients who tested negative for latent TB infection prior to initiating therapy. If a serious infection develops, interrupt XELJANZ/XELJANZ XR until the infection is controlled [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
|
MEDICATION GUIDE |
||
|
XELJANZ (ZEL' JANS')
|
XELJANZ XR (ZEL' JANS' EKS-AHR)
|
XELJANZ (ZEL' JANS')
|
|
What is the most important information I should know about XELJANZ/XELJANZ XR/XELJANZ oral solution?
|
||
|
1. |
Serious infections. XELJANZ/XELJANZ XR/XELJANZ oral solution are medicines that affect your immune system. XELJANZ/XELJANZ XR/XELJANZ oral solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking XELJANZ/XELJANZ XR/XELJANZ oral solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
You should not start taking XELJANZ/XELJANZ XR/XELJANZ oral solution if you have any kind of infection unless your healthcare provider tells you it is okay. You may be at a higher risk of developing shingles (herpes zoster).
|
|
|
|
|
After starting XELJANZ/XELJANZ XR/XELJANZ oral solution, call your healthcare provider right away if you have any symptoms of an infection. XELJANZ/XELJANZ XR/XELJANZ oral solution can make you more likely to get infections or make worse any infection that you have. |
||
|
2. |
Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg or 10 mg twice daily. |
|
|
3. |
Cancer and immune system problems. XELJANZ/XELJANZ XR/XELJANZ oral solution may increase your risk of certain cancers by changing the way your immune system works.
|
|
|
4. |
Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking XELJANZ 5 mg or 10 mg twice daily, especially if you are a current or past smoker.
|
|
|
5. |
Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking XELJANZ 5 mg or 10 mg twice daily. Blood clots in the lungs have also happened in people with ulcerative colitis. Some people have died from these blood clots.
|
|
|
6. |
Tears (perforation) in the stomach or intestines.
|
|
|
7. |
Allergic reactions.
|
|
|
8. |
Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking XELJANZ/XELJANZ XR/XELJANZ oral solution and while you take XELJANZ/XELJANZ XR/XELJANZ oral solution to check for the following side effects:
Your healthcare provider should routinely check certain liver tests. |
|
|
See " What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution? " for more information about side effects. |
||
|
What is XELJANZ/XELJANZ XR/XELJANZ oral solution?
It is not known if XELJANZ/XELJANZ XR/XELJANZ oral solution is safe and effective in people with Hepatitis B or C. |
||
|
What should I tell my healthcare provider before taking XELJANZ/XELJANZ XR/XELJANZ oral solution?
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. XELJANZ/XELJANZ XR/XELJANZ oral solution and other medicines may affect each other causing side effects.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||
|
How should I take XELJANZ/XELJANZ XR/XELJANZ oral solution?
|
||
|
What are the possible side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution? XELJANZ/XELJANZ XR/XELJANZ oral solution may cause serious side effects, including:
|
||
|
|
|
|
Common side effects of XELJANZ/XELJANZ XR/XELJANZ oral solution in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
Common side effects of XELJANZ/XELJANZ XR in people with ulcerative colitis include:
Common side effects of XELJANZ/XELJANZ oral solution in children with polyarticular course juvenile arthritis and psoriatic arthritis include:
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. |
||
|
How should I store XELJANZ/XELJANZ XR/XELJANZ oral solution?
Keep XELJANZ/XELJANZ XR/XELJANZ oral solution and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of XELJANZ/XELJANZ XR/XELJANZ oral solution.
|
||
|
What are the ingredients in XELJANZ 5 mg?
This Medication Guide may have been updated. For the most recent Medication Guide, please visit www.pfizer.com. LAB-0535-16.0 |
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: October 2025
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for overdose with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). In case of an overdose, it is recommended that the patient be monitored for signs and symptoms of adverse reactions.
In a study in patients with end-stage renal disease (ESRD) undergoing hemodialysis, plasma tofacitinib concentrations declined more rapidly during the period of hemodialysis and dialyzer efficiency, calculated as dialyzer clearance/blood flow entering the dialyzer, was high [mean (SD) = 0.73 (0.15)]. However, due to the significant non-renal clearance of tofacitinib, the fraction of total elimination occurring by hemodialysis was small, and thus, limits the value of hemodialysis for treatment of overdose with XELJANZ/XELJANZ XR.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description (11 DESCRIPTION)
XELJANZ (tofacitinib) tablets, XELJANZ XR (tofacitinib) extended-release tablets and XELJANZ (tofacitinib) oral solution are formulated with the citrate salt of tofacitinib, a JAK inhibitor.
Tofacitinib citrate is a white to off-white powder with the following chemical name: (3R,4R)-4-methyl-3-(methyl-7H-pyrrolo [2,3-d]pyrimidin-4-ylamino)-ß-oxo-1-piperidinepropanenitrile, 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
The solubility of tofacitinib citrate in water is 2.9 mg/mL.
Tofacitinib citrate has a molecular weight of 504.5 Daltons (or 312.4 Daltons as the tofacitinib free base) and a molecular formula of C16H20N6O•C6H8O7. The chemical structure of tofacitinib citrate is:
XELJANZ tablets is supplied for oral administration as a:
-
•5 mg white round, immediate-release film-coated tablet. Each tablet contains 5 mg of tofacitinib (equivalent to 8.08 mg of tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
-
•10 mg blue round, immediate-release film-coated tablet. Each tablet contains 10 mg of tofacitinib (equivalent to 16.16 mg of tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
XELJANZ XR is supplied for oral administration as a:
-
•11 mg pink, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each tablet contains 11 mg of tofacitinib (equivalent to 17.77 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide and triacetin. Printing ink contains, ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
-
•22 mg beige, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each tablet contains 22 mg of tofacitinib (equivalent to 35.54 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
XELJANZ oral solution is supplied for oral administration as a 1 mg/mL clear, colorless solution. Each 1 mL contains 1 mg of tofacitinib (equivalent to 1.62 mg of tofacitinib citrate) and the following inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
5.5 Thrombosis
Thrombosis may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Thrombosis, including pulmonary embolism (PE), deep venous thrombosis (DVT), and arterial thrombosis, have occurred in patients treated with XELJANZ and other Janus kinase (JAK) inhibitors used to treat inflammatory conditions. Many of these events were serious and some resulted in death [see Warnings and Precautions (5.2)].
Patients with RA 50 years of age and older with at least one cardiovascular risk factor treated with XELJANZ tablets 5 mg or 10 mg twice daily compared to TNF blockers in RA Safety Study 1 had an observed increase in incidence of these thrombotic events. The incidence rate of DVT per 100 patient-years was 0.28 for XELJANZ tablets 10 mg twice a day, 0.22 for XELJANZ tablets 5 mg twice a day, and 0.16 for TNF blockers. The incidence rate of PE per 100 patient-years was 0.49 for XELJANZ tablets 10 mg twice a day, 0.18 for XELJANZ tablets 5 mg twice a day, and 0.05 for TNF blockers [see Clinical Studies (14.6)].
XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
In a long-term extension study in patients with UC, five cases of pulmonary embolism were reported in patients taking XELJANZ 10 mg twice daily, including one death in a patient with advanced cancer.
Promptly evaluate patients with symptoms of thrombosis and discontinue XELJANZ/XELJANZ XR in patients with symptoms of thrombosis.
Avoid XELJANZ/XELJANZ XR in patients that may be at increased risk of thrombosis. For the treatment of UC, use XELJANZ tablets or XELJANZ XR at the lowest effective dose and for the shortest duration needed to achieve/maintain therapeutic response [see Dosage and Administration (2.5) ].
5.9 Vaccinations
Avoid use of live vaccines concurrently with XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets). Prior to initiating XELJANZ/XELJANZ XR therapy, update immunizations in agreement with current immunization guidelines. The interval between live vaccinations and initiation of XELJANZ/XELJANZ XR therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
8.4 Pediatric Use
The safety and effectiveness of XELJANZ (tablets and oral solution) in pediatric patients for indications, other than in patients with active pcJIA and PsA, have not been established.
The safety and effectiveness of XELJANZ have not been established in pediatric patients less than 2 years of age.
The safety and effectiveness of XELJANZ XR (extended-release tablets) in pediatric patients have not been established.
Polyarticular Course Juvenile Idiopathic Arthritis (pcJIA)
The safety and effectiveness of XELJANZ (tablets and oral solution) for the treatment of active pcJIA have been established in pediatric patients 2 years of age and older who have had an inadequate response or intolerance to one or more TNF blockers.
Use of XELJANZ for this indication is supported by evidence from adequate and well-controlled studies of XELJANZ tablets in adults with RA, pharmacokinetic (PK) data from adult patients with RA, and with additional safety, efficacy, and PK data from a clinical trial of XELJANZ in pediatric patients 2 years and older with active pcJIA (Study pcJIA-I) [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1, 14.4)].
Adverse reactions observed in pediatric patients with pcJIA who received XELJANZ were consistent with those reported in adults with RA [see Adverse Reactions (6.1)].
Psoriatic Arthritis
The safety and effectiveness of XELJANZ (tablets and oral solution) for the treatment of active PsA have been established in pediatric patients 2 years of age and older who have had an inadequate response or intolerance to one or more TNF blockers.
Use of XELJANZ for this indication is supported by evidence from well-controlled studies of XELJANZ tablets in adults with PsA, PK data from adults with PsA, and PK data from a clinical trial of XELJANZ in 225 pediatric patients with JIA, and safety data from 280 pediatric patients 2 years of age and older with JIA [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)]. Following administration of the recommended XELJANZ dosage in pediatric patients 2 years of age and older with PsA, tofacitinib plasma exposures are predicted to be comparable to those observed in adults with PsA based on population PK modeling and simulation [see Clinical Pharmacology (12.3)].
Systemic Juvenile Idiopathic Arthritis
The safety and effectiveness of XELJANZ for the treatment of pediatric patients with systemic juvenile idiopathic arthritis (sJIA) have not been established.
The results from a two-part study (an open-label, run-in phase, followed by a double-blind, placebo-controlled, randomized event-driven withdrawal phase) in 100 patients 2 years to 17 years of age with sJIA with active systemic features did not demonstrate that XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) was efficacious in the treatment of sJIA with active systemic features.
Of the 100 patients enrolled in the open-label run-in phase, 59 (59%) patients achieved a clinical response and were eligible for the double-blind withdrawal phase. There were 28 patients randomized to XELJANZ and 31 patients to placebo. The study data were insufficient to demonstrate efficacy and, therefore, XELJANZ is not recommended for the treatment of sJIA.
Adverse reactions observed in pediatric patients with sJIA receiving XELJANZ/XELJANZ oral solution were consistent with those reported in pcJIA and RA patients [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the 3315 adults who were enrolled in clinical trials with RA (Studies RA-I to V), a total of 505 patients were 65 years of age and older, including 71 patients 75 years and older. The frequency of serious infection among XELJANZ tablets-treated patients 65 years of age and older was higher than among those adults under the age of 65.
Of the 1156 XELJANZ tablet-treated patients in clinical trials of patients with UC, a total of 77 patients (7%) were 65 years of age or older. Clinical studies of XELJANZ in patients with UC did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger adult patients.
Of the 783 XELJANZ tablet-treated patients in clinical trials of patients with PsA, a total of 72 (9.2%) patients were 65 years of age and older, including 2 (0.3%) patients 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with PsA to determine if they respond differently from younger adult patients.
Of the 420 XELJANZ tablet-treated patients in clinical trials of patients with AS, a total of 12 (2.9%) patients were 65 years of age and older, including 1 (0.2%) patient 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with AS to determine if they respond differently from younger adult patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Increased Risk of Mortality [see Warnings and Precautions (5.2)]
-
•Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3)]
-
•Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4)]
-
•Thrombosis [see Warnings and Precautions (5.5)]
-
•Gastrointestinal Perforations [see Warnings and Precautions (5.6)]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
-
•Laboratory Abnormalities [see Warnings and Precautions (5.8)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 7 includes drugs with clinically significant drug interactions when concomitantly used with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) and instructions for preventing or managing them.
|
Strong CYP3A4 Inhibitors (e.g., ketoconazole) |
|
|
Clinical Impact |
Increased exposure to tofacitinib |
|
Intervention |
Dosage modification of XELJANZ/XELJANZ XR is recommended |
|
Moderate CYP3A4 Inhibitors Concomitantly Used with Strong CYP2C19 Inhibitors (e.g., fluconazole) |
|
|
Clinical Impact |
Increased exposure to tofacitinib |
|
Intervention |
Dosage modification of XELJANZ/XELJANZ XR is recommended |
|
Strong CYP3A4 Inducers (e.g., rifampin) |
|
|
Clinical Impact |
Decreased exposure to tofacitinib and may result in loss of or reduced clinical response |
|
Intervention |
Concomitant use with XELJANZ/XELJANZ XR is not recommended |
|
Immunosuppressive Drugs (e.g., azathioprine, tacrolimus, cyclosporine) |
|
|
Clinical Impact |
Risk of added immunosuppression; concomitant use of XELJANZ/XELJANZ XR with biologic DMARDs or potent immunosuppressants has not been studied in patients with RA, PsA, AS, UC, or pcJIA. |
|
Intervention |
Concomitant use with XELJANZ/XELJANZ XR is not recommended |
8.6 Renal Impairment
Moderate and Severe Renal Impairment
XELJANZ-treated patients with moderate renal impairment (RI) (CLcr ≥30 and ≤50 mL/minute) or severe RI (<30 mL/minute) had greater tofacitinib blood concentrations than XELJANZ-treated patients with normal renal function (CLcr >80 mL/minute). The recommended dosage of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) in patients with moderate or severe RI (including those with severe RI who are undergoing hemodialysis) is lower than the recommended dosage in patients with normal renal function [see Dosage and Administration (2.3, 2.4, 2.5)].
Mild Renal Impairment
The recommended dosage in patients with mild RI (CLcr >50 and ≤80 mL/minute) is the same as patients with normal renal function.
Instructions for Use (INSTRUCTIONS FOR USE)
XELJANZ
®
(ZEL' JANS')
(tofacitinib)
oral solution
Read this Instructions for Use before you start taking XELJANZ oral solution and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring XELJANZ oral solution:
Always use the oral dosing syringe that comes with XELJANZ oral solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store XELJANZ?
-
•Store XELJANZ oral solution at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Always store XELJANZ oral solution in the original bottle and carton to protect from light.
Keep XELJANZ and all medicines out of the reach of children.
Use XELJANZ oral solution within 60 days of opening the bottle. Throw away (discard) remaining XELJANZ oral solution after 60 days.
To help you remember when to throw away your bottle of XELJANZ oral solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
Each carton of XELJANZ oral solution contains:
-
•1 press-in bottle adapter
-
•1 bottle of XELJANZ oral solution
-
•1 oral dosing syringe
Step 1. Remove bottle from carton
Open the carton and remove the bottle of XELJANZ oral solution.
Step 2. Open bottle
Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)
Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe
Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe
Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle
With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe
Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose
Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of XELJANZ
Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle
Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect XELJANZ oral solution from light.
Step 11. Clean oral dosing syringe
Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the XELJANZ oral solution.
Do not throw away the oral dosing syringe.
LAB-1422-3.0
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: October 2025
This Instructions for Use may have been updated. For the most recent Instructions for Use, please visit www.pfizer.com.
12.2 Pharmacodynamics
Treatment with XELJANZ was associated with dose-dependent reductions of circulating CD16/56+ natural killer cells, with estimated maximum reductions occurring at approximately 8-10 weeks after initiation of therapy. These changes generally resolved within 2-6 weeks after discontinuation of treatment. Treatment with XELJANZ was associated with dose-dependent increases in B cell counts. Changes in circulating T-lymphocyte counts and T-lymphocyte subsets (CD3+, CD4+ and CD8+) were small and inconsistent. The clinical significance of these changes is unknown.
Total serum IgG, IgM, and IgA levels after 6-month dosing in patients with rheumatoid arthritis (RA) were lower than in patients who received placebo; however, changes were small and not dose-dependent.
After treatment with XELJANZ in patients with RA, rapid decreases in serum C-reactive protein (CRP) were observed and maintained throughout dosing. Changes in CRP observed with XELJANZ treatment do not reverse fully within 2 weeks after discontinuation, indicating a longer duration of pharmacodynamic activity compared to the pharmacokinetic half-life.
Similar changes in T cells, B cells, and serum CRP have been observed in patients with active psoriatic arthritis (PsA) although reversibility was not assessed. Total serum immunoglobulins were not assessed in patients with active PsA.
1.5 Ulcerative Colitis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with moderately to severely active ulcerative colitis (UC), who have an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
5.1 Serious Infections
Serious and sometimes fatal infections may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, or other opportunistic pathogens have been reported in patients receiving XELJANZ. The most common serious infections reported with XELJANZ included pneumonia, cellulitis, herpes zoster, urinary tract infection, diverticulitis, and appendicitis. Among opportunistic infections, tuberculosis and other mycobacterial infections, cryptococcosis, histoplasmosis, esophageal candidiasis, pneumocystosis, multi-dermatomal herpes zoster, cytomegalovirus infections, BK virus infection, and listeriosis were reported with XELJANZ. Some patients have presented with disseminated rather than localized disease, and were often taking concomitant immunomodulating agents such as methotrexate or corticosteroids.
In the UC population, treatment with XELJANZ tablets 10 mg twice daily was associated with greater risk of serious infections compared to 5 mg twice daily. Additionally, opportunistic herpes zoster infections (including meningoencephalitis, ophthalmologic, and disseminated cutaneous) were seen in patients who were treated with XELJANZ tablets 10 mg twice daily.
Other serious infections that were not reported in clinical studies may also occur (e.g., coccidioidomycosis).
Avoid use of XELJANZ/XELJANZ XR in patients with an active, serious infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating XELJANZ/XELJANZ XR in patients:
-
•with chronic or recurrent infection
-
•who have been exposed to tuberculosis
-
•with a history of a serious or an opportunistic infection
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
-
•with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with XELJANZ/XELJANZ XR. Interrupt XELJANZ/XELJANZ XR if a patient develops a serious infection, an opportunistic infection, or sepsis. In patients who develop a new infection during treatment with XELJANZ/XELJANZ XR, promptly complete diagnostic testing appropriate for an immunocompromised patient; initiate appropriate antimicrobial therapy, and monitor the patients closely.
Caution is also recommended in patients with a history of chronic lung disease, or in those who develop interstitial lung disease, as they may be more prone to infections.
Risk of infection may be higher with increasing degrees of lymphopenia and consideration should be given to lymphocyte counts when assessing individual patient risk of infection. Discontinuation and monitoring criteria for lymphopenia are recommended [see Dosage and Administration (2.3 , 2.4 , 2.5) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) are Janus kinase (JAK) inhibitors.
XELJANZ tablets and XELJANZ XR are indicated for the treatment of adult patients with:
-
•Moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Moderately to severely active ulcerative colitis (UC), who have had an inadequate response or intolerance to one or more TNF blockers.
XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with:
-
•Active PsA, who have had an inadequate response or intolerance to one or more TNF blockers.
-
•Active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use:
-
•Use of XELJANZ/XELJANZ XR for RA, AS, PsA, or pcJIA in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended. (1.1, 1.2, 1.3, 1.4)
-
•Use of XELJANZ tablets and XELJANZ XR for UC in combination with biological therapies for UC or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended. (1.5)
1.2 Psoriatic Arthritis
XELJANZ (tablets and oral solution) is indicated for the treatment of adult and pediatric patients 2 years of age and older with active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers.
XELJANZ XR (extended-release tablets) is indicated for the treatment of adults with active PsA who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ or XELJANZ XR in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
1.1 Rheumatoid Arthritis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biologic disease-modifying antirheumatic drugs (DMARDs) or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
12.1 Mechanism of Action
Tofacitinib is a Janus kinase (JAK) inhibitor. JAKs are intracellular enzymes which transmit signals arising from cytokine or growth factor-receptor interactions on the cellular membrane to influence cellular processes of hematopoiesis and immune cell function. Within the signaling pathway, JAKs phosphorylate and activate Signal Transducers and Activators of Transcription (STATs) which modulate intracellular activity including gene expression. Tofacitinib modulates the signaling pathway at the point of JAKs, preventing the phosphorylation and activation of STATs. JAK enzymes transmit cytokine signaling through pairing of JAKs (e.g., JAK1/JAK3, JAK1/JAK2, JAK1/TyK2, JAK2/JAK2). Tofacitinib inhibited the in vitro activities of JAK1/JAK2, JAK1/JAK3, and JAK2/JAK2 combinations with IC50 of 406, 56, and 1377 nM, respectively. However, the relevance of specific JAK combinations to therapeutic effectiveness is not known.
1.3 Ankylosing Spondylitis
XELJANZ tablets and XELJANZ XR (extended-release tablets) are indicated for the treatment of adult patients with active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ tablets or XELJANZ XR in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Serious Infections: Avoid use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets) during an active serious infection, including localized infections. (5.1)
-
•Gastrointestinal Perforations: Promptly evaluate patients at increased risk for gastrointestinal perforation who present with new onset abdominal symptoms. (5.6)
-
•Laboratory Monitoring: Recommended due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes and lipids. (5.8)
-
•Vaccinations: Avoid use of live vaccines concurrently with XELJANZ or XELJANZ XR. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Recommended Evaluations and Immunization Prior to Treatment Initiation
-
•Prior to initiating XELJANZ/XELJANZ XR, consider performing an active and latent TB evaluation, viral hepatitis screening, a complete blood count, and updating immunizations. Avoid XELJANZ or XELJANZ XR initiation if absolute lymphocyte count <500 cells/mm3, an absolute neutrophil count (ANC) <1000 cells/mm3 or hemoglobin <9 g/dL. (2.1)
Important Administration Instructions
-
•XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). (2.2)
-
•Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider. (2.2)
Recommended Dosage
Adult Patients with RA, PsA or AS
-
•XELJANZ tablets 5 mg twice daily or XELJANZ XR (extended-release tablets) 11 mg once daily. (2.3)
Pediatric Patients 2 Years of Age and Older with PsA or pcJIA Who Weigh At Least 10 kg
-
•XELJANZ (tablets or oral solution) 5 mg twice daily for those ≥40 kg or weight-based equivalent twice daily for those <40 kg. (2.4)
Adult Patients with UC
-
•Induction: XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed, continue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily for a maximum of 16 weeks. Discontinue XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. (2.5)
-
•Maintenance: XELJANZ tablets 5 mg twice daily or XELJANZ XR 11 mg once daily. For patients with loss of response during maintenance treatment, XELJANZ tablets 10 mg twice daily or XELJANZ XR 22 mg once daily may be considered and limited to the shortest duration, with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. (2.5)
Dosage in Patients with Renal Impairment or Hepatic Impairment
-
•Use of XELJANZ (tablets and oral solution) or XELJANZ XR in patients with severe HI is not recommended. (2.3, 2.4, 2.5, 8.7)
-
•See full prescribing information (FPI) for recommended dosage in patients with moderate or severe RI or moderate HI. (2.3, 2.4, 2.5, 8.6, 8.7)
Dosage Modification
See the full prescribing information for dosage modification by indication for patients who concomitantly use CYP2C19 and/or CYP3A4 inhibitors and patients with lymphopenia, neutropenia, or anemia. (2.3, 2.4, 2.5, 7)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
5.8 Laboratory Abnormalities
Laboratory abnormalities may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Drug hypersensitivity (events such as angioedema and urticaria have been observed)
Skin and subcutaneous tissue disorders: Acne
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
5.7 Hypersensitivity Reactions
Hypersensitivity reactions may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Reactions such as angioedema and urticaria that may reflect drug hypersensitivity have been observed in patients receiving XELJANZ/XELJANZ XR. Some events were serious. If a serious hypersensitivity reaction occurs, promptly discontinue XELJANZ/XELJANZ XR while evaluating the potential cause or causes of the reaction [see Adverse Reactions (6.2)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
The clinical studies described in this subsection were conducted using XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) and/or XELJANZ oral solution.
5.2 Increased Risk of Mortality
Increased risk of mortality may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Adult patients with rheumatoid arthritis (RA), 50 years of age and older, with at least one cardiovascular risk factor treated with XELJANZ tablets 5 mg or 10 mg twice a day had a higher observed rate of all-cause mortality, including sudden cardiovascular death, compared to those treated with TNF blockers in a large, randomized, postmarketing safety study (RA Safety Study 1). The incidence rate of all-cause mortality per 100 patient-years was 1.23 for XELJANZ tablets 10 mg twice a day, 0.88 for XELJANZ tablets 5 mg twice a day, and 0.69 for TNF blockers [see Clinical Studies (14.6)]. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR.
XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
For the treatment of UC, use XELJANZ/XELJANZ XR at the lowest effective dose and for the shortest duration needed to achieve/maintain therapeutic response [see Dosage and Administration (2.5) ].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
5.6 Gastrointestinal Perforations
Gastrointestinal perforations may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Events of gastrointestinal perforation have been reported in clinical studies with XELJANZ tablets, although the role of JAK inhibition in these events is not known. In these studies, many patients with RA received background therapy with nonsteroidal anti-inflammatory drugs (NSAIDs).
There was no discernable difference in frequency of gastrointestinal perforation between the placebo and the XELJANZ tablets treatment groups in clinical trials of patients with UC, and many of them were receiving background corticosteroids.
Promptly evaluate patients treated with XELJANZ/XELJANZ XR who may be at increased risk for gastrointestinal perforation (e.g., patients with a history of diverticulitis or taking NSAIDs) and who present with new onset abdominal symptoms for early identification of gastrointestinal perforation [see Adverse Reactions (6.1)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied Information for XELJANZ Tablets and XELJANZ XR
How supplied information for XELJANZ tablets and XELJANZ XR (extended-release tablets) is shown in Table 23.
| Dosage Form, Strength, and Description |
Bottle Size
(number of tablets) |
NDC Number |
|---|---|---|
|
XELJANZ (tofacitinib) tablets, |
60 |
NDC 0069-1001-01 |
|
XELJANZ (tofacitinib) tablets, 10 mg
|
60 |
NDC 0069-1002-01 |
|
XELJANZ XR (tofacitinib) extended-release tablets, 11 mg
|
30 |
NDC 0069-0501-30 |
|
XELJANZ XR (tofacitinib) extended-release tablets, 22 mg
|
30 |
NDC 0069-0502-30 |
How Supplied Information for XELJANZ Oral Solution
XELJANZ (tofacitinib) oral solution, 1 mg/mL is supplied in bottles (240 mL fill volume) (NDC 0069-1029-02) and is a clear, colorless solution that contains 1 mg of tofacitinib. Each high-density polyethylene (HDPE) bottle contains one press-in bottle adapter and one 5 mL oral dosing syringe with 3.2 mL, 4 mL, and 5 mL gradations. The press-in bottle adapter and oral dosing syringe are not made with natural rubber latex.
Storage and Handling for XELJANZ Tablets/XELJANZ XR
Store XELJANZ tablets and XELJANZ XR at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature].
Do not repackage.
Storage and Handling for XELJANZ Oral Solution
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). [See USP Controlled Room Temperature].
Store in the original bottle and carton to protect from light.
Use contents of bottle within 60 days of opening.
Discard unused oral solution after 60 days.
5.4 Major Adverse Cardiovascular Events
Major adverse cardiovascular events may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). In RA Safety Study 1, patients with RA who were 50 years of age and older with at least one cardiovascular risk factor and treated with XELJANZ tablets 5 mg or 10 mg twice daily had a higher rate of major adverse cardiovascular events (MACE) defined as cardiovascular death, non-fatal myocardial infarction (MI), and non-fatal stroke, compared to those treated with TNF blockers. The incidence rate of MACE per 100 patient-years was 1.11 for XELJANZ tablets 10 mg twice a day, 0.91 for XELJANZ tablets 5 mg twice a day, and 0.79 for TNF blockers. The incidence rate of fatal or non-fatal myocardial infarction per 100 patient-years was 0.39 for XELJANZ tablets 10 mg twice a day, 0.36 for XELJANZ tablets 5 mg twice a day, and 0.2 for TNF blockers [see Clinical Studies (14.6)]. Patients who are current or past smokers are at additional increased risk.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Inform patients about the symptoms of serious cardiovascular events and the steps to take if they occur. Discontinue XELJANZ/XELJANZ XR in patients that have experienced a MI or stroke. XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
2.2 Important Administration Instructions
-
•XELJANZ XR (extended-release tablets) is not substitutable with XELJANZ (tablets and oral solution). Switching between XELJANZ and XELJANZ XR should be made by the healthcare provider.
-
•Dose interruption is recommended for management of lymphopenia, neutropenia, and anemia [see Warnings and Precautions (5.8) and Adverse Reactions (6.1)].
-
•Interrupt use of XELJANZ/XELJANZ XR if a patient develops a serious infection until the infection is controlled [see Warnings and Precautions (5.1)].
-
•Take XELJANZ/XELJANZ XR with or without food [see Clinical Pharmacology (12.3)].
-
•Swallow XELJANZ XR whole and intact. Do not crush, split, or chew the extended-release tablets [see Clinical Pharmacology (12.3)].
14.2 Clinical Studies in Psoriatic Arthritis
The psoriatic arthritis (PsA) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included 2 multicenter, randomized, double-blind, placebo-controlled trials in 816 adults with active PsA (Studies PsA-I and PsA-II).
Trial Designs and Population
All patients had active PsA for at least 6 months based upon the Classification Criteria for Psoriatic Arthritis (CASPAR), at least 3 tender/painful joints and at least 3 swollen joints, and active plaque psoriasis. Patients randomized and treated across the 2 clinical trials represented different PsA subtypes at screening, including <5 joints or asymmetric involvement (21%), ≥5 joints involved (90%), distal interphalangeal (DIP) joint involvement (61%), arthritis mutilans (8%), and spondylitis (19%). Patients in these clinical trials had a diagnosis of PsA for a mean (SD) of 7.7 (7.2) years. At baseline, 80% and 53% of patients had enthesitis and dactylitis, respectively. At baseline, all patients were required to receive treatment with a stable dose of a nonbiologic DMARD (79% received methotrexate, 13% received sulfasalazine, 7% received leflunomide, 1% received other nonbiologic DMARDs). In both clinical trials, the primary endpoints were the ACR20 response and the change from baseline in HAQ-DI at Month 3.
-
•Study PsA-I was a 12-month clinical trial in 422 patients who had an inadequate response to a nonbiologic DMARD (67% and 33% were inadequate responders to 1 nonbiologic DMARD and ≥2 nonbiologic DMARDs, respectively) and who were naïve to treatment with a TNF blocker. Although Study PsA-1 included patients who are TNF blocker-naïve, XELJANZ and XELJANZ XR are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)]. Patients were randomized in a 2:2:2:1:1 ratio to receive XELJANZ 5 mg twice daily, XELJANZ 10 mg twice daily, adalimumab 40 mg subcutaneously once every 2 weeks, placebo to XELJANZ 5 mg twice daily treatment sequence, or placebo to XELJANZ 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a predetermined XELJANZ dosage of 5 mg or 10 mg twice daily. Study PsA-I was not designed to demonstrate noninferiority or superiority to adalimumab.
-
•Study PsA-II was a 6-month clinical trial in 394 patients who had an inadequate response to at least 1 approved TNF blocker (66%, 19%, and 15% were inadequate responders to 1 TNF blocker, 2 TNF blockers and ≥3 TNF blockers, respectively). Patients were randomized in a 2:2:1:1 ratio to receive XELJANZ 5 mg twice daily, XELJANZ 10 mg twice daily, placebo to XELJANZ 5 mg twice daily treatment sequence, or placebo to XELJANZ 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, placebo patients were switched in a blinded fashion to a predetermined XELJANZ dosage of 5 mg or 10 mg twice daily as in Study PsA-I.
Although other dosages have been studied, the recommended dosage of XELJANZ is 5 mg twice daily. XELJANZ 10 mg twice daily is not recommended for treatment of PsA [see Dosage and Administration (2.3)].
14.1 Clinical Studies in Rheumatoid Arthritis
The rheumatoid arthritis (RA) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included six randomized controlled trials in adults with moderate to severe active RA.
Trial Design
-
•Study RA-I (NCT00814307) was a 6-month monotherapy trial in which 610 patients with moderate to severe active RA who had an inadequate response to a DMARD (nonbiologic or biologic) received XELJANZ 5 mg or 10 mg twice daily or placebo added to their background DMARD. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 mg or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, changes in Health Assessment Questionnaire – Disability Index (HAQ-DI), and rates of Disease Activity Score DAS28-4(ESR) less than 2.6.
-
•Study RA-II (NCT00856544) was a 12-month trial in which 792 patients with moderate to severe active RA who had an inadequate response to a nonbiologic DMARD received XELJANZ 5 mg or 10 mg twice daily or placebo added to background DMARD treatment (excluding potent immunosuppressive treatments such as azathioprine or cyclosporine). At the Month 3 visit, nonresponding patients were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 mg or 10 mg twice daily. At the end of Month 6, all patients treated with placebo were switched to their second predetermined XELJANZ treatment in a blinded fashion. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, changes in HAQ-DI at Month 3, and rates of DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-III (NCT00853385) was a 12-month trial in 717 patients with moderate to severe active RA who had an inadequate response to methotrexate (MTX). Patients received XELJANZ 5 mg or 10 mg orally twice daily, adalimumab 40 mg subcutaneously every other week, or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-IV (NCT00847613) was a 2-year trial with a planned analysis at 1 year in which 797 patients with moderate to severe active RA who had an inadequate response to MTX received XELJANZ 5 mg or 10 mg twice daily or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, mean change from baseline in van der Heijde-modified total Sharp Score (mTSS) at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
-
•Study RA-V (NCT00960440) was a 6-month trial in which 399 patients with moderate to severe active RA who had an inadequate response to at least one approved TNF blocking biological product received XELJANZ 5 mg or 10 mg twice daily or placebo added to background MTX. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of XELJANZ 5 or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, HAQ-DI, and DAS28-4(ESR) less than 2.6.
-
•Study RA-VI (NCT01039688) was a 2-year monotherapy trial with a planned analysis at 1 year in which 952 MTX-naïve patients with moderate to severe active RA received XELJANZ 5 or 10 mg twice daily or MTX dose-titrated over 8 weeks to 20 mg weekly. The primary endpoints were mean change from baseline in van der Heijde-modified Total Sharp Score (mTSS) at Month 6 and the proportion of patients who achieved an ACR70 response at Month 6.
Although other dosages have been studied, the recommended dosage of XELJANZ is 5 mg twice daily. XELJANZ 10 mg twice daily is not recommended for the treatment of RA [see Dosage and Administration (2.3)].
Clinical Response
The percentages of XELJANZ-treated patients who achieved ACR20, ACR50, and ACR70 responses in Studies RA-I, IV, and V are shown in Table 10. Similar results were observed with Studies RA-II and III. In trials RA-I through V, patients treated with 5 mg twice daily XELJANZ had higher ACR20, ACR50, and ACR70 response rates versus patients treated with placebo, with or without background DMARD treatment, at Month 3 and Month 6. Higher ACR20 response rates were observed within 2 weeks compared to placebo. In the 12-month trials, ACR response rates in XELJANZ-treated patients were consistent at 6 and 12 months.
|
Monotherapy in Nonbiologic or Biologic DMARD Inadequate Responders Inadequate response to at least one DMARD (biologic or nonbiologic) due to lack of efficacy or toxicity.
|
MTX Inadequate Responders Inadequate response to MTX defined as the presence of sufficient residual disease activity to meet the entry criteria.
|
TNF Blocker Inadequate Responders Inadequate response to a least one TNF blocker due to lack of efficacy and/or intolerance.
|
||||
|
Study RA-I |
Study RA-IV |
Study RA-V |
||||
|
N N is number of randomized and treated patients.
|
Placebo + background DMARD |
XELJANZ 5 mg Twice Daily + background DMARD |
Placebo + background MTX |
XELJANZ 5 mg Twice Daily + background MTX |
Placebo + background MTX |
XELJANZ 5 mg Twice Daily + background MTX |
|
122 |
243 |
160 |
321 |
132 |
133 |
|
|
ACR20 Month 3 Month 6 |
26% NA NA (not applicable), as data for placebo treatment is not available beyond 3 months in Studies RA-I and RA-V due to placebo advancement.
|
59% 69% |
27% 25% |
55% 50% |
24% NA |
41% 51% |
|
ACR50 Month 3 Month 6 |
12% NA |
31% 42% |
8% 9% |
29% 32% |
8% NA |
26% 37% |
|
ACR70 Month 3 Month 6 |
6% NA |
15% 22% |
3% 1% |
11% 14% |
2% NA |
14% 16% |
In Study RA-IV, a greater proportion of patients treated with XELJANZ 5 mg twice daily plus MTX achieved a low level of disease activity as measured by a DAS28-4(ESR) less than 2.6 at 6 months compared to those treated with MTX alone (Table 11).
|
Study RA-IV |
||
|
DAS28-4(ESR) Less Than 2.6 |
Placebo + MTX 160 |
XELJANZ 5 mg Twice Daily + MTX 321 |
|
Proportion of responders at Month 6 (n) |
1% (2) |
6% (19) |
|
Of responders, proportion with 0 active joints (n) |
50% (1) |
42% (8) |
|
Of responders, proportion with 1 active joint (n) |
0 |
5% (1) |
|
Of responders, proportion with 2 active joints (n) |
0 |
32% (6) |
|
Of responders, proportion with 3 or more active joints (n) |
50% (1) |
21% (4) |
The results of the components of the ACR response criteria for Study RA-IV are shown in Table 12. Similar results were observed for XELJANZ in Studies RA-I, II, III, V, and VI.
|
Study RA-IV |
||||
|
Placebo + MTX N=160 |
XELJANZ 5 mg Twice Daily + MTX N=321 |
|||
|
Component (mean) Data shown is mean (Standard Deviation) at Month 3.
|
Baseline |
Month 3 |
Baseline |
Month 3 |
|
Number of tender joints (0-68) |
23 (13) |
18 (14) |
24 (14) |
13 (14) |
|
Number of swollen joints (0-66) |
14 (9) |
10 (9) |
14 (8) |
6 (8) |
|
Pain Visual analog scale: 0 = best, 100 = worst.
|
55 (24) |
47 (24) |
58 (23) |
34 (23) |
|
Patient global assessment |
54 (23) |
47 (24) |
58 (24) |
35 (23) |
|
Disability index (HAQ-DI) Health Assessment Questionnaire Disability Index: 0 = best, 3 = worst; 20 questions; categories: dressing and grooming, arising, eating, walking, hygiene, reach, grip, and activities.
|
1.32 (0.67) |
1.19 (0.68) |
1.41 (0.68) |
0.99 (0.65) |
|
Physician global assessment |
56 (18) |
43 (22) |
59 (16) |
30 (19) |
|
CRP (mg/L) |
13.7 (14.9) |
14.6 (18.7) |
15.3 (19.0) |
7.1 (19.1) |
The percent of ACR20 responders by visit for Study RA-IV is shown in Figure 4. Similar responses were observed for XELJANZ in Studies RA-I, II, III, V, and VI.
Figure 4: Percentage of ACR20 Responders by Visit Through Month 6 in Study RA-IV
Principal Display Panel 240 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 240 mL Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
NDC 63539-029-01
ALWAYS DISPENSE WITH
MEDICATION GUIDE
Pfizer
Xeljanz®
(tofacitinib) oral solution
1 mg/mL
240 mL
Rx only
Principal Display Panel 240 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 240 mL Bottle Carton)
PROFESSIONAL SAMPLE – NOT FOR SALE
NDC 63539-029-02
ALWAYS DISPENSE WITH MEDICATION GUIDE
Pfizer
Xeljanz®
(tofacitinib) oral solution
1 mg/mL
Contents:
-
•Oral solution bottle
-
•1 Oral dosing syringe
-
•1 Press-in bottle adapter
-
•Prescribing Information
-
•Medication Guide
-
•Instructions for Use
240 mL
Rx only
14.3 Clinical Studies in Ankylosing Spondylitis
The ankylosing spondylitis (AS) clinical development program with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) included one placebo-controlled trial (Study AS-I) in adults with active AS. Patients had active disease as defined by both Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and back pain score (BASDAI question 2) of greater or equal to 4 despite non-steroidal anti-inflammatory drug (NSAID), corticosteroid or disease modifying anti-rheumatic drug (DMARD) therapy.
5.3 Malignancy and Lymphoproliferative Disorders
Malignancies and lymphoproliferative disorders may occur with XELJANZ (tablets and oral solution) and XELJANZ XR (extended-release tablets). Malignancies, including lymphomas and solid cancers, were observed in clinical studies of XELJANZ [see Adverse Reactions (6.1)].
Other malignancies were observed in XELJANZ clinical studies and the postmarketing setting, including, but not limited to, lung cancer, breast cancer, melanoma, prostate cancer, and pancreatic cancer.
In RA Safety Study 1, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in patients treated with XELJANZ tablets 5 mg or 10 mg twice a day compared with TNF blockers. The incidence rate of malignancies (excluding NMSC) per 100 patient-years was 1.13 for XELJANZ tablets 10 mg twice a day, 1.13 for XELJANZ tablets 5 mg twice a day, and 0.77 for TNF blockers. Patients who are current or past smokers are at additional increased risk [see Clinical Studies (14.6)].
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with XELJANZ tablets 5 mg twice a day and XELJANZ tablets 10 mg twice a day compared to those treated with TNF blockers. The incidence rate of lymphomas per 100 patient-years was 0.11 for XELJANZ tablets 10 mg twice a day, 0.07 for XELJANZ tablets 5 mg twice a day, and 0.02 for TNF blockers. The incidence rate of lung cancers per 100 patient-years among current and past smokers was 0.59 for XELJANZ tablets 10 mg twice a day, 0.48 for XELJANZ tablets 5 mg twice a day, and 0.27 for TNF blockers [see Clinical Studies (14.6)].
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with XELJANZ/XELJANZ XR, particularly in patients with a known malignancy (other than a successfully treated NMSC), patients who develop a malignancy while on treatment, and patients who are current or past smokers. XELJANZ 10 mg twice daily (or XELJANZ XR 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3 , 2.4) ].
Principal Display Panel 5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-012-02
Pfizer
Xeljanz
™
(tofacitinib tablets)
5 mg*
60 Tablets
Rx only
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-016-02
Pfizer
Xeljanz
®
(tofacitinib) tablets
10 mg*
10mg Is Recommended Only In
Ulcerative Colitis
60 Tablets
Rx only
Principal Display Panel 11 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 11 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
NDC 63539-501-30
Pfizer
Xeljanz
®
XR
(tofacitinib) tablets
Extended Release Tablets
11 mg*
30 Tablets
Rx only
Principal Display Panel 22 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 22 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE
Pfizer
NDC 63539-502-30
Xeljanz® XR
(tofacitinib) tablets
Extended Release Tablets
22 mg*
22mg Is Recommended Only In
Ulcerative Colitis
30 Tablets
Rx only
1.4 Polyarticular Course Juvenile Idiopathic Arthritis
XELJANZ (tablets and oral solution) are indicated for the treatment of pediatric patients 2 years of age and older with of active polyarticular course juvenile idiopathic arthritis (pcJIA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of XELJANZ in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
2.5 Recommended Dosage in Adults With Ulcerative Colitis (2.5 Recommended Dosage in Adults with Ulcerative Colitis)
Table 3 displays the recommended dosage of XELJANZ tablets and XELJANZ XR (extended-release tablets) in adult patients with ulcerative colitis (UC) [see Indications and Usage (1.5)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. Table 4 displays the recommended dosage modification for patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and patients with lymphopenia, neutropenia, or anemia.
| Adults |
XELJANZ
tablets |
XELJANZ XR
(extended-release tablets) |
|---|---|---|
|
Patients with Normal Renal and Hepatic Function |
Induction: 10 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 10 mg twice daily for a maximum of 16 weeks. Discontinue 10 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 22 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 22 mg once daily for a maximum of 16 weeks. Discontinue 22 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Maintenance: 5 mg twice daily. For patients with loss of response during maintenance treatment, may consider a dosage of 10 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: 11 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 22 mg once daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dose needed to maintain response. |
|
|
Recommended Dosage in Patients with Renal Impairment (RI) Tofacitinib PK was evaluated in subjects with varying degrees of renal impairment, where the severity of renal impairment was defined based on creatinine clearance (CLcr) estimated using the Cockcroft‑Gault equation: CLcr >80 mL/min (normal renal function); CLcr >50 and ≤80 mL/min (mild renal impairment); ≥30 and ≤50 mL/min (moderate renal impairment); <30 mL/min (severe renal impairment).
|
||
|
Mild RI |
Same as patients with normal renal function. |
|
|
Moderate RI |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Severe RI |
Maintenance: 5 mg once daily.
|
Maintenance: XELJANZ XR is not recommended. See Maintenance Dosage for XELJANZ tablets for Moderate or Severe RI.
|
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
||
|
Mild HI |
Same as patients with normal hepatic function. |
|
|
Moderate HI |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Maintenance: 5 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 5 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: XELJANZ XR is not recommended. See Maintenance Dosage for XELJANZ tablets for Moderate HI. |
|
|
Severe HI |
Use of XELJANZ tablets/XELJANZ XR is not recommended. |
| Adults |
XELJANZ
Tablets |
XELJANZ XR
(extended-release tablets) |
|---|---|---|
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
||
|
Strong CYP2C19 inhibitor(s) |
No dosage modification is recommended. |
|
|
Moderate CYP2C19 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
Induction: 5 mg twice daily for at least 8 weeks [see Clinical Studies (14.5)]; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 5 mg twice daily for a maximum of 16 weeks. Discontinue 5 mg twice daily after 16 weeks if adequate therapeutic response is not achieved. |
Induction: 11 mg once daily for at least 8 weeks; evaluate patients and transition to maintenance therapy depending on therapeutic response. If needed continue 11 mg once daily for a maximum of 16 weeks. Discontinue 11 mg once daily after 16 weeks if adequate therapeutic response is not achieved. |
|
Strong CYP3A4 inhibitor(s) |
||
|
Maintenance: 5 mg once daily. For patients with loss of response during maintenance treatment, may consider a dosage of 5 mg twice daily (limited to the shortest duration), with careful consideration of the benefits and risks for the individual patient. Use the lowest effective dosage needed to maintain response. |
Maintenance: XELJANZ XR is not recommended. see Maintenance Dosage for XELJANZ tablets for Strong CYP3A4 inhibitors. |
|
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
||
|
Lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
|
ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
|
ANC 500 to 1000 cells/mm3 |
If taking:
|
If taking:
|
|
Hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 39-week toxicology study in monkeys, tofacitinib at exposure levels approximately 6 times the recommended dose of 5 mg twice daily, and approximately 3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 5 mg/kg twice daily) produced lymphomas. No lymphomas were observed in this study at exposure levels 1 times the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg twice daily).
The carcinogenic potential of tofacitinib was assessed in 6-month rasH2 transgenic mouse carcinogenicity and 2-year rat carcinogenicity studies. Tofacitinib, at exposure levels approximately 34 times the recommended dose of 5 mg twice daily, and approximately 17 times the 10 mg twice daily dose (on an AUC basis at oral doses of 200 mg/kg/day) was not carcinogenic in mice.
In the 24-month oral carcinogenicity study in Sprague-Dawley rats, tofacitinib caused benign Leydig cell tumors, hibernomas (malignancy of brown adipose tissue), and benign thymomas at doses greater than or equal to 30 mg/kg/day (approximately 42 times the exposure levels at the recommended dose of 5 mg twice daily, and approximately 21 times the 10 mg twice daily dose on an AUC basis). The relevance of benign Leydig cell tumors to human risk is not known.
Tofacitinib was not mutagenic in the bacterial reverse mutation assay. It was positive for clastogenicity in the in vitro chromosome aberration assay with human lymphocytes in the presence of metabolic enzymes, but negative in the absence of metabolic enzymes. Tofacitinib was negative in the in vivo rat micronucleus assay and in the in vitro CHO-HGPRT assay and the in vivo rat hepatocyte unscheduled DNA synthesis assay.
In rats, tofacitinib at exposure levels approximately 17 times the recommended dose of 5 mg twice daily, and approximately 8.3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 10 mg/kg/day) reduced female fertility due to increased post-implantation loss. There was no impairment of female rat fertility at exposure levels of tofacitinib equal to the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg/day). Tofacitinib exposure levels at approximately 133 times the recommended dose of 5 mg twice daily, and approximately 67 times the 10 mg twice daily dose (on an AUC basis at oral doses of 100 mg/kg/day) had no effect on male fertility, sperm motility, or sperm concentration.
2.1 Recommended Evaluations and Immunization Prior to Treatment Initiation
Prior to initiating XELJANZ (tablets and oral solution) or XELJANZ XR (extended-release tablets), consider performing the following:
-
•Active and latent tuberculosis (TB) infection evaluation: If the patient has latent TB, treat for TB prior to XELJANZ/XELJANZ XR treatment [see Warnings and Precautions (5.1)].
-
•Viral hepatitis screening in accordance with clinical guidelines [see Warnings and Precautions (5.1)].
-
•A complete blood count: Avoid initiation of XELJANZ/XELJANZ XR treatment in patients with a lymphocyte count less than 500 cells/mm3, absolute neutrophil count less than 1000 cells/mm3, or hemoglobin level less than 9 g/dL [see Warnings and Precautions (5.8)].
-
•Baseline hepatic function evaluation: XELJANZ/XELJANZ XR is not recommended for patients with severe hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
-
•Update immunizations according to current immunization guidelines. The interval between live vaccinations and initiation of XELJANZ/XELJANZ XR should be in accordance with current vaccination guidelines regarding immunosuppressive agents [see Warnings and Precautions (5.9)].
14.4 Clinical Studies in Polyarticular Course Juvenile Idiopathic Arthritis
The efficacy of XELJANZ (tablets and oral solution) for pcJIA was assessed in Study pcJIA-I (NCT02592434). This was a 44-week, two-part study (that consisted of an 18-week, open-label, run-in phase, followed by a 26-week double-blind, placebo-controlled, randomized withdrawal phase) in pediatric patients 2 years to 17 years of age with active rheumatoid factor (RF) negative polyarthritis, RF positive polyarthritis, extended oligoarthritis, and systemic JIA without systemic manifestations who had an inadequate response or intolerance to at least one DMARD which could have included MTX or biologic agents. This study also included patients ages 2 years to 17 years of age with active juvenile psoriatic arthritis (jPsA) and enthesitis-related arthritis (ERA) who had an inadequate response to NSAIDs. Although the clinical studies included some patients who are TNF blocker-naïve, XELJANZ is not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.4)].
Patients received XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) for 18 weeks (run-in phase) followed by randomization to either XELJANZ (dosed at 5 mg twice daily or body weight-based equivalent twice daily) or placebo for 26 weeks (double-blind phase). Only patients who achieved at least a JIA ACR30 response at the end of the run-in phase were randomized (1:1) to the double-blind phase. Treatment with a stable dose of MTX was permitted but was not required during the study. Concurrent use of biologics or DMARDs other than MTX was not permitted in the study.
Baseline Disease Characteristics
A total of 225 pediatric patients with JIA (56 male and 169 female) with active polyarthritis were enrolled in the run-in phase including RF negative (104), RF positive (39), extended oligoarthritis (28), systemic JIA without systemic manifestations (13), jPsA (20), and ERA (21). Patients had a mean (SD) disease duration of 3.8 ± 3.5 years, and a mean (SD) number of active joints of 12.2 ± 8.1.
Efficacy Results
Of the 225 patients, 173 (77%) patients achieved JIA ACR30 response at Week 18 and were randomized into the double-blind phase to either XELJANZ (n=88) or placebo (n=85). At the conclusion of the 18-week, open-label, run-in phase, pediatric ACR 30/50/70 responses were 77%, 70%, and 49%, respectively.
In both the run-in and double-blind phases, approximately one-third of the patients were taking concomitant oral corticosteroids, and approximately two-thirds were taking concomitant MTX.
The primary endpoint was the occurrence of disease flare at Week 44 relative to the double-blind phase baseline at Week 18. Disease flare was defined (according to Pediatric Rheumatology Collaborative Study Group (PRCSG)/Pediatric Rheumatology International Trials Organization (PRINTO) Disease Flare criteria) as worsening of ≥30% in 3 or more of the 6 JIA core response variables with no more than 1 of the remaining JIA core response variables improving by ≥30%.
XELJANZ-treated patients experienced significantly fewer disease flares at Week 44 compared to placebo-treated patients (31% [27/88] vs. 55% [47/85]; difference in proportions -25% [95% CI: -39%, -10%]; p=0.0007). The occurrence of disease flare by visit in Study pcJIA-I is shown in Figure 7.
| BID = twice daily; SE = standard error; N = total number of patients. |
| The 26-week double-blind phase is from Week 18 through Week 44 on and after randomization day. |
14.6 Safety Study in Adults With Rheumatoid Arthritis (xeljanz Versus Tnf Blocker) (14.6 Safety Study in Adults with Rheumatoid Arthritis (XELJANZ Versus TNF-blocker))
A randomized open-label trial (RA Safety Study 1; NCT02092467) was conducted to evaluate safety with XELJANZ tablets (referred to as “XELJANZ” in this subsection of labeling) at two doses, 5 mg twice daily (N=1455) and 10 mg twice daily (N=1456), versus the TNF-blocker control (N=1451) in RA patients 50 years of age and older with at least one cardiovascular risk factor. The co-primary endpoints were adjudicated MACE (defined as cardiovascular death, non-fatal MI, and non-fatal stroke) and adjudicated malignancy (excluding non-melanoma skin cancer). The study was designed to exclude a prespecified risk margin of 1.8 for the hazard ratio of combined XELJANZ regimens versus the TNF-blocker control for each co-primary endpoint. An independent committee conducted a blinded evaluation of the co-primary endpoints according to predefined criteria (adjudication). The study was event-driven and patients were followed until a sufficient number of primary outcome events accrued. Other endpoints included mortality, serious infections, and thromboembolic events. The median on-study follow-up time was 4 years.
The mean age of the population was 61 years (range: 50 to 88 years). Most patients were female (78%) and Caucasian (77%). Patients had a diagnosis of RA for a mean of 10 years, and a median swollen and tender joint count of 11 and 15 respectively. Cardiovascular risk factors included cigarette smoking (current or past) (48%), hypertension (66%), high density lipoprotein <40 mg/dL (12%), diabetes mellitus (17%), family history of premature coronary heart disease (15%), extra-articular disease associated with RA (37%), and history of coronary artery disease (11%).
The non-inferiority criterion was not met for the primary comparison of the combined XELJANZ dosages to TNF blockers since the upper limit of the 95% CI exceeded the pre-specified non-inferiority criterion of 1.8 (for MACE, the upper limit of the 95% CI was 1.94; for malignancies excluding NMSC, the upper limit of the 95% CI was 2.09).
Table 22 shows the study results for each of the co-primary endpoints, and other endpoints. There was an increased risk of death, MACE, malignancies, serious infections, and thromboembolic events associated with both dosages of XELJANZ.
| Endpoint |
TNF Blocker
N=1451 PY=5468 |
XELJANZ
5 mg Twice Daily N=1455 PY=5490 |
XELJANZ
10 mg Twice Daily N=1456 PY=5298 |
|---|---|---|---|
| Note: XELJANZ 10 mg twice daily was discontinued by the Data Monitoring Committee due to safety concerns, and ongoing patients switched from XELJANZ 10 mg to XELJANZ 5 mg. The column “XELJANZ 10 mg Twice Daily” includes all events and follow-up for patients randomized to XELJANZ 10 mg twice daily. A XELJANZ (refers to tablets and oral solution) 10 mg twice daily (or a XELJANZ XR 22 mg once daily) dosage is not recommended for the treatment of RA, PsA, AS, or pcJIA [see Dosage and Administration (2.3)]. N indicates number of patients; n indicates number of patients with events. IR indicates incidence rate per 100 person-year (PY). NMSC: Non-melanoma Skin Cancer; MACE: Major Adverse Cardiac Events; HR: Hazard Ratio; DVT: Deep Vein Thrombosis; PE: Pulmonary Embolism; VTE: Venous Thromboembolism, first occurrence of a VTE, defined as the composite of adjudicated DVT and adjudicated PE; ATE: Arterial Thromboembolism; TE: Thromboembolism, first occurrence of a TE, defined as the composite of adjudicated VTE and unadjudicated ATE. |
|||
|
MACE, n [IR] |
43 [0.79] |
50 [0.91] |
59 [1.11] |
|
HR (95% CI) HR (95%) CI for XELJANZ vs. TNF Blocker (Univariate Cox Proportional Hazard Model).
|
1.16 (0.77, 1.74) |
1.41 (0.95, 2.10) |
|
|
MI, MI and Stroke include fatal and non-fatal events. n [IR]
|
11 [0.20] |
20 [0.36] |
21 [0.39] |
|
HR (95% CI) |
1.81 (0.87, 3.79) |
1.97 (0.95, 4.09) |
|
|
Stroke, n [IR] |
20 [0.36] |
18 [0.33] |
21 [0.39] |
|
HR (95% CI) |
0.89 (0.47, 1.69) |
1.08 (0.59, 2.00) |
|
|
Cardiovascular Death, n [IR] |
15 [0.27] |
18 [0.32] |
25 [0.47] |
|
HR (95% CI) |
1.20 (0.60, 2.37) |
1.71 (0.90, 3.24) |
|
|
Malignancies Excl. NMSC, n [IR] |
42 [0.77] |
62 [1.13] |
60 [1.13] |
|
HR (95% CI) |
1.47 (1.00, 2.18) |
1.48 (1.00, 2.19) |
|
|
Malignancies Excl. NMSC
Data and analyses for Malignancies excluding NMSC for current and ex-smokers are included. There were 720 current and ex-smokers randomized to XELJANZ 5 mg, 704 to XELJANZ 10 mg, and 679 to TNF blockers.
|
25 [0.99] |
41 [1.53] |
48 [1.91] |
|
HR (95% CI) |
1.55 (0.94, 2.55) |
1.94 (1.19, 3.14) |
|
|
All Death |
38 [0.69] |
49 [0.88] |
66 [1.23] |
|
HR (95% CI) |
1.29 (0.84, 1.96) |
1.79 (1.20, 2.66) |
|
|
Serious Infections |
133 [2.52] |
155 [2.95] |
184 [3.65] |
|
HR (95% CI) |
1.17 (0.93, 1.47) |
1.44 (1.15, 1.80) |
|
|
DVT |
9 [0.16] |
12 [0.22] |
15 [0.28] |
|
HR (95% CI) |
1.33 (0.56, 3.15) |
1.72 (0.75, 3.92) |
|
|
PE |
3 [0.05] |
10 [0.18] |
26 [0.49] |
|
HR (95% CI) |
3.32 (0.91, 12.08) |
8.95 (2.71, 29.56) |
|
|
VTE |
12 [0.22] |
18 [0.33] |
36 [0.68] |
|
HR (95% CI) |
1.50 (0.72, 3.10) |
3.10 (1.61, 5.96) |
|
|
ATE |
45 [0.83] |
51 [0.93] |
55 [1.04] |
|
HR (95% CI) |
1.13 (0.76, 1.69) |
1.26 (0.85, 1.87) |
|
|
TE |
56 [1.03] |
67 [1.23] |
86 [1.65] |
|
HR (95% CI) |
1.19 (0.84, 1.70) |
1.60 (1.14, 2.23) |
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with XELJANZ 5 mg twice a day and XELJANZ 10 mg twice a day compared to those treated with TNF blockers. Lymphoma was reported for 4 patients who received XELJANZ 5 mg twice a day, 6 patients who received XELJANZ 10 mg twice a day, and 1 patient who received TNF blockers (Incidence Rate [IR] of 0.07, 0.11, and 0.02 per 100 patient-years, respectively). Among current and past smokers, lung cancer was reported for 13 patients who received XELJANZ 5 mg twice a day, 15 patients who received XELJANZ 10 mg twice a day, and 7 patients who received TNF blockers (IR of 0.48, 0.59, and 0.27 per 100 patient-years, respectively).
Given these increased risks, XELJANZ (tablets and oral solution) 10 mg twice daily (or XELJANZ XR (extended-release tablets) 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, AS or pcJIA [see Dosage and Administration (2.3, 2.4)].
Warning: Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis (WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS)
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS
See full prescribing information for complete boxed warning.
-
•Increased risk of serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), leading to hospitalization or death. Interrupt XELJANZ/XELJANZ XR treatment if serious infection occurs until the infection is controlled. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test. (5.1)
-
•Higher rate of all-cause mortality, including sudden cardiovascular (CV) death with XELJANZ vs. TNF blockers in rheumatoid arthritis (RA) patients. (5.2)
-
•Malignancies have occurred in patients treated with XELJANZ. Higher rate of lymphomas and lung cancers with XELJANZ vs. TNF blockers in RA patients. (5.3)
-
•Higher rate of major adverse CV events (defined as CV death, myocardial infarction, and stroke) with XELJANZ vs. TNF blockers in RA patients. (5.4)
-
•Thrombosis has occurred in patients treated with XELJANZ. Increased incidence of pulmonary embolism, venous and arterial thrombosis with XELJANZ vs. TNF blockers in RA patients. (5.5)
5.10 Risk of Gastrointestinal Obstruction With Xeljanz Xr A Non Deformable Extended Release Formulation (5.10 Risk of Gastrointestinal Obstruction with XELJANZ XR - A Non-Deformable Extended-Release Formulation)
Gastrointestinal obstruction may occur with XELJANZ XR (extended-release tablets). Avoid use of XELJANZ XR in patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs utilizing a non-deformable extended-release formulation.
2.3 Recommended Dosage in Adults With Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis (2.3 Recommended Dosage in Adults with Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis)
Table 1 displays the recommended dosage of XELJANZ tablets and XELJANZ XR (extended-release tablets) for adults with RA, PsA, and AS [see Indication and Usage (1.1, 1.2, 1.3)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. The table also displays the recommended dosage modifications for patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and patients with lymphopenia, neutropenia, or anemia.
|
Adults |
XELJANZ Tablets |
XELJANZ XR (extended-release tablets) |
|
Patients with Normal Renal and Hepatic Function Excludes patients who concomitantly use XELJANZ tablets/XELJANZ XR with strong CYP3A4 inhibitor(s) or moderate CYP3A4 inhibitor(s) and strong CYP2C19 inhibitor(s), as well as patients with lymphocyte count less than 500 cells/mm3, ANC <1000 cells/mm3, or hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL.
|
5 mg twice daily |
11 mg once daily |
|
Recommended Dosage in Patients with Renal Impairment (RI) Tofacitinib PK was evaluated in subjects with varying degrees of renal impairment, where the severity of renal impairment was defined based on creatinine clearance (CLcr) estimated using the Cockcroft‑Gault equation: CLcr >80 mL/min (normal renal function); >50 and ≤80 mL/min (mild renal impairment); ≥30 and ≤50 mL/min (moderate renal impairment); <30 mL/min (severe renal impairment).
|
||
|
Mild RI (CLcr >50 and ≤80 mL/min) |
5 mg twice daily |
11 mg once daily |
|
Moderate RI (CLcr ≥30 and ≤50 mL/min) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Severe RI (CLcr <30 mL/min) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
For patients undergoing hemodialysis, administer the dose after the dialysis session on dialysis days. If a dose was taken before the dialysis procedure, supplemental doses are not recommended after dialysis. |
||
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
||
|
Mild HI (Child-Pugh A) |
5 mg twice daily |
11 mg once daily |
|
Moderate HI (Child-Pugh B) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Severe HI (Child-Pugh C) |
Use of XELJANZ tablets/XELJANZ XR is not recommended. |
|
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
||
|
Strong CYP2C19 inhibitor(s) |
5 mg twice daily |
11 mg once daily |
|
Moderate CYP2C19 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) |
||
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
5 mg once daily |
XELJANZ tablets 5 mg once daily |
|
Strong CYP3A4 inhibitor(s) |
||
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
||
|
Patients with lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
|
Patients with ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
|
Patients with ANC 500 to 1000 cells/mm3 |
Interrupt dosing. When ANC is greater than 1000, resume 5 mg twice daily. |
Interrupt dosing. When ANC is greater than 1000, resume 11 mg once daily. |
|
Patients with hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
2.4 Recommended Dosage in Pediatric Patients 2 Years of Age and Older With Psoriatic Arthritis Or Polyarticular Course Juvenile Idiopathic Arthritis (2.4 Recommended Dosage in Pediatric Patients 2 Years of Age and Older with Psoriatic Arthritis or Polyarticular Course Juvenile Idiopathic Arthritis)
Table 2 displays the recommended body weight-based dosages for XELJANZ tablets and XELJANZ oral solution in pediatric patients 2 years of age and older with PsA or pcJIA [see Indication and Usage (1.2, 1.4)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. The table also includes recommended dosage modification for pediatric patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and pediatric patients with lymphopenia, neutropenia, or anemia.
Administer XELJANZ oral solution using the included press-in bottle adapter and oral dosing syringe [see Instructions for Use].
| Pediatric Patients 2 Years of Age and Older | XELJANZ tablets and XELJANZ oral solution |
|---|---|
|
Patients with Normal Renal and Hepatic Function
|
|
|
Recommended Dosage in Patients with Renal Impairment (RI) |
|
|
Mild RI |
Same as patients with normal renal function. |
|
Moderate RI |
|
|
Severe RI |
|
|
For patients undergoing hemodialysis, administer the dose after the dialysis session on dialysis days. If a dose was taken before the dialysis procedure, supplemental doses are not recommended after dialysis. |
|
|
Recommended Dosage in Patients with Hepatic Impairment (HI) |
|
|
Mild HI |
Same as patients with normal hepatic function. |
|
Moderate HI |
|
|
Severe HI |
Use of XELJANZ tablets/XELJANZ oral solution is not recommended. |
|
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s) |
|
|
Strong CYP2C19 inhibitor(s) |
No dosage modification is recommended. |
|
Moderate CYP2C19 inhibitor(s) |
|
|
Moderate CYP3A4 inhibitor(s) |
|
|
Moderate CYP3A4 inhibitor(s) with strong CYP2C19 inhibitor(s) (e.g., fluconazole) |
|
|
Strong CYP3A4 inhibitor(s) |
|
|
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia |
|
|
Patients with lymphocyte count less than 500 cells/mm3, confirmed by repeat testing |
Discontinue dosing. |
|
Patients with ANC less than 500 cells/mm3 |
Discontinue dosing. |
|
Patients with ANC 500 to 1000 cells/mm3 |
Interrupt dosing until ANC is greater than 1000 cells/mm3. |
|
Patients with hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL |
Interrupt dosing until hemoglobin values have normalized. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:00.236747 · Updated: 2026-03-14T22:45:11.004418