Drug Facts
68e3174e-cb96-4739-a723-c36b2389f4f6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
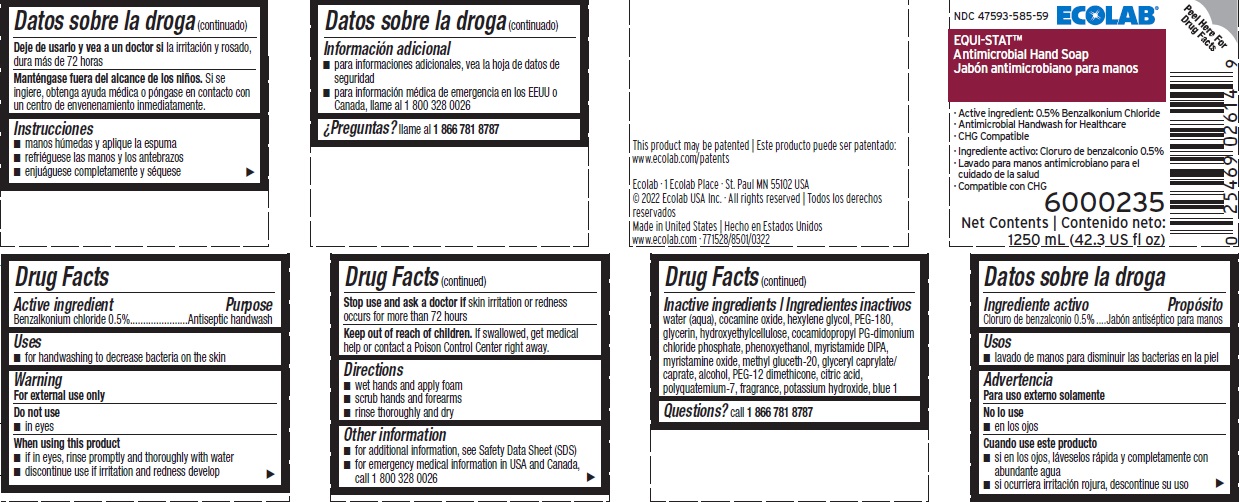
Benzalkonium Chloride, 0.5%
Purpose
Antiseptic handwash
Medication Information
Purpose
Antiseptic handwash
Description
Benzalkonium Chloride, 0.5%
Uses
for handwashing to decrease bacteria on the skin
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3
Stop use and ask a doctor if skin irritation or redness occurs for more than 72 hours
Section 51727-6
Inactive ingredients water (aqua), cocamine oxide, hexylene glycol, PEG-180, glycerin, hydroxyethylcellulose, cocamidopropyl PG-dimonium chloride phosphate, phenoxyethanol, myristamide DIPA, myristamine oxide, methyl gluceth-20, glyceryl caprylate/caprate, alcohol, PEG-12 dimethicone, citric acid, polyquatemium-7, fragrance, potassium hydroxide, blue 1
Section 53413-1
Questions? call 1-866-781-8787
Warnings
For external use only
Directions
- wet hands and apply product
- scrub hands and forearms
- rinse thoroughly and dry
Do Not Use
- in eyes
Active Ingredient
Benzalkonium Chloride, 0.5%
Other Information
- for additional information, see Safety Data Sheet (SDS)
- for emergency medical information in USA and Canada, call 1 800 328 0026
When Using the Product
- if in eyes, rinse promptly and thoroughly with water
- discontinue use if irritation and redness develop
Principal Display Panel and Representative Label
NDC 47593-585-59 ECOLAB
EQUI-STAT ™
Antimicrobial Hand Soap
Active Ingredient: 0.5% Benzalkonium Chloride
Antimicrobial Handwash for Healthcare
CHG Compatible
Net Contents
1250 mL (42.3 US fl oz) 6000235
This product may be patented | Este producto puede ser patentado: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA · tel: 1 800 35 CLEAN (352 5326)
© 2022 Ecolab USA Inc. · All rights reserved | Todos los derechos reservados
Made in U.S.A. | Hecho en EE.UU.
www.ecolab.com · 771528/8501/0322
Structured Label Content
Uses
for handwashing to decrease bacteria on the skin
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3 (50569-3)
Stop use and ask a doctor if skin irritation or redness occurs for more than 72 hours
Section 51727-6 (51727-6)
Inactive ingredients water (aqua), cocamine oxide, hexylene glycol, PEG-180, glycerin, hydroxyethylcellulose, cocamidopropyl PG-dimonium chloride phosphate, phenoxyethanol, myristamide DIPA, myristamine oxide, methyl gluceth-20, glyceryl caprylate/caprate, alcohol, PEG-12 dimethicone, citric acid, polyquatemium-7, fragrance, potassium hydroxide, blue 1
Section 53413-1 (53413-1)
Questions? call 1-866-781-8787
Purpose
Antiseptic handwash
Warnings
For external use only
Directions
- wet hands and apply product
- scrub hands and forearms
- rinse thoroughly and dry
Do Not Use (Do not use)
- in eyes
Active Ingredient (Active ingredient)
Benzalkonium Chloride, 0.5%
Other Information (Other information)
- for additional information, see Safety Data Sheet (SDS)
- for emergency medical information in USA and Canada, call 1 800 328 0026
When Using the Product (When using the product)
- if in eyes, rinse promptly and thoroughly with water
- discontinue use if irritation and redness develop
Principal Display Panel and Representative Label (Principal display panel and representative label)
NDC 47593-585-59 ECOLAB
EQUI-STAT ™
Antimicrobial Hand Soap
Active Ingredient: 0.5% Benzalkonium Chloride
Antimicrobial Handwash for Healthcare
CHG Compatible
Net Contents
1250 mL (42.3 US fl oz) 6000235
This product may be patented | Este producto puede ser patentado: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA · tel: 1 800 35 CLEAN (352 5326)
© 2022 Ecolab USA Inc. · All rights reserved | Todos los derechos reservados
Made in U.S.A. | Hecho en EE.UU.
www.ecolab.com · 771528/8501/0322
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:22.179523 · Updated: 2026-03-14T23:03:24.276357