These Highlights Do Not Include All The Information Needed To Use Diclofenac Potassium For Oral Solution Safely And Effectively. See Full Prescribing Information For Diclofenac Potassium For Oral Solution.
68a926a2-b818-420f-9c9a-fd91dc5a8f36
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1 ) Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4 , 5.1 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2 )
Indications and Usage
Diclofenac potassium for oral solution is indicated for the acute treatment of migraine attacks with or without aura in adults (18 years of age or older).
Dosage and Administration
Single 50 mg dose; mix single packet contents with 1 to 2 ounces or 2 to 4 tablespoons (30 to 60 mL) of water prior to administration Use the lowest effective dose for shortest duration consistent with individual patient treatment goals ( 2.1 )
Warnings and Precautions
Hepatotoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop ( 5.3 , 8.6 , 12.3 ) Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.4 , 7 ) Heart Failure and Edema: Avoid use of diclofenac potassium for oral solution in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.5 ) Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac potassium for oral solution in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.6 ) Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs ( 5.7 ) Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac potassium for oral solution is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity) ( 5.8 ) Serious Skin Reactions: Discontinue diclofenac potassium for oral solution at first appearance of skin rash or other signs of hypersensitivity ( 5.9 ). Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue and evaluate clinically ( 5.10 ) Medication Overuse Headache: Detoxification may be necessary. ( 5.11 ) Fetal Toxicity: Limit use of NSAIDs, including diclofenac potassium for oral solution, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal dysfunction and premature closure of the fetal ductus arteriosus ( 5.12 , 8.1 ) Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.13 , 7 )
Contraindications
Diclofenac potassium for oral solution is contraindicated in the following patients: Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [see Warnings and Precautions (5.7 , 5.9) ] History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7 , 5.8) ] In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1) ]
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1) ] GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2) ] Hepatotoxicity [see Warnings and Precautions (5.3) ] Hypertension [see Warnings and Precautions (5.4) ] Heart Failure and Edema [see Warnings and Precautions (5.5) ] Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6) ] Anaphylactic Reactions [see Warnings and Precautions (5.7) ] Serious Skin Reactions [see Warnings and Precautions (5.9) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.10) ] Medication Overuse Headache [see Warnings and Precautions (5.11) ] Hematologic Toxicity [see Warnings and Precautions (5.13) ]
Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac. Table 2: Clinically Significant Drug Interactions with Diclofenac Drugs That Interfere with Hemostasis Clinical Impact: Diclofenac and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of diclofenac and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of diclofenac potassium for oral solution with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.13) ]. Aspirin Clinical Impact: Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3) ] . Intervention: Concomitant use of diclofenac potassium for oral solution and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.13) ]. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of diclofenac potassium for oral solution and ACE-inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of diclofenac potassium for oral solution and ACE-inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6) ] . Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac potassium for oral solution with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6) ] . Digoxin Clinical Impact: The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of diclofenac potassium for oral solution and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac potassium for oral solution and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Intervention: During concomitant use of diclofenac potassium for oral solution and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of diclofenac potassium for oral solution and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of diclofenac potassium for oral solution and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2) ] . Intervention: The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of diclofenac potassium for oral solution and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of NSAIDs and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. Inhibitors of Cytochrome P450 2C9 Clinical Impact: Diclofenac is metabolized predominantly by Cytochrome P-450 CYP2C9. Co-administration of medications that inhibit CYP2C9 may affect the pharmacokinetics of diclofenac [see Clinical Pharmacology (12.3) ]. Intervention: During concomitant use of diclofenac potassium for oral solution and drugs that inhibit CYP2C9, an increase in the duration between diclofenac potassium for oral solution doses for subsequent migraine attacks may be necessary.
Storage and Handling
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water. NDC 51672-4240-8 Individual Diclofenac Potassium for Oral Solution Packet NDC 51672-4240-6 Carton of nine (9) Diclofenac Potassium for Oral Solution Packets
How Supplied
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water. NDC 51672-4240-8 Individual Diclofenac Potassium for Oral Solution Packet NDC 51672-4240-6 Carton of nine (9) Diclofenac Potassium for Oral Solution Packets
Medication Information
Warnings and Precautions
Hepatotoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop ( 5.3 , 8.6 , 12.3 ) Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.4 , 7 ) Heart Failure and Edema: Avoid use of diclofenac potassium for oral solution in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.5 ) Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac potassium for oral solution in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.6 ) Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs ( 5.7 ) Exacerbation of Asthma Related to Aspirin Sensitivity: Diclofenac potassium for oral solution is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity) ( 5.8 ) Serious Skin Reactions: Discontinue diclofenac potassium for oral solution at first appearance of skin rash or other signs of hypersensitivity ( 5.9 ). Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue and evaluate clinically ( 5.10 ) Medication Overuse Headache: Detoxification may be necessary. ( 5.11 ) Fetal Toxicity: Limit use of NSAIDs, including diclofenac potassium for oral solution, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal dysfunction and premature closure of the fetal ductus arteriosus ( 5.12 , 8.1 ) Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.13 , 7 )
Indications and Usage
Diclofenac potassium for oral solution is indicated for the acute treatment of migraine attacks with or without aura in adults (18 years of age or older).
Dosage and Administration
Single 50 mg dose; mix single packet contents with 1 to 2 ounces or 2 to 4 tablespoons (30 to 60 mL) of water prior to administration Use the lowest effective dose for shortest duration consistent with individual patient treatment goals ( 2.1 )
Contraindications
Diclofenac potassium for oral solution is contraindicated in the following patients: Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [see Warnings and Precautions (5.7 , 5.9) ] History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7 , 5.8) ] In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1) ]
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1) ] GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2) ] Hepatotoxicity [see Warnings and Precautions (5.3) ] Hypertension [see Warnings and Precautions (5.4) ] Heart Failure and Edema [see Warnings and Precautions (5.5) ] Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6) ] Anaphylactic Reactions [see Warnings and Precautions (5.7) ] Serious Skin Reactions [see Warnings and Precautions (5.9) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.10) ] Medication Overuse Headache [see Warnings and Precautions (5.11) ] Hematologic Toxicity [see Warnings and Precautions (5.13) ]
Drug Interactions
See Table 2 for clinically significant drug interactions with diclofenac. Table 2: Clinically Significant Drug Interactions with Diclofenac Drugs That Interfere with Hemostasis Clinical Impact: Diclofenac and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of diclofenac and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of diclofenac potassium for oral solution with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.13) ]. Aspirin Clinical Impact: Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3) ] . Intervention: Concomitant use of diclofenac potassium for oral solution and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.13) ]. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of diclofenac potassium for oral solution and ACE-inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of diclofenac potassium for oral solution and ACE-inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6) ] . Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac potassium for oral solution with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6) ] . Digoxin Clinical Impact: The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of diclofenac potassium for oral solution and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of diclofenac potassium for oral solution and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Intervention: During concomitant use of diclofenac potassium for oral solution and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of diclofenac potassium for oral solution and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of diclofenac potassium for oral solution and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2) ] . Intervention: The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of diclofenac potassium for oral solution and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of NSAIDs and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. Inhibitors of Cytochrome P450 2C9 Clinical Impact: Diclofenac is metabolized predominantly by Cytochrome P-450 CYP2C9. Co-administration of medications that inhibit CYP2C9 may affect the pharmacokinetics of diclofenac [see Clinical Pharmacology (12.3) ]. Intervention: During concomitant use of diclofenac potassium for oral solution and drugs that inhibit CYP2C9, an increase in the duration between diclofenac potassium for oral solution doses for subsequent migraine attacks may be necessary.
Storage and Handling
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water. NDC 51672-4240-8 Individual Diclofenac Potassium for Oral Solution Packet NDC 51672-4240-6 Carton of nine (9) Diclofenac Potassium for Oral Solution Packets
How Supplied
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water. NDC 51672-4240-8 Individual Diclofenac Potassium for Oral Solution Packet NDC 51672-4240-6 Carton of nine (9) Diclofenac Potassium for Oral Solution Packets
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1 ) Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4 , 5.1 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2 )
Section 42229-5
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.1)] .
- Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4)and Warnings and Precautions (5.1)] .
Section 42231-1
Dispense with Medication Guide available at: www.taro.com/usa-medication-guides
| Medication Guide
Diclofenac Potassium (dye-KLOE-fen-ak poe-TAS-ee-um) for Oral Solution, USP |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: June 2024 5240062-0624-01 |
||
|
What is the most important information I should know about diclofenac potassium for oral solution?
Diclofenac potassium for oral solution contains diclofenac (a non-steroidal anti-inflammatory drug or NSAID). NSAIDs, including diclofenac potassium for oral solution, can cause serious side effects, including:
Avoid taking NSAIDs, including diclofenac potassium for oral solution, after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.
|
||
|
||
|
|
|
| Diclofenac potassium for oral solution should only be used: | ||
|
||
|
What is diclofenac potassium for oral solution?
Diclofenac potassium for oral solution is a prescription medicine used to treat migraine attacks in adults. It does not prevent or lessen the number of migraines you have, and it is not for other types of headaches. Diclofenac potassium for oral solution contains diclofenac potassium (a non-steroidal anti-inflammatory drug or NSAID). |
||
|
How should I take diclofenac potassium for oral solution?
Take diclofenac potassium for oral solution exactly as your healthcare provider tells you to take it. Take 1 dose of diclofenac potassium for oral solution to treat your migraine headache:
|
||
|
Who should not take diclofenac potassium for oral solution?
Do not take diclofenac potassium for oral solution:
|
||
Before taking diclofenac potassium for oral solution, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your doctor if you take:
|
||
|
What are the possible side effects of diclofenac potassium for oral solution?
Diclofenac potassium for oral solution can cause serious side effects, including: See " What is the most important information I should know about diclofenac potassium for oral solution?" |
||
|
||
|
||
|
|
|
| Stop taking diclofenac potassium for oral solution and call your healthcare provider right away if you get any of the following symptoms | ||
|
|
|
|
If you take too much of your NSAID, call your healthcare provider or get medical help right away.
These are not all the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
Other information about NSAIDs
|
||
|
General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals. For more information, call Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or visit ww.taro.com |
||
| Manufactured by: Taro Pharmaceutical Industries Ltd., Haifa Bay, Israel 2624761
Distributed by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 All trademarks are the property of their respective owners. |
Section 44425-7
Storage
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
10 Overdosage
Symptoms following acute NSAID overdoses have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression and coma have occurred, but were rare [see Warnings and Precautions (5.1, 5.2, 5.4, 5.6)] .
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Consider emesis and/or activated charcoal (60 to 100 grams in adults, 1 to 2 grams per kg of body weight in pediatric patients) and/or osmotic cathartic in symptomatic patients seen within four hours of ingestion or in patients with a large overdosage (5 to 10 times the recommended dosage). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment contact a poison control center (1-800-222-1222).
Anaphylactic reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
11 Description
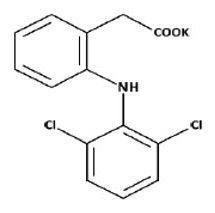
Diclofenac potassium for oral solution, USP is a nonsteroidal anti-inflammatory drug, available as a buffered soluble powder, designed to be mixed with water prior to oral administration. Diclofenac potassium, USP is a white to off-white, buffered, flavored powder for oral solution packaged in individual unit dose packets.
The chemical name is benzeneacetic acid, 2-[(2,6-dichlorophenyl)amino]-, monopotassium salt.
The molecular weight is 334.2 g/mole. Its molecular formula is C
14H
10Cl
2KNO
2, and it has the following chemical structure.
The inactive ingredients in diclofenac potassium for oral solution, USP include: aspartame (equivalent to 25 mg phenylalanine), flavoring agent (peppermint), glyceryl dibehenate, mannitol, potassium bicarbonate, and saccharin sodium.
5.4 Hypertension
NSAIDs, including diclofenac potassium for oral solution, can lead to new onset of hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Use NSAIDs, including diclofenac potassium for oral solution, with caution in patients with hypertension. Monitor blood pressure closely during the initiation of NSAID treatment and throughout the course of therapy.
Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazides, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.15)] .
Clinical studies of diclofenac potassium for oral solution did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
5.3 Hepatotoxicity
Elevations of one or more liver tests may occur during therapy with diclofenac potassium for oral solution. These laboratory abnormalities may progress, may persist, or may only be transient with continued therapy. Borderline elevations (less than 3 times the upper limit of the normal [ULN] range) or greater elevations of transaminases occurred in about 15% of diclofenac-treated patients. Of the markers of hepatic function, ALT (SGPT) is recommended for the monitoring of liver injury.
In clinical trials, meaningful elevations (i.e., more than 3 times the ULN) of AST (SGOT) occurred in about 2% of approximately 5,700 patients at some time during treatment (ALT was not measured in all studies). In an open-label, controlled trial of 3,700 patients treated for 2 to 6 months, patients were monitored at 8 weeks and 1,200 patients were monitored again at 24 weeks. Meaningful elevations of ALT and/or AST occurred in about 4% of the 3,700 patients and included marked elevations (>8 times the ULN) in about 1% of the 3,700 patients. In this open-label study, a higher incidence of borderline (less than 3 times the ULN), moderate (3 to 8 times the ULN), and marked (>8 times the ULN) elevations of ALT or AST was observed in patients receiving diclofenac when compared to other NSAIDs. Almost all meaningful elevations in transaminases were detected before patients became symptomatic [see Warnings and Precautions (5.15)].
Abnormal tests occurred during the first 2 months of therapy with diclofenac in 42 of the 51 patients in all trials who developed marked transaminase elevations. In postmarketing reports, cases of drug-induced hepatotoxicity have been reported in the first month, and in some cases, the first 2 months of NSAID therapy, but can occur at any time during treatment with diclofenac.
Postmarketing surveillance has reported cases of severe hepatic reactions, including liver necrosis, jaundice, fulminant hepatitis with and without jaundice, and liver failure. Some of these reported cases resulted in fatalities or liver transplantation.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue diclofenac potassium for oral solution immediately, and perform a clinical evaluation of the patient.
To minimize the potential risk for an adverse liver-related event in patients treated with diclofenac potassium for oral solution, use the lowest effective dose for the shortest duration possible. Exercise caution when prescribing diclofenac potassium for oral solution with concomitant drugs that are known to be potentially hepatotoxic (e.g., acetaminophen, certain antibiotics, antiepileptics). Caution patients to avoid taking nonprescription acetaminophen-containing products while using diclofenac potassium for oral solution.
14 Clinical Studies
The efficacy of diclofenac potassium for oral solution in the acute treatment of migraine headache was demonstrated in two randomized, double-blind, placebo-controlled trials.
Patients enrolled in these two trials were predominantly female (85%) and white (86%), with a mean age of 40 years (range: 18 to 65). Patients were instructed to treat a migraine of moderate to severe pain with 1 dose of study medication. Patients evaluated their headache pain 2 hours later. Associated symptoms of nausea, photophobia, and phonophobia were also evaluated. In addition, the proportion of patients who were "sustained pain free", defined as a reduction in headache severity from moderate or severe pain to no pain at 2 hours post-dose without a return of mild, moderate, or severe pain and no use of rescue medication for 24 hours post-dose, was also evaluated. In these studies, the percentage of patients achieving pain freedom 2 hours after treatment and sustained pain freedom from 2 to 24 hours post-dose was significantly greater in patients who received diclofenac potassium for oral solution compared with those who received placebo (see Table 3). The percentage of patients achieving pain relief 2 hours after treatment (defined as a reduction in headache severity from moderate or severe pain to mild or no pain) was also significantly greater in patients who received diclofenac potassium for oral solution compared with those who received placebo (see Table 3).
| Study 1 |
Diclofenac Potassium for Oral Solution
(n=265) |
Placebo
(n=257) |
| 2-Hour Pain Free | 24% | 13% |
| 2 to 24h Sustained Pain Free | 22% | 10% |
| 2-Hour Pain Relief | 48% | 27% |
| Study 2 |
Diclofenac Potassium for Oral Solution
(n=343) |
Placebo
(n=347) |
| 2-Hour Pain Free | 25% | 10% |
| 2 to 24h Sustained Pain Free | 19% | 7% |
| 2-Hour Pain Relief | 65% | 41% |
The estimated probability of achieving migraine headache pain freedom within 2 hours following treatment with diclofenac potassium for oral solution is shown in Figure 1.
Figure 1: Percentage of Patients with Initial Headache Pain Freedom within 2 Hours
There was a decreased incidence of nausea, photophobia and phonophobia following administration of diclofenac potassium for oral solution, compared to placebo. The efficacy and safety of diclofenac potassium for oral solution was unaffected by age or gender of the patient.
4 Contraindications
Diclofenac potassium for oral solution is contraindicated in the following patients:
- Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [see Warnings and Precautions (5.7, 5.9)]
- History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7, 5.8)]
- In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)]
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1)]
- GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Hypertension [see Warnings and Precautions (5.4)]
- Heart Failure and Edema [see Warnings and Precautions (5.5)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6)]
- Anaphylactic Reactions [see Warnings and Precautions (5.7)]
- Serious Skin Reactions [see Warnings and Precautions (5.9)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.10)]
- Medication Overuse Headache [see Warnings and Precautions (5.11)]
- Hematologic Toxicity [see Warnings and Precautions (5.13)]
7 Drug Interactions
See Table 2for clinically significant drug interactions with diclofenac.
| Drugs That Interfere with Hemostasis | |
|---|---|
| Clinical Impact: |
|
| Intervention: | Monitor patients with concomitant use of diclofenac potassium for oral solution with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.13)]. |
| Aspirin | |
| Clinical Impact: | Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)and Clinical Pharmacology (12.3)] . |
| Intervention: | Concomitant use of diclofenac potassium for oral solution and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.13)]. |
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6)] . |
| Digoxin | |
| Clinical Impact: | The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and lithium, monitor patients for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of diclofenac potassium for oral solution and cyclosporine may increase cyclosporine's nephrotoxicity. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2)] . |
| Intervention: | The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of diclofenac potassium for oral solution and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of NSAIDs and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity.
NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. |
| Inhibitors of Cytochrome P450 2C9 | |
| Clinical Impact: | Diclofenac is metabolized predominantly by Cytochrome P-450 CYP2C9.
Co-administration of medications that inhibit CYP2C9 may affect the pharmacokinetics of diclofenac [see Clinical Pharmacology (12.3)]. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and drugs that inhibit CYP2C9, an increase in the duration between diclofenac potassium for oral solution doses for subsequent migraine attacks may be necessary. |
8.7 Renal Impairment
No information is available from controlled clinical studies regarding the use of diclofenac potassium for oral solution in patients with advanced renal disease. Therefore, treatment with diclofenac potassium for oral solution is not recommended in patients with advanced renal disease. If diclofenac potassium for oral solution therapy must be initiated, close monitoring of the patient's renal function is advisable.
5.16 Phenylketonurics
Diclofenac potassium for oral solution contains phenylalanine 25 mg per each 50 mg packet.
8.6 Hepatic Impairment
Because hepatic metabolism accounts for almost 100% of diclofenac elimination, patients with hepatic impairment should be considered for treatment with diclofenac potassium for oral solution only if the benefits outweigh the risks. There is insufficient information available to support dosing recommendations for diclofenac potassium for oral solution in patients with hepatic insufficiency [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
Diclofenac potassium for oral solution is indicated for the acute treatment of migraine attacks with or without aura in adults (18 years of age or older).
12.1 Mechanism of Action
Diclofenac potassium for oral solution has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of diclofenac potassium for oral solution, like that of other NSAIDs, is not completely understood but involves inhibition of cyclooxygenase (COX-1 and COX-2).
Diclofenac is a potent inhibitor of prostaglandin synthesis in vitro. Diclofenac concentrations reached during therapy have produced in vivoeffects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Because diclofenac is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
5.13 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect upon erythropoiesis. If a patient treated with diclofenac potassium for oral solution has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including diclofenac potassium for oral solution, may increase the risk of bleeding events. Concomitant use of warfarin and other anticoagulants, antiplatelet agents (e.g., aspirin), and serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients and any patient who may be adversely affected by alterations in platelet function for signs of bleeding [see Drug Interactions (7)].
5 Warnings and Precautions
- Hepatotoxicity:Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop ( 5.3, 8.6, 12.3)
- Hypertension:Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.4, 7)
- Heart Failure and Edema:Avoid use of diclofenac potassium for oral solution in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.5)
- Renal Toxicity:Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac potassium for oral solution in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.6)
- Anaphylactic Reactions:Seek emergency help if an anaphylactic reaction occurs ( 5.7)
- Exacerbation of Asthma Related to Aspirin Sensitivity:Diclofenac potassium for oral solution is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity) ( 5.8)
- Serious Skin Reactions:Discontinue diclofenac potassium for oral solution at first appearance of skin rash or other signs of hypersensitivity ( 5.9).
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS):Discontinue and evaluate clinically ( 5.10)
- Medication Overuse Headache:Detoxification may be necessary. ( 5.11)
- Fetal Toxicity:Limit use of NSAIDs, including diclofenac potassium for oral solution, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal dysfunction and premature closure of the fetal ductus arteriosus ( 5.12, 8.1)
- Hematologic Toxicity:Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.13, 7)
5.15 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6)].
Discontinue diclofenac potassium for oral solution if abnormal liver tests or renal tests persist or worsen.
5.7 Anaphylactic Reactions
Diclofenac has been associated with anaphylactic reactions in patients with and without known hypersensitivity to diclofenac and in patients with aspirin-sensitive asthma [see Contraindications (4)and Warnings and Precautions (5.8)] .
Seek emergency help if an anaphylactic reaction occurs.
5.9 Serious Skin Reactions
NSAIDs, including diclofenac, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of diclofenac potassium for oral solution at the first appearance of skin rash or any other sign of hypersensitivity.
Diclofenac potassium for oral solution is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)] .
2 Dosage and Administration
Single 50 mg dose; mix single packet contents with 1 to 2 ounces or 2 to 4 tablespoons (30 to 60 mL) of water prior to administration
- Use the lowest effective dose for shortest duration consistent with individual patient treatment goals ( 2.1)
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of diclofenac may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)].
Avoid the use of diclofenac potassium for oral solution in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If diclofenac potassium for oral solution is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
3 Dosage Forms and Strengths
Diclofenac potassium for oral solution, USP is available in individual packets each designed to deliver a 50 mg dose when mixed in water.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of diclofenac or other NSAIDs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
- Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of diclofenac potassium for oral solution in women who have difficulties conceiving ( 8.3)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of a single dose of diclofenac potassium for oral solution was evaluated in 2 placebo-controlled trials with a total of 634 migraine patients treated with diclofenac potassium for oral solution for a single migraine headache. Following treatment with diclofenac potassium (either diclofenac potassium for oral solution or diclofenac potassium immediate-release tablets [as a control]), 5 subjects (0.8%) withdrew from the studies; following placebo exposure, 1 subject (0.2%) withdrew.
The most common adverse reactions (i.e., that occurred in 1% or more of diclofenac potassium for oral solution-treated patients) and more frequent with diclofenac potassium for oral solution than with placebo were nausea and dizziness (see Table 1).
| Adverse Reactions | Diclofenac Potassium for Oral Solution
n=634 |
Placebo
n=646 |
|---|---|---|
| Gastrointestinal | ||
| Nausea | 3% | 2% |
| Nervous System | ||
| Dizziness | 1% | 0.5% |
The most common adverse events resulting in discontinuation of patients following diclofenac potassium for oral solution dosing in controlled clinical trials were urticaria (0.2%) and flushing (0.2%). No withdrawals were due to a serious reaction.
2.1 Acute Treatment of Migraine
Administer one packet (50 mg) of diclofenac potassium for oral solution for the acute treatment of migraine. Empty the contents of one packet into a cup containing 1 to 2 ounces (30 to 60 mL) of water, mix well and drink immediately.
Do not use liquids other than water.
Taking diclofenac potassium for oral solution with food may cause a reduction in effectiveness compared to taking diclofenac potassium for oral solution on an empty stomach [see Clinical Pharmacology (12.3)] .
Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals. The safety and effectiveness of a second dose have not been established.
5.11 Medication Overuse Headache
Overuse of acute migraine drugs (e.g., ergotamine, triptans, opioids, nonsteroidal anti-inflammatory drugs or combination of these drugs for 10 or more days per month) may lead to exacerbation of headache (medication overuse headache). Medication overuse headache may present as migraine-like daily headaches or as a marked increase in frequency of migraine attacks. Detoxification of patients, including withdrawal of the overused drugs and treatment of withdrawal symptoms (which often includes a transient worsening of headache) may be necessary.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed. Inform patients, families, or their caregivers of the following information before initiating therapy with diclofenac potassium for oral solution and periodically during the course of ongoing therapy.
16 How Supplied/storage and Handling
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water.
| NDC 51672-4240-8 | Individual Diclofenac Potassium for Oral Solution Packet |
| NDC 51672-4240-6 | Carton of nine (9) Diclofenac Potassium for Oral Solution Packets |
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur. There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as diclofenac, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)] .
5.14 Masking of Inflammation and Fever
The pharmacological activity of diclofenac potassium for oral solution in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
Principal Display Panel 50 Mg Packet Carton
NDC 51672-4240-6
Diclofenac Potassium
for Oral Solution
USP, 50 mg
PHARMACIST: Dispense the Medication Guide provided separately
to each patient. Print Medication Guides at: www.taro.com/usa-medication-guides
WARNING: Phenylketonurics: Contains Phenylalanine 25 mg per each 50 mg packet.
Contains nine (9) single use packets (50 mg per packet)
Keep this and all medications out of the reach of children.
Rx only
TARO
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, diclofenac potassium for oral solution is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)] . When diclofenac potassium for oral solution is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including diclofenac, cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
2.2 Non Interchangeability With Other Formulations of Diclofenac
Different formulations of oral diclofenac (e.g., diclofenac potassium for oral solution, diclofenac sodium enteric-coated tablets, diclofenac sodium extended-release tablets, or diclofenac potassium immediate-release tablets) may not be bioequivalent even if the milligram strength is the same. Therefore, it is not possible to convert dosing from any other formulation of diclofenac to diclofenac potassium for oral solution.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as diclofenac potassium for oral solution. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue diclofenac potassium for oral solution and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1)
- Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4, 5.1)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2)
Structured Label Content
Section 42229-5 (42229-5)
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.1)] .
- Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4)and Warnings and Precautions (5.1)] .
Section 42231-1 (42231-1)
Dispense with Medication Guide available at: www.taro.com/usa-medication-guides
| Medication Guide
Diclofenac Potassium (dye-KLOE-fen-ak poe-TAS-ee-um) for Oral Solution, USP |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: June 2024 5240062-0624-01 |
||
|
What is the most important information I should know about diclofenac potassium for oral solution?
Diclofenac potassium for oral solution contains diclofenac (a non-steroidal anti-inflammatory drug or NSAID). NSAIDs, including diclofenac potassium for oral solution, can cause serious side effects, including:
Avoid taking NSAIDs, including diclofenac potassium for oral solution, after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.
|
||
|
||
|
|
|
| Diclofenac potassium for oral solution should only be used: | ||
|
||
|
What is diclofenac potassium for oral solution?
Diclofenac potassium for oral solution is a prescription medicine used to treat migraine attacks in adults. It does not prevent or lessen the number of migraines you have, and it is not for other types of headaches. Diclofenac potassium for oral solution contains diclofenac potassium (a non-steroidal anti-inflammatory drug or NSAID). |
||
|
How should I take diclofenac potassium for oral solution?
Take diclofenac potassium for oral solution exactly as your healthcare provider tells you to take it. Take 1 dose of diclofenac potassium for oral solution to treat your migraine headache:
|
||
|
Who should not take diclofenac potassium for oral solution?
Do not take diclofenac potassium for oral solution:
|
||
Before taking diclofenac potassium for oral solution, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your doctor if you take:
|
||
|
What are the possible side effects of diclofenac potassium for oral solution?
Diclofenac potassium for oral solution can cause serious side effects, including: See " What is the most important information I should know about diclofenac potassium for oral solution?" |
||
|
||
|
||
|
|
|
| Stop taking diclofenac potassium for oral solution and call your healthcare provider right away if you get any of the following symptoms | ||
|
|
|
|
If you take too much of your NSAID, call your healthcare provider or get medical help right away.
These are not all the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
Other information about NSAIDs
|
||
|
General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals. For more information, call Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or visit ww.taro.com |
||
| Manufactured by: Taro Pharmaceutical Industries Ltd., Haifa Bay, Israel 2624761
Distributed by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 All trademarks are the property of their respective owners. |
Section 44425-7 (44425-7)
Storage
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Symptoms following acute NSAID overdoses have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression and coma have occurred, but were rare [see Warnings and Precautions (5.1, 5.2, 5.4, 5.6)] .
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Consider emesis and/or activated charcoal (60 to 100 grams in adults, 1 to 2 grams per kg of body weight in pediatric patients) and/or osmotic cathartic in symptomatic patients seen within four hours of ingestion or in patients with a large overdosage (5 to 10 times the recommended dosage). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment contact a poison control center (1-800-222-1222).
Anaphylactic reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
11 Description (11 DESCRIPTION)
Diclofenac potassium for oral solution, USP is a nonsteroidal anti-inflammatory drug, available as a buffered soluble powder, designed to be mixed with water prior to oral administration. Diclofenac potassium, USP is a white to off-white, buffered, flavored powder for oral solution packaged in individual unit dose packets.
The chemical name is benzeneacetic acid, 2-[(2,6-dichlorophenyl)amino]-, monopotassium salt.
The molecular weight is 334.2 g/mole. Its molecular formula is C
14H
10Cl
2KNO
2, and it has the following chemical structure.
The inactive ingredients in diclofenac potassium for oral solution, USP include: aspartame (equivalent to 25 mg phenylalanine), flavoring agent (peppermint), glyceryl dibehenate, mannitol, potassium bicarbonate, and saccharin sodium.
5.4 Hypertension
NSAIDs, including diclofenac potassium for oral solution, can lead to new onset of hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Use NSAIDs, including diclofenac potassium for oral solution, with caution in patients with hypertension. Monitor blood pressure closely during the initiation of NSAID treatment and throughout the course of therapy.
Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazides, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.15)] .
Clinical studies of diclofenac potassium for oral solution did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
5.3 Hepatotoxicity
Elevations of one or more liver tests may occur during therapy with diclofenac potassium for oral solution. These laboratory abnormalities may progress, may persist, or may only be transient with continued therapy. Borderline elevations (less than 3 times the upper limit of the normal [ULN] range) or greater elevations of transaminases occurred in about 15% of diclofenac-treated patients. Of the markers of hepatic function, ALT (SGPT) is recommended for the monitoring of liver injury.
In clinical trials, meaningful elevations (i.e., more than 3 times the ULN) of AST (SGOT) occurred in about 2% of approximately 5,700 patients at some time during treatment (ALT was not measured in all studies). In an open-label, controlled trial of 3,700 patients treated for 2 to 6 months, patients were monitored at 8 weeks and 1,200 patients were monitored again at 24 weeks. Meaningful elevations of ALT and/or AST occurred in about 4% of the 3,700 patients and included marked elevations (>8 times the ULN) in about 1% of the 3,700 patients. In this open-label study, a higher incidence of borderline (less than 3 times the ULN), moderate (3 to 8 times the ULN), and marked (>8 times the ULN) elevations of ALT or AST was observed in patients receiving diclofenac when compared to other NSAIDs. Almost all meaningful elevations in transaminases were detected before patients became symptomatic [see Warnings and Precautions (5.15)].
Abnormal tests occurred during the first 2 months of therapy with diclofenac in 42 of the 51 patients in all trials who developed marked transaminase elevations. In postmarketing reports, cases of drug-induced hepatotoxicity have been reported in the first month, and in some cases, the first 2 months of NSAID therapy, but can occur at any time during treatment with diclofenac.
Postmarketing surveillance has reported cases of severe hepatic reactions, including liver necrosis, jaundice, fulminant hepatitis with and without jaundice, and liver failure. Some of these reported cases resulted in fatalities or liver transplantation.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue diclofenac potassium for oral solution immediately, and perform a clinical evaluation of the patient.
To minimize the potential risk for an adverse liver-related event in patients treated with diclofenac potassium for oral solution, use the lowest effective dose for the shortest duration possible. Exercise caution when prescribing diclofenac potassium for oral solution with concomitant drugs that are known to be potentially hepatotoxic (e.g., acetaminophen, certain antibiotics, antiepileptics). Caution patients to avoid taking nonprescription acetaminophen-containing products while using diclofenac potassium for oral solution.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of diclofenac potassium for oral solution in the acute treatment of migraine headache was demonstrated in two randomized, double-blind, placebo-controlled trials.
Patients enrolled in these two trials were predominantly female (85%) and white (86%), with a mean age of 40 years (range: 18 to 65). Patients were instructed to treat a migraine of moderate to severe pain with 1 dose of study medication. Patients evaluated their headache pain 2 hours later. Associated symptoms of nausea, photophobia, and phonophobia were also evaluated. In addition, the proportion of patients who were "sustained pain free", defined as a reduction in headache severity from moderate or severe pain to no pain at 2 hours post-dose without a return of mild, moderate, or severe pain and no use of rescue medication for 24 hours post-dose, was also evaluated. In these studies, the percentage of patients achieving pain freedom 2 hours after treatment and sustained pain freedom from 2 to 24 hours post-dose was significantly greater in patients who received diclofenac potassium for oral solution compared with those who received placebo (see Table 3). The percentage of patients achieving pain relief 2 hours after treatment (defined as a reduction in headache severity from moderate or severe pain to mild or no pain) was also significantly greater in patients who received diclofenac potassium for oral solution compared with those who received placebo (see Table 3).
| Study 1 |
Diclofenac Potassium for Oral Solution
(n=265) |
Placebo
(n=257) |
| 2-Hour Pain Free | 24% | 13% |
| 2 to 24h Sustained Pain Free | 22% | 10% |
| 2-Hour Pain Relief | 48% | 27% |
| Study 2 |
Diclofenac Potassium for Oral Solution
(n=343) |
Placebo
(n=347) |
| 2-Hour Pain Free | 25% | 10% |
| 2 to 24h Sustained Pain Free | 19% | 7% |
| 2-Hour Pain Relief | 65% | 41% |
The estimated probability of achieving migraine headache pain freedom within 2 hours following treatment with diclofenac potassium for oral solution is shown in Figure 1.
Figure 1: Percentage of Patients with Initial Headache Pain Freedom within 2 Hours
There was a decreased incidence of nausea, photophobia and phonophobia following administration of diclofenac potassium for oral solution, compared to placebo. The efficacy and safety of diclofenac potassium for oral solution was unaffected by age or gender of the patient.
4 Contraindications (4 CONTRAINDICATIONS)
Diclofenac potassium for oral solution is contraindicated in the following patients:
- Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [see Warnings and Precautions (5.7, 5.9)]
- History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7, 5.8)]
- In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1)]
- GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Hypertension [see Warnings and Precautions (5.4)]
- Heart Failure and Edema [see Warnings and Precautions (5.5)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6)]
- Anaphylactic Reactions [see Warnings and Precautions (5.7)]
- Serious Skin Reactions [see Warnings and Precautions (5.9)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.10)]
- Medication Overuse Headache [see Warnings and Precautions (5.11)]
- Hematologic Toxicity [see Warnings and Precautions (5.13)]
7 Drug Interactions (7 DRUG INTERACTIONS)
See Table 2for clinically significant drug interactions with diclofenac.
| Drugs That Interfere with Hemostasis | |
|---|---|
| Clinical Impact: |
|
| Intervention: | Monitor patients with concomitant use of diclofenac potassium for oral solution with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.13)]. |
| Aspirin | |
| Clinical Impact: | Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)and Clinical Pharmacology (12.3)] . |
| Intervention: | Concomitant use of diclofenac potassium for oral solution and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.13)]. |
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6)] . |
| Digoxin | |
| Clinical Impact: | The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and lithium, monitor patients for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of diclofenac potassium for oral solution and cyclosporine may increase cyclosporine's nephrotoxicity. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2)] . |
| Intervention: | The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of diclofenac potassium for oral solution and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of NSAIDs and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity.
NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. |
| Inhibitors of Cytochrome P450 2C9 | |
| Clinical Impact: | Diclofenac is metabolized predominantly by Cytochrome P-450 CYP2C9.
Co-administration of medications that inhibit CYP2C9 may affect the pharmacokinetics of diclofenac [see Clinical Pharmacology (12.3)]. |
| Intervention: | During concomitant use of diclofenac potassium for oral solution and drugs that inhibit CYP2C9, an increase in the duration between diclofenac potassium for oral solution doses for subsequent migraine attacks may be necessary. |
8.7 Renal Impairment
No information is available from controlled clinical studies regarding the use of diclofenac potassium for oral solution in patients with advanced renal disease. Therefore, treatment with diclofenac potassium for oral solution is not recommended in patients with advanced renal disease. If diclofenac potassium for oral solution therapy must be initiated, close monitoring of the patient's renal function is advisable.
5.16 Phenylketonurics
Diclofenac potassium for oral solution contains phenylalanine 25 mg per each 50 mg packet.
8.6 Hepatic Impairment
Because hepatic metabolism accounts for almost 100% of diclofenac elimination, patients with hepatic impairment should be considered for treatment with diclofenac potassium for oral solution only if the benefits outweigh the risks. There is insufficient information available to support dosing recommendations for diclofenac potassium for oral solution in patients with hepatic insufficiency [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Diclofenac potassium for oral solution is indicated for the acute treatment of migraine attacks with or without aura in adults (18 years of age or older).
12.1 Mechanism of Action
Diclofenac potassium for oral solution has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of diclofenac potassium for oral solution, like that of other NSAIDs, is not completely understood but involves inhibition of cyclooxygenase (COX-1 and COX-2).
Diclofenac is a potent inhibitor of prostaglandin synthesis in vitro. Diclofenac concentrations reached during therapy have produced in vivoeffects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Because diclofenac is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
5.13 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect upon erythropoiesis. If a patient treated with diclofenac potassium for oral solution has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including diclofenac potassium for oral solution, may increase the risk of bleeding events. Concomitant use of warfarin and other anticoagulants, antiplatelet agents (e.g., aspirin), and serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients and any patient who may be adversely affected by alterations in platelet function for signs of bleeding [see Drug Interactions (7)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatotoxicity:Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop ( 5.3, 8.6, 12.3)
- Hypertension:Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.4, 7)
- Heart Failure and Edema:Avoid use of diclofenac potassium for oral solution in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.5)
- Renal Toxicity:Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of diclofenac potassium for oral solution in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.6)
- Anaphylactic Reactions:Seek emergency help if an anaphylactic reaction occurs ( 5.7)
- Exacerbation of Asthma Related to Aspirin Sensitivity:Diclofenac potassium for oral solution is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity) ( 5.8)
- Serious Skin Reactions:Discontinue diclofenac potassium for oral solution at first appearance of skin rash or other signs of hypersensitivity ( 5.9).
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS):Discontinue and evaluate clinically ( 5.10)
- Medication Overuse Headache:Detoxification may be necessary. ( 5.11)
- Fetal Toxicity:Limit use of NSAIDs, including diclofenac potassium for oral solution, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal dysfunction and premature closure of the fetal ductus arteriosus ( 5.12, 8.1)
- Hematologic Toxicity:Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.13, 7)
5.15 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6)].
Discontinue diclofenac potassium for oral solution if abnormal liver tests or renal tests persist or worsen.
5.7 Anaphylactic Reactions
Diclofenac has been associated with anaphylactic reactions in patients with and without known hypersensitivity to diclofenac and in patients with aspirin-sensitive asthma [see Contraindications (4)and Warnings and Precautions (5.8)] .
Seek emergency help if an anaphylactic reaction occurs.
5.9 Serious Skin Reactions
NSAIDs, including diclofenac, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of diclofenac potassium for oral solution at the first appearance of skin rash or any other sign of hypersensitivity.
Diclofenac potassium for oral solution is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)] .
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Single 50 mg dose; mix single packet contents with 1 to 2 ounces or 2 to 4 tablespoons (30 to 60 mL) of water prior to administration
- Use the lowest effective dose for shortest duration consistent with individual patient treatment goals ( 2.1)
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of diclofenac may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)].
Avoid the use of diclofenac potassium for oral solution in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If diclofenac potassium for oral solution is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Diclofenac potassium for oral solution, USP is available in individual packets each designed to deliver a 50 mg dose when mixed in water.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of diclofenac or other NSAIDs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of diclofenac potassium for oral solution in women who have difficulties conceiving ( 8.3)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of a single dose of diclofenac potassium for oral solution was evaluated in 2 placebo-controlled trials with a total of 634 migraine patients treated with diclofenac potassium for oral solution for a single migraine headache. Following treatment with diclofenac potassium (either diclofenac potassium for oral solution or diclofenac potassium immediate-release tablets [as a control]), 5 subjects (0.8%) withdrew from the studies; following placebo exposure, 1 subject (0.2%) withdrew.
The most common adverse reactions (i.e., that occurred in 1% or more of diclofenac potassium for oral solution-treated patients) and more frequent with diclofenac potassium for oral solution than with placebo were nausea and dizziness (see Table 1).
| Adverse Reactions | Diclofenac Potassium for Oral Solution
n=634 |
Placebo
n=646 |
|---|---|---|
| Gastrointestinal | ||
| Nausea | 3% | 2% |
| Nervous System | ||
| Dizziness | 1% | 0.5% |
The most common adverse events resulting in discontinuation of patients following diclofenac potassium for oral solution dosing in controlled clinical trials were urticaria (0.2%) and flushing (0.2%). No withdrawals were due to a serious reaction.
2.1 Acute Treatment of Migraine
Administer one packet (50 mg) of diclofenac potassium for oral solution for the acute treatment of migraine. Empty the contents of one packet into a cup containing 1 to 2 ounces (30 to 60 mL) of water, mix well and drink immediately.
Do not use liquids other than water.
Taking diclofenac potassium for oral solution with food may cause a reduction in effectiveness compared to taking diclofenac potassium for oral solution on an empty stomach [see Clinical Pharmacology (12.3)] .
Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals. The safety and effectiveness of a second dose have not been established.
5.11 Medication Overuse Headache
Overuse of acute migraine drugs (e.g., ergotamine, triptans, opioids, nonsteroidal anti-inflammatory drugs or combination of these drugs for 10 or more days per month) may lead to exacerbation of headache (medication overuse headache). Medication overuse headache may present as migraine-like daily headaches or as a marked increase in frequency of migraine attacks. Detoxification of patients, including withdrawal of the overused drugs and treatment of withdrawal symptoms (which often includes a transient worsening of headache) may be necessary.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed. Inform patients, families, or their caregivers of the following information before initiating therapy with diclofenac potassium for oral solution and periodically during the course of ongoing therapy.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Diclofenac Potassium for Oral Solution USP 50 mg, is a white to off-white, buffered, flavored powder for oral solution, supplied as individual dose packets. Each individual packet is designed to deliver a dose of 50 mg diclofenac potassium when mixed in water.
| NDC 51672-4240-8 | Individual Diclofenac Potassium for Oral Solution Packet |
| NDC 51672-4240-6 | Carton of nine (9) Diclofenac Potassium for Oral Solution Packets |
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur. There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as diclofenac, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)] .
5.14 Masking of Inflammation and Fever
The pharmacological activity of diclofenac potassium for oral solution in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
Principal Display Panel 50 Mg Packet Carton (PRINCIPAL DISPLAY PANEL - 50 mg Packet Carton)
NDC 51672-4240-6
Diclofenac Potassium
for Oral Solution
USP, 50 mg
PHARMACIST: Dispense the Medication Guide provided separately
to each patient. Print Medication Guides at: www.taro.com/usa-medication-guides
WARNING: Phenylketonurics: Contains Phenylalanine 25 mg per each 50 mg packet.
Contains nine (9) single use packets (50 mg per packet)
Keep this and all medications out of the reach of children.
Rx only
TARO
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, diclofenac potassium for oral solution is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)] . When diclofenac potassium for oral solution is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including diclofenac, cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
2.2 Non Interchangeability With Other Formulations of Diclofenac (2.2 Non-Interchangeability with Other Formulations of Diclofenac)
Different formulations of oral diclofenac (e.g., diclofenac potassium for oral solution, diclofenac sodium enteric-coated tablets, diclofenac sodium extended-release tablets, or diclofenac potassium immediate-release tablets) may not be bioequivalent even if the milligram strength is the same. Therefore, it is not possible to convert dosing from any other formulation of diclofenac to diclofenac potassium for oral solution.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress) (5.10 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS))
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as diclofenac potassium for oral solution. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue diclofenac potassium for oral solution and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events (WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS)
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1)
- Diclofenac potassium for oral solution is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4, 5.1)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:15.607086 · Updated: 2026-03-14T22:14:14.773616