These Highlights Do Not Include All The Information Needed To Use Glycopyrrolate Injection Safely And Effectively. See Full Prescribing Information For Glycopyrrolate Injection.
685aaf64-1a64-42f9-a96f-4764d650c26f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in: anesthesia (all ages) for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation, intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents. peptic ulcer (adults) as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
Indications and Usage
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in: anesthesia (all ages) for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation, intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents. peptic ulcer (adults) as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
Dosage and Administration
Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution, in the following indications: Adults ( 2.2 ) Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia Intraoperative Medication: single doses of 0.1 mg IV and repeated, as needed, at intervals of 2 to 3 minutes Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine Peptic Ulcer: 0.1 mg IV or IM at 4-hour intervals, 3 or 4 times daily Pediatric patients ( 2.3 ) Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia. Patients under 2 years of age may require up to 0.009 mg/kg Intraoperative Medication: 0.004 mg/kg IV, not to exceed 0.1 mg in a single dose and repeated, as needed, at intervals of 2 to 3 minutes Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine Peptic Ulcer: Glycopyrrolate Injection is not indicated for the treatment of peptic ulcer in pediatric patients Do not use prefilled syringe to administer a dose of less than 0.1 mg (0.5 mL). ( 2.3 ) See Full Prescribing Information for preparation, handling, and instructions for use of pre-filled syringe ( 2.4 , 2.5 )
Warnings and Precautions
Precipitation of Acute Glaucoma : Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care if they experience symptoms of acute angle closure glaucoma. ( 5.1 ) Drowsiness or Blurred Vision : May cause drowsiness or blurred vision. Advise patients not to drive or perform hazardous work until resolved. ( 5.2 ) Heat Prostration : Advise patients to avoid exertion and high environmental temperatures after receiving Glycopyrrolate Injection. ( 5.3 ) Intestinal Obstruction : Diarrhea may be an early symptom of incomplete intestinal obstruction. Avoid use in patients with diarrhea and ileostomy or colostomy. ( 5.4 ) Tachycardia : Increase in heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, or hyperthyroidism. ( 5.5 )
Contraindications
Glycopyrrolate Injection is contraindicated in: patients with known hypersensitivity to Glycopyrrolate Injection or any of its inactive ingredients. peptic ulcer patients with the following concurrent conditions: glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus, intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.
Adverse Reactions
The following adverse reactions were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse reactions to anticholinergics include xerostomia (dry mouth); urinary hesitancy and retention; blurred vision and photophobia due to mydriasis (dilation of the pupil); cycloplegia; increased ocular tension; tachycardia; palpitation; decreased sweating; loss of taste; headache; nervousness; drowsiness; weakness; dizziness; insomnia; nausea; vomiting; impotence; suppression of lactation; constipation; bloated feeling; severe allergic reactions including anaphylactic/anaphylactoid reactions; hypersensitivity; urticaria, pruritus, dry skin, and other dermal manifestations; some degree of mental confusion and/or excitement, especially in elderly persons. The following adverse events have been reported from post-marketing experience with glycopyrrolate: malignant hyperthermia; cardiac arrhythmias (including bradycardia, ventricular tachycardia, ventricular fibrillation); cardiac arrest; hypertension; hypotension; seizures; and respiratory arrest. Post-marketing reports have included cases of heart block and QTc interval prolongation associated with the combined use of glycopyrrolate and an anticholinesterase. Injection site reactions including pruritus, edema, erythema, and pain have also been reported.
Drug Interactions
The concurrent use of Glycopyrrolate Injection with other anticholinergics or medications with anticholinergic activity, such as phenothiazines, antiparkinson drugs, or tricyclic antidepressants, may intensify the antimuscarinic effects and result in an increase in anticholinergic side effects. Concomitant administration of Glycopyrrolate Injection and potassium chloride in a wax matrix may increase the severity of potassium chloride-induced gastrointestinal lesions as a result of a slower gastrointestinal transit time.
Storage and Handling
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as: Product Code Unit of Sale Strength Each 720330 NDC 76045-023-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe RF720330 NDC 76045-223-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.] Sensitive to heat – Do not autoclave. Discard unused portion. Do not place syringe on a sterile field.
How Supplied
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as: Product Code Unit of Sale Strength Each 720330 NDC 76045-023-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe RF720330 NDC 76045-223-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.] Sensitive to heat – Do not autoclave. Discard unused portion. Do not place syringe on a sterile field.
Medication Information
Warnings and Precautions
Precipitation of Acute Glaucoma : Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care if they experience symptoms of acute angle closure glaucoma. ( 5.1 ) Drowsiness or Blurred Vision : May cause drowsiness or blurred vision. Advise patients not to drive or perform hazardous work until resolved. ( 5.2 ) Heat Prostration : Advise patients to avoid exertion and high environmental temperatures after receiving Glycopyrrolate Injection. ( 5.3 ) Intestinal Obstruction : Diarrhea may be an early symptom of incomplete intestinal obstruction. Avoid use in patients with diarrhea and ileostomy or colostomy. ( 5.4 ) Tachycardia : Increase in heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, or hyperthyroidism. ( 5.5 )
Indications and Usage
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in: anesthesia (all ages) for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation, intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents. peptic ulcer (adults) as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
Dosage and Administration
Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution, in the following indications: Adults ( 2.2 ) Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia Intraoperative Medication: single doses of 0.1 mg IV and repeated, as needed, at intervals of 2 to 3 minutes Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine Peptic Ulcer: 0.1 mg IV or IM at 4-hour intervals, 3 or 4 times daily Pediatric patients ( 2.3 ) Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia. Patients under 2 years of age may require up to 0.009 mg/kg Intraoperative Medication: 0.004 mg/kg IV, not to exceed 0.1 mg in a single dose and repeated, as needed, at intervals of 2 to 3 minutes Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine Peptic Ulcer: Glycopyrrolate Injection is not indicated for the treatment of peptic ulcer in pediatric patients Do not use prefilled syringe to administer a dose of less than 0.1 mg (0.5 mL). ( 2.3 ) See Full Prescribing Information for preparation, handling, and instructions for use of pre-filled syringe ( 2.4 , 2.5 )
Contraindications
Glycopyrrolate Injection is contraindicated in: patients with known hypersensitivity to Glycopyrrolate Injection or any of its inactive ingredients. peptic ulcer patients with the following concurrent conditions: glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus, intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.
Adverse Reactions
The following adverse reactions were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse reactions to anticholinergics include xerostomia (dry mouth); urinary hesitancy and retention; blurred vision and photophobia due to mydriasis (dilation of the pupil); cycloplegia; increased ocular tension; tachycardia; palpitation; decreased sweating; loss of taste; headache; nervousness; drowsiness; weakness; dizziness; insomnia; nausea; vomiting; impotence; suppression of lactation; constipation; bloated feeling; severe allergic reactions including anaphylactic/anaphylactoid reactions; hypersensitivity; urticaria, pruritus, dry skin, and other dermal manifestations; some degree of mental confusion and/or excitement, especially in elderly persons. The following adverse events have been reported from post-marketing experience with glycopyrrolate: malignant hyperthermia; cardiac arrhythmias (including bradycardia, ventricular tachycardia, ventricular fibrillation); cardiac arrest; hypertension; hypotension; seizures; and respiratory arrest. Post-marketing reports have included cases of heart block and QTc interval prolongation associated with the combined use of glycopyrrolate and an anticholinesterase. Injection site reactions including pruritus, edema, erythema, and pain have also been reported.
Drug Interactions
The concurrent use of Glycopyrrolate Injection with other anticholinergics or medications with anticholinergic activity, such as phenothiazines, antiparkinson drugs, or tricyclic antidepressants, may intensify the antimuscarinic effects and result in an increase in anticholinergic side effects. Concomitant administration of Glycopyrrolate Injection and potassium chloride in a wax matrix may increase the severity of potassium chloride-induced gastrointestinal lesions as a result of a slower gastrointestinal transit time.
Storage and Handling
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as: Product Code Unit of Sale Strength Each 720330 NDC 76045-023-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe RF720330 NDC 76045-223-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.] Sensitive to heat – Do not autoclave. Discard unused portion. Do not place syringe on a sterile field.
How Supplied
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as: Product Code Unit of Sale Strength Each 720330 NDC 76045-023-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe RF720330 NDC 76045-223-30 Unit of 10 0.6 mg/3 mL (0.2 mg/mL) NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.] Sensitive to heat – Do not autoclave. Discard unused portion. Do not place syringe on a sterile field.
Description
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in: anesthesia (all ages) for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation, intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents. peptic ulcer (adults) as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
Section 42229-5
Preanesthetic Medication
The recommended dose of Glycopyrrolate Injection is 0.004 mg/kg by intramuscular injection, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia or at the time the preanesthetic narcotic and/or sedative are administered.
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY – Glycopyrrolate Injection, USP 3 mL Syringe Label
3 mL Single-Dose Prefilled Syringe. For IM or IV Use. Rx only
Glycopyrrolate Injection, USP
0.6 mg / 3 mL (0.2 mg /mL)
10 Overdosage
To combat peripheral anticholinergic effects, a quaternary ammonium anticholinesterase such as neostigmine methylsulfate (which does not cross the blood-brain barrier) may be given intravenously in increments of 0.25 mg in adults. This dosage may be repeated every five to ten minutes until anticholinergic overactivity is reversed or up to a maximum of 2.5 mg. Proportionately smaller doses should be used in pediatric patients. Indication for repetitive doses of neostigmine should be based on close monitoring of the decrease in heart rate and the return of bowel sounds.
If CNS symptoms (e.g., excitement, restlessness, convulsions, psychotic behavior) occur, physostigmine (which does cross the blood–brain barrier) may be used. Physostigmine 0.5 to 2 mg should be slowly administered intravenously and repeated as necessary up to a total of 5 mg in adults. Proportionately smaller doses should be used in pediatric patients.
To combat hypotension, administer IV fluids and/or pressor agents along with supportive care.
Fever should be treated symptomatically.
Following overdosage, a curare-like action may occur, i.e., neuromuscular blockade leading to muscular weakness and possible paralysis. In the event of a curare-like effect on respiratory muscles, artificial respiration should be instituted and maintained until effective respiratory action returns.
11 Description
Glycopyrrolate Injection, USP is a synthetic anticholinergic agent. It is intended for intramuscular or intravenous administration. Each 1 mL contains:
Glycopyrrolate, USP 0.2 mg, Water for Injection, USP q.s., pH adjusted, when necessary, with hydrochloric acid and/or sodium hydroxide. Solution does not contain preservatives.
Glycopyrrolate is a quaternary ammonium salt with the following chemical name:
3[(cyclopentylhydroxyphenylacetyl)oxy]-1,1-dimethyl pyrrolidinium bromide.
Its structural formula is as follows:
Glycopyrrolate occurs as a white, odorless crystalline powder. It is soluble in water and alcohol, and practically insoluble in chloroform and ether. It completely ionized at physiological pH values. Glycopyrrolate Injection, USP, is a clear, colorless, sterile liquid; pH 2.0 to 3.0. The partition coefficient of glycopyrrolate in a n-octanol/water system is 0.304 (log10 P= -1.52) at ambient room temperature (24°C).
5.5 Tachycardia
Investigate any tachycardia before giving Glycopyrrolate Injection because an increase in the heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, and/or hyperthyroidism.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established for the management of peptic ulcer.
Dysrhythmias associated with the use of glycopyrrolate intravenously as a premedicant or during anesthesia have been observed in pediatric patients.
Infants, patients with Down's syndrome, and pediatric patients with spastic paralysis or brain damage may experience an increased response to anticholinergics, thus increasing the potential for side effects.
A paradoxical reaction characterized by hyperexcitability may occur in pediatric patients taking large doses of anticholinergics including Glycopyrrolate Injection. Infants and young children are especially susceptible to the toxic effects of anticholinergics.
8.5 Geriatric Use
Clinical Studies of Glycopyrrolate Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other therapy.
4 Contraindications
Glycopyrrolate Injection is contraindicated in:
- patients with known hypersensitivity to Glycopyrrolate Injection or any of its inactive ingredients.
- peptic ulcer patients with the following concurrent conditions: glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus, intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.
6 Adverse Reactions
The following adverse reactions were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to anticholinergics include xerostomia (dry mouth); urinary hesitancy and retention; blurred vision and photophobia due to mydriasis (dilation of the pupil); cycloplegia; increased ocular tension; tachycardia; palpitation; decreased sweating; loss of taste; headache; nervousness; drowsiness; weakness; dizziness; insomnia; nausea; vomiting; impotence; suppression of lactation; constipation; bloated feeling; severe allergic reactions including anaphylactic/anaphylactoid reactions; hypersensitivity; urticaria, pruritus, dry skin, and other dermal manifestations; some degree of mental confusion and/or excitement, especially in elderly persons.
The following adverse events have been reported from post-marketing experience with glycopyrrolate: malignant hyperthermia; cardiac arrhythmias (including bradycardia, ventricular tachycardia, ventricular fibrillation); cardiac arrest; hypertension; hypotension; seizures; and respiratory arrest. Post-marketing reports have included cases of heart block and QTc interval prolongation associated with the combined use of glycopyrrolate and an anticholinesterase. Injection site reactions including pruritus, edema, erythema, and pain have also been reported.
7 Drug Interactions
The concurrent use of Glycopyrrolate Injection with other anticholinergics or medications with anticholinergic activity, such as phenothiazines, antiparkinson drugs, or tricyclic antidepressants, may intensify the antimuscarinic effects and result in an increase in anticholinergic side effects.
Concomitant administration of Glycopyrrolate Injection and potassium chloride in a wax matrix may increase the severity of potassium chloride-induced gastrointestinal lesions as a result of a slower gastrointestinal transit time.
5.3 Heat Prostration
In the presence of fever, high environmental temperature, and/or during physical exercise, heat prostration can occur with use of anticholinergic agents including Glycopyrrolate Injection (due to decreased sweating), particularly in children and the elderly. Advise patients to avoid exertion and high environmental temperature after receiving Glycopyrrolate Injection.
8.6 Renal Impairment
Renal elimination of glycopyrrolate may be severely impaired in patients with renal failure. Dosage adjustments may be necessary [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Glycopyrrolate antagonizes muscarinic symptoms (e.g., bronchorrhea, bronchospasm, bradycardia, and intestinal hypermotility) induced by cholinergic drugs such as the anticholinesterases. The highly polar quaternary ammonium group of glycopyrrolate limits its passage across lipid membranes, such as the blood-brain barrier, in contrast to atropine sulfate and scopolamine hydrobromide, which are highly non-polar tertiary amines which penetrate lipid barriers easily. For this reason, the occurrence of CNS-related side effects is lower, in comparison to their incidence following administration of anticholinergics which are chemically tertiary amines that can cross this barrier readily. With intravenous injection, the onset of action is generally evident within one minute. Following intramuscular administration, the onset of action is noted in 15 to 30 minutes, with peak effects occurring within approximately 30 to 45 minutes. The vagal blocking effects persist for 2 to 3 hours and the antisialagogue effects persist up to 7 hours, periods longer than for atropine.
12.3 Pharmacokinetics
The following pharmacokinetic information and conclusions were obtained from published studies that used nonspecific assay methods.
5.9 Light Sensitivity
Patients may experience sensitivity of the eyes to light. Advise patients to protect their eyes from light after receiving Glycopyrrolate Injection.
1 Indications and Usage
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in:
anesthesia (all ages)
- for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation,
- intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and
- for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents.
peptic ulcer (adults)
- as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
12.1 Mechanism of Action
Glycopyrrolate, like other anticholinergic (antimuscarinic) agents, inhibits the action of acetylcholine on structures innervated by postganglionic cholinergic nerves and on smooth muscles that respond to acetylcholine but lack cholinergic innervation. These peripheral cholinergic receptors are present in the autonomic effector cells of smooth muscle, cardiac muscle, the sinoatrial node, the atrioventricular node, exocrine glands and, to a limited degree, in the autonomic ganglia. Thus, it diminishes the volume and free acidity of gastric secretions and controls excessive pharyngeal, tracheal, and bronchial secretions.
5 Warnings and Precautions
- Precipitation of Acute Glaucoma: Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care if they experience symptoms of acute angle closure glaucoma. (5.1)
- Drowsiness or Blurred Vision: May cause drowsiness or blurred vision. Advise patients not to drive or perform hazardous work until resolved. (5.2)
- Heat Prostration: Advise patients to avoid exertion and high environmental temperatures after receiving Glycopyrrolate Injection. (5.3)
- Intestinal Obstruction: Diarrhea may be an early symptom of incomplete intestinal obstruction. Avoid use in patients with diarrhea and ileostomy or colostomy. (5.4)
- Tachycardia: Increase in heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, or hyperthyroidism. (5.5)
5.4 Intestinal Obstruction
Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance treatment with Glycopyrrolate Injection is inappropriate and possibly harmful. Avoid use in patients with these conditions.
2 Dosage and Administration
Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution, in the following indications:
Adults (2.2)
Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia
Intraoperative Medication: single doses of 0.1 mg IV and repeated, as needed, at intervals of 2 to 3 minutes
Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine
Peptic Ulcer: 0.1 mg IV or IM at 4-hour intervals, 3 or 4 times daily
Pediatric patients (2.3)
Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia. Patients under 2 years of age may require up to 0.009 mg/kg
Intraoperative Medication: 0.004 mg/kg IV, not to exceed 0.1 mg in a single dose and repeated, as needed, at intervals of 2 to 3 minutes
Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine
Peptic Ulcer: Glycopyrrolate Injection is not indicated for the treatment of peptic ulcer in pediatric patients
Do not use prefilled syringe to administer a dose of less than 0.1 mg (0.5 mL). (2.3)
See Full Prescribing Information for preparation, handling, and instructions for use of pre-filled syringe (2.4, 2.5)
3 Dosage Forms and Strengths
Glycopyrrolate Injection, USP, is a clear, colorless solution available in 0.6 mg/3 mL (0.2 mg/mL) single-dose, prefilled, disposable syringes.
8 Use in Specific Populations
- Pediatric Use: Infants, patients with Down's Syndrome, and pediatric patients with spastic paralysis or brain damage may experience an increased response to anticholinergics, thus increasing the potential for side effects. Large doses may cause hyperexcitability. (8.4)
5.2 Drowsiness Or Blurred Vision
Glycopyrrolate Injection may cause drowsiness or blurred vision. Warn patients not to participate in activities requiring mental alertness, such as operating a motor vehicle or other machinery, or performing hazardous work, until these issues resolve.
5.1 Precipitation of Acute Glaucoma
Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care in the event that they experience symptoms of acute angle closure glaucoma (pain and reddening of the eyes, accompanied by dilated pupils).
16 How Supplied/storage and Handling
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as:
|
Product
Code |
Unit of Sale | Strength | Each |
| 720330 | NDC 76045-023-30 Unit of 10 |
0.6 mg/3 mL (0.2 mg/mL) |
NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe |
| RF720330 | NDC 76045-223-30 Unit of 10 |
0.6 mg/3 mL (0.2 mg/mL) |
NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.]
Sensitive to heat – Do not autoclave. Discard unused portion.
Do not place syringe on a sterile field.
5.8 Delayed Gastric Emptying/gastric Stasis
The use of anticholinergetic drugs, including Glycopyrrolate Injection, in the treatment of gastric ulcer may produce a delay in gastric emptying due to antral statis. Monitor patients for symptoms such as vomiting, dyspepsia, early satiety, abdominal distention, and increased abdominal pain. Discontinue Glycopyrrolate Injection treatment if these symptoms develop or worsen on treatment.
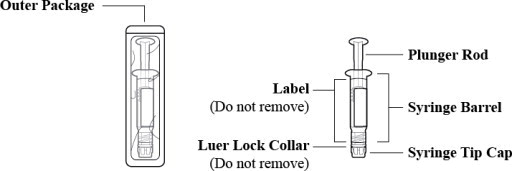
2.5 Instructions for Use of Pre Filled Syringe:
Figure 1: Outer Packaging and Prefilled Syringe
- Inspect the outer packaging (blister pack) to confirm the integrity of the packaging. Do not use if the blister pack or the prefilled syringe has been damaged.
- Remove the syringe from the outer packaging. (See Figure 2)
Figure 2
- Visually inspect the syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Twist off the syringe tip cap. Do not remove the label around the luer lock collar. (See Figure 3)
Figure 3
- Expel air bubble(s). Adjust the dose (if applicable).
- Administer the dose ensuring that pressure is maintained on the plunger rod during the entire administration.
- Discard the used syringe into an appropriate receptacle.
2.1 General Dosing and Administration Information
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
- Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution.
- Do not introduce any other fluid into the syringe at any time.
- Do not dilute for IV push.
- Do not re-sterilize the syringe.
- Do not use this product on a sterile field.
- This product is for single dose only.
5.6 Risk of Use in Patients With Renal Impairment
Renal elimination of glycopyrrolate may be severely impaired in patients with renal failure. Dosage adjustments may be necessary in this population [see Pharmacokinetics (12.3)]
5.7 Autonomic Neuropathy, Hepatic Disease, Ulcerative Colitis, Prostatic Hypertrophy, Or Hiatal Hernia
Use Glycopyrrolate Injection with caution in the elderly and in all patients with autonomic neuropathy, hepatic disease, ulcerative colitis, prostatic hypertrophy, or hiatal hernia, because anticholinergic drugs may aggravate these conditions. Consider dose reduction and closely monitor the elderly and patients with autonomic neuropathy, hepatic disease, ulcerative colitis, prostatic hypertrophy, or hiatal hernia.
Structured Label Content
Section 42229-5 (42229-5)
Preanesthetic Medication
The recommended dose of Glycopyrrolate Injection is 0.004 mg/kg by intramuscular injection, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia or at the time the preanesthetic narcotic and/or sedative are administered.
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY – Glycopyrrolate Injection, USP 3 mL Syringe Label
3 mL Single-Dose Prefilled Syringe. For IM or IV Use. Rx only
Glycopyrrolate Injection, USP
0.6 mg / 3 mL (0.2 mg /mL)
10 Overdosage (10 OVERDOSAGE)
To combat peripheral anticholinergic effects, a quaternary ammonium anticholinesterase such as neostigmine methylsulfate (which does not cross the blood-brain barrier) may be given intravenously in increments of 0.25 mg in adults. This dosage may be repeated every five to ten minutes until anticholinergic overactivity is reversed or up to a maximum of 2.5 mg. Proportionately smaller doses should be used in pediatric patients. Indication for repetitive doses of neostigmine should be based on close monitoring of the decrease in heart rate and the return of bowel sounds.
If CNS symptoms (e.g., excitement, restlessness, convulsions, psychotic behavior) occur, physostigmine (which does cross the blood–brain barrier) may be used. Physostigmine 0.5 to 2 mg should be slowly administered intravenously and repeated as necessary up to a total of 5 mg in adults. Proportionately smaller doses should be used in pediatric patients.
To combat hypotension, administer IV fluids and/or pressor agents along with supportive care.
Fever should be treated symptomatically.
Following overdosage, a curare-like action may occur, i.e., neuromuscular blockade leading to muscular weakness and possible paralysis. In the event of a curare-like effect on respiratory muscles, artificial respiration should be instituted and maintained until effective respiratory action returns.
11 Description (11 DESCRIPTION)
Glycopyrrolate Injection, USP is a synthetic anticholinergic agent. It is intended for intramuscular or intravenous administration. Each 1 mL contains:
Glycopyrrolate, USP 0.2 mg, Water for Injection, USP q.s., pH adjusted, when necessary, with hydrochloric acid and/or sodium hydroxide. Solution does not contain preservatives.
Glycopyrrolate is a quaternary ammonium salt with the following chemical name:
3[(cyclopentylhydroxyphenylacetyl)oxy]-1,1-dimethyl pyrrolidinium bromide.
Its structural formula is as follows:
Glycopyrrolate occurs as a white, odorless crystalline powder. It is soluble in water and alcohol, and practically insoluble in chloroform and ether. It completely ionized at physiological pH values. Glycopyrrolate Injection, USP, is a clear, colorless, sterile liquid; pH 2.0 to 3.0. The partition coefficient of glycopyrrolate in a n-octanol/water system is 0.304 (log10 P= -1.52) at ambient room temperature (24°C).
5.5 Tachycardia
Investigate any tachycardia before giving Glycopyrrolate Injection because an increase in the heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, and/or hyperthyroidism.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established for the management of peptic ulcer.
Dysrhythmias associated with the use of glycopyrrolate intravenously as a premedicant or during anesthesia have been observed in pediatric patients.
Infants, patients with Down's syndrome, and pediatric patients with spastic paralysis or brain damage may experience an increased response to anticholinergics, thus increasing the potential for side effects.
A paradoxical reaction characterized by hyperexcitability may occur in pediatric patients taking large doses of anticholinergics including Glycopyrrolate Injection. Infants and young children are especially susceptible to the toxic effects of anticholinergics.
8.5 Geriatric Use
Clinical Studies of Glycopyrrolate Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other therapy.
4 Contraindications (4 CONTRAINDICATIONS)
Glycopyrrolate Injection is contraindicated in:
- patients with known hypersensitivity to Glycopyrrolate Injection or any of its inactive ingredients.
- peptic ulcer patients with the following concurrent conditions: glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus, intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to anticholinergics include xerostomia (dry mouth); urinary hesitancy and retention; blurred vision and photophobia due to mydriasis (dilation of the pupil); cycloplegia; increased ocular tension; tachycardia; palpitation; decreased sweating; loss of taste; headache; nervousness; drowsiness; weakness; dizziness; insomnia; nausea; vomiting; impotence; suppression of lactation; constipation; bloated feeling; severe allergic reactions including anaphylactic/anaphylactoid reactions; hypersensitivity; urticaria, pruritus, dry skin, and other dermal manifestations; some degree of mental confusion and/or excitement, especially in elderly persons.
The following adverse events have been reported from post-marketing experience with glycopyrrolate: malignant hyperthermia; cardiac arrhythmias (including bradycardia, ventricular tachycardia, ventricular fibrillation); cardiac arrest; hypertension; hypotension; seizures; and respiratory arrest. Post-marketing reports have included cases of heart block and QTc interval prolongation associated with the combined use of glycopyrrolate and an anticholinesterase. Injection site reactions including pruritus, edema, erythema, and pain have also been reported.
7 Drug Interactions (7 DRUG INTERACTIONS)
The concurrent use of Glycopyrrolate Injection with other anticholinergics or medications with anticholinergic activity, such as phenothiazines, antiparkinson drugs, or tricyclic antidepressants, may intensify the antimuscarinic effects and result in an increase in anticholinergic side effects.
Concomitant administration of Glycopyrrolate Injection and potassium chloride in a wax matrix may increase the severity of potassium chloride-induced gastrointestinal lesions as a result of a slower gastrointestinal transit time.
5.3 Heat Prostration
In the presence of fever, high environmental temperature, and/or during physical exercise, heat prostration can occur with use of anticholinergic agents including Glycopyrrolate Injection (due to decreased sweating), particularly in children and the elderly. Advise patients to avoid exertion and high environmental temperature after receiving Glycopyrrolate Injection.
8.6 Renal Impairment
Renal elimination of glycopyrrolate may be severely impaired in patients with renal failure. Dosage adjustments may be necessary [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Glycopyrrolate antagonizes muscarinic symptoms (e.g., bronchorrhea, bronchospasm, bradycardia, and intestinal hypermotility) induced by cholinergic drugs such as the anticholinesterases. The highly polar quaternary ammonium group of glycopyrrolate limits its passage across lipid membranes, such as the blood-brain barrier, in contrast to atropine sulfate and scopolamine hydrobromide, which are highly non-polar tertiary amines which penetrate lipid barriers easily. For this reason, the occurrence of CNS-related side effects is lower, in comparison to their incidence following administration of anticholinergics which are chemically tertiary amines that can cross this barrier readily. With intravenous injection, the onset of action is generally evident within one minute. Following intramuscular administration, the onset of action is noted in 15 to 30 minutes, with peak effects occurring within approximately 30 to 45 minutes. The vagal blocking effects persist for 2 to 3 hours and the antisialagogue effects persist up to 7 hours, periods longer than for atropine.
12.3 Pharmacokinetics
The following pharmacokinetic information and conclusions were obtained from published studies that used nonspecific assay methods.
5.9 Light Sensitivity
Patients may experience sensitivity of the eyes to light. Advise patients to protect their eyes from light after receiving Glycopyrrolate Injection.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Glycopyrrolate Injection, USP (0.2 mg/mL) is an anticholinergic indicated for use in:
anesthesia (all ages)
- for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation,
- intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and
- for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents.
peptic ulcer (adults)
- as adjunctive therapy for the treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
12.1 Mechanism of Action
Glycopyrrolate, like other anticholinergic (antimuscarinic) agents, inhibits the action of acetylcholine on structures innervated by postganglionic cholinergic nerves and on smooth muscles that respond to acetylcholine but lack cholinergic innervation. These peripheral cholinergic receptors are present in the autonomic effector cells of smooth muscle, cardiac muscle, the sinoatrial node, the atrioventricular node, exocrine glands and, to a limited degree, in the autonomic ganglia. Thus, it diminishes the volume and free acidity of gastric secretions and controls excessive pharyngeal, tracheal, and bronchial secretions.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Precipitation of Acute Glaucoma: Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care if they experience symptoms of acute angle closure glaucoma. (5.1)
- Drowsiness or Blurred Vision: May cause drowsiness or blurred vision. Advise patients not to drive or perform hazardous work until resolved. (5.2)
- Heat Prostration: Advise patients to avoid exertion and high environmental temperatures after receiving Glycopyrrolate Injection. (5.3)
- Intestinal Obstruction: Diarrhea may be an early symptom of incomplete intestinal obstruction. Avoid use in patients with diarrhea and ileostomy or colostomy. (5.4)
- Tachycardia: Increase in heart rate may occur. Use with caution in patients with coronary artery disease, congestive heart failure, cardiac arrhythmias, hypertension, or hyperthyroidism. (5.5)
5.4 Intestinal Obstruction
Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance treatment with Glycopyrrolate Injection is inappropriate and possibly harmful. Avoid use in patients with these conditions.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution, in the following indications:
Adults (2.2)
Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia
Intraoperative Medication: single doses of 0.1 mg IV and repeated, as needed, at intervals of 2 to 3 minutes
Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine
Peptic Ulcer: 0.1 mg IV or IM at 4-hour intervals, 3 or 4 times daily
Pediatric patients (2.3)
Preanesthetic Medication: 0.004 mg/kg IM, given 30 to 60 minutes prior to the anticipated time of induction of anesthesia. Patients under 2 years of age may require up to 0.009 mg/kg
Intraoperative Medication: 0.004 mg/kg IV, not to exceed 0.1 mg in a single dose and repeated, as needed, at intervals of 2 to 3 minutes
Reversal of Neuromuscular Blockade: 0.2 mg for each 1 mg of neostigmine or 5 mg of pyridostigmine
Peptic Ulcer: Glycopyrrolate Injection is not indicated for the treatment of peptic ulcer in pediatric patients
Do not use prefilled syringe to administer a dose of less than 0.1 mg (0.5 mL). (2.3)
See Full Prescribing Information for preparation, handling, and instructions for use of pre-filled syringe (2.4, 2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Glycopyrrolate Injection, USP, is a clear, colorless solution available in 0.6 mg/3 mL (0.2 mg/mL) single-dose, prefilled, disposable syringes.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatric Use: Infants, patients with Down's Syndrome, and pediatric patients with spastic paralysis or brain damage may experience an increased response to anticholinergics, thus increasing the potential for side effects. Large doses may cause hyperexcitability. (8.4)
5.2 Drowsiness Or Blurred Vision (5.2 Drowsiness or Blurred Vision)
Glycopyrrolate Injection may cause drowsiness or blurred vision. Warn patients not to participate in activities requiring mental alertness, such as operating a motor vehicle or other machinery, or performing hazardous work, until these issues resolve.
5.1 Precipitation of Acute Glaucoma
Glycopyrrolate Injection may cause mydriasis and increase intraocular pressure in patients with glaucoma. Advise patients with glaucoma to promptly seek medical care in the event that they experience symptoms of acute angle closure glaucoma (pain and reddening of the eyes, accompanied by dilated pupils).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Glycopyrrolate Injection, USP, 0.2 mg per mL without preservative is available as:
|
Product
Code |
Unit of Sale | Strength | Each |
| 720330 | NDC 76045-023-30 Unit of 10 |
0.6 mg/3 mL (0.2 mg/mL) |
NDC 76045-023-00 3 mL single-dose pre-filled disposable syringe |
| RF720330 | NDC 76045-223-30 Unit of 10 |
0.6 mg/3 mL (0.2 mg/mL) |
NDC 76045-223-03 3 mL single-dose pre-filled disposable syringe This product contains an RFID. |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.]
Sensitive to heat – Do not autoclave. Discard unused portion.
Do not place syringe on a sterile field.
5.8 Delayed Gastric Emptying/gastric Stasis (5.8 Delayed Gastric Emptying/Gastric Stasis)
The use of anticholinergetic drugs, including Glycopyrrolate Injection, in the treatment of gastric ulcer may produce a delay in gastric emptying due to antral statis. Monitor patients for symptoms such as vomiting, dyspepsia, early satiety, abdominal distention, and increased abdominal pain. Discontinue Glycopyrrolate Injection treatment if these symptoms develop or worsen on treatment.
2.5 Instructions for Use of Pre Filled Syringe: (2.5 Instructions for Use of Pre-filled Syringe:)
Figure 1: Outer Packaging and Prefilled Syringe
- Inspect the outer packaging (blister pack) to confirm the integrity of the packaging. Do not use if the blister pack or the prefilled syringe has been damaged.
- Remove the syringe from the outer packaging. (See Figure 2)
Figure 2
- Visually inspect the syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Twist off the syringe tip cap. Do not remove the label around the luer lock collar. (See Figure 3)
Figure 3
- Expel air bubble(s). Adjust the dose (if applicable).
- Administer the dose ensuring that pressure is maintained on the plunger rod during the entire administration.
- Discard the used syringe into an appropriate receptacle.
2.1 General Dosing and Administration Information
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
- Glycopyrrolate Injection may be administered intramuscularly, or intravenously, without dilution.
- Do not introduce any other fluid into the syringe at any time.
- Do not dilute for IV push.
- Do not re-sterilize the syringe.
- Do not use this product on a sterile field.
- This product is for single dose only.
5.6 Risk of Use in Patients With Renal Impairment (5.6 Risk of Use in Patients with Renal Impairment)
Renal elimination of glycopyrrolate may be severely impaired in patients with renal failure. Dosage adjustments may be necessary in this population [see Pharmacokinetics (12.3)]
5.7 Autonomic Neuropathy, Hepatic Disease, Ulcerative Colitis, Prostatic Hypertrophy, Or Hiatal Hernia (5.7 Autonomic Neuropathy, Hepatic Disease, Ulcerative Colitis, Prostatic Hypertrophy, or Hiatal Hernia)
Use Glycopyrrolate Injection with caution in the elderly and in all patients with autonomic neuropathy, hepatic disease, ulcerative colitis, prostatic hypertrophy, or hiatal hernia, because anticholinergic drugs may aggravate these conditions. Consider dose reduction and closely monitor the elderly and patients with autonomic neuropathy, hepatic disease, ulcerative colitis, prostatic hypertrophy, or hiatal hernia.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:23.580336 · Updated: 2026-03-14T22:05:39.947280