These Highlights Do Not Include All The Information Needed To Use Daybue Or Daybue Stix Safely And Effectively. See Full Prescribing Information For Daybue And Daybue Stix.
67e6f2d9-21f6-466f-9def-826c6a4b8257
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.6 ) 12/2025
Indications and Usage
DAYBUE and DAYBUE STIX are indicated for the treatment of Rett syndrome in adults and pediatric patients 2 years of age and older.
Dosage and Administration
Recommended dosage is twice daily, morning and evening, according to patient weight. DAYBUE or DAYBUE STIX can be given with or without food. ( 2.1 ) Patient Weight Recommended Dosage 9 kg to less than 12 kg 5,000 mg twice daily 12 kg to less than 20 kg 6,000 mg twice daily 20 kg to less than 35 kg 8,000 mg twice daily 35 kg to less than 50 kg 10,000 mg twice daily 50 kg or more 12,000 mg twice daily Can be given orally or via gastrostomy (G) tube; doses administered via gastrojejunal (GJ) tubes must be administered through the G-port. ( 2.1 ) See Full Prescribing Information for instruction on dissolving DAYBUE STIX for oral solution powder. ( 2.3 ) See Full Prescribing Information for dosage recommendations in patients with renal impairment. ( 2.6 , 8.6 )
Warnings and Precautions
Diarrhea: Most patients experience diarrhea during treatment with DAYBUE. Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should start antidiarrheal treatment, increase oral fluids, and notify their healthcare provider. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected. ( 2.4 , 5.1 ) Weight Loss: Weight loss may occur in patients treated with DAYBUE or DAYBUE STIX. Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs. ( 2.4 , 5.2 ) Vomiting: Aspiration and aspiration pneumonia have occurred after vomiting in patients treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management. ( 2.5 , 5.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling: Diarrhea [see Warnings and Precautions (5.1) ] Weight Loss [see Warnings and Precautions (5.2) ] Vomiting [see Warnings and Precautions (5.3) ]
Drug Interactions
Orally administered CYP3A and/or P-gp sensitive substrates for which a small change in substrate plasma concentration may lead to serious adverse reactions: closely monitor for adverse reactions with concomitant use. ( 7.1 )
How Supplied
DAYBUE (trofinetide) 200 mg/mL oral solution is a pink to red, strawberry flavored solution supplied in a round high-density polyethylene (HDPE) multi-dose bottle with a child-resistant closure containing 450 mL of oral solution (NDC 63090-660-01). DAYBUE STIX (trofinetide) for oral solution is a white, off-white to pinkish powder with a strawberry flavor supplied in multi-layer aluminum packets as follows: Package Configuration and NDC Number Strength Individual Packet Carton of 60 Packets 5,000 mg 63090-663-01 63090-663-60 6,000 mg 63090-664-01 63090-664-60 8,000 mg 63090-665-01 63090-665-60
Medication Information
Warnings and Precautions
Diarrhea: Most patients experience diarrhea during treatment with DAYBUE. Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should start antidiarrheal treatment, increase oral fluids, and notify their healthcare provider. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected. ( 2.4 , 5.1 ) Weight Loss: Weight loss may occur in patients treated with DAYBUE or DAYBUE STIX. Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs. ( 2.4 , 5.2 ) Vomiting: Aspiration and aspiration pneumonia have occurred after vomiting in patients treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management. ( 2.5 , 5.3 )
Indications and Usage
DAYBUE and DAYBUE STIX are indicated for the treatment of Rett syndrome in adults and pediatric patients 2 years of age and older.
Dosage and Administration
Recommended dosage is twice daily, morning and evening, according to patient weight. DAYBUE or DAYBUE STIX can be given with or without food. ( 2.1 ) Patient Weight Recommended Dosage 9 kg to less than 12 kg 5,000 mg twice daily 12 kg to less than 20 kg 6,000 mg twice daily 20 kg to less than 35 kg 8,000 mg twice daily 35 kg to less than 50 kg 10,000 mg twice daily 50 kg or more 12,000 mg twice daily Can be given orally or via gastrostomy (G) tube; doses administered via gastrojejunal (GJ) tubes must be administered through the G-port. ( 2.1 ) See Full Prescribing Information for instruction on dissolving DAYBUE STIX for oral solution powder. ( 2.3 ) See Full Prescribing Information for dosage recommendations in patients with renal impairment. ( 2.6 , 8.6 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling: Diarrhea [see Warnings and Precautions (5.1) ] Weight Loss [see Warnings and Precautions (5.2) ] Vomiting [see Warnings and Precautions (5.3) ]
Drug Interactions
Orally administered CYP3A and/or P-gp sensitive substrates for which a small change in substrate plasma concentration may lead to serious adverse reactions: closely monitor for adverse reactions with concomitant use. ( 7.1 )
How Supplied
DAYBUE (trofinetide) 200 mg/mL oral solution is a pink to red, strawberry flavored solution supplied in a round high-density polyethylene (HDPE) multi-dose bottle with a child-resistant closure containing 450 mL of oral solution (NDC 63090-660-01). DAYBUE STIX (trofinetide) for oral solution is a white, off-white to pinkish powder with a strawberry flavor supplied in multi-layer aluminum packets as follows: Package Configuration and NDC Number Strength Individual Packet Carton of 60 Packets 5,000 mg 63090-663-01 63090-663-60 6,000 mg 63090-664-01 63090-664-60 8,000 mg 63090-665-01 63090-665-60
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.6 ) 12/2025
Section 42229-5
Preparation of DAYBUE STIX
- Determine the correct dosage as shown in Table 1 or Table 4 (for patients with moderate renal impairment).
- Select the appropriate packet strength and number of packets required for each dose, as shown in Table 3.
- Determine the appropriate volume of liquid within the recommended volume range as shown in Table 3, based on individual patient factors (e.g., age, palatability).
- Measure the volume of liquid determined in Step 3 by using a calibrated measuring device obtained from the pharmacy.
- Empty the entire contents of the DAYBUE STIX packet(s) into the measured liquid. Do not attempt to use partial packets to prepare a dose.
- Stir until the powder is completely dissolved.
- Administer the prepared oral solution immediately; do not store for future use.
- Discard any prepared oral solution that is not immediately administered.
| Dose | DAYBUE STIX Packet(s) Needed to Prepare Each Dose | Volume of Liquid Required to Dissolve Entire Dose
Liquid types include water or water-based beverage.
,
Volume should be selected within the recommended range based on individual patient factors.
|
|---|---|---|
| 5,000 mg | One 5,000 mg packet | 15 mL to 60 mL |
| 6,000 mg | One 6,000 mg packet | 15 mL to 60 mL |
| 8,000 mg | One 8,000 mg packet | 25 mL to 120 mL |
| 10,000 mg | Two 5,000 mg packets | 30 mL to 120 mL (each packet requires 15 mL to 60 mL) |
| 12,000 mg | Two 6,000 mg packets | 30 mL to 120 mL (each packet requires 15 mL to 60 mL) |
Section 42230-3
| PATIENT INFORMATION | |
|---|---|
| DAYBUE® (day-BYOO) (trofinetide) oral solution |
DAYBUE® STIX (day-BYOO STIX) (trofinetide) for oral solution |
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 12/2025 |
What is DAYBUE or DAYBUE STIX?
|
|
Before taking DAYBUE or DAYBUE STIX, tell your healthcare provider about all of your medical conditions, including if you:
Taking DAYBUE or DAYBUE STIX with certain medicines may affect the way other medicines work and can cause serious side effects. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take DAYBUE or DAYBUE STIX?
|
|
|
What are the possible side effects of DAYBUE or DAYBUE STIX? DAYBUE or DAYBUE STIX may cause side effects, including:
These are not all the possible side effects of DAYBUE or DAYBUE STIX. Tell your healthcare provider if you have any side effects that bother you or do not go away. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store DAYBUE or DAYBUE STIX?
|
|
|
General information about the safe and effective use of DAYBUE or DAYBUE STIX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DAYBUE or DAYBUE STIX for a condition for which it was not prescribed. Do not give DAYBUE or DAYBUE STIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DAYBUE or DAYBUE STIX that is written for health professionals. |
|
|
What are the ingredients in DAYBUE or DAYBUE STIX?
Active ingredient: trofinetide DAYBUE oral solution inactive ingredients: FD&C Red No. 40, maltitol, methylparaben sodium, propylparaben sodium, purified water, strawberry flavor, and sucralose. DAYBUE STIX for oral solution inactive ingredients: natural strawberry flavor and sucralose. Marketed by Acadia Pharmaceuticals Inc., San Diego, CA 92130 USA DAYBUE is a registered trademark of Acadia Pharmaceuticals Inc. ©2025 Acadia Pharmaceuticals Inc. All rights reserved. For more information, go to www.daybue.com or call 1-844-422-2342. |
5.1 Diarrhea
In Study 1 [see Clinical Studies (14)] and in long-term studies, 85% of patients treated with DAYBUE experienced diarrhea. In those treated with DAYBUE, 49% either had persistent diarrhea or recurrence after resolution despite dose interruptions, reductions, or concomitant antidiarrheal therapy. Diarrhea severity was of mild or moderate severity in 96% of cases. In Study 1, antidiarrheal medication was used in 51% of patients treated with DAYBUE.
Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should notify their healthcare provider, consider starting antidiarrheal treatment, and monitor hydration status and increase oral fluids, if needed. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected [see Dosage and Administration (2.4)].
5.3 Vomiting
In Study 1, vomiting occurred in 29% of patients treated with DAYBUE and in 12% of patients who received placebo [see Adverse Reactions (6.1)].
Patients with Rett syndrome are at risk for aspiration and aspiration pneumonia. Aspiration and aspiration pneumonia have been reported following vomiting in patients being treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management [see Dosage and Administration (2.5)].
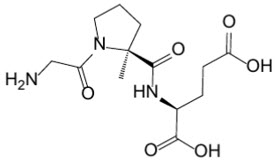
11 Description
DAYBUE oral solution and DAYBUE STIX for oral solution contain the active moiety trofinetide. The chemical name of trofinetide is (2S)-2-{[(2S)-1-(2-aminoacetyl)-2-methylpyrrolidine-2-carbonyl]amino}pentanedioic acid (IUPAC). The molecular formula of trofinetide is C13H21N3O6 and its molecular weight is 315.33 g/mol. The chemical structure is shown below.
Trofinetide is a white to off-white solid and is freely soluble in water.
DAYBUE oral solution is pink to red in color and contains 1 g of trofinetide in each 5 mL of solution (200 mg/mL). The oral solution also contains FD&C Red No. 40, maltitol, methylparaben sodium, propylparaben sodium, purified water, strawberry flavor, and sucralose as inactive ingredients.
DAYBUE STIX for oral solution is a white, off-white to pinkish powder to be dissolved in a cold to room temperature water or water-based beverage before administration and contains 5,000 mg, 6,000 mg, or 8,000 mg of trofinetide in each packet. The for oral solution powder contains natural strawberry flavor and sucralose as inactive ingredients.
2.7 Missed Dose
If a dose of DAYBUE or DAYBUE STIX is missed, the next dose should be taken as scheduled. Doses should not be doubled.
5.2 Weight Loss
In Study 1, 12% of patients treated with DAYBUE experienced weight loss of greater than 7% from baseline, compared to 4% of patients who received placebo. In long-term studies, 2.2% of patients discontinued treatment with DAYBUE due to weight loss.
Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs [see Dosage and Administration (2.4)].
16.1 How Supplied
DAYBUE (trofinetide) 200 mg/mL oral solution is a pink to red, strawberry flavored solution supplied in a round high-density polyethylene (HDPE) multi-dose bottle with a child-resistant closure containing 450 mL of oral solution (NDC 63090-660-01).
DAYBUE STIX (trofinetide) for oral solution is a white, off-white to pinkish powder with a strawberry flavor supplied in multi-layer aluminum packets as follows:
| Package Configuration and NDC Number | ||
|---|---|---|
| Strength | Individual Packet | Carton of 60 Packets |
| 5,000 mg | 63090-663-01 | 63090-663-60 |
| 6,000 mg | 63090-664-01 | 63090-664-60 |
| 8,000 mg | 63090-665-01 | 63090-665-60 |
8.4 Pediatric Use
The safety and effectiveness of DAYBUE and DAYBUE STIX for the treatment of Rett syndrome have been established in pediatric patients aged 2 years and older. The safety and effectiveness of DAYBUE for the treatment of Rett syndrome in pediatric patients 5 years of age and older was established in a randomized, double-blind, placebo-controlled, 12-week study (Study 1), which included 108 pediatric patients age 5 to less than 12 years of age and 47 pediatric patients age 12 to less than 17 years of age [see Adverse Reactions (6.1) and Clinical Studies (14)]. Use of DAYBUE or DAYBUE STIX in patients 2 to 4 years of age is supported by evidence from Study 1 and pharmacokinetic and safety data in 13 pediatric patients 2 to 4 years of age treated with DAYBUE for 12 weeks [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients less than 2 years of age have not been established.
8.5 Geriatric Use
Clinical studies of DAYBUE did not include patients 65 years of age and older to determine whether or not they respond differently from younger patients. This drug is known to be substantially excreted by the kidney. Because elderly patients are more likely to have decreased renal function, it may be useful to monitor renal function.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling:
- Diarrhea [see Warnings and Precautions (5.1)]
- Weight Loss [see Warnings and Precautions (5.2)]
- Vomiting [see Warnings and Precautions (5.3)]
7 Drug Interactions
- Orally administered CYP3A and/or P-gp sensitive substrates for which a small change in substrate plasma concentration may lead to serious adverse reactions: closely monitor for adverse reactions with concomitant use. (7.1)
8.6 Renal Impairment
Mild renal impairment is not expected to impact the exposure of trofinetide; therefore, dosage adjustment is not necessary. Dosage adjustment of DAYBUE or DAYBUE STIX is recommended in patients with moderate renal impairment (adult: eGFR 30 to 59 mL/min; pediatric: eGFR 30 to 59 mL/min/1.73 m2) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)]. Administration of DAYBUE or DAYBUE STIX to patients with severe renal impairment (eGFR less than 30 mL/min for adults or less than 30 mL/min/1.73 m2 for pediatrics) is not recommended.
12.3 Pharmacokinetics
DAYBUE STIX for oral solution demonstrated comparable bioavailability to DAYBUE oral solution in adult healthy subjects under fasted state.
Trofinetide exhibits linear kinetics with no time- or dose-dependent effect on pharmacokinetic parameters. Systemic exposure to trofinetide was dose-proportional across the studied dose range. Minimal to no accumulation was observed following multiple-dose administration.
2.1 Recommended Dosage
The recommended dosage of DAYBUE or DAYBUE STIX is based on patient weight as shown in Table 1. Administer DAYBUE or DAYBUE STIX orally or via gastrostomy (G) tube twice daily, in the morning and evening, with or without food. Doses administered via gastrojejunal (GJ) tubes must be administered through the G-port.
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 5,000 mg twice daily |
| 12 kg to less than 20 kg | 6,000 mg twice daily |
| 20 kg to less than 35 kg | 8,000 mg twice daily |
| 35 kg to less than 50 kg | 10,000 mg twice daily |
| 50 kg or more | 12,000 mg twice daily |
1 Indications and Usage
DAYBUE and DAYBUE STIX are indicated for the treatment of Rett syndrome in adults and pediatric patients 2 years of age and older.
12.1 Mechanism of Action
The mechanism by which trofinetide exerts therapeutic effects in patients with Rett syndrome is unknown.
5 Warnings and Precautions
- Diarrhea: Most patients experience diarrhea during treatment with DAYBUE. Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should start antidiarrheal treatment, increase oral fluids, and notify their healthcare provider. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected. (2.4, 5.1)
- Weight Loss: Weight loss may occur in patients treated with DAYBUE or DAYBUE STIX. Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs. (2.4, 5.2)
- Vomiting: Aspiration and aspiration pneumonia have occurred after vomiting in patients treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management. (2.5, 5.3)
2 Dosage and Administration
- Recommended dosage is twice daily, morning and evening, according to patient weight. DAYBUE or DAYBUE STIX can be given with or without food. (2.1)
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 5,000 mg twice daily |
| 12 kg to less than 20 kg | 6,000 mg twice daily |
| 20 kg to less than 35 kg | 8,000 mg twice daily |
| 35 kg to less than 50 kg | 10,000 mg twice daily |
| 50 kg or more | 12,000 mg twice daily |
- Can be given orally or via gastrostomy (G) tube; doses administered via gastrojejunal (GJ) tubes must be administered through the G-port. (2.1)
- See Full Prescribing Information for instruction on dissolving DAYBUE STIX for oral solution powder. (2.3)
- See Full Prescribing Information for dosage recommendations in patients with renal impairment. (2.6, 8.6)
3 Dosage Forms and Strengths
Oral solution: 200 mg/mL of a pink to red, strawberry flavored solution.
For oral solution: 5,000 mg, 6,000 mg, or 8,000 mg of white, off-white to pinkish powder with strawberry flavor, packaged in individual packets.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of DAYBUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Aspiration and aspiration pneumonia secondary to vomiting [see Warnings and Precautions (5.3)].
8 Use in Specific Populations
Severe renal impairment: DAYBUE and DAYBUE STIX are not recommended. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled and uncontrolled trials in patients with Rett syndrome, 260 patients ages 2 to 40 years were treated with DAYBUE, including 109 patients treated for more than 6 months, 69 patients treated for more than 1 year, and 4 patients treated for more than 2 years.
The safety of DAYBUE STIX has been established from an adequate, well-controlled study, and open-label studies of DAYBUE [see Clinical Studies (14)]. Below is a display of the adverse reactions of DAYBUE in these studies.
17 Patient Counseling Information
Advise the caregiver or patient to read the FDA-approved patient labeling (Patient Information).
2.2 Daybue Oral Solution Preparation
Table 2 includes the volume of DAYBUE oral solution to administer for the corresponding recommended dosage [see Dosage and Administration (2.1, 2.6)]. A calibrated measuring device, such as an oral syringe or oral dosing cup, should be obtained from the pharmacy to measure and deliver the prescribed dose accurately. A household measuring cup, teaspoon, or tablespoon is not an adequate measuring device.
| Dosage | DAYBUE Oral Solution Volume |
|---|---|
|
2,500 mg twice daily Dosage for patients with moderate renal impairment [see Dosage and Administration (2.6)]
|
12.5 mL twice daily |
| 3,000 mg twice daily | 15 mL twice daily |
| 4,000 mg twice daily | 20 mL twice daily |
| 5,000 mg twice daily | 25 mL twice daily |
| 6,000 mg twice daily | 30 mL twice daily |
| 8,000 mg twice daily | 40 mL twice daily |
| 10,000 mg twice daily | 50 mL twice daily |
| 12,000 mg twice daily | 60 mL twice daily |
Discard any unused DAYBUE oral solution after 14 days of first opening the bottle [see How Supplied/Storage and Handling (16.2)].
2.3 Daybue Stix for Oral Solution Preparation
Prior to administration, DAYBUE STIX for oral solution powder must be dissolved in a cold to room temperature water or water-based beverage (juice, tea, lemonade, limeade, or liquid hydration).
2.4 Dose Modification for Diarrhea Or Weight Loss
Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs, if dehydration is suspected, or if significant weight loss occurs [see Warnings and Precautions (5.1, 5.2)].
2.5 Dose Modification for Vomiting After Administration
If vomiting occurs after DAYBUE or DAYBUE STIX administration, an additional dose should not be taken. Instead, continue with the next scheduled dose. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management [see Warnings and Precautions (5.3)].
2.6 Dosage Recommendations in Patients With Renal Impairment
No dosage adjustment is recommended for patients with mild renal impairment (estimated glomerular filtration rate [eGFR] 60 to 89 mL/min for adult patients or 60 to 89 mL/min/1.73 m2 for pediatric patients). The recommended dosage of DAYBUE or DAYBUE STIX for patients with moderate renal impairment (eGFR 30 to 59 mL/min for adult patients or 30 to 59 mL/min/1.73 m2 for pediatric patients) is described in Table 4 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)]. DAYBUE and DAYBUE STIX are not recommended for patients with severe renal impairment (eGFR less than 30 mL/min for adult patients or less than 30 mL/min/1.73 m2 for pediatric patients).
For patients with moderate renal impairment who require a dose less than 5,000 mg, DAYBUE oral solution is recommended. Do not attempt to use partial packets of DAYBUE STIX to prepare a dose.
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 2,500 mg twice daily
Use DAYBUE oral solution to administer this dosage.
|
| 12 kg to less than 20 kg | 3,000 mg twice daily |
| 20 kg to less than 35 kg | 4,000 mg twice daily |
| 35 kg to less than 50 kg | 5,000 mg twice daily |
| 50 kg or more | 6,000 mg twice daily |
Principal Display Panel – 450 Ml Oral Solution Bottle Label
NDC 63090-660-01
450 mL
Rx only
Daybue™
(trofinetide)
oral solution
200 mg/mL
Recommended Dosage:
See prescribing information.
For oral or G-tube administration only.
Principal Display Panel – 450 Ml Oral Solution Bottle Carton
NDC 63090-660-01
Daybue™
(trofinetide)
oral solution
200 mg/mL
Recommended Dosage:
See prescribing information.
For oral or G-tube administration only.
450 mL
Rx only
Principal Display Panel – 5,000 Mg for Oral Solution Powder Carton
NDC 63090-663-60
Daybue® Stix
(trofinetide)
for oral solution
5,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 5,000 Mg for Oral Solution Powder Packet
NDC 63090-663-01
Daybue® Stix
(trofinetide)
for oral solution
5,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Principal Display Panel – 6,000 Mg for Oral Solution Powder Carton
NDC 63090-664-60
Daybue® Stix
(trofinetide)
for oral solution
6,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 6,000 Mg for Oral Solution Powder Packet
NDC 63090-664-01
Daybue® Stix
(trofinetide)
for oral solution
6,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Principal Display Panel – 8,000 Mg for Oral Solution Powder Carton
NDC 63090-665-60
Daybue® Stix
(trofinetide)
for oral solution
8,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 8,000 Mg for Oral Solution Powder Packet
NDC 63090-665-01
Daybue® Stix
(trofinetide)
for oral solution
8,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Structured Label Content
Section 42229-5 (42229-5)
Preparation of DAYBUE STIX
- Determine the correct dosage as shown in Table 1 or Table 4 (for patients with moderate renal impairment).
- Select the appropriate packet strength and number of packets required for each dose, as shown in Table 3.
- Determine the appropriate volume of liquid within the recommended volume range as shown in Table 3, based on individual patient factors (e.g., age, palatability).
- Measure the volume of liquid determined in Step 3 by using a calibrated measuring device obtained from the pharmacy.
- Empty the entire contents of the DAYBUE STIX packet(s) into the measured liquid. Do not attempt to use partial packets to prepare a dose.
- Stir until the powder is completely dissolved.
- Administer the prepared oral solution immediately; do not store for future use.
- Discard any prepared oral solution that is not immediately administered.
| Dose | DAYBUE STIX Packet(s) Needed to Prepare Each Dose | Volume of Liquid Required to Dissolve Entire Dose
Liquid types include water or water-based beverage.
,
Volume should be selected within the recommended range based on individual patient factors.
|
|---|---|---|
| 5,000 mg | One 5,000 mg packet | 15 mL to 60 mL |
| 6,000 mg | One 6,000 mg packet | 15 mL to 60 mL |
| 8,000 mg | One 8,000 mg packet | 25 mL to 120 mL |
| 10,000 mg | Two 5,000 mg packets | 30 mL to 120 mL (each packet requires 15 mL to 60 mL) |
| 12,000 mg | Two 6,000 mg packets | 30 mL to 120 mL (each packet requires 15 mL to 60 mL) |
Section 42230-3 (42230-3)
| PATIENT INFORMATION | |
|---|---|
| DAYBUE® (day-BYOO) (trofinetide) oral solution |
DAYBUE® STIX (day-BYOO STIX) (trofinetide) for oral solution |
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 12/2025 |
What is DAYBUE or DAYBUE STIX?
|
|
Before taking DAYBUE or DAYBUE STIX, tell your healthcare provider about all of your medical conditions, including if you:
Taking DAYBUE or DAYBUE STIX with certain medicines may affect the way other medicines work and can cause serious side effects. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take DAYBUE or DAYBUE STIX?
|
|
|
What are the possible side effects of DAYBUE or DAYBUE STIX? DAYBUE or DAYBUE STIX may cause side effects, including:
These are not all the possible side effects of DAYBUE or DAYBUE STIX. Tell your healthcare provider if you have any side effects that bother you or do not go away. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store DAYBUE or DAYBUE STIX?
|
|
|
General information about the safe and effective use of DAYBUE or DAYBUE STIX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DAYBUE or DAYBUE STIX for a condition for which it was not prescribed. Do not give DAYBUE or DAYBUE STIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DAYBUE or DAYBUE STIX that is written for health professionals. |
|
|
What are the ingredients in DAYBUE or DAYBUE STIX?
Active ingredient: trofinetide DAYBUE oral solution inactive ingredients: FD&C Red No. 40, maltitol, methylparaben sodium, propylparaben sodium, purified water, strawberry flavor, and sucralose. DAYBUE STIX for oral solution inactive ingredients: natural strawberry flavor and sucralose. Marketed by Acadia Pharmaceuticals Inc., San Diego, CA 92130 USA DAYBUE is a registered trademark of Acadia Pharmaceuticals Inc. ©2025 Acadia Pharmaceuticals Inc. All rights reserved. For more information, go to www.daybue.com or call 1-844-422-2342. |
5.1 Diarrhea
In Study 1 [see Clinical Studies (14)] and in long-term studies, 85% of patients treated with DAYBUE experienced diarrhea. In those treated with DAYBUE, 49% either had persistent diarrhea or recurrence after resolution despite dose interruptions, reductions, or concomitant antidiarrheal therapy. Diarrhea severity was of mild or moderate severity in 96% of cases. In Study 1, antidiarrheal medication was used in 51% of patients treated with DAYBUE.
Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should notify their healthcare provider, consider starting antidiarrheal treatment, and monitor hydration status and increase oral fluids, if needed. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected [see Dosage and Administration (2.4)].
5.3 Vomiting
In Study 1, vomiting occurred in 29% of patients treated with DAYBUE and in 12% of patients who received placebo [see Adverse Reactions (6.1)].
Patients with Rett syndrome are at risk for aspiration and aspiration pneumonia. Aspiration and aspiration pneumonia have been reported following vomiting in patients being treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management [see Dosage and Administration (2.5)].
11 Description (11 DESCRIPTION)
DAYBUE oral solution and DAYBUE STIX for oral solution contain the active moiety trofinetide. The chemical name of trofinetide is (2S)-2-{[(2S)-1-(2-aminoacetyl)-2-methylpyrrolidine-2-carbonyl]amino}pentanedioic acid (IUPAC). The molecular formula of trofinetide is C13H21N3O6 and its molecular weight is 315.33 g/mol. The chemical structure is shown below.
Trofinetide is a white to off-white solid and is freely soluble in water.
DAYBUE oral solution is pink to red in color and contains 1 g of trofinetide in each 5 mL of solution (200 mg/mL). The oral solution also contains FD&C Red No. 40, maltitol, methylparaben sodium, propylparaben sodium, purified water, strawberry flavor, and sucralose as inactive ingredients.
DAYBUE STIX for oral solution is a white, off-white to pinkish powder to be dissolved in a cold to room temperature water or water-based beverage before administration and contains 5,000 mg, 6,000 mg, or 8,000 mg of trofinetide in each packet. The for oral solution powder contains natural strawberry flavor and sucralose as inactive ingredients.
2.7 Missed Dose
If a dose of DAYBUE or DAYBUE STIX is missed, the next dose should be taken as scheduled. Doses should not be doubled.
5.2 Weight Loss
In Study 1, 12% of patients treated with DAYBUE experienced weight loss of greater than 7% from baseline, compared to 4% of patients who received placebo. In long-term studies, 2.2% of patients discontinued treatment with DAYBUE due to weight loss.
Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs [see Dosage and Administration (2.4)].
16.1 How Supplied
DAYBUE (trofinetide) 200 mg/mL oral solution is a pink to red, strawberry flavored solution supplied in a round high-density polyethylene (HDPE) multi-dose bottle with a child-resistant closure containing 450 mL of oral solution (NDC 63090-660-01).
DAYBUE STIX (trofinetide) for oral solution is a white, off-white to pinkish powder with a strawberry flavor supplied in multi-layer aluminum packets as follows:
| Package Configuration and NDC Number | ||
|---|---|---|
| Strength | Individual Packet | Carton of 60 Packets |
| 5,000 mg | 63090-663-01 | 63090-663-60 |
| 6,000 mg | 63090-664-01 | 63090-664-60 |
| 8,000 mg | 63090-665-01 | 63090-665-60 |
8.4 Pediatric Use
The safety and effectiveness of DAYBUE and DAYBUE STIX for the treatment of Rett syndrome have been established in pediatric patients aged 2 years and older. The safety and effectiveness of DAYBUE for the treatment of Rett syndrome in pediatric patients 5 years of age and older was established in a randomized, double-blind, placebo-controlled, 12-week study (Study 1), which included 108 pediatric patients age 5 to less than 12 years of age and 47 pediatric patients age 12 to less than 17 years of age [see Adverse Reactions (6.1) and Clinical Studies (14)]. Use of DAYBUE or DAYBUE STIX in patients 2 to 4 years of age is supported by evidence from Study 1 and pharmacokinetic and safety data in 13 pediatric patients 2 to 4 years of age treated with DAYBUE for 12 weeks [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients less than 2 years of age have not been established.
8.5 Geriatric Use
Clinical studies of DAYBUE did not include patients 65 years of age and older to determine whether or not they respond differently from younger patients. This drug is known to be substantially excreted by the kidney. Because elderly patients are more likely to have decreased renal function, it may be useful to monitor renal function.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in labeling:
- Diarrhea [see Warnings and Precautions (5.1)]
- Weight Loss [see Warnings and Precautions (5.2)]
- Vomiting [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Orally administered CYP3A and/or P-gp sensitive substrates for which a small change in substrate plasma concentration may lead to serious adverse reactions: closely monitor for adverse reactions with concomitant use. (7.1)
8.6 Renal Impairment
Mild renal impairment is not expected to impact the exposure of trofinetide; therefore, dosage adjustment is not necessary. Dosage adjustment of DAYBUE or DAYBUE STIX is recommended in patients with moderate renal impairment (adult: eGFR 30 to 59 mL/min; pediatric: eGFR 30 to 59 mL/min/1.73 m2) [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)]. Administration of DAYBUE or DAYBUE STIX to patients with severe renal impairment (eGFR less than 30 mL/min for adults or less than 30 mL/min/1.73 m2 for pediatrics) is not recommended.
12.3 Pharmacokinetics
DAYBUE STIX for oral solution demonstrated comparable bioavailability to DAYBUE oral solution in adult healthy subjects under fasted state.
Trofinetide exhibits linear kinetics with no time- or dose-dependent effect on pharmacokinetic parameters. Systemic exposure to trofinetide was dose-proportional across the studied dose range. Minimal to no accumulation was observed following multiple-dose administration.
2.1 Recommended Dosage
The recommended dosage of DAYBUE or DAYBUE STIX is based on patient weight as shown in Table 1. Administer DAYBUE or DAYBUE STIX orally or via gastrostomy (G) tube twice daily, in the morning and evening, with or without food. Doses administered via gastrojejunal (GJ) tubes must be administered through the G-port.
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 5,000 mg twice daily |
| 12 kg to less than 20 kg | 6,000 mg twice daily |
| 20 kg to less than 35 kg | 8,000 mg twice daily |
| 35 kg to less than 50 kg | 10,000 mg twice daily |
| 50 kg or more | 12,000 mg twice daily |
1 Indications and Usage (1 INDICATIONS AND USAGE)
DAYBUE and DAYBUE STIX are indicated for the treatment of Rett syndrome in adults and pediatric patients 2 years of age and older.
12.1 Mechanism of Action
The mechanism by which trofinetide exerts therapeutic effects in patients with Rett syndrome is unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Diarrhea: Most patients experience diarrhea during treatment with DAYBUE. Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. If diarrhea occurs, patients should start antidiarrheal treatment, increase oral fluids, and notify their healthcare provider. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs or if dehydration is suspected. (2.4, 5.1)
- Weight Loss: Weight loss may occur in patients treated with DAYBUE or DAYBUE STIX. Monitor weight and interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if significant weight loss occurs. (2.4, 5.2)
- Vomiting: Aspiration and aspiration pneumonia have occurred after vomiting in patients treated with DAYBUE. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management. (2.5, 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage is twice daily, morning and evening, according to patient weight. DAYBUE or DAYBUE STIX can be given with or without food. (2.1)
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 5,000 mg twice daily |
| 12 kg to less than 20 kg | 6,000 mg twice daily |
| 20 kg to less than 35 kg | 8,000 mg twice daily |
| 35 kg to less than 50 kg | 10,000 mg twice daily |
| 50 kg or more | 12,000 mg twice daily |
- Can be given orally or via gastrostomy (G) tube; doses administered via gastrojejunal (GJ) tubes must be administered through the G-port. (2.1)
- See Full Prescribing Information for instruction on dissolving DAYBUE STIX for oral solution powder. (2.3)
- See Full Prescribing Information for dosage recommendations in patients with renal impairment. (2.6, 8.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Oral solution: 200 mg/mL of a pink to red, strawberry flavored solution.
For oral solution: 5,000 mg, 6,000 mg, or 8,000 mg of white, off-white to pinkish powder with strawberry flavor, packaged in individual packets.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of DAYBUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Aspiration and aspiration pneumonia secondary to vomiting [see Warnings and Precautions (5.3)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Severe renal impairment: DAYBUE and DAYBUE STIX are not recommended. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled and uncontrolled trials in patients with Rett syndrome, 260 patients ages 2 to 40 years were treated with DAYBUE, including 109 patients treated for more than 6 months, 69 patients treated for more than 1 year, and 4 patients treated for more than 2 years.
The safety of DAYBUE STIX has been established from an adequate, well-controlled study, and open-label studies of DAYBUE [see Clinical Studies (14)]. Below is a display of the adverse reactions of DAYBUE in these studies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the caregiver or patient to read the FDA-approved patient labeling (Patient Information).
2.2 Daybue Oral Solution Preparation (2.2 DAYBUE Oral Solution Preparation)
Table 2 includes the volume of DAYBUE oral solution to administer for the corresponding recommended dosage [see Dosage and Administration (2.1, 2.6)]. A calibrated measuring device, such as an oral syringe or oral dosing cup, should be obtained from the pharmacy to measure and deliver the prescribed dose accurately. A household measuring cup, teaspoon, or tablespoon is not an adequate measuring device.
| Dosage | DAYBUE Oral Solution Volume |
|---|---|
|
2,500 mg twice daily Dosage for patients with moderate renal impairment [see Dosage and Administration (2.6)]
|
12.5 mL twice daily |
| 3,000 mg twice daily | 15 mL twice daily |
| 4,000 mg twice daily | 20 mL twice daily |
| 5,000 mg twice daily | 25 mL twice daily |
| 6,000 mg twice daily | 30 mL twice daily |
| 8,000 mg twice daily | 40 mL twice daily |
| 10,000 mg twice daily | 50 mL twice daily |
| 12,000 mg twice daily | 60 mL twice daily |
Discard any unused DAYBUE oral solution after 14 days of first opening the bottle [see How Supplied/Storage and Handling (16.2)].
2.3 Daybue Stix for Oral Solution Preparation (2.3 DAYBUE STIX for Oral Solution Preparation)
Prior to administration, DAYBUE STIX for oral solution powder must be dissolved in a cold to room temperature water or water-based beverage (juice, tea, lemonade, limeade, or liquid hydration).
2.4 Dose Modification for Diarrhea Or Weight Loss (2.4 Dose Modification for Diarrhea or Weight Loss)
Advise patients to stop laxatives before starting DAYBUE or DAYBUE STIX. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if severe diarrhea occurs, if dehydration is suspected, or if significant weight loss occurs [see Warnings and Precautions (5.1, 5.2)].
2.5 Dose Modification for Vomiting After Administration
If vomiting occurs after DAYBUE or DAYBUE STIX administration, an additional dose should not be taken. Instead, continue with the next scheduled dose. Interrupt, reduce dose, or discontinue DAYBUE or DAYBUE STIX if vomiting is severe or occurs despite medical management [see Warnings and Precautions (5.3)].
2.6 Dosage Recommendations in Patients With Renal Impairment (2.6 Dosage Recommendations in Patients with Renal Impairment)
No dosage adjustment is recommended for patients with mild renal impairment (estimated glomerular filtration rate [eGFR] 60 to 89 mL/min for adult patients or 60 to 89 mL/min/1.73 m2 for pediatric patients). The recommended dosage of DAYBUE or DAYBUE STIX for patients with moderate renal impairment (eGFR 30 to 59 mL/min for adult patients or 30 to 59 mL/min/1.73 m2 for pediatric patients) is described in Table 4 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)]. DAYBUE and DAYBUE STIX are not recommended for patients with severe renal impairment (eGFR less than 30 mL/min for adult patients or less than 30 mL/min/1.73 m2 for pediatric patients).
For patients with moderate renal impairment who require a dose less than 5,000 mg, DAYBUE oral solution is recommended. Do not attempt to use partial packets of DAYBUE STIX to prepare a dose.
| Patient Weight | Recommended Dosage |
|---|---|
| 9 kg to less than 12 kg | 2,500 mg twice daily
Use DAYBUE oral solution to administer this dosage.
|
| 12 kg to less than 20 kg | 3,000 mg twice daily |
| 20 kg to less than 35 kg | 4,000 mg twice daily |
| 35 kg to less than 50 kg | 5,000 mg twice daily |
| 50 kg or more | 6,000 mg twice daily |
Principal Display Panel – 450 Ml Oral Solution Bottle Label (PRINCIPAL DISPLAY PANEL – 450 ML ORAL SOLUTION BOTTLE LABEL)
NDC 63090-660-01
450 mL
Rx only
Daybue™
(trofinetide)
oral solution
200 mg/mL
Recommended Dosage:
See prescribing information.
For oral or G-tube administration only.
Principal Display Panel – 450 Ml Oral Solution Bottle Carton (PRINCIPAL DISPLAY PANEL – 450 ML ORAL SOLUTION BOTTLE CARTON)
NDC 63090-660-01
Daybue™
(trofinetide)
oral solution
200 mg/mL
Recommended Dosage:
See prescribing information.
For oral or G-tube administration only.
450 mL
Rx only
Principal Display Panel – 5,000 Mg for Oral Solution Powder Carton (PRINCIPAL DISPLAY PANEL – 5,000 MG FOR ORAL SOLUTION POWDER CARTON)
NDC 63090-663-60
Daybue® Stix
(trofinetide)
for oral solution
5,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 5,000 Mg for Oral Solution Powder Packet (PRINCIPAL DISPLAY PANEL – 5,000 MG FOR ORAL SOLUTION POWDER PACKET)
NDC 63090-663-01
Daybue® Stix
(trofinetide)
for oral solution
5,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Principal Display Panel – 6,000 Mg for Oral Solution Powder Carton (PRINCIPAL DISPLAY PANEL – 6,000 MG FOR ORAL SOLUTION POWDER CARTON)
NDC 63090-664-60
Daybue® Stix
(trofinetide)
for oral solution
6,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 6,000 Mg for Oral Solution Powder Packet (PRINCIPAL DISPLAY PANEL – 6,000 MG FOR ORAL SOLUTION POWDER PACKET)
NDC 63090-664-01
Daybue® Stix
(trofinetide)
for oral solution
6,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Principal Display Panel – 8,000 Mg for Oral Solution Powder Carton (PRINCIPAL DISPLAY PANEL – 8,000 MG FOR ORAL SOLUTION POWDER CARTON)
NDC 63090-665-60
Daybue® Stix
(trofinetide)
for oral solution
8,000 mg
per packet
For Oral or G-tube
administration only
60 Packets
Rx Only
Principal Display Panel – 8,000 Mg for Oral Solution Powder Packet (PRINCIPAL DISPLAY PANEL – 8,000 MG FOR ORAL SOLUTION POWDER PACKET)
NDC 63090-665-01
Daybue® Stix
(trofinetide)
for oral solution
8,000 mg
For Oral or G-tube administration only
Keep out of reach of children
Rx Only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:34.461923 · Updated: 2026-03-14T22:40:50.579212