drug facts
66fd73be-05c8-4905-96f5-f73fa1447b3f
34390-5
HUMAN OTC DRUG LABEL
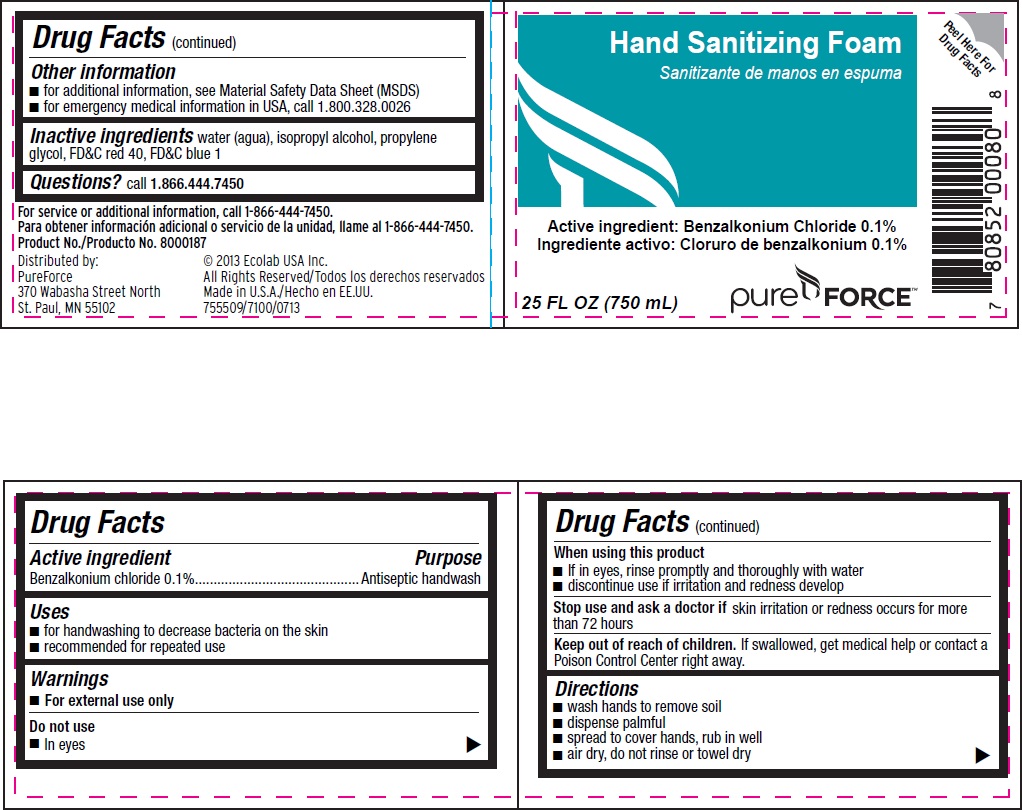
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antiseptic handwash
Description
Benzalkonium chloride 0.1%
Medication Information
Warnings
- For external use only
Uses
- for handwashing to decrease bacteria on the skin
- recommended for repeated use
Purpose
Antiseptic handwash
Directions
- wash hands to remove soil
- dispense palmful
- spread to cover hands, rub in well
- air dry, do not rinse or towel dry
Other Information
- for additional information, see Material Safety Data Sheet (MSDS)
- for emergency medical information in USA, call 1.800.328.0026
Description
Benzalkonium chloride 0.1%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3
Stop use and ask a doctor if skin irritation or redness occurs for more than 72 hours
Section 51727-6
Inactive ingredients water (aqua), isopropoyl alcohol, propylene glycol, FDC red 40, FDC blue 1
Section 53413-1
Questions? call 1.866.444.7450
Do Not Use
- In eyes
Active Ingredient
Benzalkonium chloride 0.1%
When Using This Product
- If in eyes, rinse promptly and thoroughly with water
- discontinue use if irritation and redness develop
Principal Display Panel With Representative Label
Hand Sanitizing Foam
Active ingredient: Benzalkonium chloride 0.1%
25 FL OZ (750 mL)
Pure Force™
Distributed by: © 2013 Ecolab USA Inc.
PureForce All Rights Reserved
370 Wabasha Street North Made in U.S.A.
St. Paul, MN 55102 755509/7100/0713
Structured Label Content
Warnings
- For external use only
Uses
- for handwashing to decrease bacteria on the skin
- recommended for repeated use
Purpose
Antiseptic handwash
Directions
- wash hands to remove soil
- dispense palmful
- spread to cover hands, rub in well
- air dry, do not rinse or towel dry
Other Information (Other information)
- for additional information, see Material Safety Data Sheet (MSDS)
- for emergency medical information in USA, call 1.800.328.0026
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3 (50569-3)
Stop use and ask a doctor if skin irritation or redness occurs for more than 72 hours
Section 51727-6 (51727-6)
Inactive ingredients water (aqua), isopropoyl alcohol, propylene glycol, FDC red 40, FDC blue 1
Section 53413-1 (53413-1)
Questions? call 1.866.444.7450
Do Not Use (Do not use)
- In eyes
Active Ingredient
Benzalkonium chloride 0.1%
When Using This Product (When using this product)
- If in eyes, rinse promptly and thoroughly with water
- discontinue use if irritation and redness develop
Principal Display Panel With Representative Label (Principal Display Panel with Representative Label)
Hand Sanitizing Foam
Active ingredient: Benzalkonium chloride 0.1%
25 FL OZ (750 mL)
Pure Force™
Distributed by: © 2013 Ecolab USA Inc.
PureForce All Rights Reserved
370 Wabasha Street North Made in U.S.A.
St. Paul, MN 55102 755509/7100/0713
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:16.166199 · Updated: 2026-03-14T23:03:15.788850