Mupirocin Ointment
66db1071-024f-aa19-e053-2a91aa0a8e39
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes) .
Dosage and Administration
For Topical Use Only. Apply a small amount of mupirocin ointment, with a cotton swab or gauze pad, to the affected area 3 times daily for up to 10 days. Cover the treated area with gauze dressing if desired. Re-evaluate patients not showing a clinical response within 3 to 5 days. Mupirocin ointment is not for intranasal, ophthalmic, or other mucosal use [see Warnings and Precautions (5.2 , 5.6) ] . Do not apply mupirocin ointment concurrently with any other lotions, creams, or ointments [see Clinical Pharmacology (12.3) ] .
Contraindications
Mupirocin ointment is contraindicated in patients with known hypersensitivity to mupirocin or any of the excipients of mupirocin ointment.
Warnings and Precautions
Severe Allergic Reactions: Anaphylaxis, urticaria, angioedema, and generalized rash have been reported in patients treated with formulations of mupirocin, including mupirocin ointment. ( 5.1 ) Eye Irritation: Avoid contact with eyes. ( 5.2 ) Local Irritation: Discontinue in the event of sensitization or severe local irritation. ( 5.3 ) Clostridium difficile- Associated Diarrhea (CDAD): If diarrhea occurs, evaluate patients for CDAD. ( 5.4 ) Potential for Microbial Overgrowth: Prolonged use may result in overgrowth of nonsusceptible microorganisms, including fungi. ( 5.5 ) Risk Associated with Mucosal Use: Mupirocin ointment is not formulated for use on mucosal surfaces. A separate formulation, mupirocin nasal ointment, is available for intranasal use. ( 5.6 ) Risk of Polyethylene Glycol Absorption: Mupirocin ointment should not be used where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment. ( 5.7 ) Risk Associated with Use at Intravenous Sites: Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance. ( 5.8 )
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Severe Allergic Reactions [see Warnings and Precautions (5.1) ] Eye Irritation [see Warnings and Precautions (5.2) ] Local Irritation [see Warnings and Precautions (5.3) ] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ]
How Supplied
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base. Mupirocin Ointment USP, 2% is supplied as follows: NDC 68071-4321-2
Storage and Handling
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base. Mupirocin Ointment USP, 2% is supplied as follows: NDC 68071-4321-2
Description
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes) .
Medication Information
Warnings and Precautions
Severe Allergic Reactions: Anaphylaxis, urticaria, angioedema, and generalized rash have been reported in patients treated with formulations of mupirocin, including mupirocin ointment. ( 5.1 ) Eye Irritation: Avoid contact with eyes. ( 5.2 ) Local Irritation: Discontinue in the event of sensitization or severe local irritation. ( 5.3 ) Clostridium difficile- Associated Diarrhea (CDAD): If diarrhea occurs, evaluate patients for CDAD. ( 5.4 ) Potential for Microbial Overgrowth: Prolonged use may result in overgrowth of nonsusceptible microorganisms, including fungi. ( 5.5 ) Risk Associated with Mucosal Use: Mupirocin ointment is not formulated for use on mucosal surfaces. A separate formulation, mupirocin nasal ointment, is available for intranasal use. ( 5.6 ) Risk of Polyethylene Glycol Absorption: Mupirocin ointment should not be used where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment. ( 5.7 ) Risk Associated with Use at Intravenous Sites: Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance. ( 5.8 )
Indications and Usage
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes) .
Dosage and Administration
For Topical Use Only. Apply a small amount of mupirocin ointment, with a cotton swab or gauze pad, to the affected area 3 times daily for up to 10 days. Cover the treated area with gauze dressing if desired. Re-evaluate patients not showing a clinical response within 3 to 5 days. Mupirocin ointment is not for intranasal, ophthalmic, or other mucosal use [see Warnings and Precautions (5.2 , 5.6) ] . Do not apply mupirocin ointment concurrently with any other lotions, creams, or ointments [see Clinical Pharmacology (12.3) ] .
Contraindications
Mupirocin ointment is contraindicated in patients with known hypersensitivity to mupirocin or any of the excipients of mupirocin ointment.
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Severe Allergic Reactions [see Warnings and Precautions (5.1) ] Eye Irritation [see Warnings and Precautions (5.2) ] Local Irritation [see Warnings and Precautions (5.3) ] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ]
Storage and Handling
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base. Mupirocin Ointment USP, 2% is supplied as follows: NDC 68071-4321-2
How Supplied
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base. Mupirocin Ointment USP, 2% is supplied as follows: NDC 68071-4321-2
Description
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes) .
Principal Display Panel
Section 42229-5
Immune System Disorders
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash [see Warnings and Precautions (5.1)] .
Section 42230-3
| PATIENT INFORMATION MUPIROCIN (mue pir`oh sin) OINTMENT | ||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: June, 2017 PK-4402-4 2 |
||
|
What is mupirocin ointment?
Mupirocin ointment is a prescription medicine used on the skin (topical use) to treat a skin infection called impetigo that is caused by bacteria called Staphylococcus aureus and Streptococcus pyogenes. It is not known if mupirocin ointment is safe and effective in children under 2 months of age. |
||
|
Who should not use mupirocin ointment?
Do not use mupirocin ointment if:
|
||
|
What should I tell my healthcare provider before using mupirocin ointment?
Before using mupirocin ointment, tell your healthcare provider about all of your medical conditions including if you:
|
||
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Do not mix mupirocin ointment with other lotions, creams, or ointments. | ||
How should I use mupirocin ointment?
|
||
|
What are the possible side effects of mupirocin ointment?
Mupirocin ointment may cause serious side effects, including:
|
||
|
|
|
|
||
How should I store mupirocin ointment?
|
||
|
General information about the safe and effective use of mupirocin ointment.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use mupirocin ointment for a condition for which it was not prescribed. Do not give mupirocin ointment to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about mupirocin ointment that is written for health professionals. |
||
|
What are the ingredients in mupirocin ointment?
Active Ingredient: mupirocin Inactive Ingredients: polyethylene glycol 400 and polyethylene glycol 3350 Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1 Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 For more information, call 1-886-923-4914. |
Section 44425-7
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
15 References
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-sixth Informational Supplement. CLSI document M100-S26. Clinical and Laboratory Standards Institute, 950 West Valley Rd., Suite 2500, Wayne, PA 19087, USA, 2016.
- Patel J, Gorwitz RJ, et al. Mupirocin Resistance. Clinical Infectious Diseases. 2009; 49(6): 935-41.
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard – Tenth Edition. CLSI document M07-A10. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard – Twelfth Edition. CLSI document M02-A12. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Finlay JE, Miller LA, Poupard JA. Interpretive criteria for testing susceptibility of staphylococci to mupirocin. Antimicrob Agents Chemother. 1997; 41(5):1137-1139.
11 Description
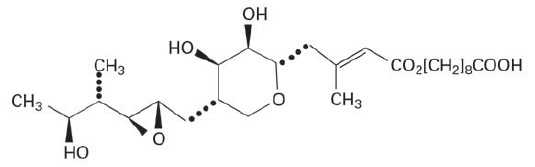
Mupirocin Ointment USP, 2% contains the RNA synthetase inhibitor antibacterial, mupirocin. The chemical name is ( E)-(2 S,3 R,4 R,5 S)-5-[(2 S,3 S,4 S,5 S)-2,3-epoxy-5-hydroxy-4-methylhexyl] tetrahydro-3,4-dihydroxy-ß-methyl-2 H-pyran-2-crotonic acid, ester with 9-hydroxynonanoic acid. The molecular formula of mupirocin is C 26H 44O 9, and the molecular weight is 500.6. The structural formula of mupirocin is:
Each gram of mupirocin ointment, 2% contains 20 mg mupirocin in a water-miscible ointment base (polyethylene glycol ointment) consisting of polyethylene glycol 400 and polyethylene glycol 3350.
12.4 Microbiology
Mupirocin is an RNA synthetase inhibitor antibacterial produced by fermentation using the organism Pseudomonas fluorescens.
8.4 Pediatric Use
The safety and effectiveness of mupirocin ointment have been established in the age range of 2 months to 16 years. Use of mupirocin ointment in these age-groups is supported by evidence from adequate and well-controlled trials of mupirocin ointment in impetigo in pediatric subjects studied as a part of the pivotal clinical trials in adults [see Clinical Studies (14)] .
5.2 Eye Irritation
Avoid contact with the eyes. In case of accidental contact, rinse well with water.
14 Clinical Studies
The efficacy of topical mupirocin ointment in impetigo was tested in 2 trials. In the first, subjects with impetigo were randomized to receive either mupirocin ointment or vehicle placebo 3 times daily for 8 to 12 days. Clinical efficacy rates at end of therapy in the evaluable populations (adults and pediatric subjects included) were 71% for mupirocin ointment (n = 49) and 35% for vehicle placebo (n = 51). Pathogen eradication rates in the evaluable populations were 94% for mupirocin ointment and 62% for vehicle placebo.
In the second trial, subjects with impetigo were randomized to receive either mupirocin ointment 3 times daily or 30 mg per kg to 40 mg per kg oral erythromycin ethylsuccinate per day (this was an unblinded trial) for 8 days. There was a follow-up visit 1 week after treatment ended. Clinical efficacy rates at the follow-up visit in the evaluable populations (adults and pediatric subjects included) were 93% for mupirocin ointment (n = 29) and 78.5% for erythromycin (n = 28). Pathogen eradication rates in the evaluable populations were 100% for both test groups.
4 Contraindications
Mupirocin ointment is contraindicated in patients with known hypersensitivity to mupirocin or any of the excipients of mupirocin ointment.
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Severe Allergic Reactions [see Warnings and Precautions (5.1)]
- Eye Irritation [see Warnings and Precautions (5.2)]
- Local Irritation [see Warnings and Precautions (5.3)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
5.3 Local Irritation
In the event of a sensitization or severe local irritation from mupirocin ointment, usage should be discontinued, and appropriate alternative therapy for the infection instituted.
1 Indications and Usage
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes).
12.1 Mechanism of Action
Mupirocin is an RNA synthetase inhibitor antibacterial [see Microbiology (12.4)] .
5.4 Clostridium Difficile
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5 Warnings and Precautions
- Severe Allergic Reactions: Anaphylaxis, urticaria, angioedema, and generalized rash have been reported in patients treated with formulations of mupirocin, including mupirocin ointment. ( 5.1)
- Eye Irritation: Avoid contact with eyes. ( 5.2)
- Local Irritation: Discontinue in the event of sensitization or severe local irritation. ( 5.3)
- Clostridium difficile-Associated Diarrhea (CDAD): If diarrhea occurs, evaluate patients for CDAD. ( 5.4)
- Potential for Microbial Overgrowth: Prolonged use may result in overgrowth of nonsusceptible microorganisms, including fungi. ( 5.5)
- Risk Associated with Mucosal Use: Mupirocin ointment is not formulated for use on mucosal surfaces. A separate formulation, mupirocin nasal ointment, is available for intranasal use. ( 5.6)
- Risk of Polyethylene Glycol Absorption: Mupirocin ointment should not be used where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment. ( 5.7)
- Risk Associated with Use at Intravenous Sites: Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance. ( 5.8)
2 Dosage and Administration
- For Topical Use Only.
- Apply a small amount of mupirocin ointment, with a cotton swab or gauze pad, to the affected area 3 times daily for up to 10 days.
- Cover the treated area with gauze dressing if desired.
- Re-evaluate patients not showing a clinical response within 3 to 5 days.
- Mupirocin ointment is not for intranasal, ophthalmic, or other mucosal use [see Warnings and Precautions (5.2, 5.6)] .
- Do not apply mupirocin ointment concurrently with any other lotions, creams, or ointments [see Clinical Pharmacology (12.3)] .
3 Dosage Forms and Strengths
Each gram of mupirocin ointment contains 20 mg mupirocin in a water-miscible ointment base supplied in 15-gram, 22-gram and 30-gram tubes.
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following reactions have been identified during postmarketing use of mupirocin ointment. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal relationship to mupirocin ointment.
5.1 Severe Allergic Reactions
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash, have been reported in patients treated with formulations of mupirocin, including mupirocin ointment [see Adverse Reactions (6.2)] .
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following local adverse reactions were reported by at least 1% of subjects in connection with the use of mupirocin ointment in clinical trials: burning, stinging, or pain in 1.5% of subjects; itching in 1% of subjects. Rash, nausea, erythema, dry skin, tenderness, swelling, contact dermatitis, and increased exudate were reported in less than 1% of subjects.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise the patient to administer mupirocin ointment as follows:
- Use mupirocin ointment only as directed by the healthcare provider. It is for external use only. Avoid contact of mupirocin ointment with the eyes. If mupirocin ointment gets in the eyes, rinse thoroughly with water.
- Do not use mupirocin ointment in the nose.
- Wash your hands before and after applying mupirocin ointment.
- Use a gauze pad or cotton swab to apply a small amount of mupirocin ointment to the affected area. The treated area may be covered by gauze dressing if desired.
- Report to the healthcare provider any signs of local adverse reactions. Mupirocin ointment should be stopped and the healthcare provider contacted if irritation, severe itching, or rash occurs.
- Report to the healthcare provider or go to the nearest emergency room if severe allergic reactions, such as swelling of the lips, face, or tongue, or wheezing occur [see Warnings and Precautions (5.1)] .
- If impetigo has not improved in 3 to 5 days, contact the healthcare provider.
16 How Supplied/storage and Handling
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base.
Mupirocin Ointment USP, 2% is supplied as follows:
NDC 68071-4321-2
5.6 Risk Associated With Mucosal Use
Mupirocin ointment is not formulated for use on mucosal surfaces. Intranasal use has been associated with isolated reports of stinging and drying. A separate formulation, mupirocin calcium nasal ointment, is available for intranasal use.
5.5 Potential for Microbial Overgrowth
As with other antibacterial products, prolonged use of mupirocin ointment may result in overgrowth of nonsusceptible microorganisms, including fungi [see Dosage and Administration (2)] .
5.7 Risk of Polyethylene Glycol Absorption
Polyethylene glycol can be absorbed from open wounds and damaged skin and is excreted by the kidneys. In common with other polyethylene glycol-based ointments, mupirocin ointment should not be used in conditions where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment.
5.8 Risk Associated With Use At Intravenous Sites
Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential of mupirocin have not been conducted.
Results of the following studies performed with mupirocin calcium or mupirocin sodium in vitro and in vivo did not indicate a potential for genotoxicity: rat primary hepatocyte unscheduled DNA synthesis, sediment analysis for DNA strand breaks, Salmonella reversion test (Ames), Escherichia coli mutation assay, metaphase analysis of human lymphocytes, mouse lymphoma assay, and bone marrow micronuclei assay in mice.
In a fertility/reproductive performance study (with dosing through lactation), mupirocin administered subcutaneously to male and female rats at doses up to 100 mg per kg per day which is 14 times the human topical dose (approximately 60 mg mupirocin per day) based on calculations of dose divided by the entire body surface area, did not result in impaired fertility or impaired reproductive performance attributable to mupirocin.
Structured Label Content
Principal Display Panel (PRINCIPAL DISPLAY PANEL -)
Section 42229-5 (42229-5)
Immune System Disorders
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash [see Warnings and Precautions (5.1)] .
Section 42230-3 (42230-3)
| PATIENT INFORMATION MUPIROCIN (mue pir`oh sin) OINTMENT | ||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: June, 2017 PK-4402-4 2 |
||
|
What is mupirocin ointment?
Mupirocin ointment is a prescription medicine used on the skin (topical use) to treat a skin infection called impetigo that is caused by bacteria called Staphylococcus aureus and Streptococcus pyogenes. It is not known if mupirocin ointment is safe and effective in children under 2 months of age. |
||
|
Who should not use mupirocin ointment?
Do not use mupirocin ointment if:
|
||
|
What should I tell my healthcare provider before using mupirocin ointment?
Before using mupirocin ointment, tell your healthcare provider about all of your medical conditions including if you:
|
||
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Do not mix mupirocin ointment with other lotions, creams, or ointments. | ||
How should I use mupirocin ointment?
|
||
|
What are the possible side effects of mupirocin ointment?
Mupirocin ointment may cause serious side effects, including:
|
||
|
|
|
|
||
How should I store mupirocin ointment?
|
||
|
General information about the safe and effective use of mupirocin ointment.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use mupirocin ointment for a condition for which it was not prescribed. Do not give mupirocin ointment to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about mupirocin ointment that is written for health professionals. |
||
|
What are the ingredients in mupirocin ointment?
Active Ingredient: mupirocin Inactive Ingredients: polyethylene glycol 400 and polyethylene glycol 3350 Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1 Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 For more information, call 1-886-923-4914. |
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
15 References (15 REFERENCES)
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-sixth Informational Supplement. CLSI document M100-S26. Clinical and Laboratory Standards Institute, 950 West Valley Rd., Suite 2500, Wayne, PA 19087, USA, 2016.
- Patel J, Gorwitz RJ, et al. Mupirocin Resistance. Clinical Infectious Diseases. 2009; 49(6): 935-41.
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard – Tenth Edition. CLSI document M07-A10. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard – Twelfth Edition. CLSI document M02-A12. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Finlay JE, Miller LA, Poupard JA. Interpretive criteria for testing susceptibility of staphylococci to mupirocin. Antimicrob Agents Chemother. 1997; 41(5):1137-1139.
11 Description (11 DESCRIPTION)
Mupirocin Ointment USP, 2% contains the RNA synthetase inhibitor antibacterial, mupirocin. The chemical name is ( E)-(2 S,3 R,4 R,5 S)-5-[(2 S,3 S,4 S,5 S)-2,3-epoxy-5-hydroxy-4-methylhexyl] tetrahydro-3,4-dihydroxy-ß-methyl-2 H-pyran-2-crotonic acid, ester with 9-hydroxynonanoic acid. The molecular formula of mupirocin is C 26H 44O 9, and the molecular weight is 500.6. The structural formula of mupirocin is:
Each gram of mupirocin ointment, 2% contains 20 mg mupirocin in a water-miscible ointment base (polyethylene glycol ointment) consisting of polyethylene glycol 400 and polyethylene glycol 3350.
12.4 Microbiology
Mupirocin is an RNA synthetase inhibitor antibacterial produced by fermentation using the organism Pseudomonas fluorescens.
8.4 Pediatric Use
The safety and effectiveness of mupirocin ointment have been established in the age range of 2 months to 16 years. Use of mupirocin ointment in these age-groups is supported by evidence from adequate and well-controlled trials of mupirocin ointment in impetigo in pediatric subjects studied as a part of the pivotal clinical trials in adults [see Clinical Studies (14)] .
5.2 Eye Irritation
Avoid contact with the eyes. In case of accidental contact, rinse well with water.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of topical mupirocin ointment in impetigo was tested in 2 trials. In the first, subjects with impetigo were randomized to receive either mupirocin ointment or vehicle placebo 3 times daily for 8 to 12 days. Clinical efficacy rates at end of therapy in the evaluable populations (adults and pediatric subjects included) were 71% for mupirocin ointment (n = 49) and 35% for vehicle placebo (n = 51). Pathogen eradication rates in the evaluable populations were 94% for mupirocin ointment and 62% for vehicle placebo.
In the second trial, subjects with impetigo were randomized to receive either mupirocin ointment 3 times daily or 30 mg per kg to 40 mg per kg oral erythromycin ethylsuccinate per day (this was an unblinded trial) for 8 days. There was a follow-up visit 1 week after treatment ended. Clinical efficacy rates at the follow-up visit in the evaluable populations (adults and pediatric subjects included) were 93% for mupirocin ointment (n = 29) and 78.5% for erythromycin (n = 28). Pathogen eradication rates in the evaluable populations were 100% for both test groups.
4 Contraindications (4 CONTRAINDICATIONS)
Mupirocin ointment is contraindicated in patients with known hypersensitivity to mupirocin or any of the excipients of mupirocin ointment.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Severe Allergic Reactions [see Warnings and Precautions (5.1)]
- Eye Irritation [see Warnings and Precautions (5.2)]
- Local Irritation [see Warnings and Precautions (5.3)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
5.3 Local Irritation
In the event of a sensitization or severe local irritation from mupirocin ointment, usage should be discontinued, and appropriate alternative therapy for the infection instituted.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to susceptible isolates of Staphylococcus aureus (S. aureus) and Streptococcus pyogenes (S. pyogenes).
12.1 Mechanism of Action
Mupirocin is an RNA synthetase inhibitor antibacterial [see Microbiology (12.4)] .
5.4 Clostridium Difficile (5.4 Clostridium difficile)
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Severe Allergic Reactions: Anaphylaxis, urticaria, angioedema, and generalized rash have been reported in patients treated with formulations of mupirocin, including mupirocin ointment. ( 5.1)
- Eye Irritation: Avoid contact with eyes. ( 5.2)
- Local Irritation: Discontinue in the event of sensitization or severe local irritation. ( 5.3)
- Clostridium difficile-Associated Diarrhea (CDAD): If diarrhea occurs, evaluate patients for CDAD. ( 5.4)
- Potential for Microbial Overgrowth: Prolonged use may result in overgrowth of nonsusceptible microorganisms, including fungi. ( 5.5)
- Risk Associated with Mucosal Use: Mupirocin ointment is not formulated for use on mucosal surfaces. A separate formulation, mupirocin nasal ointment, is available for intranasal use. ( 5.6)
- Risk of Polyethylene Glycol Absorption: Mupirocin ointment should not be used where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment. ( 5.7)
- Risk Associated with Use at Intravenous Sites: Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance. ( 5.8)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For Topical Use Only.
- Apply a small amount of mupirocin ointment, with a cotton swab or gauze pad, to the affected area 3 times daily for up to 10 days.
- Cover the treated area with gauze dressing if desired.
- Re-evaluate patients not showing a clinical response within 3 to 5 days.
- Mupirocin ointment is not for intranasal, ophthalmic, or other mucosal use [see Warnings and Precautions (5.2, 5.6)] .
- Do not apply mupirocin ointment concurrently with any other lotions, creams, or ointments [see Clinical Pharmacology (12.3)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Each gram of mupirocin ointment contains 20 mg mupirocin in a water-miscible ointment base supplied in 15-gram, 22-gram and 30-gram tubes.
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following reactions have been identified during postmarketing use of mupirocin ointment. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal relationship to mupirocin ointment.
5.1 Severe Allergic Reactions
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash, have been reported in patients treated with formulations of mupirocin, including mupirocin ointment [see Adverse Reactions (6.2)] .
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following local adverse reactions were reported by at least 1% of subjects in connection with the use of mupirocin ointment in clinical trials: burning, stinging, or pain in 1.5% of subjects; itching in 1% of subjects. Rash, nausea, erythema, dry skin, tenderness, swelling, contact dermatitis, and increased exudate were reported in less than 1% of subjects.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise the patient to administer mupirocin ointment as follows:
- Use mupirocin ointment only as directed by the healthcare provider. It is for external use only. Avoid contact of mupirocin ointment with the eyes. If mupirocin ointment gets in the eyes, rinse thoroughly with water.
- Do not use mupirocin ointment in the nose.
- Wash your hands before and after applying mupirocin ointment.
- Use a gauze pad or cotton swab to apply a small amount of mupirocin ointment to the affected area. The treated area may be covered by gauze dressing if desired.
- Report to the healthcare provider any signs of local adverse reactions. Mupirocin ointment should be stopped and the healthcare provider contacted if irritation, severe itching, or rash occurs.
- Report to the healthcare provider or go to the nearest emergency room if severe allergic reactions, such as swelling of the lips, face, or tongue, or wheezing occur [see Warnings and Precautions (5.1)] .
- If impetigo has not improved in 3 to 5 days, contact the healthcare provider.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Each gram of mupirocin ointment contains 22 mg mupirocin in a water-miscible ointment base.
Mupirocin Ointment USP, 2% is supplied as follows:
NDC 68071-4321-2
5.6 Risk Associated With Mucosal Use (5.6 Risk Associated with Mucosal Use)
Mupirocin ointment is not formulated for use on mucosal surfaces. Intranasal use has been associated with isolated reports of stinging and drying. A separate formulation, mupirocin calcium nasal ointment, is available for intranasal use.
5.5 Potential for Microbial Overgrowth
As with other antibacterial products, prolonged use of mupirocin ointment may result in overgrowth of nonsusceptible microorganisms, including fungi [see Dosage and Administration (2)] .
5.7 Risk of Polyethylene Glycol Absorption
Polyethylene glycol can be absorbed from open wounds and damaged skin and is excreted by the kidneys. In common with other polyethylene glycol-based ointments, mupirocin ointment should not be used in conditions where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment.
5.8 Risk Associated With Use At Intravenous Sites (5.8 Risk Associated with Use at Intravenous Sites)
Mupirocin ointment should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential of mupirocin have not been conducted.
Results of the following studies performed with mupirocin calcium or mupirocin sodium in vitro and in vivo did not indicate a potential for genotoxicity: rat primary hepatocyte unscheduled DNA synthesis, sediment analysis for DNA strand breaks, Salmonella reversion test (Ames), Escherichia coli mutation assay, metaphase analysis of human lymphocytes, mouse lymphoma assay, and bone marrow micronuclei assay in mice.
In a fertility/reproductive performance study (with dosing through lactation), mupirocin administered subcutaneously to male and female rats at doses up to 100 mg per kg per day which is 14 times the human topical dose (approximately 60 mg mupirocin per day) based on calculations of dose divided by the entire body surface area, did not result in impaired fertility or impaired reproductive performance attributable to mupirocin.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:24.725860 · Updated: 2026-03-14T22:05:47.396240