These Highlights Do Not Include All The Information Needed To Use Gattex Safely And Effectively. See Full Prescribing Information For Gattex.
66b69c1e-b25c-44d3-b5ff-1c1de9a516fa
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
GATTEX ® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support .
Indications and Usage
GATTEX ® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support .
Dosage and Administration
Important Administration Information GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested. Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg. ( 2.1 ) Evaluation Testing within 6 Months Prior to Starting GATTEX Adult Patients : Perform a colonoscopy and upper GI endoscopy with removal of polyps. ( 2.1 , 5.1 ) Pediatric Patients : Perform fecal occult blood testing. If new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and upper GI endoscopy. ( 2.1 , 5.1 ) Adult and Pediatric Patients : Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase). ( 2.1 , 5.3 ) Dosage and Administration For subcutaneous use only. ( 2.2 ) The recommended dosage of GATTEX for both adults and pediatric patients is 0.05 mg/kg once daily by subcutaneous injection. ( 2.2 ) Alternate sites between 1 of the 4 quadrants of the abdomen, or into alternating thighs or alternating arms. ( 2.2 ) Dosage Adjustment for Renal Impairment For adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate less than 60 mL/min/1.73 m 2 ) the recommended dosage is 0.025 mg/kg once daily. ( 2.3 ) Discontinuation When treatment is discontinued, monitor for fluid and electrolyte imbalances. ( 2.5 , 5.4 ) Preparation See full prescribing information for instructions on reconstitution. ( 2.6 )
Warnings and Precautions
Acceleration of Neoplastic Growth : In case of intestinal malignancy, discontinue GATTEX. The decision to continue GATTEX in patients with non-gastrointestinal malignancy should be made based on benefit risk considerations. ( 5.1 ) In adult patients, colonoscopy and upper GI endoscopy (or alternate imaging) is recommended after 1 year of treatment. Perform subsequent colonoscopies and upper GI endoscopes (or alternate imaging) no less frequently than every 5 years. ( 5.1 ) In pediatric patients, perform fecal occult blood testing annually. Colonoscopy/sigmoidoscopy is recommended after 1 year of treatment and every 5 years thereafter on treatment. Consider upper GI endoscopy (or alternate imaging) during treatment with GATTEX. ( 5.1 ) Intestinal Obstruction : In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX pending further clinical evaluation and management. ( 5.2 ) Biliary and Pancreatic Disease : Obtain bilirubin, alkaline phosphatase, lipase, amylase every 6 months. If clinically meaningful changes are seen, further evaluation is recommended including imaging, and reassess continued GATTEX treatment. ( 5.3 ) Fluid Overload, Including Congestive Heart Failure : If fluid overload occurs, adjust parenteral support, and reassess continued GATTEX treatment. ( 5.4 ) Potential for Increased Absorption of Oral Medications : Monitor patients on concomitant oral medications (e.g., benzodiazepines) for adverse reactions related to the concomitant drug; dosage reduction of the other drug may be required. ( 5.5 , 7.1 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Acceleration of Neoplastic Growth [see Warnings and Precautions ( 5.1 )] Intestinal Obstruction [see Warnings and Precautions ( 5.2 )] Biliary and Pancreatic Disease [see Warnings and Precautions ( 5.3 )] Fluid Imbalance and Fluid Overload [see Warnings and Precautions ( 5.4 )]
Storage and Handling
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
How Supplied
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
Medication Information
Warnings and Precautions
Acceleration of Neoplastic Growth : In case of intestinal malignancy, discontinue GATTEX. The decision to continue GATTEX in patients with non-gastrointestinal malignancy should be made based on benefit risk considerations. ( 5.1 ) In adult patients, colonoscopy and upper GI endoscopy (or alternate imaging) is recommended after 1 year of treatment. Perform subsequent colonoscopies and upper GI endoscopes (or alternate imaging) no less frequently than every 5 years. ( 5.1 ) In pediatric patients, perform fecal occult blood testing annually. Colonoscopy/sigmoidoscopy is recommended after 1 year of treatment and every 5 years thereafter on treatment. Consider upper GI endoscopy (or alternate imaging) during treatment with GATTEX. ( 5.1 ) Intestinal Obstruction : In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX pending further clinical evaluation and management. ( 5.2 ) Biliary and Pancreatic Disease : Obtain bilirubin, alkaline phosphatase, lipase, amylase every 6 months. If clinically meaningful changes are seen, further evaluation is recommended including imaging, and reassess continued GATTEX treatment. ( 5.3 ) Fluid Overload, Including Congestive Heart Failure : If fluid overload occurs, adjust parenteral support, and reassess continued GATTEX treatment. ( 5.4 ) Potential for Increased Absorption of Oral Medications : Monitor patients on concomitant oral medications (e.g., benzodiazepines) for adverse reactions related to the concomitant drug; dosage reduction of the other drug may be required. ( 5.5 , 7.1 )
Indications and Usage
GATTEX ® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support .
Dosage and Administration
Important Administration Information GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested. Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg. ( 2.1 ) Evaluation Testing within 6 Months Prior to Starting GATTEX Adult Patients : Perform a colonoscopy and upper GI endoscopy with removal of polyps. ( 2.1 , 5.1 ) Pediatric Patients : Perform fecal occult blood testing. If new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and upper GI endoscopy. ( 2.1 , 5.1 ) Adult and Pediatric Patients : Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase). ( 2.1 , 5.3 ) Dosage and Administration For subcutaneous use only. ( 2.2 ) The recommended dosage of GATTEX for both adults and pediatric patients is 0.05 mg/kg once daily by subcutaneous injection. ( 2.2 ) Alternate sites between 1 of the 4 quadrants of the abdomen, or into alternating thighs or alternating arms. ( 2.2 ) Dosage Adjustment for Renal Impairment For adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate less than 60 mL/min/1.73 m 2 ) the recommended dosage is 0.025 mg/kg once daily. ( 2.3 ) Discontinuation When treatment is discontinued, monitor for fluid and electrolyte imbalances. ( 2.5 , 5.4 ) Preparation See full prescribing information for instructions on reconstitution. ( 2.6 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Acceleration of Neoplastic Growth [see Warnings and Precautions ( 5.1 )] Intestinal Obstruction [see Warnings and Precautions ( 5.2 )] Biliary and Pancreatic Disease [see Warnings and Precautions ( 5.3 )] Fluid Imbalance and Fluid Overload [see Warnings and Precautions ( 5.4 )]
Storage and Handling
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
How Supplied
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
Description
GATTEX ® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support .
Section 42229-5
Colonoscopy and Upper GI Endoscopy in Adults
- A follow-up colonoscopy and upper GI endoscopy (or alternate imaging) is recommended at the end of 1 year of GATTEX. If no polyp is found, subsequent colonoscopies and upper GI endoscopies (or alternate imaging) should be done no less frequently than every 5 years. If a polyp is found, adherence to current polyp follow-up guidelines is recommended [see Warnings and Precautions (5.1)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 09/2025 | ||
|
MEDICATION GUIDE GATTEX® (Ga'-tex) (teduglutide) for injection, for subcutaneous use |
|||
| Read this Medication Guide carefully before you start taking GATTEX and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
|
What is the most important information I should know about GATTEX? GATTEX may cause serious side effects, including:
For Adult Patients
For Pediatric Patients
For Adult Patients
|
|||
|
|
||
If a blockage is found, your healthcare provider may temporarily stop GATTEX.
|
|||
|
|
||
| These are not all the side effects of GATTEX. For more information, see "What are the possible side effects of GATTEX?" | |||
What is GATTEX?
|
|||
|
What should I tell my healthcare provider before using GATTEX? Before using GATTEX, tell your healthcare provider about all your medical conditions, including if you or your child:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I use GATTEX?
|
|||
|
What are the possible side effects of GATTEX? GATTEX may cause serious side effects, including:
|
|||
|
|
||
| The side effects of GATTEX in pediatric patients are similar to those seen in adults. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of GATTEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store GATTEX?
|
|||
|
General information about the safe and effective use of GATTEX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GATTEX for a condition for which it was not prescribed. Do not give GATTEX to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about GATTEX talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about GATTEX that is written for health professionals. |
|||
|
What are the ingredients in GATTEX? Active ingredient: teduglutide
Inactive ingredients: dibasic sodium phosphate heptahydrate, L-histidine, mannitol, and monobasic sodium phosphate monohydrate. Sterile Water for Injection is provided as a diluent. ©2025 Takeda Pharmaceutical Company Limited. All rights reserved. Patented: see www.takeda.com/en-us/patents. |
Section 44425-7
Storage and Handling of One-Vial Kit
Prior to Dispensing:
- Store GATTEX 5 mg Strength one-vial kits refrigerated at 2°C to 8°C (36°F to 46°F). Do not freeze. Do not use beyond the expiration date on the label.
After Dispensing by the Pharmacist:
- Store GATTEX 5 mg Strength one-vial kits at room temperature up to 25°C (77°F). Do not freeze. Dispense with a 90-day "use by" dating.
GATTEX is also supplied in 30-vial cartons to be assembled by the dispensing pharmacist into a 30-vial kit by transferring the trays containing 30 vials from a Carton of Drug Vials into a Carton of Ancillary Supplies:
GATTEX 5 mg Carton of Drug Vials (NDC 68875-0101-2):
- Thirty single-dose vials of GATTEX 5 mg (NDC 68875-0101-1)
-
Carton of Ancillary Supplies:
- Thirty disposable prefilled syringes containing diluent (0.5 mL Sterile Water for Injection USP) for reconstitution
- Thirty separate needles (23G × 1½ in) to attach to the syringes for reconstitution
- Thirty sterile disposable 1 mL syringes with needle (27G × 1/2 in)
- Sixty alcohol swabs
The final assembled 30-Vial Kit should contain the items:
GATTEX 5 mg Strength 30-Vial Kit (NDC 68875-0102-1):
- Thirty single-dose vials of 5 mg teduglutide (NDC 68875-0101-1)
- Thirty disposable prefilled syringes containing 0.5 mL Sterile Water for Injection USP for reconstitution, with 30 separate needles (23G × 1½ in) to attach to the syringes
- Thirty sterile disposable 1 mL syringes with needle (27G × 1/2 in) for dosing
- Sixty alcohol swabs
10 Overdosage
The maximum dose of GATTEX studied during clinical development was 80 mg/day for 8 days. No unexpected systemic adverse reactions were seen. In the event of overdose, the patient should be carefully monitored by the medical professional.
11 Description
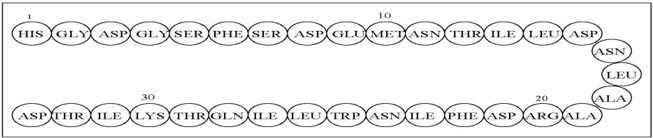
The active ingredient in GATTEX (teduglutide) for injection is teduglutide, which is a 33 amino acid glucagon-like peptide-2 (GLP-2) analog manufactured using a strain of Escherichia coli modified by recombinant DNA technology. The chemical composition of teduglutide is L-histidyl-L-glycyl-L-aspartyl-L-glycyl-L-seryl-L-phenylalanyl-L-seryl-L-aspartyl-L-glutamyl-L-methionyl-L-asparaginyl-L-threonyl-L-isoleucyl-L-leucyl-L-aspartyl-L-asparaginyl-L-leucyl-L-alanyl-L-alanyl-L-arginyl-L-aspartyl-L-phenylalanyl-L-isoleucyl-L-asparaginyl-L-tryptophanyl-L-leucyl-L-isoleucyl-L-glutaminyl-L-threonyl-L-lysyl-L-isoleucyl-L-threonyl-L-aspartic acid. The structural formula is:
Teduglutide has a molecular weight of 3752 Daltons. Teduglutide drug substance is a clear, colorless to light-straw–colored liquid.
Each single-dose vial of GATTEX contains 5 mg of teduglutide as a white lyophilized powder for reconstitution and administration by subcutaneous injection. In addition to the active pharmaceutical ingredient (teduglutide), each vial of GATTEX contains 3.434 mg dibasic sodium phosphate heptahydrate, 3.88 mg L-histidine, 15 mg mannitol, and 0.644 mg monobasic sodium phosphate monohydrate as excipients. No preservatives are present.
At the time of administration, the lyophilized powder is reconstituted with 0.5 mL of Sterile Water for Injection, which is provided in a single-dose prefilled syringe. A 10 mg/mL sterile solution is obtained after reconstitution. Up to 0.38 mL of the reconstituted solution which contains 3.8 mg of teduglutide can be withdrawn for subcutaneous injection upon reconstitution.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients less than 1 year of age have not been established.
The safety and effectiveness of GATTEX have been established in pediatric patients 1 year to less than 17 years of age who are dependent on parenteral support for the treatment of SBS. Use of GATTEX in this population is supported by evidence from adequate and well-controlled studies in adults, with additional efficacy, safety, pharmacokinetic and pharmacodynamic data in pediatric patients 1 year to less than 17 years of age [see Dosage and Administration (2), Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.2)]. These data were derived from two studies of 24-week (Study 5) and 12-week (NCT01952080) duration in which 41 pediatric patients were treated with GATTEX in the following groups: 1 infant (1 year to less than 2 years), 37 children (2 years to less than 12 years) and 3 adolescents (12 years to less than 17 years).
In these 2 studies and the corresponding extension studies (Study 6 and NCT02949362), 29 pediatric patients were administered GATTEX prospectively for up to 94 weeks [see Clinical Studies (14.2)]. Adverse reactions in pediatric patients were similar to those seen in adults [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the 134 patients with SBS that were treated with GATTEX at the recommended dosage of 0.05 mg/kg/day in the clinical studies, 19 patients were 65 years or older while 5 patients were 75 years of age or older. No overall differences in safety or efficacy were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of GATTEX or of other teduglutide products.
Adults
Based on integrated data from two studies in adults with SBS (a 6-month randomized placebo-controlled study, followed by a 24-month open-label study), the development of anti-teduglutide antibodies in patients who received subcutaneous administration of 0.05 mg/kg GATTEX once daily was 3% (2/60) at Month 3, 17% (13/77) at Month 6, 24% (16/67) at Month 12, 33% (11/33) at Month 24, and 48% (14/29) at Month 30. Anti-teduglutide antibodies were cross-reactive to native glucagon-like peptide (GLP-2) in 5 of the 6 patients (83%) who had anti-teduglutide antibodies and were tested for cross-reactivity. In the same two studies, a total of 36 patients were tested for neutralizing antibodies: one patient developed borderline positive neutralizing antibody responses at month 24 of the extension study. The antibody formation has not been associated with clinically relevant safety findings, reduced efficacy or changed pharmacokinetics of GATTEX.
Pediatric Patients 1 Year to Less than 17 Years of Age
In pediatric patients who received subcutaneous administration of 0.05 mg/kg GATTEX once daily for 24 weeks, the rate of anti-teduglutide antibody formation at Month 6 was 19% (5/26) and was similar to the rate of antibody formation in adult patients (17%). Of the 5 pediatric patients who had developed antibodies to teduglutide at Month 6, 2 patients had neutralizing antibodies. However, with the longer duration of treatment, the rate of anti-teduglutide formation at Month 12 was higher in pediatric patients with 54% (14/26), compared to that of adults (24%). Of the 14 pediatric patients who had developed antibodies to teduglutide at Month 12, 1 patient had neutralizing antibodies.
Among the small number of pediatric patients who developed anti-teduglutide antibodies, no association with adverse events or lack of efficacy was observed.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
8.6 Renal Impairment
In adult subjects with moderate to severe renal impairment or end-stage renal disease (ESRD) (creatinine clearance <60 mL/min), the exposure to teduglutide increased with the degree of renal impairment [see Clinical Pharmacology (12.3)]. Reduce the dosage of GATTEX by half in both pediatric and adult patients with eGFR less than 60 mL/min/1.73 m2 [see Dosage and Administration (2.3)].
Instructions for Use
This Instructions for Use contains information on how to inject GATTEX.
Read this Instructions for Use before you start using GATTEX and each time you get a refill. There may be new information. Your healthcare provider or nurse should show you how to prepare, measure your dose, and give your injection of GATTEX the right way.
If you cannot give yourself the injection:
- ask your healthcare provider or nurse to help you, or
- ask someone who has been trained by a healthcare provider or nurse to give your injections.
Self-administration is not recommended in pediatric patients. In pediatric patients, GATTEX should be injected by:
- a healthcare provider or nurse, or
- a parent or adult caregiver who has been trained by a healthcare provider or nurse to give injections of GATTEX to pediatric patients.
Important information:
- Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 22 pounds (10 kg).
- Before you start, check the "Use By" date on your GATTEX kit. Make sure that the "Use By" date has not passed. Do not use anything in the GATTEX kit after the "Use By" date on the kit.
- Give GATTEX within 3 hours after you mix the powder with the Diluent (Sterile Water for Injection).
- Use the syringes and needles provided in the GATTEX kit.
- Do not use a GATTEX vial more than 1 time, even if there is medicine left in the vial.
- Throw away (dispose of) any unused GATTEX after you give your injection.
- Safely throw away GATTEX vials after use.
- Do not re-use syringes or needles. See "Step 7: Dispose of syringes and needles" for information about how to safely throw away needles and syringes.
- To help avoid needle-stick injuries, do not recap needles.
Gather the supplies you will need to prepare GATTEX and to give your injection (See Figure A).
|
Figure A |
From your GATTEX kit you will need:
|
Step 1: Prepare the injection.
- Choose a well-lit, clean, flat work surface.
- Wash your hands with soap and water.
Step 2: Preparing the Diluent syringe.
- Put the Diluent syringe (See Figure B1) and the 23G, 1½ inch needle in front of you on your work surface.
| Figure B1 |
|
- Hold the Diluent syringe by the barrel. Snap off the white cap (bend the cap sideways until the cap comes off). Only the top portion of the white cap should be snapped off. The lower portion of the cap will remain in place (See Figure B2). Throw the cap away.
| Figure B2 |
|
- Remove the 23G, 1½ inch needle from the package. Use the fold in the package to peel back the plastic cover (See Figure C). Leave the plastic cap on the needle.
| Figure C |
|
- Push the open end of the needle onto the end of the Diluent syringe (See Figure D).
Twist the needle clockwise (to the right) until it stops turning.
| Figure D |
|
- When the needle is tightly in place, put the Diluent syringe and needle on your work surface.
Step 3: Mix GATTEX powder with Diluent.
- Remove the green cap from the GATTEX vial. Throw away the green cap.
- Find the gray rubber seal on top of the GATTEX vial (See Figure E).
| Figure E |
|
- Use an alcohol swab pad to clean the gray rubber seal (See Figure F).
- Do not touch the gray rubber seal after you clean it.
| Figure F |
|
- Pick up the Diluent syringe with the needle attached.
- Remove the plastic cap that covers the needle (See Figure G). Throw the cap away.
| Figure G |
|
- Hold the GATTEX vial between your thumb and index (pointer) finger (See Figure H). Be careful not to touch the gray rubber seal.
- Push the needle down through the center of the gray rubber seal.
- Slowly push down on the plunger of the Diluent syringe. Empty all the Diluent into the GATTEX vial.
- Leave the needle and Diluent syringe in place.
| Figure H |
|
- Gently tap the barrel of the Diluent syringe with a finger (See Figure I).
- Make sure all the Diluent has gone into the GATTEX vial.
| Figure I |
|
- Remove the Diluent syringe and needle from the GATTEX vial. Let the vial sit for about 30 seconds.
- Do not put the needle cap back on the needle.
- Throw away (dispose of) the Diluent syringe and needle in your sharps disposal container.
- After 30 seconds, place the GATTEX vial between the palms of your hands. Gently roll the vial for about 15 seconds (See Figure J).
- Do not shake the GATTEX vial.
- Do not touch the gray rubber seal. If you do, clean it again with a new alcohol pad.
- Let the GATTEX vial stand on your work surface for about 2 minutes.
| Figure J |
|
Step 4: Check the mixed GATTEX.
- After 2 minutes, look at the vial of GATTEX. The liquid in the vial should be clear and colorless to pale yellow, and should not have any particles in it.
- If there is any powder in the GATTEX vial that did not dissolve, gently roll the vial between your hands for 15 seconds more.
- Do not shake the GATTEX vial.
- Check the GATTEX vial again for anything that did not dissolve.
- Do not use the GATTEX vial if there is anything in it that did not dissolve. Start from the beginning of this Instructions for Use to prepare a new vial. Use a new GATTEX vial, new Diluent syringe, and a new needle.
Step 5: Draw up your dose of GATTEX.
- Remove the plastic dosing syringe from the package. Use the fold in the package to peel back the plastic cover (See Figure K).
| Figure K |
|
- Remove the needle cap from the plastic dosing syringe (See Figure L ).
- Throw the needle cap away. Do not touch the needle or allow it to touch anything.
| Figure L |
|
- Carefully pull back on the plunger to the line that matches the dose prescribed by your healthcare provider.
- Use 1 hand to hold the GATTEX vial steady. Use your other hand to insert the needle straight down into the middle of the gray rubber seal on the GATTEX vial (See Figure M). You may feel some resistance as the needle passes through the rubber seal.
- Gently push down the plunger until all of the air has gone from the plastic dosing syringe into the GATTEX vial.
- Turn the GATTEX vial and plastic dosing syringe upside down (See Figure N).
| Figure M |
|
| Figure N |
|
- Hold the GATTEX vial with 1 hand.
- Slowly pull back the plunger of the plastic dosing syringe with your other hand.
- Fill the plastic dosing syringe until the black tip of the plunger lines up with the mark that matches your prescribed dose (See Figure O).
- Keep the plastic dosing syringe and needle in the GATTEX vial.
| Figure O |
|
- You may see some bubbles inside the GATTEX vial when the plastic dosing syringe is filled. This is normal. With the needle still in the vial, gently tap the side of the plastic dosing syringe with a finger to make any air bubbles rise to the top (See Figure P).
| Figure P |
|
- Slowly push the plunger up until all air bubbles are out of the plastic dosing syringe. Make sure the tip of the needle is in the fluid. Slowly pull back the plunger to draw up the right dose of GATTEX into the plastic dosing syringe.
- Remove the plastic dosing syringe and needle from the GATTEX vial (See Figure Q). Do not touch the needle or allow it to touch anything.
| Figure Q |
|
Step 6: Inject GATTEX.
- Choose an injection site on the stomach area (abdomen), thighs, or upper arms.
- Choose a different site to give the injection each day. Do not inject into areas where the skin is tender, bruised, red, or hard. (See Figure R and Figure S)
| Figure R |
|
|
Figure S |
- Clean the skin where you plan to give the injection with a new alcohol swab pad. Do not touch this area again before giving the injection.
- Use 1 hand to gently pinch up a fold of skin around the injection site (See Figure T).
| Figure T |
|
- Use your other hand to hold the plastic dosing syringe. Insert the full length of the needle into the skin at a 45-degree angle with a quick, "dart-like" motion (See Figure U).
| Figure U |
|
- Let go of the skin. Hold the syringe barrel with 1 hand while you slowly push down the plunger until the plastic dosing syringe is empty (See Figure V).
| Figure V |
|
- When the plastic dosing syringe is empty, quickly pull the needle out of your skin. There may be a little bleeding at the injection site. Apply an adhesive bandage to the injection site if needed.
Step 7: Dispose of syringes and needles.
- Do not re-use a syringe or needle.
- To help avoid needle-stick injuries, do not recap a needle.
- Put your needles and syringes in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharp items being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container.
There may be local or state laws about how to throw away syringes and needles. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. - Do not dispose of your sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your sharps disposal container.
- Throw away the GATTEX vial into the container where you put the syringes and needles.
- If you have any questions, talk to your healthcare provider or pharmacist.
How should I store GATTEX?
- Store GATTEX powder at room temperature up to 77°F (25°C).
- Do not freeze GATTEX.
- Use the GATTEX powder by the expiration date on the "Use By" sticker on the kit.
- Use GATTEX within 3 hours after mixing it.
- Throw away any unused GATTEX that has been mixed, even if there is medicine left in the vial.
- Do not store any GATTEX you have mixed.
Keep GATTEX and all medicines out of the reach of children.
Distributed by:
Takeda Pharmaceuticals America, Inc.
Cambridge, MA 02142
USA
1-877-825-3327
GATTEX and
are registered trademarks of Takeda Pharmaceuticals U.S.A., Inc.©2025 Takeda Pharmaceutical Company Limited. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 09/2025
8.7 Hepatic Impairment
GATTEX has not been studied in patients with severe hepatic impairment (Child-Pugh grade C). No dosage adjustment is recommended for patients with mild and moderate hepatic impairment (Child-Pugh grade A and B) [see Clinical Pharmacology (12.3)].
1 Indications and Usage
GATTEX® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support.
12.1 Mechanism of Action
Teduglutide is an analog of naturally occurring human glucagon-like peptide-2 (GLP-2), a peptide secreted by L-cells of the distal intestine. GLP-2 is known to increase intestinal and portal blood flow and inhibit gastric acid secretion. Teduglutide binds to the glucagon-like peptide-2 receptors located in intestinal subpopulations of enteroendocrine cells, subepithelial myofibroblasts and enteric neurons of the submucosal and myenteric plexus. Activation of these receptors results in the local release of multiple mediators including insulin-like growth factor (IGF)-1, nitric oxide and keratinocyte growth factor (KGF).
5 Warnings and Precautions
-
Acceleration of Neoplastic Growth: In case of intestinal malignancy, discontinue GATTEX. The decision to continue GATTEX in patients with non-gastrointestinal malignancy should be made based on benefit risk considerations. (5.1)
- In adult patients, colonoscopy and upper GI endoscopy (or alternate imaging) is recommended after 1 year of treatment. Perform subsequent colonoscopies and upper GI endoscopes (or alternate imaging) no less frequently than every 5 years. (5.1)
- In pediatric patients, perform fecal occult blood testing annually. Colonoscopy/sigmoidoscopy is recommended after 1 year of treatment and every 5 years thereafter on treatment. Consider upper GI endoscopy (or alternate imaging) during treatment with GATTEX. (5.1)
- Intestinal Obstruction: In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX pending further clinical evaluation and management. (5.2)
- Biliary and Pancreatic Disease: Obtain bilirubin, alkaline phosphatase, lipase, amylase every 6 months. If clinically meaningful changes are seen, further evaluation is recommended including imaging, and reassess continued GATTEX treatment. (5.3)
- Fluid Overload, Including Congestive Heart Failure: If fluid overload occurs, adjust parenteral support, and reassess continued GATTEX treatment. (5.4)
- Potential for Increased Absorption of Oral Medications: Monitor patients on concomitant oral medications (e.g., benzodiazepines) for adverse reactions related to the concomitant drug; dosage reduction of the other drug may be required. (5.5, 7.1)
5.2 Intestinal Obstruction
Intestinal obstruction has been reported in clinical studies [see Adverse Reactions (6.1)] and postmarketing. In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX while the patient is clinically managed. GATTEX may be restarted when the obstructive presentation resolves, if clinically indicated.
2 Dosage and Administration
Important Administration Information
GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested. Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg. (2.1)
Evaluation Testing within 6 Months Prior to Starting GATTEX
- Adult Patients: Perform a colonoscopy and upper GI endoscopy with removal of polyps. (2.1, 5.1)
- Pediatric Patients: Perform fecal occult blood testing. If new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and upper GI endoscopy. (2.1, 5.1)
- Adult and Pediatric Patients: Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase). (2.1, 5.3)
Dosage and Administration
- For subcutaneous use only. (2.2)
- The recommended dosage of GATTEX for both adults and pediatric patients is 0.05 mg/kg once daily by subcutaneous injection. (2.2)
- Alternate sites between 1 of the 4 quadrants of the abdomen, or into alternating thighs or alternating arms. (2.2)
Dosage Adjustment for Renal Impairment
- For adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate less than 60 mL/min/1.73 m2) the recommended dosage is 0.025 mg/kg once daily. (2.3)
Discontinuation
Preparation
- See full prescribing information for instructions on reconstitution. (2.6)
2.6 Preparation Instructions
- Reconstitute each vial of GATTEX by slowly injecting the 0.5 mL of preservative-free Sterile Water for Injection provided in the prefilled syringe. A 10 mg/mL sterile solution is obtained after reconstitution.
- Allow the vial containing GATTEX and water to stand for approximately 30 seconds and then gently roll the vial between the palms for about 15 seconds. Do not shake the vial.
- Allow the mixed contents to stand for about 2 minutes. Inspect the vial for any undissolved powder. If undissolved powder is observed, gently roll the vial again until all material is dissolved. Do not shake the vial.
- Reconstituted GATTEX is a sterile, clear, colorless to light straw-colored solution, which should be free from particulates. If there is any discoloration or particulates, discard the solution.
- A maximum of 0.38 mL of the reconstituted solution, containing 3.8 mg of teduglutide, can be withdrawn from the vial for dosing.
- If the product remains undissolved after the second attempt, do not use.
3 Dosage Forms and Strengths
For Injection: 5 mg teduglutide as a white lyophilized powder for reconstitution in a single-dose vial supplied with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of teduglutide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Neoplasia: colorectal polyps, gastric polyps, small intestinal polyps (duodenum, ileum, and jejunum)
Injection Site Reactions: injection site induration, inflammation, pruritus, urticaria, and warmth
8 Use in Specific Populations
Lactation: Breastfeeding not recommended. (8.2)
2.5 Discontinuation of Treatment
Discontinuation of treatment with GATTEX may result in fluid and electrolyte imbalance. Monitor fluid and electrolyte status in patients who discontinue GATTEX treatment [see Warnings and Precautions (5.4)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
16 How Supplied/storage and Handling
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
5.1 Acceleration of Neoplastic Growth
Based on the pharmacologic activity and tumor findings in the rat and mouse carcinogenicity studies, GATTEX has the potential to cause hyperplastic changes including neoplasia [see Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. In patients at increased risk for malignancy, the clinical decision to use GATTEX should be considered only if the benefits outweigh the risks. In patients who develop active gastrointestinal malignancy (GI tract, hepatobiliary, pancreatic) while on GATTEX, discontinue GATTEX treatment. In patients who develop active non-gastrointestinal malignancy while on GATTEX, the clinical decision to continue GATTEX should be made based on benefit-risk considerations.
2.1 Important Administration Information
GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested.
Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg.
Evaluation and testing prior to starting treatment with GATTEX:
Within 6 months prior to treatment:
Adult patients
- Perform a colonoscopy and an upper gastrointestinal (GI) endoscopy with removal of polyps [see Warnings and Precautions (5.1)].
- Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase) [see Warnings and Precautions (5.3)].
Pediatric patients
- Perform fecal occult blood testing; if there is new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and an upper GI endoscopy [see Warnings and Precautions (5.1)].
- Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase) [see Warnings and Precautions (5.3)].
2.3 Dosage Adjustment for Renal Impairment
The recommended dosage in adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2) is 0.025 mg/kg once daily [see Use in Specific Populations (8.6)].
Principal Display Panel 5 Mg Vial Carton
NDC 68875-0101-2
Gattex®
(teduglutide) for injection
5 mg
5 mg per vial.
For subcutaneous use only.
EACH CARTON CONTAINS:
- Thirty single-dose vials of Gattex®
- Package Insert
- Medication Guide
ATTENTION PHARMACIST:
- For dispensing, transfer the product with vial trays
from this carton into the 30-count patient kit.
Takeda
Principal Display Panel Kit Carton 68875 0102
Rx only
Pharmacy:
Apply Use By dating
sticker here
NDC 68875-0102-1
Gattex®
(teduglutide) for injection
5 mg
5 mg per vial.
For subcutaneous use only.
Takeda
Principal Display Panel Kit Carton 68875 0103
NDC 68875-0103-1
Rx only
Gattex®
(teduglutide) for injection
5 mg
Pharmacy:
Apply Use By dating
sticker here
5 mg per vial.
For subcutaneous use only.
Attention Patients: Store at room temperature up to 25°C (77°F). Do not freeze.
Takeda
5.5 Increased Absorption of Concomitant Oral Medication
In the adult placebo-controlled studies, an analysis of episodes of cognition and attention disturbances was performed for patients on benzodiazepines. One patient receiving prazepam concomitantly with GATTEX 0.05 mg/kg once daily experienced a dramatic deterioration in mental status progressing to coma during the first week of GATTEX therapy. The patient was admitted to the ICU and the prazepam blood concentration was >300 mcg/L. GATTEX and prazepam were discontinued, and coma resolved 5 days later.
Monitor patients receiving concomitant oral drugs requiring titration or with a narrow therapeutic index, for adverse reactions due to potential increased absorption of the concomitant drug. The concomitant drug may require a reduction in dosage [see Drug Interactions (7.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic potential of GATTEX was assessed in 2-year subcutaneous carcinogenicity studies in rats and mice. In a 2-year carcinogenicity study in Wistar Han rats at subcutaneous doses of 3, 10, and 35 mg/kg/day (about 15, 41, and 199 times the exposures [AUC] achieved at the recommended daily human dose of 0.05 mg/kg, respectively), teduglutide caused statistically significant increases in the incidences of adenomas in the bile duct and jejunum of male rats. In a 2-year carcinogenicity study in Crl:CD1(ICR) mice at subcutaneous doses of 1, 3.5, and 12.5 mg/kg/day (about 32, 66, and 244 times the exposures [AUC] achieved at the recommended daily human dose of 0.05 mg/kg, respectively), teduglutide caused a significant increase in papillary adenomas in the gall bladder; it also caused adenocarcinomas in the jejunum in male mice at the high dose of 12.5 mg/kg/day.
Teduglutide was negative in the Ames test, chromosomal aberration test in Chinese hamster ovary cells, and in vivo mouse micronucleus assay.
Teduglutide at subcutaneous doses up to 25 mg/kg twice daily (50 mg/kg/day or at least 202 times the clinical exposure (AUC) at the recommended daily human dose of 0.05 mg/kg) was found to have no adverse effect on fertility and reproductive performance of male and female rats.
7.1 Potential for Increased Absorption of Oral Medications
Based upon the pharmacodynamic effect of GATTEX, there is a potential for increased absorption of concomitant oral medications. Altered mental status has been observed in patients taking GATTEX and benzodiazepines in the adult clinical studies [see Warnings and Precautions (5.5)].
Monitor patients on concomitant oral drugs requiring titration or with a narrow therapeutic index for adverse reactions related to the concomitant drug while on GATTEX. The concomitant drug may require a reduction in dosage.
2.2 Recommended Dosage and Administration for Adults and Pediatric Patients 1 Year of Age and Older
GATTEX is for subcutaneous injection only. Not for intravenous or intramuscular administration.
The recommended dosage of GATTEX is 0.05 mg/kg once daily administered by subcutaneous injection.
If a dose is missed, that dose should be taken as soon as possible on that day. Do not take 2 doses on the same day.
Alternation of sites for subcutaneous injection is recommended, and can include the thighs, upper arms, and the abdomen.
Structured Label Content
Section 42229-5 (42229-5)
Colonoscopy and Upper GI Endoscopy in Adults
- A follow-up colonoscopy and upper GI endoscopy (or alternate imaging) is recommended at the end of 1 year of GATTEX. If no polyp is found, subsequent colonoscopies and upper GI endoscopies (or alternate imaging) should be done no less frequently than every 5 years. If a polyp is found, adherence to current polyp follow-up guidelines is recommended [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 09/2025 | ||
|
MEDICATION GUIDE GATTEX® (Ga'-tex) (teduglutide) for injection, for subcutaneous use |
|||
| Read this Medication Guide carefully before you start taking GATTEX and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
|
What is the most important information I should know about GATTEX? GATTEX may cause serious side effects, including:
For Adult Patients
For Pediatric Patients
For Adult Patients
|
|||
|
|
||
If a blockage is found, your healthcare provider may temporarily stop GATTEX.
|
|||
|
|
||
| These are not all the side effects of GATTEX. For more information, see "What are the possible side effects of GATTEX?" | |||
What is GATTEX?
|
|||
|
What should I tell my healthcare provider before using GATTEX? Before using GATTEX, tell your healthcare provider about all your medical conditions, including if you or your child:
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I use GATTEX?
|
|||
|
What are the possible side effects of GATTEX? GATTEX may cause serious side effects, including:
|
|||
|
|
||
| The side effects of GATTEX in pediatric patients are similar to those seen in adults. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of GATTEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store GATTEX?
|
|||
|
General information about the safe and effective use of GATTEX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GATTEX for a condition for which it was not prescribed. Do not give GATTEX to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information about GATTEX talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about GATTEX that is written for health professionals. |
|||
|
What are the ingredients in GATTEX? Active ingredient: teduglutide
Inactive ingredients: dibasic sodium phosphate heptahydrate, L-histidine, mannitol, and monobasic sodium phosphate monohydrate. Sterile Water for Injection is provided as a diluent. ©2025 Takeda Pharmaceutical Company Limited. All rights reserved. Patented: see www.takeda.com/en-us/patents. |
Section 44425-7 (44425-7)
Storage and Handling of One-Vial Kit
Prior to Dispensing:
- Store GATTEX 5 mg Strength one-vial kits refrigerated at 2°C to 8°C (36°F to 46°F). Do not freeze. Do not use beyond the expiration date on the label.
After Dispensing by the Pharmacist:
- Store GATTEX 5 mg Strength one-vial kits at room temperature up to 25°C (77°F). Do not freeze. Dispense with a 90-day "use by" dating.
GATTEX is also supplied in 30-vial cartons to be assembled by the dispensing pharmacist into a 30-vial kit by transferring the trays containing 30 vials from a Carton of Drug Vials into a Carton of Ancillary Supplies:
GATTEX 5 mg Carton of Drug Vials (NDC 68875-0101-2):
- Thirty single-dose vials of GATTEX 5 mg (NDC 68875-0101-1)
-
Carton of Ancillary Supplies:
- Thirty disposable prefilled syringes containing diluent (0.5 mL Sterile Water for Injection USP) for reconstitution
- Thirty separate needles (23G × 1½ in) to attach to the syringes for reconstitution
- Thirty sterile disposable 1 mL syringes with needle (27G × 1/2 in)
- Sixty alcohol swabs
The final assembled 30-Vial Kit should contain the items:
GATTEX 5 mg Strength 30-Vial Kit (NDC 68875-0102-1):
- Thirty single-dose vials of 5 mg teduglutide (NDC 68875-0101-1)
- Thirty disposable prefilled syringes containing 0.5 mL Sterile Water for Injection USP for reconstitution, with 30 separate needles (23G × 1½ in) to attach to the syringes
- Thirty sterile disposable 1 mL syringes with needle (27G × 1/2 in) for dosing
- Sixty alcohol swabs
10 Overdosage (10 OVERDOSAGE)
The maximum dose of GATTEX studied during clinical development was 80 mg/day for 8 days. No unexpected systemic adverse reactions were seen. In the event of overdose, the patient should be carefully monitored by the medical professional.
11 Description (11 DESCRIPTION)
The active ingredient in GATTEX (teduglutide) for injection is teduglutide, which is a 33 amino acid glucagon-like peptide-2 (GLP-2) analog manufactured using a strain of Escherichia coli modified by recombinant DNA technology. The chemical composition of teduglutide is L-histidyl-L-glycyl-L-aspartyl-L-glycyl-L-seryl-L-phenylalanyl-L-seryl-L-aspartyl-L-glutamyl-L-methionyl-L-asparaginyl-L-threonyl-L-isoleucyl-L-leucyl-L-aspartyl-L-asparaginyl-L-leucyl-L-alanyl-L-alanyl-L-arginyl-L-aspartyl-L-phenylalanyl-L-isoleucyl-L-asparaginyl-L-tryptophanyl-L-leucyl-L-isoleucyl-L-glutaminyl-L-threonyl-L-lysyl-L-isoleucyl-L-threonyl-L-aspartic acid. The structural formula is:
Teduglutide has a molecular weight of 3752 Daltons. Teduglutide drug substance is a clear, colorless to light-straw–colored liquid.
Each single-dose vial of GATTEX contains 5 mg of teduglutide as a white lyophilized powder for reconstitution and administration by subcutaneous injection. In addition to the active pharmaceutical ingredient (teduglutide), each vial of GATTEX contains 3.434 mg dibasic sodium phosphate heptahydrate, 3.88 mg L-histidine, 15 mg mannitol, and 0.644 mg monobasic sodium phosphate monohydrate as excipients. No preservatives are present.
At the time of administration, the lyophilized powder is reconstituted with 0.5 mL of Sterile Water for Injection, which is provided in a single-dose prefilled syringe. A 10 mg/mL sterile solution is obtained after reconstitution. Up to 0.38 mL of the reconstituted solution which contains 3.8 mg of teduglutide can be withdrawn for subcutaneous injection upon reconstitution.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients less than 1 year of age have not been established.
The safety and effectiveness of GATTEX have been established in pediatric patients 1 year to less than 17 years of age who are dependent on parenteral support for the treatment of SBS. Use of GATTEX in this population is supported by evidence from adequate and well-controlled studies in adults, with additional efficacy, safety, pharmacokinetic and pharmacodynamic data in pediatric patients 1 year to less than 17 years of age [see Dosage and Administration (2), Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.2)]. These data were derived from two studies of 24-week (Study 5) and 12-week (NCT01952080) duration in which 41 pediatric patients were treated with GATTEX in the following groups: 1 infant (1 year to less than 2 years), 37 children (2 years to less than 12 years) and 3 adolescents (12 years to less than 17 years).
In these 2 studies and the corresponding extension studies (Study 6 and NCT02949362), 29 pediatric patients were administered GATTEX prospectively for up to 94 weeks [see Clinical Studies (14.2)]. Adverse reactions in pediatric patients were similar to those seen in adults [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the 134 patients with SBS that were treated with GATTEX at the recommended dosage of 0.05 mg/kg/day in the clinical studies, 19 patients were 65 years or older while 5 patients were 75 years of age or older. No overall differences in safety or efficacy were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of GATTEX or of other teduglutide products.
Adults
Based on integrated data from two studies in adults with SBS (a 6-month randomized placebo-controlled study, followed by a 24-month open-label study), the development of anti-teduglutide antibodies in patients who received subcutaneous administration of 0.05 mg/kg GATTEX once daily was 3% (2/60) at Month 3, 17% (13/77) at Month 6, 24% (16/67) at Month 12, 33% (11/33) at Month 24, and 48% (14/29) at Month 30. Anti-teduglutide antibodies were cross-reactive to native glucagon-like peptide (GLP-2) in 5 of the 6 patients (83%) who had anti-teduglutide antibodies and were tested for cross-reactivity. In the same two studies, a total of 36 patients were tested for neutralizing antibodies: one patient developed borderline positive neutralizing antibody responses at month 24 of the extension study. The antibody formation has not been associated with clinically relevant safety findings, reduced efficacy or changed pharmacokinetics of GATTEX.
Pediatric Patients 1 Year to Less than 17 Years of Age
In pediatric patients who received subcutaneous administration of 0.05 mg/kg GATTEX once daily for 24 weeks, the rate of anti-teduglutide antibody formation at Month 6 was 19% (5/26) and was similar to the rate of antibody formation in adult patients (17%). Of the 5 pediatric patients who had developed antibodies to teduglutide at Month 6, 2 patients had neutralizing antibodies. However, with the longer duration of treatment, the rate of anti-teduglutide formation at Month 12 was higher in pediatric patients with 54% (14/26), compared to that of adults (24%). Of the 14 pediatric patients who had developed antibodies to teduglutide at Month 12, 1 patient had neutralizing antibodies.
Among the small number of pediatric patients who developed anti-teduglutide antibodies, no association with adverse events or lack of efficacy was observed.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
8.6 Renal Impairment
In adult subjects with moderate to severe renal impairment or end-stage renal disease (ESRD) (creatinine clearance <60 mL/min), the exposure to teduglutide increased with the degree of renal impairment [see Clinical Pharmacology (12.3)]. Reduce the dosage of GATTEX by half in both pediatric and adult patients with eGFR less than 60 mL/min/1.73 m2 [see Dosage and Administration (2.3)].
Instructions for Use
This Instructions for Use contains information on how to inject GATTEX.
Read this Instructions for Use before you start using GATTEX and each time you get a refill. There may be new information. Your healthcare provider or nurse should show you how to prepare, measure your dose, and give your injection of GATTEX the right way.
If you cannot give yourself the injection:
- ask your healthcare provider or nurse to help you, or
- ask someone who has been trained by a healthcare provider or nurse to give your injections.
Self-administration is not recommended in pediatric patients. In pediatric patients, GATTEX should be injected by:
- a healthcare provider or nurse, or
- a parent or adult caregiver who has been trained by a healthcare provider or nurse to give injections of GATTEX to pediatric patients.
Important information:
- Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 22 pounds (10 kg).
- Before you start, check the "Use By" date on your GATTEX kit. Make sure that the "Use By" date has not passed. Do not use anything in the GATTEX kit after the "Use By" date on the kit.
- Give GATTEX within 3 hours after you mix the powder with the Diluent (Sterile Water for Injection).
- Use the syringes and needles provided in the GATTEX kit.
- Do not use a GATTEX vial more than 1 time, even if there is medicine left in the vial.
- Throw away (dispose of) any unused GATTEX after you give your injection.
- Safely throw away GATTEX vials after use.
- Do not re-use syringes or needles. See "Step 7: Dispose of syringes and needles" for information about how to safely throw away needles and syringes.
- To help avoid needle-stick injuries, do not recap needles.
Gather the supplies you will need to prepare GATTEX and to give your injection (See Figure A).
|
Figure A |
From your GATTEX kit you will need:
|
Step 1: Prepare the injection.
- Choose a well-lit, clean, flat work surface.
- Wash your hands with soap and water.
Step 2: Preparing the Diluent syringe.
- Put the Diluent syringe (See Figure B1) and the 23G, 1½ inch needle in front of you on your work surface.
| Figure B1 |
|
- Hold the Diluent syringe by the barrel. Snap off the white cap (bend the cap sideways until the cap comes off). Only the top portion of the white cap should be snapped off. The lower portion of the cap will remain in place (See Figure B2). Throw the cap away.
| Figure B2 |
|
- Remove the 23G, 1½ inch needle from the package. Use the fold in the package to peel back the plastic cover (See Figure C). Leave the plastic cap on the needle.
| Figure C |
|
- Push the open end of the needle onto the end of the Diluent syringe (See Figure D).
Twist the needle clockwise (to the right) until it stops turning.
| Figure D |
|
- When the needle is tightly in place, put the Diluent syringe and needle on your work surface.
Step 3: Mix GATTEX powder with Diluent.
- Remove the green cap from the GATTEX vial. Throw away the green cap.
- Find the gray rubber seal on top of the GATTEX vial (See Figure E).
| Figure E |
|
- Use an alcohol swab pad to clean the gray rubber seal (See Figure F).
- Do not touch the gray rubber seal after you clean it.
| Figure F |
|
- Pick up the Diluent syringe with the needle attached.
- Remove the plastic cap that covers the needle (See Figure G). Throw the cap away.
| Figure G |
|
- Hold the GATTEX vial between your thumb and index (pointer) finger (See Figure H). Be careful not to touch the gray rubber seal.
- Push the needle down through the center of the gray rubber seal.
- Slowly push down on the plunger of the Diluent syringe. Empty all the Diluent into the GATTEX vial.
- Leave the needle and Diluent syringe in place.
| Figure H |
|
- Gently tap the barrel of the Diluent syringe with a finger (See Figure I).
- Make sure all the Diluent has gone into the GATTEX vial.
| Figure I |
|
- Remove the Diluent syringe and needle from the GATTEX vial. Let the vial sit for about 30 seconds.
- Do not put the needle cap back on the needle.
- Throw away (dispose of) the Diluent syringe and needle in your sharps disposal container.
- After 30 seconds, place the GATTEX vial between the palms of your hands. Gently roll the vial for about 15 seconds (See Figure J).
- Do not shake the GATTEX vial.
- Do not touch the gray rubber seal. If you do, clean it again with a new alcohol pad.
- Let the GATTEX vial stand on your work surface for about 2 minutes.
| Figure J |
|
Step 4: Check the mixed GATTEX.
- After 2 minutes, look at the vial of GATTEX. The liquid in the vial should be clear and colorless to pale yellow, and should not have any particles in it.
- If there is any powder in the GATTEX vial that did not dissolve, gently roll the vial between your hands for 15 seconds more.
- Do not shake the GATTEX vial.
- Check the GATTEX vial again for anything that did not dissolve.
- Do not use the GATTEX vial if there is anything in it that did not dissolve. Start from the beginning of this Instructions for Use to prepare a new vial. Use a new GATTEX vial, new Diluent syringe, and a new needle.
Step 5: Draw up your dose of GATTEX.
- Remove the plastic dosing syringe from the package. Use the fold in the package to peel back the plastic cover (See Figure K).
| Figure K |
|
- Remove the needle cap from the plastic dosing syringe (See Figure L ).
- Throw the needle cap away. Do not touch the needle or allow it to touch anything.
| Figure L |
|
- Carefully pull back on the plunger to the line that matches the dose prescribed by your healthcare provider.
- Use 1 hand to hold the GATTEX vial steady. Use your other hand to insert the needle straight down into the middle of the gray rubber seal on the GATTEX vial (See Figure M). You may feel some resistance as the needle passes through the rubber seal.
- Gently push down the plunger until all of the air has gone from the plastic dosing syringe into the GATTEX vial.
- Turn the GATTEX vial and plastic dosing syringe upside down (See Figure N).
| Figure M |
|
| Figure N |
|
- Hold the GATTEX vial with 1 hand.
- Slowly pull back the plunger of the plastic dosing syringe with your other hand.
- Fill the plastic dosing syringe until the black tip of the plunger lines up with the mark that matches your prescribed dose (See Figure O).
- Keep the plastic dosing syringe and needle in the GATTEX vial.
| Figure O |
|
- You may see some bubbles inside the GATTEX vial when the plastic dosing syringe is filled. This is normal. With the needle still in the vial, gently tap the side of the plastic dosing syringe with a finger to make any air bubbles rise to the top (See Figure P).
| Figure P |
|
- Slowly push the plunger up until all air bubbles are out of the plastic dosing syringe. Make sure the tip of the needle is in the fluid. Slowly pull back the plunger to draw up the right dose of GATTEX into the plastic dosing syringe.
- Remove the plastic dosing syringe and needle from the GATTEX vial (See Figure Q). Do not touch the needle or allow it to touch anything.
| Figure Q |
|
Step 6: Inject GATTEX.
- Choose an injection site on the stomach area (abdomen), thighs, or upper arms.
- Choose a different site to give the injection each day. Do not inject into areas where the skin is tender, bruised, red, or hard. (See Figure R and Figure S)
| Figure R |
|
|
Figure S |
- Clean the skin where you plan to give the injection with a new alcohol swab pad. Do not touch this area again before giving the injection.
- Use 1 hand to gently pinch up a fold of skin around the injection site (See Figure T).
| Figure T |
|
- Use your other hand to hold the plastic dosing syringe. Insert the full length of the needle into the skin at a 45-degree angle with a quick, "dart-like" motion (See Figure U).
| Figure U |
|
- Let go of the skin. Hold the syringe barrel with 1 hand while you slowly push down the plunger until the plastic dosing syringe is empty (See Figure V).
| Figure V |
|
- When the plastic dosing syringe is empty, quickly pull the needle out of your skin. There may be a little bleeding at the injection site. Apply an adhesive bandage to the injection site if needed.
Step 7: Dispose of syringes and needles.
- Do not re-use a syringe or needle.
- To help avoid needle-stick injuries, do not recap a needle.
- Put your needles and syringes in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharp items being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container.
There may be local or state laws about how to throw away syringes and needles. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. - Do not dispose of your sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your sharps disposal container.
- Throw away the GATTEX vial into the container where you put the syringes and needles.
- If you have any questions, talk to your healthcare provider or pharmacist.
How should I store GATTEX?
- Store GATTEX powder at room temperature up to 77°F (25°C).
- Do not freeze GATTEX.
- Use the GATTEX powder by the expiration date on the "Use By" sticker on the kit.
- Use GATTEX within 3 hours after mixing it.
- Throw away any unused GATTEX that has been mixed, even if there is medicine left in the vial.
- Do not store any GATTEX you have mixed.
Keep GATTEX and all medicines out of the reach of children.
Distributed by:
Takeda Pharmaceuticals America, Inc.
Cambridge, MA 02142
USA
1-877-825-3327
GATTEX and
are registered trademarks of Takeda Pharmaceuticals U.S.A., Inc.©2025 Takeda Pharmaceutical Company Limited. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 09/2025
8.7 Hepatic Impairment
GATTEX has not been studied in patients with severe hepatic impairment (Child-Pugh grade C). No dosage adjustment is recommended for patients with mild and moderate hepatic impairment (Child-Pugh grade A and B) [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
GATTEX® is indicated for the treatment of adults and pediatric patients 1 year of age and older with Short Bowel Syndrome (SBS) who are dependent on parenteral support.
12.1 Mechanism of Action
Teduglutide is an analog of naturally occurring human glucagon-like peptide-2 (GLP-2), a peptide secreted by L-cells of the distal intestine. GLP-2 is known to increase intestinal and portal blood flow and inhibit gastric acid secretion. Teduglutide binds to the glucagon-like peptide-2 receptors located in intestinal subpopulations of enteroendocrine cells, subepithelial myofibroblasts and enteric neurons of the submucosal and myenteric plexus. Activation of these receptors results in the local release of multiple mediators including insulin-like growth factor (IGF)-1, nitric oxide and keratinocyte growth factor (KGF).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
Acceleration of Neoplastic Growth: In case of intestinal malignancy, discontinue GATTEX. The decision to continue GATTEX in patients with non-gastrointestinal malignancy should be made based on benefit risk considerations. (5.1)
- In adult patients, colonoscopy and upper GI endoscopy (or alternate imaging) is recommended after 1 year of treatment. Perform subsequent colonoscopies and upper GI endoscopes (or alternate imaging) no less frequently than every 5 years. (5.1)
- In pediatric patients, perform fecal occult blood testing annually. Colonoscopy/sigmoidoscopy is recommended after 1 year of treatment and every 5 years thereafter on treatment. Consider upper GI endoscopy (or alternate imaging) during treatment with GATTEX. (5.1)
- Intestinal Obstruction: In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX pending further clinical evaluation and management. (5.2)
- Biliary and Pancreatic Disease: Obtain bilirubin, alkaline phosphatase, lipase, amylase every 6 months. If clinically meaningful changes are seen, further evaluation is recommended including imaging, and reassess continued GATTEX treatment. (5.3)
- Fluid Overload, Including Congestive Heart Failure: If fluid overload occurs, adjust parenteral support, and reassess continued GATTEX treatment. (5.4)
- Potential for Increased Absorption of Oral Medications: Monitor patients on concomitant oral medications (e.g., benzodiazepines) for adverse reactions related to the concomitant drug; dosage reduction of the other drug may be required. (5.5, 7.1)
5.2 Intestinal Obstruction
Intestinal obstruction has been reported in clinical studies [see Adverse Reactions (6.1)] and postmarketing. In patients who develop intestinal or stomal obstruction, temporarily discontinue GATTEX while the patient is clinically managed. GATTEX may be restarted when the obstructive presentation resolves, if clinically indicated.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Important Administration Information
GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested. Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg. (2.1)
Evaluation Testing within 6 Months Prior to Starting GATTEX
- Adult Patients: Perform a colonoscopy and upper GI endoscopy with removal of polyps. (2.1, 5.1)
- Pediatric Patients: Perform fecal occult blood testing. If new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and upper GI endoscopy. (2.1, 5.1)
- Adult and Pediatric Patients: Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase). (2.1, 5.3)
Dosage and Administration
- For subcutaneous use only. (2.2)
- The recommended dosage of GATTEX for both adults and pediatric patients is 0.05 mg/kg once daily by subcutaneous injection. (2.2)
- Alternate sites between 1 of the 4 quadrants of the abdomen, or into alternating thighs or alternating arms. (2.2)
Dosage Adjustment for Renal Impairment
- For adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate less than 60 mL/min/1.73 m2) the recommended dosage is 0.025 mg/kg once daily. (2.3)
Discontinuation
Preparation
- See full prescribing information for instructions on reconstitution. (2.6)
2.6 Preparation Instructions
- Reconstitute each vial of GATTEX by slowly injecting the 0.5 mL of preservative-free Sterile Water for Injection provided in the prefilled syringe. A 10 mg/mL sterile solution is obtained after reconstitution.
- Allow the vial containing GATTEX and water to stand for approximately 30 seconds and then gently roll the vial between the palms for about 15 seconds. Do not shake the vial.
- Allow the mixed contents to stand for about 2 minutes. Inspect the vial for any undissolved powder. If undissolved powder is observed, gently roll the vial again until all material is dissolved. Do not shake the vial.
- Reconstituted GATTEX is a sterile, clear, colorless to light straw-colored solution, which should be free from particulates. If there is any discoloration or particulates, discard the solution.
- A maximum of 0.38 mL of the reconstituted solution, containing 3.8 mg of teduglutide, can be withdrawn from the vial for dosing.
- If the product remains undissolved after the second attempt, do not use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For Injection: 5 mg teduglutide as a white lyophilized powder for reconstitution in a single-dose vial supplied with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of teduglutide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Neoplasia: colorectal polyps, gastric polyps, small intestinal polyps (duodenum, ileum, and jejunum)
Injection Site Reactions: injection site induration, inflammation, pruritus, urticaria, and warmth
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Breastfeeding not recommended. (8.2)
2.5 Discontinuation of Treatment
Discontinuation of treatment with GATTEX may result in fluid and electrolyte imbalance. Monitor fluid and electrolyte status in patients who discontinue GATTEX treatment [see Warnings and Precautions (5.4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
GATTEX (teduglutide) for injection is supplied as 5 mg of teduglutide as a white, lyophilized powder for reconstitution in a sterile, single-dose glass vial with 0.5 mL Sterile Water for Injection in a single-dose prefilled syringe. The product to be dispensed is either a one-vial kit or a 30-vial kit.
5.1 Acceleration of Neoplastic Growth
Based on the pharmacologic activity and tumor findings in the rat and mouse carcinogenicity studies, GATTEX has the potential to cause hyperplastic changes including neoplasia [see Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. In patients at increased risk for malignancy, the clinical decision to use GATTEX should be considered only if the benefits outweigh the risks. In patients who develop active gastrointestinal malignancy (GI tract, hepatobiliary, pancreatic) while on GATTEX, discontinue GATTEX treatment. In patients who develop active non-gastrointestinal malignancy while on GATTEX, the clinical decision to continue GATTEX should be made based on benefit-risk considerations.
2.1 Important Administration Information
GATTEX is for adult self-administration or caregiver administration. Self-administration in pediatric patients has not been tested.
Use of the GATTEX 5 mg kit is not recommended in pediatric patients weighing less than 10 kg.
Evaluation and testing prior to starting treatment with GATTEX:
Within 6 months prior to treatment:
Adult patients
- Perform a colonoscopy and an upper gastrointestinal (GI) endoscopy with removal of polyps [see Warnings and Precautions (5.1)].
- Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase) [see Warnings and Precautions (5.3)].
Pediatric patients
- Perform fecal occult blood testing; if there is new or unexplained blood in the stool, perform colonoscopy/sigmoidoscopy and an upper GI endoscopy [see Warnings and Precautions (5.1)].
- Obtain baseline laboratory assessments (bilirubin, alkaline phosphatase, lipase and amylase) [see Warnings and Precautions (5.3)].
2.3 Dosage Adjustment for Renal Impairment
The recommended dosage in adult and pediatric patients with moderate and severe renal impairment and end-stage renal disease (estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2) is 0.025 mg/kg once daily [see Use in Specific Populations (8.6)].
Principal Display Panel 5 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 5 mg Vial Carton)
NDC 68875-0101-2
Gattex®
(teduglutide) for injection
5 mg
5 mg per vial.
For subcutaneous use only.
EACH CARTON CONTAINS:
- Thirty single-dose vials of Gattex®
- Package Insert
- Medication Guide
ATTENTION PHARMACIST:
- For dispensing, transfer the product with vial trays
from this carton into the 30-count patient kit.
Takeda
Principal Display Panel Kit Carton 68875 0102 (PRINCIPAL DISPLAY PANEL - Kit Carton - 68875-0102)
Rx only
Pharmacy:
Apply Use By dating
sticker here
NDC 68875-0102-1
Gattex®
(teduglutide) for injection
5 mg
5 mg per vial.
For subcutaneous use only.
Takeda
Principal Display Panel Kit Carton 68875 0103 (PRINCIPAL DISPLAY PANEL - Kit Carton - 68875-0103)
NDC 68875-0103-1
Rx only
Gattex®
(teduglutide) for injection
5 mg
Pharmacy:
Apply Use By dating
sticker here
5 mg per vial.
For subcutaneous use only.
Attention Patients: Store at room temperature up to 25°C (77°F). Do not freeze.
Takeda
5.5 Increased Absorption of Concomitant Oral Medication
In the adult placebo-controlled studies, an analysis of episodes of cognition and attention disturbances was performed for patients on benzodiazepines. One patient receiving prazepam concomitantly with GATTEX 0.05 mg/kg once daily experienced a dramatic deterioration in mental status progressing to coma during the first week of GATTEX therapy. The patient was admitted to the ICU and the prazepam blood concentration was >300 mcg/L. GATTEX and prazepam were discontinued, and coma resolved 5 days later.
Monitor patients receiving concomitant oral drugs requiring titration or with a narrow therapeutic index, for adverse reactions due to potential increased absorption of the concomitant drug. The concomitant drug may require a reduction in dosage [see Drug Interactions (7.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic potential of GATTEX was assessed in 2-year subcutaneous carcinogenicity studies in rats and mice. In a 2-year carcinogenicity study in Wistar Han rats at subcutaneous doses of 3, 10, and 35 mg/kg/day (about 15, 41, and 199 times the exposures [AUC] achieved at the recommended daily human dose of 0.05 mg/kg, respectively), teduglutide caused statistically significant increases in the incidences of adenomas in the bile duct and jejunum of male rats. In a 2-year carcinogenicity study in Crl:CD1(ICR) mice at subcutaneous doses of 1, 3.5, and 12.5 mg/kg/day (about 32, 66, and 244 times the exposures [AUC] achieved at the recommended daily human dose of 0.05 mg/kg, respectively), teduglutide caused a significant increase in papillary adenomas in the gall bladder; it also caused adenocarcinomas in the jejunum in male mice at the high dose of 12.5 mg/kg/day.
Teduglutide was negative in the Ames test, chromosomal aberration test in Chinese hamster ovary cells, and in vivo mouse micronucleus assay.
Teduglutide at subcutaneous doses up to 25 mg/kg twice daily (50 mg/kg/day or at least 202 times the clinical exposure (AUC) at the recommended daily human dose of 0.05 mg/kg) was found to have no adverse effect on fertility and reproductive performance of male and female rats.
7.1 Potential for Increased Absorption of Oral Medications
Based upon the pharmacodynamic effect of GATTEX, there is a potential for increased absorption of concomitant oral medications. Altered mental status has been observed in patients taking GATTEX and benzodiazepines in the adult clinical studies [see Warnings and Precautions (5.5)].
Monitor patients on concomitant oral drugs requiring titration or with a narrow therapeutic index for adverse reactions related to the concomitant drug while on GATTEX. The concomitant drug may require a reduction in dosage.
2.2 Recommended Dosage and Administration for Adults and Pediatric Patients 1 Year of Age and Older
GATTEX is for subcutaneous injection only. Not for intravenous or intramuscular administration.
The recommended dosage of GATTEX is 0.05 mg/kg once daily administered by subcutaneous injection.
If a dose is missed, that dose should be taken as soon as possible on that day. Do not take 2 doses on the same day.
Alternation of sites for subcutaneous injection is recommended, and can include the thighs, upper arms, and the abdomen.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:34.871404 · Updated: 2026-03-14T22:33:41.553358