Dragon®
65cbdda6-b555-49fb-86e7-ce458c64cae0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Topical analgesic
Medication Information
Purpose
Topical analgesic
Description
Drug Facts
Use
- For the temporary relief of pain
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children to avoid accidental ingestion. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
For external use only
Directions
- use only as directed
- adults and children 12 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
Do Not Use
- on large areas of the body, particularly over raw surfaces or blistered areas
- on wounds or damaged skin
Active Ingredient
Lidocaine HCl 4%
Other Information
- store at room temperature.
- close cap tightly after use.
Inactive Ingredients
Acrylates / C10-30 Alkyl Acrylate Crosspolymer, Alcohol Denat., Aminomethyl Propanol, C30-45 Alkyl Cetearyl Dimethicone Crosspolymer, Caprylyl Methicone, Cetearyl Alcohol, Ceteth-20 Phosphate, Decolorized Aloe Barbadensis Leaf Juice, Dicetyl Phosphate, Dimethicone, Disodium EDTA, Ethylhexylglycerin, Glyceryl StearateSE, Phenoxyethanol, Steareth-21, Water
Questions Or Comments?
Call toll free 1-877-994-3666
When Using This Product
- use only as directed
- avoid contact with eyes
- do not apply tight bandages
- do not apply a heating pad to treated skin
- do not use in large quantities
Stop Use and Ask A Doctor If
- condition worsens or symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
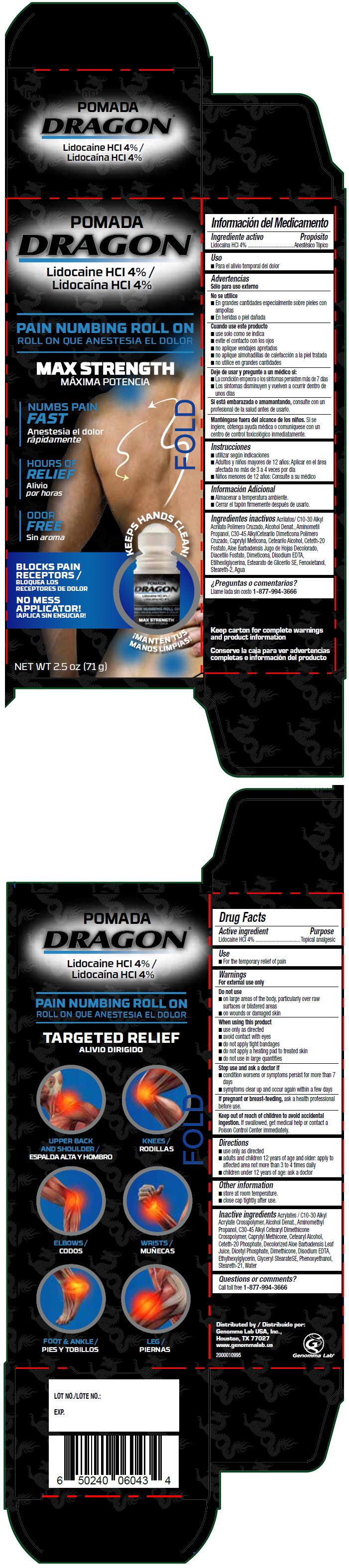
Principal Display Panel 71 G Container Carton
POMADA
DRAGON®
Lidocaine HCl 4%
PAIN NUMBING ROLL ON
MAX STRENGTH
NUMBS PAIN
FAST
HOURS OF
RELIEF
ODOR
FREE
KEEPS HANDS CLEAN!
BLOCKS PAIN
RECEPTORS
NO MESS
APPLICATOR!
NET WT 2.5 oz (71 g)
Structured Label Content
Use
- For the temporary relief of pain
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children to avoid accidental ingestion. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Topical analgesic
Warnings
For external use only
Directions
- use only as directed
- adults and children 12 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
Do Not Use (Do not use)
- on large areas of the body, particularly over raw surfaces or blistered areas
- on wounds or damaged skin
Active Ingredient (Active ingredient)
Lidocaine HCl 4%
Other Information (Other information)
- store at room temperature.
- close cap tightly after use.
Inactive Ingredients (Inactive ingredients)
Acrylates / C10-30 Alkyl Acrylate Crosspolymer, Alcohol Denat., Aminomethyl Propanol, C30-45 Alkyl Cetearyl Dimethicone Crosspolymer, Caprylyl Methicone, Cetearyl Alcohol, Ceteth-20 Phosphate, Decolorized Aloe Barbadensis Leaf Juice, Dicetyl Phosphate, Dimethicone, Disodium EDTA, Ethylhexylglycerin, Glyceryl StearateSE, Phenoxyethanol, Steareth-21, Water
Questions Or Comments? (Questions or comments?)
Call toll free 1-877-994-3666
When Using This Product (When using this product)
- use only as directed
- avoid contact with eyes
- do not apply tight bandages
- do not apply a heating pad to treated skin
- do not use in large quantities
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens or symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
Principal Display Panel 71 G Container Carton (PRINCIPAL DISPLAY PANEL - 71 g Container Carton)
POMADA
DRAGON®
Lidocaine HCl 4%
PAIN NUMBING ROLL ON
MAX STRENGTH
NUMBS PAIN
FAST
HOURS OF
RELIEF
ODOR
FREE
KEEPS HANDS CLEAN!
BLOCKS PAIN
RECEPTORS
NO MESS
APPLICATOR!
NET WT 2.5 oz (71 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:38.857353 · Updated: 2026-03-14T22:59:43.649746