These Highlights Do Not Include All The Information Needed To Use Glydo (lidocaine Hcl Jelly) 2% Safely And Effectively. See Full Prescribing Information For Glydo (lidocaine Hcl Jelly) 2%.
65760723-75cd-472c-96e1-7d05f95b4686
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated: for prevention and control of pain in procedures involving the male and female urethra for topical treatment of painful urethritis as an anesthetic lubricant for oral and nasal endotracheal intubation
Indications and Usage
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated: for prevention and control of pain in procedures involving the male and female urethra for topical treatment of painful urethritis as an anesthetic lubricant for oral and nasal endotracheal intubation
Dosage and Administration
The toxic effects of local anesthetics are additive. When using this product concomitantly with other lidocaine-containing products, consider the total dose of lidocaine and monitor patients for cardiovascular and respiratory vital signs. ( 2.1 ) For surface anesthesia of male adult urethra: 15 mL (300 mg lidocaine HCl) followed by additional 15 mL if needed ( 2.2 ) For surface anesthesia of female adult urethra: 3 mL to 5 mL (60 mg to 100 mg lidocaine HCl) ( 2.3 ) For lubrication for endotracheal intubation: sufficient amount to coat the external surface of endotracheal tube ( 2.4 ) For pediatric patients: Not more than 4.5 mg/kg body weight of lidocaine HCl ( 2.5 )
Warnings and Precautions
Dose-Related Toxicity : follow dosing instructions carefully. ( 5.1 ) Methemoglobinemia : Cases have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2 ) Familial Malignant Hyperthermia : Monitoring of patients is recommended ( 5.3 ) Endotracheal Tube Occlusion : Avoid introduction of jelly into the lumen of the endotracheal tube ( 5.4 ) Risk of Aspiration and Biting Trauma with Oral Use : Food and chewing gum should not be taken while the mouth or throat area is anesthetized ( 5.7 )
Contraindications
Known hypersensitivity to any local anesthetic agent of the amide-type or to other components of GLYDO 2% Jelly ( 4.1 ) Infected and/or Severely Traumatized Mucosa ( 4.2 ) Severe shock or heart block ( 4.3 )
Adverse Reactions
The following adverse reactions associated with the use of GLYDO 2% Jelly were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage or slow metabolic degradation. The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported. There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube.
Drug Interactions
Local Anesthetics : The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered ( 7.1 ). Drugs Associated with Methemoglobinemia : Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs ( 7.2 ). Hepatic Impairment : Consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment ( 8.6 )
Storage and Handling
GLYDO ® (lidocaine HCl jelly, USP) 2% is supplied as follows: NDC GLYDO ® (lidocaine HCl jelly, USP) 2% Package Factor (20 mg per mL) 25021-673-76 120 mg per 6 mL Single-Dose Prefilled Syringe 10 syringes per carton 25021-673-77 220 mg per 11 mL Single-Dose Prefilled Syringe 10 syringes per carton
How Supplied
GLYDO ® (lidocaine HCl jelly, USP) 2% is supplied as follows: NDC GLYDO ® (lidocaine HCl jelly, USP) 2% Package Factor (20 mg per mL) 25021-673-76 120 mg per 6 mL Single-Dose Prefilled Syringe 10 syringes per carton 25021-673-77 220 mg per 11 mL Single-Dose Prefilled Syringe 10 syringes per carton
Medication Information
Warnings and Precautions
Dose-Related Toxicity : follow dosing instructions carefully. ( 5.1 ) Methemoglobinemia : Cases have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2 ) Familial Malignant Hyperthermia : Monitoring of patients is recommended ( 5.3 ) Endotracheal Tube Occlusion : Avoid introduction of jelly into the lumen of the endotracheal tube ( 5.4 ) Risk of Aspiration and Biting Trauma with Oral Use : Food and chewing gum should not be taken while the mouth or throat area is anesthetized ( 5.7 )
Indications and Usage
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated: for prevention and control of pain in procedures involving the male and female urethra for topical treatment of painful urethritis as an anesthetic lubricant for oral and nasal endotracheal intubation
Dosage and Administration
The toxic effects of local anesthetics are additive. When using this product concomitantly with other lidocaine-containing products, consider the total dose of lidocaine and monitor patients for cardiovascular and respiratory vital signs. ( 2.1 ) For surface anesthesia of male adult urethra: 15 mL (300 mg lidocaine HCl) followed by additional 15 mL if needed ( 2.2 ) For surface anesthesia of female adult urethra: 3 mL to 5 mL (60 mg to 100 mg lidocaine HCl) ( 2.3 ) For lubrication for endotracheal intubation: sufficient amount to coat the external surface of endotracheal tube ( 2.4 ) For pediatric patients: Not more than 4.5 mg/kg body weight of lidocaine HCl ( 2.5 )
Contraindications
Known hypersensitivity to any local anesthetic agent of the amide-type or to other components of GLYDO 2% Jelly ( 4.1 ) Infected and/or Severely Traumatized Mucosa ( 4.2 ) Severe shock or heart block ( 4.3 )
Adverse Reactions
The following adverse reactions associated with the use of GLYDO 2% Jelly were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage or slow metabolic degradation. The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported. There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube.
Drug Interactions
Local Anesthetics : The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered ( 7.1 ). Drugs Associated with Methemoglobinemia : Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs ( 7.2 ). Hepatic Impairment : Consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment ( 8.6 )
Storage and Handling
GLYDO ® (lidocaine HCl jelly, USP) 2% is supplied as follows: NDC GLYDO ® (lidocaine HCl jelly, USP) 2% Package Factor (20 mg per mL) 25021-673-76 120 mg per 6 mL Single-Dose Prefilled Syringe 10 syringes per carton 25021-673-77 220 mg per 11 mL Single-Dose Prefilled Syringe 10 syringes per carton
How Supplied
GLYDO ® (lidocaine HCl jelly, USP) 2% is supplied as follows: NDC GLYDO ® (lidocaine HCl jelly, USP) 2% Package Factor (20 mg per mL) 25021-673-76 120 mg per 6 mL Single-Dose Prefilled Syringe 10 syringes per carton 25021-673-77 220 mg per 11 mL Single-Dose Prefilled Syringe 10 syringes per carton
Description
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated: for prevention and control of pain in procedures involving the male and female urethra for topical treatment of painful urethritis as an anesthetic lubricant for oral and nasal endotracheal intubation
Section 42229-5
Administration Precautions
The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects related to local anesthetic systemic toxicity when additional local anesthetics are administered with GLYDO (lidocaine HCl jelly) 2% [see Warnings and Precautions (5.1), Adverse Reactions (6), Overdosage (10)].
The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients.
Although the incidence of adverse effects with GLYDO (lidocaine HCl jelly) 2% is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
No more than 600 mg of lidocaine HCl should be given in any 12 hour period.
Section 42230-3
INSTRUCTIONS FOR USE
Please note: The blister package contains a sterile syringe.
Do not open the blister until ready to use.
- When ready to use, open the blister and drop the syringe onto a sterile field.
- Before removing the tip cap, press in the plunger to remove any resistance that may be present. This helps ensure that the syringe will empty easily and uniformly.
- Remove the tip cap from the syringe. The syringe is now ready for use.
- GLYDO (lidocaine HCl jelly, USP) 2% should be instilled slowly and evenly into the urethra. See the DOSAGE AND ADMINISTRATION section for additional details.
- Wait for a few minutes after instillation of GLYDO (lidocaine HCl jelly, USP) 2% for the anesthetic to take full effect. Full anesthetic effect will occur in 5 to 10 minutes after complete instillation.
- Any gel not used in a single application must be discarded.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Mfd. by Klosterfrau Berlin GmbH
Made in Germany
©2025 Sagent Pharmaceuticals
Revised: May 2025
This “Instructions for Use” has been approved by the U.S. Food and Drug Administration.
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Discard unused portion.
Sterile, Preservative-free, PVC-free.
The container and container closure are not made with natural rubber latex.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Syringe Label
NDC 25021-673-76
6 mL Single-Dose Prefilled Syringe
Rx only
glydo®
(lidocaine HCl jelly, USP) 2%
120 mg per 6 mL (20 mg per mL)
6 mL
Sterile TOPICAL Anesthetic
10 Overdosage
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics [see Warnings and Precautions (5) and Adverse Reactions (6)]. The use of this product concomitantly with other lidocaine products should be done with caution and appropriate patient monitoring.
11 Description
GLYDO (lidocaine HCl jelly, USP) 2% is a sterile aqueous product that contains a local anesthetic agent and is administered topically (see INDICATIONS AND USAGE for specific uses).
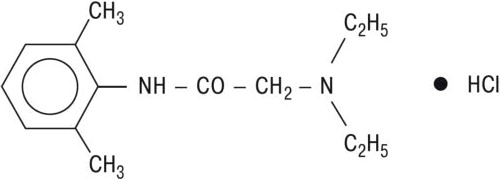
GLYDO (lidocaine HCl jelly, USP) 2% contains lidocaine HCl which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride, the molecular weight is 270.8. The molecular formula is C14H23ClN2O, and has the following structural formula:
GLYDO (lidocaine HCl jelly, USP) 2% also contains hypromellose, and the resulting mixture maximizes contact with mucosa and provides lubrication for instrumentation. The unused portion should be discarded after initial use.
Composition of GLYDO (lidocaine HCl jelly, USP) 2% 6 mL or 11 mL syringes: Each mL contains 20 mg of lidocaine HCl. The formulation also contains hypromellose, and sodium hydroxide to adjust pH to 6.0 to 7.0.
8.4 Pediatric Use
Although the safety and effectiveness of GLYDO 2% Jelly in pediatric patients have not been established, a study of 19 premature neonates (gestational age <33 weeks) found no correlation between the plasma concentration of lidocaine or monoethylglycinexylidide and infant body weight when moderate amounts of lidocaine (i.e. 0.3 mL/kg of lidocaine gel 20 mg/mL) were used for lubricating both intranasal and endotracheal tubes. No neonate had plasma levels of lidocaine above 750 mcg/L. Dosages in children should be reduced, commensurate with age, body weight, and physical condition [see Dosage and Administration (2)].
8.5 Geriatric Use
Elderly patients should be given reduced doses commensurate with their age and physical condition.
4 Contraindications
6 Adverse Reactions
The following adverse reactions associated with the use of GLYDO 2% Jelly were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported.
There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube.
7 Drug Interactions
- Local Anesthetics: The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered (7.1).
- Drugs Associated with Methemoglobinemia: Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs (7.2).
- Hepatic Impairment: Consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment (8.6)
4.1 Hypersensitivity
GLYDO 2% Jelly is contraindicated in patients with a known history of hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of GLYDO 2% Jelly.
12.2 Pharmacodynamics
The onset of action is 3 to 5 minutes. It is ineffective when applied to intact skin.
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine hydrochloride required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
5.2 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions (7.2)]. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue GLYDO 2% Jelly and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.1 Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with GLYDO 2% Jelly cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions (5.1)].
17.2 Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue [see Warnings and Precautions (5.2)].
8.6 Hepatic Impairment
Amide-type local anesthetics such as lidocaine are metabolized by the liver. Patients with severe hepatic impairment, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations and potentially local anesthetic systemic toxicity. Therefore, consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment treated with GLYDO 2% Jelly especially with repeat doses [see Warnings and Precautions (5.6)].
1 Indications and Usage
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated:
- for prevention and control of pain in procedures involving the male and female urethra
- for topical treatment of painful urethritis
- as an anesthetic lubricant for oral and nasal endotracheal intubation
17.3 Risk of Aspiration
When topical anesthetics are used in the mouth, the patient should be aware that the production of topical anesthesia may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
12.1 Mechanism of Action
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
5.1 Dose Related Toxicity
The safety and effectiveness of GLYDO 2% Jelly depends on proper dosage, correct technique, adequate precautions, and readiness for emergencies [see Adverse Reactions (6)]. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after application of GLYDO 2% Jelly. Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
Use the lowest dosage that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical status.
Patients and healthcare providers should be instructed to strictly adhere to the recommended dosage and administration guidelines as set forth in this package insert. The management of serious adverse reactions may require the use of resuscitative equipment, oxygen, and other resuscitative drugs.
17.4 Risk of Biting Trauma
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food and chewing gum should not be taken while the mouth or throat area is anesthetized.
Brands listed are the trademarks of their respective owners.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Mfd. by Klosterfrau Berlin GmbH
Made in Germany
©2025 Sagent Pharmaceuticals
Revised: May 2025
5 Warnings and Precautions
- Dose-Related Toxicity: follow dosing instructions carefully. (5.1)
- Methemoglobinemia: Cases have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. (5.2)
- Familial Malignant Hyperthermia: Monitoring of patients is recommended (5.3)
- Endotracheal Tube Occlusion: Avoid introduction of jelly into the lumen of the endotracheal tube (5.4)
- Risk of Aspiration and Biting Trauma with Oral Use: Food and chewing gum should not be taken while the mouth or throat area is anesthetized (5.7)
5.5 Anaphylactic Reactions
Anaphylactic reactions may occur following administration of lidocaine hydrochloride [see Adverse Reactions (6)]. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
2 Dosage and Administration
- The toxic effects of local anesthetics are additive. When using this product concomitantly with other lidocaine-containing products, consider the total dose of lidocaine and monitor patients for cardiovascular and respiratory vital signs. (2.1)
- For surface anesthesia of male adult urethra: 15 mL (300 mg lidocaine HCl) followed by additional 15 mL if needed (2.2)
- For surface anesthesia of female adult urethra: 3 mL to 5 mL (60 mg to 100 mg lidocaine HCl) (2.3)
- For lubrication for endotracheal intubation: sufficient amount to coat the external surface of endotracheal tube (2.4)
- For pediatric patients: Not more than 4.5 mg/kg body weight of lidocaine HCl (2.5)
17.1 Allergic Type Reactions
3 Dosage Forms and Strengths
GLYDO (lidocaine HCl jelly, USP) 2% in:
- 120 mg per 6 mL Prefilled Syringe
- 220 mg per 11 mL Prefilled Syringe
8 Use in Specific Populations
- Pediatric Use: Dose should be reduced commensurate with age, body weight and physical condition (8.4)
5.4 Endotracheal Tube Occlusion
When used for endotracheal tube lubrication, care should be taken to avoid introducing the product into the lumen of the tube. Do not use the jelly to lubricate the endotracheal stylettes. If allowed into the inner lumen, the jelly may dry on the inner surface leaving a residue which tends to clump with flexion, narrowing the lumen. There have been rare reports in which this residue has caused the lumen to occlude [see Adverse Reactions (6) and Dosage and Administration (2)].
2.5 Dosing for Pediatric Patients
A maximum dose of GLYDO (lidocaine HCl jelly) 2% for children varies based on age and weight. The maximum dose should not exceed 4.5 mg/kg of body weight. For children over 3 years of age, the maximum dose is determined by the child's age and weight. For example, in a child of 5 years weighing approximately 23 kg, the dose of lidocaine hydrochloride should not exceed approximately 75 mg to 100 mg (3.3 mg/kg to 4.4 mg/kg).
The lowest effective dose should be used.
5.3 Familial Malignant Hyperthermia
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure, and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
16 How Supplied/storage and Handling
GLYDO® (lidocaine HCl jelly, USP) 2% is supplied as follows:
| NDC | GLYDO ® (lidocaine HCl jelly, USP) 2% | Package Factor |
| (20 mg per mL) | ||
| 25021-673-76 | 120 mg per 6 mL Single-Dose Prefilled Syringe | 10 syringes per carton |
| 25021-673-77 | 220 mg per 11 mL Single-Dose Prefilled Syringe | 10 syringes per carton |
4.3 Use in Severe Shock Or Heart Block
GLYDO 2% Jelly should not be used in patients with severe shock or heart block.
2.4 Lubrication for Endotracheal Intubation
Apply a sufficient amount of jelly to coat the external surface of the endotracheal tube shortly before use. Care should be taken to avoid introducing the product into the lumen of the tube.
Do not use the jelly to lubricate endotracheal stylettes. (see WARNINGS and ADVERSE REACTIONS concerning rare reports of inner lumen occlusion). It is also recommended that use of endotracheal tubes with dried jelly on the external surface be avoided for lack of lubricating effect.
7.2 Drugs Associated With Methemoglobinemia
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
| Class | Examples |
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastics Agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase, |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | Phenobarbital, phenytoin, sodium valproate, |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
2.2 for Surface Anesthesia of the Male Adult Urethra
Slowly instill approximately 15 mL (300 mg of lidocaine HCl) into the urethra. Apply a penile clamp for several minutes at the corona. An additional dose of not more than 15 mL (300 mg) can be instilled if needed.
Prior to sounding or cystoscopy, a penile clamp should be applied for 5 to 10 minutes to obtain adequate anesthesia. A total dose of 30 mL (600 mg) is usually required to fill and dilate the male urethra.
Prior to catheterization, 5 mL to 10 mL (100 mg to 200 mg) are recommended for lubrication.
2.3 for Surface Anesthesia of the Female Adult Urethra
Slowly instill approximately 3 mL to 5 mL (60 mg to 100 mg of lidocaine HCl) into the urethra. In order to obtain adequate anesthesia, wait for several minutes prior to performing urological procedures. If desired, some jelly may be deposited on a cotton swab and introduced into the urethra.
4.2 Use On Infected And/or Severely Traumatized Mucosa
GLYDO 2% Jelly should not be used on infected and/or severely traumatized mucosa in the area of application.
5.7 Risk of Aspiration and Biting Trauma With Oral Use
When used orally (i.e., endotracheal tube lubrication), topical anesthesia may occur to oropharyngeal structures. This may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food and chewing gum should not be taken while the mouth or throat area is anesthetized.
5.6 Risk of Toxicity in Patients With Hepatic Impairment
Because amide local anesthetics such as lidocaine are metabolized by the liver, consider reduced dosing and increased monitoring for lidocaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated with GLYDO 2% Jelly, especially with repeat doses [see Use in Specific Populations (8.6)].
Structured Label Content
Section 42229-5 (42229-5)
Administration Precautions
The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects related to local anesthetic systemic toxicity when additional local anesthetics are administered with GLYDO (lidocaine HCl jelly) 2% [see Warnings and Precautions (5.1), Adverse Reactions (6), Overdosage (10)].
The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients.
Although the incidence of adverse effects with GLYDO (lidocaine HCl jelly) 2% is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
No more than 600 mg of lidocaine HCl should be given in any 12 hour period.
Section 42230-3 (42230-3)
INSTRUCTIONS FOR USE
Please note: The blister package contains a sterile syringe.
Do not open the blister until ready to use.
- When ready to use, open the blister and drop the syringe onto a sterile field.
- Before removing the tip cap, press in the plunger to remove any resistance that may be present. This helps ensure that the syringe will empty easily and uniformly.
- Remove the tip cap from the syringe. The syringe is now ready for use.
- GLYDO (lidocaine HCl jelly, USP) 2% should be instilled slowly and evenly into the urethra. See the DOSAGE AND ADMINISTRATION section for additional details.
- Wait for a few minutes after instillation of GLYDO (lidocaine HCl jelly, USP) 2% for the anesthetic to take full effect. Full anesthetic effect will occur in 5 to 10 minutes after complete instillation.
- Any gel not used in a single application must be discarded.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Mfd. by Klosterfrau Berlin GmbH
Made in Germany
©2025 Sagent Pharmaceuticals
Revised: May 2025
This “Instructions for Use” has been approved by the U.S. Food and Drug Administration.
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Discard unused portion.
Sterile, Preservative-free, PVC-free.
The container and container closure are not made with natural rubber latex.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Syringe Label
NDC 25021-673-76
6 mL Single-Dose Prefilled Syringe
Rx only
glydo®
(lidocaine HCl jelly, USP) 2%
120 mg per 6 mL (20 mg per mL)
6 mL
Sterile TOPICAL Anesthetic
10 Overdosage (10 OVERDOSAGE)
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics [see Warnings and Precautions (5) and Adverse Reactions (6)]. The use of this product concomitantly with other lidocaine products should be done with caution and appropriate patient monitoring.
11 Description (11 DESCRIPTION)
GLYDO (lidocaine HCl jelly, USP) 2% is a sterile aqueous product that contains a local anesthetic agent and is administered topically (see INDICATIONS AND USAGE for specific uses).
GLYDO (lidocaine HCl jelly, USP) 2% contains lidocaine HCl which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride, the molecular weight is 270.8. The molecular formula is C14H23ClN2O, and has the following structural formula:
GLYDO (lidocaine HCl jelly, USP) 2% also contains hypromellose, and the resulting mixture maximizes contact with mucosa and provides lubrication for instrumentation. The unused portion should be discarded after initial use.
Composition of GLYDO (lidocaine HCl jelly, USP) 2% 6 mL or 11 mL syringes: Each mL contains 20 mg of lidocaine HCl. The formulation also contains hypromellose, and sodium hydroxide to adjust pH to 6.0 to 7.0.
8.4 Pediatric Use
Although the safety and effectiveness of GLYDO 2% Jelly in pediatric patients have not been established, a study of 19 premature neonates (gestational age <33 weeks) found no correlation between the plasma concentration of lidocaine or monoethylglycinexylidide and infant body weight when moderate amounts of lidocaine (i.e. 0.3 mL/kg of lidocaine gel 20 mg/mL) were used for lubricating both intranasal and endotracheal tubes. No neonate had plasma levels of lidocaine above 750 mcg/L. Dosages in children should be reduced, commensurate with age, body weight, and physical condition [see Dosage and Administration (2)].
8.5 Geriatric Use
Elderly patients should be given reduced doses commensurate with their age and physical condition.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions associated with the use of GLYDO 2% Jelly were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported.
There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Local Anesthetics: The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered (7.1).
- Drugs Associated with Methemoglobinemia: Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs (7.2).
- Hepatic Impairment: Consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment (8.6)
4.1 Hypersensitivity
GLYDO 2% Jelly is contraindicated in patients with a known history of hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of GLYDO 2% Jelly.
12.2 Pharmacodynamics
The onset of action is 3 to 5 minutes. It is ineffective when applied to intact skin.
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine hydrochloride required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
5.2 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions (7.2)]. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue GLYDO 2% Jelly and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.1 Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with GLYDO 2% Jelly cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions (5.1)].
17.2 Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue [see Warnings and Precautions (5.2)].
8.6 Hepatic Impairment
Amide-type local anesthetics such as lidocaine are metabolized by the liver. Patients with severe hepatic impairment, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations and potentially local anesthetic systemic toxicity. Therefore, consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment treated with GLYDO 2% Jelly especially with repeat doses [see Warnings and Precautions (5.6)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
GLYDO (lidocaine HCl jelly) 2% is an amide local anesthetic indicated:
- for prevention and control of pain in procedures involving the male and female urethra
- for topical treatment of painful urethritis
- as an anesthetic lubricant for oral and nasal endotracheal intubation
17.3 Risk of Aspiration
When topical anesthetics are used in the mouth, the patient should be aware that the production of topical anesthesia may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
12.1 Mechanism of Action
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
5.1 Dose Related Toxicity (5.1 Dose-Related Toxicity)
The safety and effectiveness of GLYDO 2% Jelly depends on proper dosage, correct technique, adequate precautions, and readiness for emergencies [see Adverse Reactions (6)]. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after application of GLYDO 2% Jelly. Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
Use the lowest dosage that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical status.
Patients and healthcare providers should be instructed to strictly adhere to the recommended dosage and administration guidelines as set forth in this package insert. The management of serious adverse reactions may require the use of resuscitative equipment, oxygen, and other resuscitative drugs.
17.4 Risk of Biting Trauma
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food and chewing gum should not be taken while the mouth or throat area is anesthetized.
Brands listed are the trademarks of their respective owners.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Mfd. by Klosterfrau Berlin GmbH
Made in Germany
©2025 Sagent Pharmaceuticals
Revised: May 2025
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Dose-Related Toxicity: follow dosing instructions carefully. (5.1)
- Methemoglobinemia: Cases have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. (5.2)
- Familial Malignant Hyperthermia: Monitoring of patients is recommended (5.3)
- Endotracheal Tube Occlusion: Avoid introduction of jelly into the lumen of the endotracheal tube (5.4)
- Risk of Aspiration and Biting Trauma with Oral Use: Food and chewing gum should not be taken while the mouth or throat area is anesthetized (5.7)
5.5 Anaphylactic Reactions
Anaphylactic reactions may occur following administration of lidocaine hydrochloride [see Adverse Reactions (6)]. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The toxic effects of local anesthetics are additive. When using this product concomitantly with other lidocaine-containing products, consider the total dose of lidocaine and monitor patients for cardiovascular and respiratory vital signs. (2.1)
- For surface anesthesia of male adult urethra: 15 mL (300 mg lidocaine HCl) followed by additional 15 mL if needed (2.2)
- For surface anesthesia of female adult urethra: 3 mL to 5 mL (60 mg to 100 mg lidocaine HCl) (2.3)
- For lubrication for endotracheal intubation: sufficient amount to coat the external surface of endotracheal tube (2.4)
- For pediatric patients: Not more than 4.5 mg/kg body weight of lidocaine HCl (2.5)
17.1 Allergic Type Reactions (17.1 Allergic-Type Reactions)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
GLYDO (lidocaine HCl jelly, USP) 2% in:
- 120 mg per 6 mL Prefilled Syringe
- 220 mg per 11 mL Prefilled Syringe
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatric Use: Dose should be reduced commensurate with age, body weight and physical condition (8.4)
5.4 Endotracheal Tube Occlusion
When used for endotracheal tube lubrication, care should be taken to avoid introducing the product into the lumen of the tube. Do not use the jelly to lubricate the endotracheal stylettes. If allowed into the inner lumen, the jelly may dry on the inner surface leaving a residue which tends to clump with flexion, narrowing the lumen. There have been rare reports in which this residue has caused the lumen to occlude [see Adverse Reactions (6) and Dosage and Administration (2)].
2.5 Dosing for Pediatric Patients
A maximum dose of GLYDO (lidocaine HCl jelly) 2% for children varies based on age and weight. The maximum dose should not exceed 4.5 mg/kg of body weight. For children over 3 years of age, the maximum dose is determined by the child's age and weight. For example, in a child of 5 years weighing approximately 23 kg, the dose of lidocaine hydrochloride should not exceed approximately 75 mg to 100 mg (3.3 mg/kg to 4.4 mg/kg).
The lowest effective dose should be used.
5.3 Familial Malignant Hyperthermia
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure, and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
GLYDO® (lidocaine HCl jelly, USP) 2% is supplied as follows:
| NDC | GLYDO ® (lidocaine HCl jelly, USP) 2% | Package Factor |
| (20 mg per mL) | ||
| 25021-673-76 | 120 mg per 6 mL Single-Dose Prefilled Syringe | 10 syringes per carton |
| 25021-673-77 | 220 mg per 11 mL Single-Dose Prefilled Syringe | 10 syringes per carton |
4.3 Use in Severe Shock Or Heart Block (4.3 Use in Severe Shock or Heart Block)
GLYDO 2% Jelly should not be used in patients with severe shock or heart block.
2.4 Lubrication for Endotracheal Intubation
Apply a sufficient amount of jelly to coat the external surface of the endotracheal tube shortly before use. Care should be taken to avoid introducing the product into the lumen of the tube.
Do not use the jelly to lubricate endotracheal stylettes. (see WARNINGS and ADVERSE REACTIONS concerning rare reports of inner lumen occlusion). It is also recommended that use of endotracheal tubes with dried jelly on the external surface be avoided for lack of lubricating effect.
7.2 Drugs Associated With Methemoglobinemia (7.2 Drugs Associated with Methemoglobinemia)
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
| Class | Examples |
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastics Agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase, |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | Phenobarbital, phenytoin, sodium valproate, |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
2.2 for Surface Anesthesia of the Male Adult Urethra (2.2 For Surface Anesthesia of the Male Adult Urethra)
Slowly instill approximately 15 mL (300 mg of lidocaine HCl) into the urethra. Apply a penile clamp for several minutes at the corona. An additional dose of not more than 15 mL (300 mg) can be instilled if needed.
Prior to sounding or cystoscopy, a penile clamp should be applied for 5 to 10 minutes to obtain adequate anesthesia. A total dose of 30 mL (600 mg) is usually required to fill and dilate the male urethra.
Prior to catheterization, 5 mL to 10 mL (100 mg to 200 mg) are recommended for lubrication.
2.3 for Surface Anesthesia of the Female Adult Urethra (2.3 For Surface Anesthesia of the Female Adult Urethra)
Slowly instill approximately 3 mL to 5 mL (60 mg to 100 mg of lidocaine HCl) into the urethra. In order to obtain adequate anesthesia, wait for several minutes prior to performing urological procedures. If desired, some jelly may be deposited on a cotton swab and introduced into the urethra.
4.2 Use On Infected And/or Severely Traumatized Mucosa (4.2 Use on Infected and/or Severely Traumatized Mucosa)
GLYDO 2% Jelly should not be used on infected and/or severely traumatized mucosa in the area of application.
5.7 Risk of Aspiration and Biting Trauma With Oral Use (5.7 Risk of Aspiration and Biting Trauma with Oral Use)
When used orally (i.e., endotracheal tube lubrication), topical anesthesia may occur to oropharyngeal structures. This may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food and chewing gum should not be taken while the mouth or throat area is anesthetized.
5.6 Risk of Toxicity in Patients With Hepatic Impairment (5.6 Risk of Toxicity in Patients with Hepatic Impairment)
Because amide local anesthetics such as lidocaine are metabolized by the liver, consider reduced dosing and increased monitoring for lidocaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated with GLYDO 2% Jelly, especially with repeat doses [see Use in Specific Populations (8.6)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:07.560452 · Updated: 2026-03-14T22:51:41.355943