Lidocaine 4 Percent Cream
651710db-3c50-4bcf-b7cc-9ed24f663427
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
LIDOCAINE - L idocaine HCl 4% C ream SA3, LLC Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Lidocaine HCl 4% Topical Analgesic Cream Drug Facts
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
LIDOCAINE - L idocaine HCl 4% C ream SA3, LLC Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Lidocaine HCl 4% Topical Analgesic Cream Drug Facts
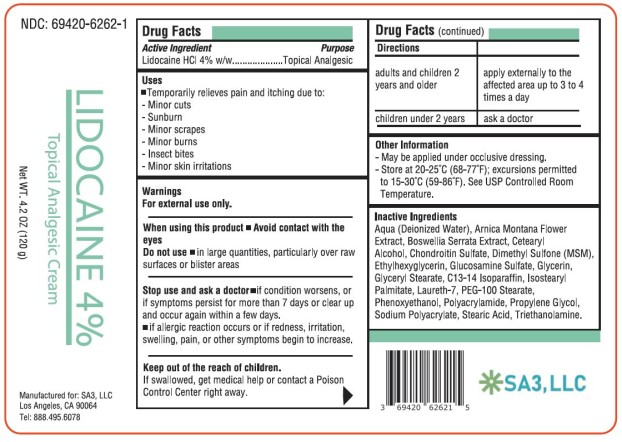
Uses
Temporarily relieves pain and itching due to:
- minor cuts
- sunburn
- minor scrapes
- minor burns
- insect bites
- minor skin irritations
Section 42229-5
LIDOCAINE
- L
idocaine
HCl 4%
C
ream
SA3, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Lidocaine HCl 4% Topical Analgesic Cream
Drug Facts
Warnings
For external use only.
Directions
| adults and children 2 years and older | apply externally to the affected area up to 3 to 4 times a day |
| children under 2 years | ask a doctor |
Active Ingredient
Lidocaine HCl 4% w/w
Other Information
- May be applied under occlusive dressing.
- Store at 20-25°C (68-77°F); excursions permitted to 15-30°C (59-86°F). See USP Controlled Room Temperature.
Inactive Ingredients
Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia Serrata Extract, Cetearyl Alcohol, Chondroitin Sulfate, Dimethyl Sulfone (MSM), Ethylhexylglycerin, Glucosamine Sulfate, Glycerin, Glyceryl Stearate, C13-14 Isoparaffin, Isostearyl Palmitate, Laureth-7, PEG-100 Stearate, Phenoxyethanol, Polyacrylamide, Propylene Glycol, Sodium Polyacrylate, Stearic Acid, Triethanolamine.
Principal Display Panel
Lidocaine HCl 4% cream
NDC 69420-6262-1
Topical Analgesic Cream
4.2 OZ ( 120 g )
SA3, LLC
When Using This Product
-
Avoid contact with the eyes
- D o not use in large quantities, particularly over raw surfaces or blistered areas
Stop Use and Ask A Doctor
- If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
- If allergic reaction occurs or if redness, irritation, swelling, pain or other symptoms begin or increase.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
Temporarily relieves pain and itching due to:
- minor cuts
- sunburn
- minor scrapes
- minor burns
- insect bites
- minor skin irritations
Section 42229-5 (42229-5)
LIDOCAINE
- L
idocaine
HCl 4%
C
ream
SA3, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Lidocaine HCl 4% Topical Analgesic Cream
Drug Facts
Purpose
Topical Analgesic
Warnings
For external use only.
Directions
| adults and children 2 years and older | apply externally to the affected area up to 3 to 4 times a day |
| children under 2 years | ask a doctor |
Active Ingredient (Active ingredient)
Lidocaine HCl 4% w/w
Other Information (Other information)
- May be applied under occlusive dressing.
- Store at 20-25°C (68-77°F); excursions permitted to 15-30°C (59-86°F). See USP Controlled Room Temperature.
Inactive Ingredients (Inactive ingredients)
Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia Serrata Extract, Cetearyl Alcohol, Chondroitin Sulfate, Dimethyl Sulfone (MSM), Ethylhexylglycerin, Glucosamine Sulfate, Glycerin, Glyceryl Stearate, C13-14 Isoparaffin, Isostearyl Palmitate, Laureth-7, PEG-100 Stearate, Phenoxyethanol, Polyacrylamide, Propylene Glycol, Sodium Polyacrylate, Stearic Acid, Triethanolamine.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Lidocaine HCl 4% cream
NDC 69420-6262-1
Topical Analgesic Cream
4.2 OZ ( 120 g )
SA3, LLC
When Using This Product (When using this product)
-
Avoid contact with the eyes
- D o not use in large quantities, particularly over raw surfaces or blistered areas
Stop Use and Ask A Doctor (Stop use and ask a doctor)
- If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
- If allergic reaction occurs or if redness, irritation, swelling, pain or other symptoms begin or increase.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:38.499536 · Updated: 2026-03-14T23:05:32.543899