Children's
64dd0b21-21c5-4d45-ac7f-9896c2a033b0
34390-5
HUMAN OTC DRUG LABEL
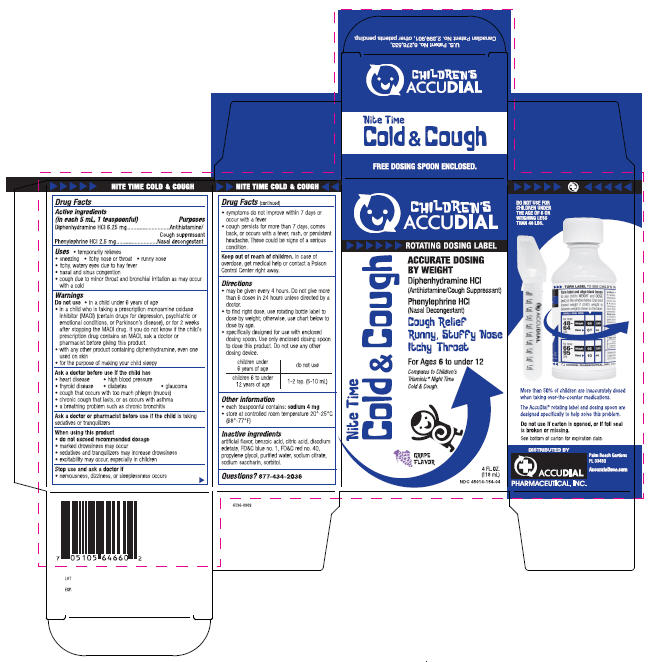
Drug Facts
Composition & Product
Identifiers & Packaging
Description
NITE TIME COLD & COUGH Drug Facts
Medication Information
Description
NITE TIME COLD & COUGH Drug Facts
Uses
- temporarily relieves
- sneezing
- itchy nose or throat
- runny nose
- itchy, watery eyes due to hay fever
- nasal and sinus congestion
- cough due to minor throat and bronchial irritation as may occur with a cold
Section 42229-5
NITE TIME COLD & COUGH
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50568-5
Ask a doctor or pharmacist before use if the child is taking sedatives or tranquilizers
Section 55105-1
|
Active ingredients (in each 5 mL, 1 teaspoonful) |
Purposes |
|---|---|
| Diphenhydramine HCl 6.25 mg | Antihistamine/Cough suppressant |
| Phenylephrine HCl 2.5 mg | Nasal decongestant |
Directions
- may be given every 4 hours. Do not give more than 6 doses in 24 hours unless directed by a doctor.
- to find right dose, use rotating bottle label to dose by weight; otherwise, use chart below to dose by age.
- specifically designed for use with enclosed dosing spoon. Use only enclosed dosing spoon to dose this product. Do not use any other dosing device.
| children under 6 years of age | do not use |
| children 6 to under 12 years of age | 1-2 tsp. (5-10 mL) |
Do Not Use
- in a child under 6 years of age
- in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if the child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
- with any other product containing diphenhydramine, even one used on skin
- for the purpose of making your child sleepy
Questions?
877-434-2036
Other Information
- each teaspoonful contains: sodium 4 mg
- store at controlled room temperature 20°-25°C (68°-77°F)
Inactive Ingredients
artificial flavor, benzoic acid, citric acid, disodium edetate, FD&C blue no. 1, FD&C red no. 40, propylene glycol, purified water, sodium citrate, sodium saccharin, sorbitol.
When Using This Product
- do not exceed recommended dosage
- marked drowsiness may occur
- sedatives and tranquilizers may increase drowsiness
- excitability may occur, especially in children
Stop Use and Ask A Doctor If
- nervousness, dizziness, or sleeplessness occurs
- symptoms do not improve within 7 days or occur with a fever
- cough persists for more than 7 days, comes back, or occurs with a fever, rash, or persistent headache. These could be signs of a serious condition.
Principal Display Panel 118 Ml Carton
CHILDREN'S
ACCUDIAL
ROTATING DOSING LABEL
Nite Time
Cold & Cough
ACCURATE DOSING
BY WEIGHT
Diphenhydramine HCl
(Antihistamine/Cough Suppressant
Phenylephrine HCl
(Nasal Decongestant)
Cough Relief
Runny, Stuffy Nose
Itchy Throat
For Ages 6 to under 12
Compares to Children's
Triaminic® Night Time
Cold & Cough.
GRAPE
FLAVOR
4 FL. OZ.
(118 mL)
NDC 45014-154-04
Ask A Doctor Before Use If the Child Has
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts, or as occurs with asthma
- a breathing problem such as chronic bronchitis
Structured Label Content
Uses
- temporarily relieves
- sneezing
- itchy nose or throat
- runny nose
- itchy, watery eyes due to hay fever
- nasal and sinus congestion
- cough due to minor throat and bronchial irritation as may occur with a cold
Section 42229-5 (42229-5)
NITE TIME COLD & COUGH
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if the child is taking sedatives or tranquilizers
Section 55105-1 (55105-1)
|
Active ingredients (in each 5 mL, 1 teaspoonful) |
Purposes |
|---|---|
| Diphenhydramine HCl 6.25 mg | Antihistamine/Cough suppressant |
| Phenylephrine HCl 2.5 mg | Nasal decongestant |
Directions
- may be given every 4 hours. Do not give more than 6 doses in 24 hours unless directed by a doctor.
- to find right dose, use rotating bottle label to dose by weight; otherwise, use chart below to dose by age.
- specifically designed for use with enclosed dosing spoon. Use only enclosed dosing spoon to dose this product. Do not use any other dosing device.
| children under 6 years of age | do not use |
| children 6 to under 12 years of age | 1-2 tsp. (5-10 mL) |
Do Not Use (Do not use)
- in a child under 6 years of age
- in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if the child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
- with any other product containing diphenhydramine, even one used on skin
- for the purpose of making your child sleepy
Questions?
877-434-2036
Other Information (Other information)
- each teaspoonful contains: sodium 4 mg
- store at controlled room temperature 20°-25°C (68°-77°F)
Inactive Ingredients (Inactive ingredients)
artificial flavor, benzoic acid, citric acid, disodium edetate, FD&C blue no. 1, FD&C red no. 40, propylene glycol, purified water, sodium citrate, sodium saccharin, sorbitol.
When Using This Product (When using this product)
- do not exceed recommended dosage
- marked drowsiness may occur
- sedatives and tranquilizers may increase drowsiness
- excitability may occur, especially in children
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- nervousness, dizziness, or sleeplessness occurs
- symptoms do not improve within 7 days or occur with a fever
- cough persists for more than 7 days, comes back, or occurs with a fever, rash, or persistent headache. These could be signs of a serious condition.
Principal Display Panel 118 Ml Carton (PRINCIPAL DISPLAY PANEL - 118 mL Carton)
CHILDREN'S
ACCUDIAL
ROTATING DOSING LABEL
Nite Time
Cold & Cough
ACCURATE DOSING
BY WEIGHT
Diphenhydramine HCl
(Antihistamine/Cough Suppressant
Phenylephrine HCl
(Nasal Decongestant)
Cough Relief
Runny, Stuffy Nose
Itchy Throat
For Ages 6 to under 12
Compares to Children's
Triaminic® Night Time
Cold & Cough.
GRAPE
FLAVOR
4 FL. OZ.
(118 mL)
NDC 45014-154-04
Ask A Doctor Before Use If the Child Has (Ask a doctor before use if the child has)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts, or as occurs with asthma
- a breathing problem such as chronic bronchitis
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:54:58.292134 · Updated: 2026-03-14T22:52:13.708968