These Highlights Do Not Include All The Information Needed To Use Mifepristone Safely And Effectively. See Full Prescribing Information For Mifepristone.
63c068cd-9370-460f-829b-70e8c7ebe91e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.5 ) 11/2019
Indications and Usage
Mifepristone is a cortisol receptor blocker indicated to control hyperglycemia secondary to hypercortisolism in adult patients with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or are not candidates for surgery. LIMITATIONS OF USE: Mifepristone should not be used in the treatment of patients with type 2 diabetes unless it is secondary to Cushing's syndrome.
Dosage and Administration
Obtain a negative pregnancy test in females of reproductive potential prior to initiating treatment with mifepristone or if treatment is interrupted for more than 14 days. ( 2.1 ) Administer once daily orally with a meal ( 2.2 ). The recommended starting dose is 300 mg once daily ( 2.2 ). Based on clinical response and tolerability, the dose may be increased in 300 mg increments to a maximum of 1200 mg once daily. Do not exceed 20 mg/kg per day ( 2.2 ). Renal impairment: do not exceed 600 mg once daily ( 2.3 ). Mild-to-moderate hepatic impairment: do not exceed 600 mg once daily. Do not use in severe hepatic impairment ( 2.4 ). Concomitant administration with strong CYP3A inhibitors: Do not exceed 900 mg once daily ( 2.5 ).
Warnings and Precautions
Adrenal insufficiency : Patients should be closely monitored for signs and symptoms of adrenal insufficiency ( 5.1 ). Hypokalemia : Hypokalemia should be corrected prior to treatment and monitored for during treatment ( 5.2 ). Vaginal bleeding and endometrial changes : Women may experience endometrial thickening or unexpected vaginal bleeding. Use with caution if patient also has a hemorrhagic disorder or is on anti-coagulant therapy ( 5.3 ). QT interval prolongation : Avoid use with QT interval-prolonging drugs, or in patients with potassium channel variants resulting in a long QT interval ( 5.4 ). Use of Strong CYP3A Inhibitors: Concomitant use can increase mifepristone plasma levels. Use only when necessary and limit mifepristone dose to 900 mg ( 5.6 ).
Contraindications
Mifepristone is contraindicated in: Pregnancy [See Dosage and Administration ( 2.1 ), Use in Specific Populations ( 8.1 , 8.3 )] Patients taking drugs metabolized by CYP3A such as simvastatin, lovastatin, and CYP3A substrates with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, due to an increased risk of adverse events. [See Drug Interactions ( 7.1 ) and Clinical Pharmacology ( 12.3 )] Patients receiving systemic corticosteroids for lifesaving purposes (e.g., immunosuppression after organ transplantation) because mifepristone antagonizes the effect of glucocorticoids. Women with a history of unexplained vaginal bleeding or with endometrial hyperplasia with atypia or endometrial carcinoma. Patients with known hypersensitivity to mifepristone or to any of the product components.
Adverse Reactions
Most common adverse reactions in Cushing's syndrome (≥ 20%): nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, endometrial hypertrophy ( 6 ). To report suspected adverse reactions, contact Corcept Therapeutics at 1-855-844-3270 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Based on the long terminal half-life of mifepristone after reaching steady state, at least 2 weeks should elapse after cessation of mifepristone before initiating or increasing the dose of any interacting concomitant medication.
Storage and Handling
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03). Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
How Supplied
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03). Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
Medication Information
Warnings and Precautions
Adrenal insufficiency : Patients should be closely monitored for signs and symptoms of adrenal insufficiency ( 5.1 ). Hypokalemia : Hypokalemia should be corrected prior to treatment and monitored for during treatment ( 5.2 ). Vaginal bleeding and endometrial changes : Women may experience endometrial thickening or unexpected vaginal bleeding. Use with caution if patient also has a hemorrhagic disorder or is on anti-coagulant therapy ( 5.3 ). QT interval prolongation : Avoid use with QT interval-prolonging drugs, or in patients with potassium channel variants resulting in a long QT interval ( 5.4 ). Use of Strong CYP3A Inhibitors: Concomitant use can increase mifepristone plasma levels. Use only when necessary and limit mifepristone dose to 900 mg ( 5.6 ).
Indications and Usage
Mifepristone is a cortisol receptor blocker indicated to control hyperglycemia secondary to hypercortisolism in adult patients with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or are not candidates for surgery. LIMITATIONS OF USE: Mifepristone should not be used in the treatment of patients with type 2 diabetes unless it is secondary to Cushing's syndrome.
Dosage and Administration
Obtain a negative pregnancy test in females of reproductive potential prior to initiating treatment with mifepristone or if treatment is interrupted for more than 14 days. ( 2.1 ) Administer once daily orally with a meal ( 2.2 ). The recommended starting dose is 300 mg once daily ( 2.2 ). Based on clinical response and tolerability, the dose may be increased in 300 mg increments to a maximum of 1200 mg once daily. Do not exceed 20 mg/kg per day ( 2.2 ). Renal impairment: do not exceed 600 mg once daily ( 2.3 ). Mild-to-moderate hepatic impairment: do not exceed 600 mg once daily. Do not use in severe hepatic impairment ( 2.4 ). Concomitant administration with strong CYP3A inhibitors: Do not exceed 900 mg once daily ( 2.5 ).
Contraindications
Mifepristone is contraindicated in: Pregnancy [See Dosage and Administration ( 2.1 ), Use in Specific Populations ( 8.1 , 8.3 )] Patients taking drugs metabolized by CYP3A such as simvastatin, lovastatin, and CYP3A substrates with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, due to an increased risk of adverse events. [See Drug Interactions ( 7.1 ) and Clinical Pharmacology ( 12.3 )] Patients receiving systemic corticosteroids for lifesaving purposes (e.g., immunosuppression after organ transplantation) because mifepristone antagonizes the effect of glucocorticoids. Women with a history of unexplained vaginal bleeding or with endometrial hyperplasia with atypia or endometrial carcinoma. Patients with known hypersensitivity to mifepristone or to any of the product components.
Adverse Reactions
Most common adverse reactions in Cushing's syndrome (≥ 20%): nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, endometrial hypertrophy ( 6 ). To report suspected adverse reactions, contact Corcept Therapeutics at 1-855-844-3270 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Based on the long terminal half-life of mifepristone after reaching steady state, at least 2 weeks should elapse after cessation of mifepristone before initiating or increasing the dose of any interacting concomitant medication.
Storage and Handling
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03). Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
How Supplied
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03). Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
Description
Dosage and Administration ( 2.5 ) 11/2019
Section 42229-5
Dosing and titration
The daily dose of mifepristone may be increased in 300 mg increments. The dose of mifepristone may be increased to a maximum of 1200 mg once daily but should not exceed 20 mg/kg per day. Increases in dose should not occur more frequently than once every 2-4 weeks. Decisions about dose increases should be based on a clinical assessment of tolerability and degree of improvement in Cushing's syndrome manifestations. Changes in glucose control, anti-diabetic medication requirements, insulin levels, and psychiatric symptoms may provide an early assessment of response (within 6 weeks) and may help guide early dose titration. Improvements in cushingoid appearance, acne, hirsutism, striae, and body weight occur over a longer period of time and, along with measures of glucose control, may be used to determine dose changes beyond the first 2 months of therapy. Careful and gradual titration of mifepristone accompanied by monitoring for recognized adverse reactions [See Warnings and Precautions (5.1) and (5.2)] may reduce the risk of severe adverse reactions. Dose reduction or even dose discontinuation may be needed in some clinical situations. If mifepristone treatment is interrupted, it should be reinitiated at the lowest dose (300 mg). If treatment was interrupted because of adverse reactions, the titration should aim for a dose lower than the one that resulted in treatment interruption.
Section 42231-1
|
Medication Guide
mifepristone tablets |
|
|
What is the most important information I should know about mifepristone?
|
|
|
What is mifepristone?
Mifepristone is a prescription medicine used to treat high blood sugar (hyperglycemia) caused by high cortisol levels in the blood (hypercortisolism) in adults with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or cannot have surgery. Mifepristone is not for people who have type 2 diabetes mellitus not caused by Cushing's syndrome. It is not known if mifepristone is safe and effective in children. |
|
Do not take mifepristone if you:
|
|
| Talk to your doctor before taking mifepristone if you have any of these conditions. | |
|
What should I tell my doctor before taking mifepristone?
Before taking mifepristone, tell your doctor if you:
|
|
|
Tell your doctor about all of the medicines you take, including prescription and nonprescription medicines, vitamins and herbal supplements. Using mifepristone with certain other medicines can affect each other. Using mifepristone with other medicines can cause serious side effects. Especially tell your doctor if you take:
|
|
| Ask your doctor or pharmacist for a list of these medicines if you are not sure. Know the medicines you take. Keep a list of them to show to your doctor and pharmacist. | |
How should I take mifepristone?
|
|
|
What should I avoid while taking mifepristone?
You should not drink grapefruit juice while you take mifepristone. Grapefruit juice may increase the amount of mifepristone in your blood and increase your chance of having side effects. |
|
|
What are the possible side effects of mifepristone?
Mifepristone can cause serious side effects including:
|
|
| The most common side effects of mifepristone include: | |
|
|
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of mifepristone. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store mifepristone?
Store mifepristone at room temperature, between 68°F to 77°F (20°C to 25°C). Keep mifepristone and all medicines out of the reach of children. |
|
|
General information about the safe and effective use of mifepristone
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use mifepristone for a condition for which it was not prescribed. Do not give mifepristone to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about mifepristone that is written for healthcare professionals. |
|
|
What are the ingredients in mifepristone?
Active ingredient: mifepristone Inactive ingredients: silicified microcrystalline cellulose, sodium starch glycolate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, hypromellose, titanium dioxide, triacetin, D&C yellow 10 aluminum lake, polysorbate 80, and FD&C yellow 6 aluminum lake. Manufactured for: Corcept Therapeutics Incorporated, Redwood City, CA 94065 ©2020 Corcept Therapeutics Incorporated. All rights reserved. M-00001 SEPTEMBER 2024 For more information, go to www.corcept.com or call 1-855-456-7596. |
This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised: 09/2024
Section 43683-2
| Dosage and Administration (2.5) | 11/2019 |
Section 51945-4
Principal Display Panel - for 280 Tablets Bottle Label (2000017691)
NDC 76346-654-03
280 Tablets
Rx only
mifepristone
300 mg tablets
Take tablets whole. Do not split, crush or chew.
ATTENTION PHARMACIST: Dispense attached Medication Guide to each patient.
10 Overdosage
There is no experience with overdosage of mifepristone.
11 Description
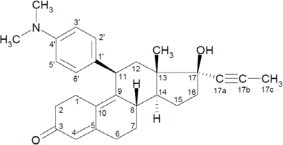
Mifepristone is a cortisol receptor blocker for oral administration. The chemical name of mifepristone is 11β-(4-dimethylaminophenyl)-17β-hydroxy-17α-(1-propynyl)-estra-4, 9-dien-3-one. The chemical formula is C29H35NO2; the molecular weight is 429.60, and the structural formula is:
Mifepristone demonstrates a pH-related solubility profile. The greatest solubility is achieved in acidic media (~ 25 mg/mL at pH 1.5) and solubility declines rapidly as the pH is increased. At pH values above 2.5 the solubility of mifepristone is less than 1 mg/mL.
Each mifepristone tablet for oral use contains 300 mg of mifepristone. The inactive ingredients of mifepristone tablets are silicified microcrystalline cellulose, sodium starch glycolate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, hypromellose, titanium dioxide, triacetin, D&C yellow 10 aluminum lake, polysorbate 80, and FD&C yellow 6 aluminum lake.
5.2 Hypokalemia
In a study of patients with Cushing's syndrome, hypokalemia was observed in 44% of subjects during treatment with mifepristone. Hypokalemia should be corrected prior to initiating mifepristone. During mifepristone administration, serum potassium should be measured 1 to 2 weeks after starting or increasing the dose of mifepristone and periodically thereafter. Hypokalemia can occur at any time during mifepristone treatment. Mifepristone-induced hypokalemia should be treated with intravenous or oral potassium supplementation based on event severity. If hypokalemia persists in spite of potassium supplementation, consider adding mineralocorticoid antagonists.
2.2 Adult Dosage
The recommended starting dose is 300 mg orally once daily. Mifepristone must be given as a single daily dose. Mifepristone should always be taken with a meal. Patients should swallow the tablet whole. Do not split, crush, or chew tablets.
8.4 Pediatric Use
Safety and effectiveness of mifepristone in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies with mifepristone did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger people.
7.3 Cyp3a Inducers
No medications that induce CYP3A have been studied when co-administered with mifepristone. Avoid co-administration of mifepristone and CYP3A inducers such as rifampin, rifabutin, rifapentin, phenobarbital, phenytoin, carbamazepine, and St. John's wort.
4 Contraindications
Mifepristone is contraindicated in:
- Pregnancy [See Dosage and Administration (2.1), Use in Specific Populations (8.1,8.3)]
- Patients taking drugs metabolized by CYP3A such as simvastatin, lovastatin, and CYP3A substrates with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, due to an increased risk of adverse events. [See Drug Interactions (7.1) and Clinical Pharmacology (12.3)]
- Patients receiving systemic corticosteroids for lifesaving purposes (e.g., immunosuppression after organ transplantation) because mifepristone antagonizes the effect of glucocorticoids.
- Women with a history of unexplained vaginal bleeding or with endometrial hyperplasia with atypia or endometrial carcinoma.
- Patients with known hypersensitivity to mifepristone or to any of the product components.
6 Adverse Reactions
Most common adverse reactions in Cushing's syndrome (≥ 20%): nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, endometrial hypertrophy (6).
To report suspected adverse reactions, contact Corcept Therapeutics at 1-855-844-3270 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Based on the long terminal half-life of mifepristone after reaching steady state, at least 2 weeks should elapse after cessation of mifepristone before initiating or increasing the dose of any interacting concomitant medication.
7.2 Cyp3a Inhibitors
Medications that inhibit CYP3A could increase plasma mifepristone concentrations and dose reduction of mifepristone may be required.
Ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir and fosamprenavir, clarithromycin, conivaptan, lopinavir /ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole may increase exposure to mifepristone. Caution should be used when strong CYP3A inhibitors are prescribed in combination with mifepristone. The benefit of concomitant use of these agents should be carefully weighed against the potential risks. The dose of mifepristone should be limited to 900 mg, and strong inhibitors of CYP3A should be used only when necessary. [See Dosage and Administration (2.4), Warnings & Precautions (5.6), and Clinical Pharmacology (12.3)]
8.6 Renal Impairment
The maximum dose should not exceed 600 mg per day in renally impaired patients. [See Clinical Pharmacology (12.3)]
12.2 Pharmacodynamics
Because mifepristone acts at the receptor level to block the effects of cortisol, its antagonistic actions affect the hypothalamic-pituitary-adrenal (HPA) axis in such a way as to further increase circulating cortisol levels while, at the same time, blocking their effects.
Mifepristone and the three active metabolites have greater affinity for the glucocorticoid receptor [100% (mifepristone), 61% (metabolite 1), 48% (metabolite 2), and 45% (metabolite 3)] than either dexamethasone (23%) or cortisol (9%).
8.7 Hepatic Impairment
In patients with mild to moderate hepatic impairment, the maximum dose should not exceed 600 mg per day. The pharmacokinetics of mifepristone in patients with severe hepatic impairment has not been studied, and mifepristone should not be used in these patients. [See Clinical Pharmacology (12.3)]
1 Indications and Usage
Mifepristone is a cortisol receptor blocker indicated to control hyperglycemia secondary to hypercortisolism in adult patients with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or are not candidates for surgery.
LIMITATIONS OF USE:
- Mifepristone should not be used in the treatment of patients with type 2 diabetes unless it is secondary to Cushing's syndrome.
14.1 Cushing's Syndrome
An uncontrolled, open-label, 24-week, multicenter clinical study was conducted to evaluate the safety and efficacy of mifepristone in the treatment of endogenous Cushing's syndrome. The study enrolled 50 subjects with clinical and biochemical evidence of hypercortisolemia despite prior surgical treatment and radiotherapy. The reasons for medical treatment were failed surgery, recurrence of disease, and poor medical candidate for surgery. Forty-three patients (86%) had Cushing's disease, four patients (8%) had ectopic ACTH secretion, and three (6%) had adrenal carcinoma. Baseline characteristics included: mean age of 45 years (range 26 to 71), mean BMI of 36 kg/m2 (range 24 to 66), mean weight 100 kg (range 61 to 199), and mean waist circumference was 119 cm (range 89 to 178); 70% were female; 84% were white and 16% were black or African American. Baseline mean urinary free cortisol level was 365 μg per 24 hr.
Patients belonged to one of two cohorts: a “diabetes” cohort (29 patients, 26 with type 2 diabetes and 3 with glucose intolerance), and a “hypertension” cohort (21 patients). Efficacy was evaluated separately in the two cohorts. Mifepristone treatment was started in all patients at a dose of 300 mg once a day. The study protocol allowed an increase in dose to 600 mg after 2 weeks, and then by additional 300 mg increments every 4 weeks to a maximum of 900 mg per day for patients <60 kg, or 1200 mg per day for patients >60 kg, based on clinical tolerance and clinical response.
12.1 Mechanism of Action
Mifepristone is a selective antagonist of the progesterone receptor at low doses and blocks the glucocorticoid receptor (GR-II) at higher doses. Mifepristone has high affinity for the GR-II receptor but little affinity for the GR-I (MR, mineralocorticoid) receptor. In addition, mifepristone appears to have little or no affinity for estrogen, muscarinic, histaminic, or monoamine receptors.
5.1 Adrenal Insufficiency
Patients receiving mifepristone may experience adrenal insufficiency. Because serum cortisol levels remain elevated and may even increase during treatment with mifepristone, serum cortisol levels do not provide an accurate assessment of hypoadrenalism in patients receiving mifepristone. Patients should be closely monitored for signs and symptoms of adrenal insufficiency, including weakness, nausea, increased fatigue, hypotension, and hypoglycemia. If adrenal insufficiency is suspected, discontinue treatment with mifepristone immediately and administer glucocorticoids without delay. High doses of supplemental glucocorticoids may be needed to overcome the glucocorticoid receptor blockade produced by mifepristone. Factors considered in deciding on the duration of glucocorticoid treatment should include the long half-life of mifepristone (85 hours).
Treatment with mifepristone at a lower dose can be resumed after resolution of adrenal insufficiency. Patients should also be evaluated for precipitating causes of hypoadrenalism (infection, trauma, etc.).
5.7 Pneumocystis Jiroveci
Patients with endogenous Cushing's syndrome are at risk for opportunistic infections such as Pneumocystis jiroveci pneumonia during mifepristone treatment. Patients may present with respiratory distress shortly after initiation of mifepristone. Appropriate diagnostic tests should be undertaken and treatment for Pneumocystis jiroveci should be considered.
5 Warnings and Precautions
- Adrenal insufficiency: Patients should be closely monitored for signs and symptoms of adrenal insufficiency (5.1).
- Hypokalemia: Hypokalemia should be corrected prior to treatment and monitored for during treatment (5.2).
- Vaginal bleeding and endometrial changes: Women may experience endometrial thickening or unexpected vaginal bleeding. Use with caution if patient also has a hemorrhagic disorder or is on anti-coagulant therapy (5.3).
- QT interval prolongation: Avoid use with QT interval-prolonging drugs, or in patients with potassium channel variants resulting in a long QT interval (5.4).
- Use of Strong CYP3A Inhibitors: Concomitant use can increase mifepristone plasma levels. Use only when necessary and limit mifepristone dose to 900 mg (5.6).
2 Dosage and Administration
- Obtain a negative pregnancy test in females of reproductive potential prior to initiating treatment with mifepristone or if treatment is interrupted for more than 14 days. (2.1)
- Administer once daily orally with a meal (2.2).
- The recommended starting dose is 300 mg once daily (2.2).
- Based on clinical response and tolerability, the dose may be increased in 300 mg increments to a maximum of 1200 mg once daily. Do not exceed 20 mg/kg per day (2.2).
- Renal impairment: do not exceed 600 mg once daily (2.3).
- Mild-to-moderate hepatic impairment: do not exceed 600 mg once daily. Do not use in severe hepatic impairment (2.4).
- Concomitant administration with strong CYP3A inhibitors: Do not exceed 900 mg once daily (2.5).
3 Dosage Forms and Strengths
Tablets: 300 mg
Oval shaped, light yellow to yellow tablets debossed with “Corcept” on one side and “300” on the other side. The tablets are not scored.
5.4 Qt Interval Prolongation
Mifepristone and its metabolites block IKr. Mifepristone prolongs the QTc interval in a dose-related manner. There is little or no experience with high exposure, concomitant dosing with other QT-prolonging drugs, or potassium channel variants resulting in a long QT interval. [See Warnings & Precautions (5.6)] To minimize risk, the lowest effective dose should always be used.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post approval use of mifepristone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
-Angioedema
2.3 Dosing in Renal Impairment
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
Safety data on the use of mifepristone are available from 50 patients with Cushing's syndrome enrolled in an uncontrolled, open-label, multi-center trial (Study 400). Forty-three patients had Cushing's disease and all except one had previously undergone pituitary surgery. Four patients had ectopic ACTH secretion, and three had adrenal carcinoma. Patients were treated for up to 24 weeks. A dose of 300 mg per day was administered for the initial 14 days; thereafter, the dose could be escalated in increments of 300 mg per day based on assessments of tolerability and clinical response. Doses were escalated up to 900 mg per day for patients <60 kg, or 1200 mg per day for patients >60 kg.
The most frequently reported adverse reactions (reported in ≥20% of patients, regardless of relationship to mifepristone) were nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, and endometrial hypertrophy. Drug-related adverse events resulted in dose interruption or reduction in study drug in 40% of patients.
The adverse reactions that occurred in ≥10% of the Cushing's syndrome patients receiving mifepristone, regardless of relationship to mifepristone, are shown in Table 2.
| Body System/Adverse Reaction |
Percent (%) of Patients Reporting Event
(n = 50) |
|---|---|
|
*The denominator was 26 females who had baseline and end-of-trial transvaginal ultrasound |
|
| Gastrointestinal disorders | |
| Nausea | 48 |
| Vomiting | 26 |
| Dry mouth | 18 |
| Diarrhea | 12 |
| Constipation | 10 |
| General disorders and administration/site conditions | |
| Fatigue | 48 |
| Edema peripheral | 26 |
| Pain | 14 |
| Nervous system disorders | |
| Headache | 44 |
| Dizziness | 22 |
| Somnolence | 10 |
| Musculoskeletal and connective tissue disorders | |
| Arthralgia | 30 |
| Back pain | 16 |
| Myalgia | 14 |
| Pain in extremity | 12 |
| Investigations | |
| Blood potassium decreased | 34 |
| Thyroid function test abnormal | 18 |
| Infections and infestations | |
| Sinusitis | 14 |
| Nasopharyngitis | 12 |
| Metabolism and nutrition disorders | |
| Decreased appetite | 20 |
| Anorexia | 10 |
| Vascular disorders | |
| Hypertension | 24 |
| Reproductive system and breast disorders | |
| Endometrial hypertrophy | 38* |
| Respiratory, thoracic, and mediastinal disorders | |
| Dyspnea | 16 |
| Psychiatric disorders | |
| Anxiety | 10 |
7.1 Drugs Metabolized By Cyp3a
Because mifepristone is an inhibitor of CYP3A, concurrent use of mifepristone with a drug whose metabolism is largely or solely mediated by CYP3A is likely to result in increased plasma concentrations of the drug. Discontinuation or dose reduction of such medications may be necessary with mifepristone co-administration.
Mifepristone increased the exposure to simvastatin and simvastatin acid significantly in healthy subjects. Concomitant use of simvastatin or lovastatin is contraindicated because of the increased risk of myopathy and rhabdomyolysis. [See Contraindications (4.2), Clinical Pharmacology 12.3]
The exposure of other substrates of CYP3A with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, may be increased by concomitant administration with mifepristone. Therefore, the concomitant use of such CYP3A substrates with mifepristone is contraindicated. [See Contraindications (4.2)]
Other drugs with similar high first pass metabolism in which CYP3A is the primary route of metabolism should be used with extreme caution if co-administered with mifepristone. The lowest possible dose and/or a decreased frequency of dosing must be used with therapeutic drug monitoring when possible. Use of alternative drugs without these metabolic characteristics is advised when possible with concomitant mifepristone.
If drugs that undergo low first pass metabolism by CYP3A or drugs in which CYP3A is not the major metabolic route are co-administered with mifepristone, use the lowest dose of concomitant medication necessary, with appropriate monitoring and follow-up. [See Clinical Pharmacology (12.3)]
7.5 Drugs Metabolized By Cyp2b6
Mifepristone is an inhibitor of CYP2B6 and may cause significant increases in exposure of drugs that are metabolized by CYP2B6 such as bupropion and efavirenz. Since no study has been conducted to evaluate the effect of mifepristone on substrates of CYP2B6, the concomitant use of bupropion and efavirenz should be undertaken with caution. [See Clinical Pharmacology (12.3)]
2.4 Dosing in Hepatic Impairment
17 Patient Counseling Information
As a part of patient counseling, doctors must review the mifepristone Medication Guide with every patient.
Warning: Termination of Pregnancy
Mifepristone is a potent antagonist of progesterone and cortisol via the progesterone and glucocorticoid (GR-II) receptors, respectively. The antiprogestational effects will result in the termination of pregnancy. Pregnancy must therefore be excluded before the initiation of treatment with mifepristone and prevented during treatment and for one month after stopping treatment by the use of a non-hormonal medically acceptable method of contraception unless the patient has had a surgical sterilization, in which case no additional contraception is needed. Pregnancy must also be excluded if treatment is interrupted for more than 14 days in females of reproductive potential.
5.6 Use of Strong Cyp3a Inhibitors
Mifepristone should be used with caution in patients taking ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir, fosamprenavir, clarithromycin, conivaptan, lopinavir/ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole, as these could increase the concentration of mifepristone in the blood. The benefit of concomitant use of these agents should be carefully weighed against the potential risks. Mifepristone should be used in combination with strong CYP3A inhibitors only when necessary, and in such cases the dose should be limited to 900 mg per day. [See Warnings & Precautions (5.4), Drug Interactions (7.2), and Clinical Pharmacology (12.3)]
7.6 Use of Hormonal Contraceptives
Mifepristone is a progesterone-receptor antagonist and will interfere with the effectiveness of hormonal contraceptives. Therefore, non-hormonal contraceptive methods should be used. [See Use In Specific Populations (8.3)]
7.4 Drugs Metabolized By Cyp2c8/2c9
Because mifepristone is an inhibitor of CYP2C8/2C9, concurrent use of mifepristone with a drug whose metabolism is largely or solely mediated by CYP2C8/2C9 is likely to result in increased plasma concentrations of the drug.
Mifepristone significantly increased exposure of fluvastatin, a typical CYP2C8/2C9 substrate, in healthy subjects. When given concomitantly with mifepristone, drugs that are substrates of CYP2C8/2C9 (including non-steroidal anti-inflammatory drugs, warfarin, and repaglinide) should be used at the smallest recommended doses, and patients should be closely monitored for adverse effects. [See Clinical Pharmacology (12.3)]
16 How Supplied/storage and Handling
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03).
Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
17.1 Importance of Preventing Pregnancy
- Advise patients that mifepristone will cause termination of pregnancy. Mifepristone is contraindicated in pregnant patients.
- Mifepristone reduces the effectiveness of hormonal contraceptives. Counsel females of reproductive potential regarding pregnancy prevention and planning with a non-hormonal contraceptive prior to use of mifepristone and up to one month after the end of treatment.
- Instruct patients to contact their physician immediately if they suspect or confirm they are pregnant.
5.8 Potential Effects of Hypercortisolemia
Mifepristone does not reduce serum cortisol levels. Elevated cortisol levels may activate mineralocorticoid receptors which are also expressed in cardiac tissues. Caution should be used in patients with underlying heart conditions including heart failure and coronary vascular disease.
5.3 Vaginal Bleeding and Endometrial Changes
Being an antagonist of the progesterone receptor, mifepristone promotes unopposed endometrial proliferation that may result in endometrium thickening, cystic dilatation of endometrial glands, and vaginal bleeding. Mifepristone should be used with caution in women who have hemorrhagic disorders or are receiving concurrent anticoagulant therapy. Women who experience vaginal bleeding during mifepristone treatment should be referred to a gynecologist for further evaluation.
2.5 Concomitant Administration With Cyp3a Inhibitors
Ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir and fosamprenavir, clarithromycin, conivaptan, lopinavir/ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole may increase exposure to mifepristone. Mifepristone should be used in combination with strong CYP3A inhibitors only when necessary. [See Warnings and Precautions (5.6), Drug Interactions (7.2)]
Administration of mifepristone to patients already being treated with strong CYP3A inhibitors:
- Start at a dose of 300 mg. If clinically indicated, titrate to a maximum of 900 mg.
Administration of strong CYP3A inhibitors to patients already being treated with mifepristone:
- Adjust the dose of mifepristone according to Table 1.
| Current dose of mifepristone |
Adjustment to dose of mifepristone
if adding a strong CYP3A inhibitor |
| 300 mg | No change |
| 600 mg | Reduce dose to 300 mg. If clinically indicated, titrate to a maximum of 600 mg |
| 900 mg | Reduce dose to 600 mg. If clinically indicated, titrate to a maximum of 900 mg |
| 1200 mg | Reduce dose to 900 mg |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Mifepristone was evaluated for carcinogenicity potential in rats and mice. Rats were dosed for up to two years at doses of 5, 25, and 125 mg/kg of mifepristone. The high dose was the maximum tolerated dose, but exposure at all doses was below exposure at the maximum clinical dose based on AUC comparison. Female rats had a statistically significant increase in follicular cell adenomas/carcinomas and liver adenomas. It is plausible that these tumors are due to drug-induced enzyme metabolism, a mechanism not considered clinically relevant, but studies confirming this mechanism were not conducted with mifepristone. Mice were also tested for up to 2 years at mifepristone doses up to the maximum tolerated dose of 125 mg/kg, which provided exposure below the maximum clinical dose based on AUC. No drug-related tumors were seen in mice.
Mifepristone was not genotoxic in a battery of bacterial, yeast, and mammalian in vitro assays, and an in vivo micronucleus study in mice.
The pharmacological activity of mifepristone disrupts the estrus cycle of adult rats at a dose of 0.3 mg/kg (less than human exposure at the maximum clinical dose, based on body surface area). However, following withdrawal of treatment and subsequent resumption of the estrus cycle, there was no effect on reproductive function when mated.
A single subcutaneous dose of mifepristone (up to 100 mg/kg) to rats on the first day after birth did not adversely affect future reproductive function in males or females, although the onset of puberty was slightly premature in dosed females. Repeated doses of mifepristone (1 mg every other day) to neonatal rats resulted in potentially adverse fertility effects, including oviduct and ovary malformations in females, delayed male puberty, deficient male sexual behavior, reduced testicular size, and lowered ejaculation frequency.
2.1 Testing Prior to and During Mifepristone Administration
5.5 Exacerbation/deterioration of Conditions Treated With Corticosteroids
Use of mifepristone in patients who receive corticosteroids for other conditions (e.g., autoimmune disorders) may lead to exacerbation or deterioration of such conditions, as mifepristone antagonizes the desired effects of glucocorticoid in these clinical settings. For medical conditions in which chronic corticosteroid therapy is lifesaving (e.g., immunosuppression in organ transplantation), mifepristone is contraindicated. [See Contraindications (4.3)]
Structured Label Content
Section 42229-5 (42229-5)
Dosing and titration
The daily dose of mifepristone may be increased in 300 mg increments. The dose of mifepristone may be increased to a maximum of 1200 mg once daily but should not exceed 20 mg/kg per day. Increases in dose should not occur more frequently than once every 2-4 weeks. Decisions about dose increases should be based on a clinical assessment of tolerability and degree of improvement in Cushing's syndrome manifestations. Changes in glucose control, anti-diabetic medication requirements, insulin levels, and psychiatric symptoms may provide an early assessment of response (within 6 weeks) and may help guide early dose titration. Improvements in cushingoid appearance, acne, hirsutism, striae, and body weight occur over a longer period of time and, along with measures of glucose control, may be used to determine dose changes beyond the first 2 months of therapy. Careful and gradual titration of mifepristone accompanied by monitoring for recognized adverse reactions [See Warnings and Precautions (5.1) and (5.2)] may reduce the risk of severe adverse reactions. Dose reduction or even dose discontinuation may be needed in some clinical situations. If mifepristone treatment is interrupted, it should be reinitiated at the lowest dose (300 mg). If treatment was interrupted because of adverse reactions, the titration should aim for a dose lower than the one that resulted in treatment interruption.
Section 42231-1 (42231-1)
|
Medication Guide
mifepristone tablets |
|
|
What is the most important information I should know about mifepristone?
|
|
|
What is mifepristone?
Mifepristone is a prescription medicine used to treat high blood sugar (hyperglycemia) caused by high cortisol levels in the blood (hypercortisolism) in adults with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or cannot have surgery. Mifepristone is not for people who have type 2 diabetes mellitus not caused by Cushing's syndrome. It is not known if mifepristone is safe and effective in children. |
|
Do not take mifepristone if you:
|
|
| Talk to your doctor before taking mifepristone if you have any of these conditions. | |
|
What should I tell my doctor before taking mifepristone?
Before taking mifepristone, tell your doctor if you:
|
|
|
Tell your doctor about all of the medicines you take, including prescription and nonprescription medicines, vitamins and herbal supplements. Using mifepristone with certain other medicines can affect each other. Using mifepristone with other medicines can cause serious side effects. Especially tell your doctor if you take:
|
|
| Ask your doctor or pharmacist for a list of these medicines if you are not sure. Know the medicines you take. Keep a list of them to show to your doctor and pharmacist. | |
How should I take mifepristone?
|
|
|
What should I avoid while taking mifepristone?
You should not drink grapefruit juice while you take mifepristone. Grapefruit juice may increase the amount of mifepristone in your blood and increase your chance of having side effects. |
|
|
What are the possible side effects of mifepristone?
Mifepristone can cause serious side effects including:
|
|
| The most common side effects of mifepristone include: | |
|
|
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of mifepristone. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store mifepristone?
Store mifepristone at room temperature, between 68°F to 77°F (20°C to 25°C). Keep mifepristone and all medicines out of the reach of children. |
|
|
General information about the safe and effective use of mifepristone
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use mifepristone for a condition for which it was not prescribed. Do not give mifepristone to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about mifepristone that is written for healthcare professionals. |
|
|
What are the ingredients in mifepristone?
Active ingredient: mifepristone Inactive ingredients: silicified microcrystalline cellulose, sodium starch glycolate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, hypromellose, titanium dioxide, triacetin, D&C yellow 10 aluminum lake, polysorbate 80, and FD&C yellow 6 aluminum lake. Manufactured for: Corcept Therapeutics Incorporated, Redwood City, CA 94065 ©2020 Corcept Therapeutics Incorporated. All rights reserved. M-00001 SEPTEMBER 2024 For more information, go to www.corcept.com or call 1-855-456-7596. |
This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised: 09/2024
Section 43683-2 (43683-2)
| Dosage and Administration (2.5) | 11/2019 |
Section 51945-4 (51945-4)
Principal Display Panel - for 280 Tablets Bottle Label (2000017691)
NDC 76346-654-03
280 Tablets
Rx only
mifepristone
300 mg tablets
Take tablets whole. Do not split, crush or chew.
ATTENTION PHARMACIST: Dispense attached Medication Guide to each patient.
10 Overdosage (10 OVERDOSAGE)
There is no experience with overdosage of mifepristone.
11 Description (11 DESCRIPTION)
Mifepristone is a cortisol receptor blocker for oral administration. The chemical name of mifepristone is 11β-(4-dimethylaminophenyl)-17β-hydroxy-17α-(1-propynyl)-estra-4, 9-dien-3-one. The chemical formula is C29H35NO2; the molecular weight is 429.60, and the structural formula is:
Mifepristone demonstrates a pH-related solubility profile. The greatest solubility is achieved in acidic media (~ 25 mg/mL at pH 1.5) and solubility declines rapidly as the pH is increased. At pH values above 2.5 the solubility of mifepristone is less than 1 mg/mL.
Each mifepristone tablet for oral use contains 300 mg of mifepristone. The inactive ingredients of mifepristone tablets are silicified microcrystalline cellulose, sodium starch glycolate, hydroxypropylcellulose, sodium lauryl sulfate, magnesium stearate, hypromellose, titanium dioxide, triacetin, D&C yellow 10 aluminum lake, polysorbate 80, and FD&C yellow 6 aluminum lake.
5.2 Hypokalemia
In a study of patients with Cushing's syndrome, hypokalemia was observed in 44% of subjects during treatment with mifepristone. Hypokalemia should be corrected prior to initiating mifepristone. During mifepristone administration, serum potassium should be measured 1 to 2 weeks after starting or increasing the dose of mifepristone and periodically thereafter. Hypokalemia can occur at any time during mifepristone treatment. Mifepristone-induced hypokalemia should be treated with intravenous or oral potassium supplementation based on event severity. If hypokalemia persists in spite of potassium supplementation, consider adding mineralocorticoid antagonists.
2.2 Adult Dosage
The recommended starting dose is 300 mg orally once daily. Mifepristone must be given as a single daily dose. Mifepristone should always be taken with a meal. Patients should swallow the tablet whole. Do not split, crush, or chew tablets.
8.4 Pediatric Use
Safety and effectiveness of mifepristone in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies with mifepristone did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger people.
7.3 Cyp3a Inducers (7.3 CYP3A Inducers)
No medications that induce CYP3A have been studied when co-administered with mifepristone. Avoid co-administration of mifepristone and CYP3A inducers such as rifampin, rifabutin, rifapentin, phenobarbital, phenytoin, carbamazepine, and St. John's wort.
4 Contraindications (4 CONTRAINDICATIONS)
Mifepristone is contraindicated in:
- Pregnancy [See Dosage and Administration (2.1), Use in Specific Populations (8.1,8.3)]
- Patients taking drugs metabolized by CYP3A such as simvastatin, lovastatin, and CYP3A substrates with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, due to an increased risk of adverse events. [See Drug Interactions (7.1) and Clinical Pharmacology (12.3)]
- Patients receiving systemic corticosteroids for lifesaving purposes (e.g., immunosuppression after organ transplantation) because mifepristone antagonizes the effect of glucocorticoids.
- Women with a history of unexplained vaginal bleeding or with endometrial hyperplasia with atypia or endometrial carcinoma.
- Patients with known hypersensitivity to mifepristone or to any of the product components.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions in Cushing's syndrome (≥ 20%): nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, endometrial hypertrophy (6).
To report suspected adverse reactions, contact Corcept Therapeutics at 1-855-844-3270 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Based on the long terminal half-life of mifepristone after reaching steady state, at least 2 weeks should elapse after cessation of mifepristone before initiating or increasing the dose of any interacting concomitant medication.
7.2 Cyp3a Inhibitors (7.2 CYP3A Inhibitors)
Medications that inhibit CYP3A could increase plasma mifepristone concentrations and dose reduction of mifepristone may be required.
Ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir and fosamprenavir, clarithromycin, conivaptan, lopinavir /ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole may increase exposure to mifepristone. Caution should be used when strong CYP3A inhibitors are prescribed in combination with mifepristone. The benefit of concomitant use of these agents should be carefully weighed against the potential risks. The dose of mifepristone should be limited to 900 mg, and strong inhibitors of CYP3A should be used only when necessary. [See Dosage and Administration (2.4), Warnings & Precautions (5.6), and Clinical Pharmacology (12.3)]
8.6 Renal Impairment
The maximum dose should not exceed 600 mg per day in renally impaired patients. [See Clinical Pharmacology (12.3)]
12.2 Pharmacodynamics
Because mifepristone acts at the receptor level to block the effects of cortisol, its antagonistic actions affect the hypothalamic-pituitary-adrenal (HPA) axis in such a way as to further increase circulating cortisol levels while, at the same time, blocking their effects.
Mifepristone and the three active metabolites have greater affinity for the glucocorticoid receptor [100% (mifepristone), 61% (metabolite 1), 48% (metabolite 2), and 45% (metabolite 3)] than either dexamethasone (23%) or cortisol (9%).
8.7 Hepatic Impairment
In patients with mild to moderate hepatic impairment, the maximum dose should not exceed 600 mg per day. The pharmacokinetics of mifepristone in patients with severe hepatic impairment has not been studied, and mifepristone should not be used in these patients. [See Clinical Pharmacology (12.3)]
1 Indications and Usage (1 INDICATIONS AND USAGE)
Mifepristone is a cortisol receptor blocker indicated to control hyperglycemia secondary to hypercortisolism in adult patients with endogenous Cushing's syndrome who have type 2 diabetes mellitus or glucose intolerance and have failed surgery or are not candidates for surgery.
LIMITATIONS OF USE:
- Mifepristone should not be used in the treatment of patients with type 2 diabetes unless it is secondary to Cushing's syndrome.
14.1 Cushing's Syndrome
An uncontrolled, open-label, 24-week, multicenter clinical study was conducted to evaluate the safety and efficacy of mifepristone in the treatment of endogenous Cushing's syndrome. The study enrolled 50 subjects with clinical and biochemical evidence of hypercortisolemia despite prior surgical treatment and radiotherapy. The reasons for medical treatment were failed surgery, recurrence of disease, and poor medical candidate for surgery. Forty-three patients (86%) had Cushing's disease, four patients (8%) had ectopic ACTH secretion, and three (6%) had adrenal carcinoma. Baseline characteristics included: mean age of 45 years (range 26 to 71), mean BMI of 36 kg/m2 (range 24 to 66), mean weight 100 kg (range 61 to 199), and mean waist circumference was 119 cm (range 89 to 178); 70% were female; 84% were white and 16% were black or African American. Baseline mean urinary free cortisol level was 365 μg per 24 hr.
Patients belonged to one of two cohorts: a “diabetes” cohort (29 patients, 26 with type 2 diabetes and 3 with glucose intolerance), and a “hypertension” cohort (21 patients). Efficacy was evaluated separately in the two cohorts. Mifepristone treatment was started in all patients at a dose of 300 mg once a day. The study protocol allowed an increase in dose to 600 mg after 2 weeks, and then by additional 300 mg increments every 4 weeks to a maximum of 900 mg per day for patients <60 kg, or 1200 mg per day for patients >60 kg, based on clinical tolerance and clinical response.
12.1 Mechanism of Action
Mifepristone is a selective antagonist of the progesterone receptor at low doses and blocks the glucocorticoid receptor (GR-II) at higher doses. Mifepristone has high affinity for the GR-II receptor but little affinity for the GR-I (MR, mineralocorticoid) receptor. In addition, mifepristone appears to have little or no affinity for estrogen, muscarinic, histaminic, or monoamine receptors.
5.1 Adrenal Insufficiency
Patients receiving mifepristone may experience adrenal insufficiency. Because serum cortisol levels remain elevated and may even increase during treatment with mifepristone, serum cortisol levels do not provide an accurate assessment of hypoadrenalism in patients receiving mifepristone. Patients should be closely monitored for signs and symptoms of adrenal insufficiency, including weakness, nausea, increased fatigue, hypotension, and hypoglycemia. If adrenal insufficiency is suspected, discontinue treatment with mifepristone immediately and administer glucocorticoids without delay. High doses of supplemental glucocorticoids may be needed to overcome the glucocorticoid receptor blockade produced by mifepristone. Factors considered in deciding on the duration of glucocorticoid treatment should include the long half-life of mifepristone (85 hours).
Treatment with mifepristone at a lower dose can be resumed after resolution of adrenal insufficiency. Patients should also be evaluated for precipitating causes of hypoadrenalism (infection, trauma, etc.).
5.7 Pneumocystis Jiroveci (5.7 Pneumocystis jiroveci)
Patients with endogenous Cushing's syndrome are at risk for opportunistic infections such as Pneumocystis jiroveci pneumonia during mifepristone treatment. Patients may present with respiratory distress shortly after initiation of mifepristone. Appropriate diagnostic tests should be undertaken and treatment for Pneumocystis jiroveci should be considered.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Adrenal insufficiency: Patients should be closely monitored for signs and symptoms of adrenal insufficiency (5.1).
- Hypokalemia: Hypokalemia should be corrected prior to treatment and monitored for during treatment (5.2).
- Vaginal bleeding and endometrial changes: Women may experience endometrial thickening or unexpected vaginal bleeding. Use with caution if patient also has a hemorrhagic disorder or is on anti-coagulant therapy (5.3).
- QT interval prolongation: Avoid use with QT interval-prolonging drugs, or in patients with potassium channel variants resulting in a long QT interval (5.4).
- Use of Strong CYP3A Inhibitors: Concomitant use can increase mifepristone plasma levels. Use only when necessary and limit mifepristone dose to 900 mg (5.6).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Obtain a negative pregnancy test in females of reproductive potential prior to initiating treatment with mifepristone or if treatment is interrupted for more than 14 days. (2.1)
- Administer once daily orally with a meal (2.2).
- The recommended starting dose is 300 mg once daily (2.2).
- Based on clinical response and tolerability, the dose may be increased in 300 mg increments to a maximum of 1200 mg once daily. Do not exceed 20 mg/kg per day (2.2).
- Renal impairment: do not exceed 600 mg once daily (2.3).
- Mild-to-moderate hepatic impairment: do not exceed 600 mg once daily. Do not use in severe hepatic impairment (2.4).
- Concomitant administration with strong CYP3A inhibitors: Do not exceed 900 mg once daily (2.5).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 300 mg
Oval shaped, light yellow to yellow tablets debossed with “Corcept” on one side and “300” on the other side. The tablets are not scored.
5.4 Qt Interval Prolongation (5.4 QT Interval Prolongation)
Mifepristone and its metabolites block IKr. Mifepristone prolongs the QTc interval in a dose-related manner. There is little or no experience with high exposure, concomitant dosing with other QT-prolonging drugs, or potassium channel variants resulting in a long QT interval. [See Warnings & Precautions (5.6)] To minimize risk, the lowest effective dose should always be used.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post approval use of mifepristone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
-Angioedema
2.3 Dosing in Renal Impairment
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
Safety data on the use of mifepristone are available from 50 patients with Cushing's syndrome enrolled in an uncontrolled, open-label, multi-center trial (Study 400). Forty-three patients had Cushing's disease and all except one had previously undergone pituitary surgery. Four patients had ectopic ACTH secretion, and three had adrenal carcinoma. Patients were treated for up to 24 weeks. A dose of 300 mg per day was administered for the initial 14 days; thereafter, the dose could be escalated in increments of 300 mg per day based on assessments of tolerability and clinical response. Doses were escalated up to 900 mg per day for patients <60 kg, or 1200 mg per day for patients >60 kg.
The most frequently reported adverse reactions (reported in ≥20% of patients, regardless of relationship to mifepristone) were nausea, fatigue, headache, decreased blood potassium, arthralgia, vomiting, peripheral edema, hypertension, dizziness, decreased appetite, and endometrial hypertrophy. Drug-related adverse events resulted in dose interruption or reduction in study drug in 40% of patients.
The adverse reactions that occurred in ≥10% of the Cushing's syndrome patients receiving mifepristone, regardless of relationship to mifepristone, are shown in Table 2.
| Body System/Adverse Reaction |
Percent (%) of Patients Reporting Event
(n = 50) |
|---|---|
|
*The denominator was 26 females who had baseline and end-of-trial transvaginal ultrasound |
|
| Gastrointestinal disorders | |
| Nausea | 48 |
| Vomiting | 26 |
| Dry mouth | 18 |
| Diarrhea | 12 |
| Constipation | 10 |
| General disorders and administration/site conditions | |
| Fatigue | 48 |
| Edema peripheral | 26 |
| Pain | 14 |
| Nervous system disorders | |
| Headache | 44 |
| Dizziness | 22 |
| Somnolence | 10 |
| Musculoskeletal and connective tissue disorders | |
| Arthralgia | 30 |
| Back pain | 16 |
| Myalgia | 14 |
| Pain in extremity | 12 |
| Investigations | |
| Blood potassium decreased | 34 |
| Thyroid function test abnormal | 18 |
| Infections and infestations | |
| Sinusitis | 14 |
| Nasopharyngitis | 12 |
| Metabolism and nutrition disorders | |
| Decreased appetite | 20 |
| Anorexia | 10 |
| Vascular disorders | |
| Hypertension | 24 |
| Reproductive system and breast disorders | |
| Endometrial hypertrophy | 38* |
| Respiratory, thoracic, and mediastinal disorders | |
| Dyspnea | 16 |
| Psychiatric disorders | |
| Anxiety | 10 |
7.1 Drugs Metabolized By Cyp3a (7.1 Drugs Metabolized by CYP3A)
Because mifepristone is an inhibitor of CYP3A, concurrent use of mifepristone with a drug whose metabolism is largely or solely mediated by CYP3A is likely to result in increased plasma concentrations of the drug. Discontinuation or dose reduction of such medications may be necessary with mifepristone co-administration.
Mifepristone increased the exposure to simvastatin and simvastatin acid significantly in healthy subjects. Concomitant use of simvastatin or lovastatin is contraindicated because of the increased risk of myopathy and rhabdomyolysis. [See Contraindications (4.2), Clinical Pharmacology 12.3]
The exposure of other substrates of CYP3A with narrow therapeutic ranges, such as cyclosporine, dihydroergotamine, ergotamine, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus, may be increased by concomitant administration with mifepristone. Therefore, the concomitant use of such CYP3A substrates with mifepristone is contraindicated. [See Contraindications (4.2)]
Other drugs with similar high first pass metabolism in which CYP3A is the primary route of metabolism should be used with extreme caution if co-administered with mifepristone. The lowest possible dose and/or a decreased frequency of dosing must be used with therapeutic drug monitoring when possible. Use of alternative drugs without these metabolic characteristics is advised when possible with concomitant mifepristone.
If drugs that undergo low first pass metabolism by CYP3A or drugs in which CYP3A is not the major metabolic route are co-administered with mifepristone, use the lowest dose of concomitant medication necessary, with appropriate monitoring and follow-up. [See Clinical Pharmacology (12.3)]
7.5 Drugs Metabolized By Cyp2b6 (7.5 Drugs Metabolized by CYP2B6)
Mifepristone is an inhibitor of CYP2B6 and may cause significant increases in exposure of drugs that are metabolized by CYP2B6 such as bupropion and efavirenz. Since no study has been conducted to evaluate the effect of mifepristone on substrates of CYP2B6, the concomitant use of bupropion and efavirenz should be undertaken with caution. [See Clinical Pharmacology (12.3)]
2.4 Dosing in Hepatic Impairment
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
As a part of patient counseling, doctors must review the mifepristone Medication Guide with every patient.
Warning: Termination of Pregnancy (WARNING: TERMINATION OF PREGNANCY)
Mifepristone is a potent antagonist of progesterone and cortisol via the progesterone and glucocorticoid (GR-II) receptors, respectively. The antiprogestational effects will result in the termination of pregnancy. Pregnancy must therefore be excluded before the initiation of treatment with mifepristone and prevented during treatment and for one month after stopping treatment by the use of a non-hormonal medically acceptable method of contraception unless the patient has had a surgical sterilization, in which case no additional contraception is needed. Pregnancy must also be excluded if treatment is interrupted for more than 14 days in females of reproductive potential.
5.6 Use of Strong Cyp3a Inhibitors (5.6 Use of Strong CYP3A Inhibitors)
Mifepristone should be used with caution in patients taking ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir, fosamprenavir, clarithromycin, conivaptan, lopinavir/ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole, as these could increase the concentration of mifepristone in the blood. The benefit of concomitant use of these agents should be carefully weighed against the potential risks. Mifepristone should be used in combination with strong CYP3A inhibitors only when necessary, and in such cases the dose should be limited to 900 mg per day. [See Warnings & Precautions (5.4), Drug Interactions (7.2), and Clinical Pharmacology (12.3)]
7.6 Use of Hormonal Contraceptives
Mifepristone is a progesterone-receptor antagonist and will interfere with the effectiveness of hormonal contraceptives. Therefore, non-hormonal contraceptive methods should be used. [See Use In Specific Populations (8.3)]
7.4 Drugs Metabolized By Cyp2c8/2c9 (7.4 Drugs Metabolized by CYP2C8/2C9)
Because mifepristone is an inhibitor of CYP2C8/2C9, concurrent use of mifepristone with a drug whose metabolism is largely or solely mediated by CYP2C8/2C9 is likely to result in increased plasma concentrations of the drug.
Mifepristone significantly increased exposure of fluvastatin, a typical CYP2C8/2C9 substrate, in healthy subjects. When given concomitantly with mifepristone, drugs that are substrates of CYP2C8/2C9 (including non-steroidal anti-inflammatory drugs, warfarin, and repaglinide) should be used at the smallest recommended doses, and patients should be closely monitored for adverse effects. [See Clinical Pharmacology (12.3)]
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Mifepristone is supplied as a light yellow to yellow, film-coated, oval-shaped tablet debossed with “Corcept” on one side and “300” on the other. Each tablet contains 300 mg of mifepristone. Mifepristone tablets are available in bottles of 280 tablets (NDC 76346-654-03).
Store at controlled room temperature, 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 – 86 °F). [See USP Controlled Room Temperature]
17.1 Importance of Preventing Pregnancy
- Advise patients that mifepristone will cause termination of pregnancy. Mifepristone is contraindicated in pregnant patients.
- Mifepristone reduces the effectiveness of hormonal contraceptives. Counsel females of reproductive potential regarding pregnancy prevention and planning with a non-hormonal contraceptive prior to use of mifepristone and up to one month after the end of treatment.
- Instruct patients to contact their physician immediately if they suspect or confirm they are pregnant.
5.8 Potential Effects of Hypercortisolemia
Mifepristone does not reduce serum cortisol levels. Elevated cortisol levels may activate mineralocorticoid receptors which are also expressed in cardiac tissues. Caution should be used in patients with underlying heart conditions including heart failure and coronary vascular disease.
5.3 Vaginal Bleeding and Endometrial Changes
Being an antagonist of the progesterone receptor, mifepristone promotes unopposed endometrial proliferation that may result in endometrium thickening, cystic dilatation of endometrial glands, and vaginal bleeding. Mifepristone should be used with caution in women who have hemorrhagic disorders or are receiving concurrent anticoagulant therapy. Women who experience vaginal bleeding during mifepristone treatment should be referred to a gynecologist for further evaluation.
2.5 Concomitant Administration With Cyp3a Inhibitors (2.5 Concomitant Administration with CYP3A Inhibitors)
Ketoconazole and other strong inhibitors of CYP3A, such as itraconazole, nefazodone, ritonavir, nelfinavir, indinavir, atazanavir, amprenavir and fosamprenavir, clarithromycin, conivaptan, lopinavir/ritonavir, posaconazole, saquinavir, telithromycin, or voriconazole may increase exposure to mifepristone. Mifepristone should be used in combination with strong CYP3A inhibitors only when necessary. [See Warnings and Precautions (5.6), Drug Interactions (7.2)]
Administration of mifepristone to patients already being treated with strong CYP3A inhibitors:
- Start at a dose of 300 mg. If clinically indicated, titrate to a maximum of 900 mg.
Administration of strong CYP3A inhibitors to patients already being treated with mifepristone:
- Adjust the dose of mifepristone according to Table 1.
| Current dose of mifepristone |
Adjustment to dose of mifepristone
if adding a strong CYP3A inhibitor |
| 300 mg | No change |
| 600 mg | Reduce dose to 300 mg. If clinically indicated, titrate to a maximum of 600 mg |
| 900 mg | Reduce dose to 600 mg. If clinically indicated, titrate to a maximum of 900 mg |
| 1200 mg | Reduce dose to 900 mg |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Mifepristone was evaluated for carcinogenicity potential in rats and mice. Rats were dosed for up to two years at doses of 5, 25, and 125 mg/kg of mifepristone. The high dose was the maximum tolerated dose, but exposure at all doses was below exposure at the maximum clinical dose based on AUC comparison. Female rats had a statistically significant increase in follicular cell adenomas/carcinomas and liver adenomas. It is plausible that these tumors are due to drug-induced enzyme metabolism, a mechanism not considered clinically relevant, but studies confirming this mechanism were not conducted with mifepristone. Mice were also tested for up to 2 years at mifepristone doses up to the maximum tolerated dose of 125 mg/kg, which provided exposure below the maximum clinical dose based on AUC. No drug-related tumors were seen in mice.
Mifepristone was not genotoxic in a battery of bacterial, yeast, and mammalian in vitro assays, and an in vivo micronucleus study in mice.
The pharmacological activity of mifepristone disrupts the estrus cycle of adult rats at a dose of 0.3 mg/kg (less than human exposure at the maximum clinical dose, based on body surface area). However, following withdrawal of treatment and subsequent resumption of the estrus cycle, there was no effect on reproductive function when mated.
A single subcutaneous dose of mifepristone (up to 100 mg/kg) to rats on the first day after birth did not adversely affect future reproductive function in males or females, although the onset of puberty was slightly premature in dosed females. Repeated doses of mifepristone (1 mg every other day) to neonatal rats resulted in potentially adverse fertility effects, including oviduct and ovary malformations in females, delayed male puberty, deficient male sexual behavior, reduced testicular size, and lowered ejaculation frequency.
2.1 Testing Prior to and During Mifepristone Administration (2.1 Testing Prior to and During mifepristone Administration)
5.5 Exacerbation/deterioration of Conditions Treated With Corticosteroids (5.5 Exacerbation/Deterioration of Conditions Treated with Corticosteroids)
Use of mifepristone in patients who receive corticosteroids for other conditions (e.g., autoimmune disorders) may lead to exacerbation or deterioration of such conditions, as mifepristone antagonizes the desired effects of glucocorticoid in these clinical settings. For medical conditions in which chronic corticosteroid therapy is lifesaving (e.g., immunosuppression in organ transplantation), mifepristone is contraindicated. [See Contraindications (4.3)]
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:08.506051 · Updated: 2026-03-14T22:36:03.395056