Improvue®
63b82129-4848-47c5-bf92-ec4b1a9e9974
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Ophthalmic Lubricant
Medication Information
Purpose
Ophthalmic Lubricant
Description
Drug Facts
Section 42229-5
Drug Facts
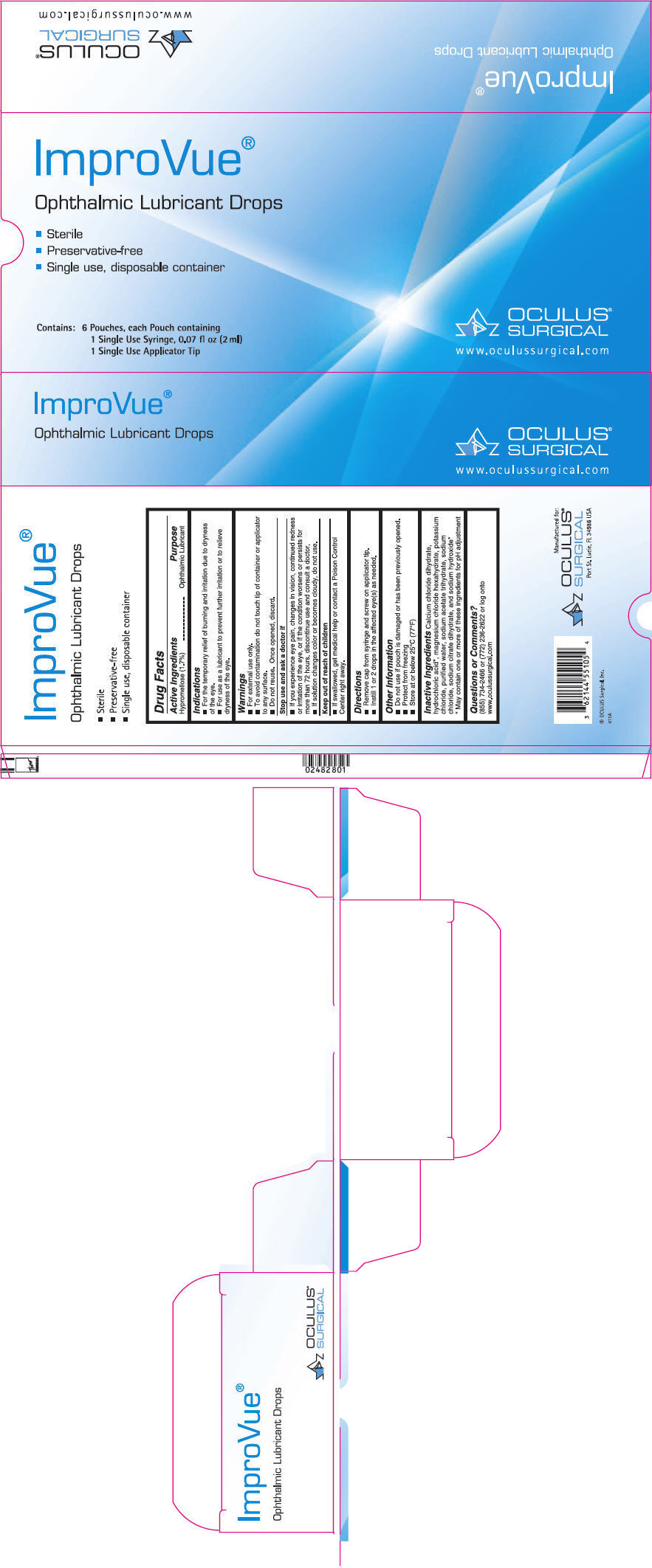
Warnings
- For external use only.
- To avoid contamination do not touch tip of container or applicator to any surface.
- Do not reuse. Once opened, discard.
Directions
- Remove cap from syringe and screw on applicator tip.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Indications
- For the temporary relief of burning and irritation due to dryness of the eye.
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Other Information
- Do not use if pouch is damaged or has been previously opened.
- Protect from freezing
- Store at or below 25°C (77°F)
Active Ingredients
Hypromellose (1.7%)
Inactive Ingredients
Calcium chloride dihydrate, hydrochloric acid
Questions Or Comments?
(855) 734-2466 or (772) 236-2622 or log onto www.oculussurgical.com
Stop Use and Ask A Doctor If
- If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
- If solution changes color or becomes cloudy, do not use.
Keep Out of Reach of Children
- If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 6 Pouch Carton
ImproVue ®
Ophthalmic Lubricant Drops
- Sterile
- Preservative-free
- Single use, disposable container
Contains:
6 Pouches, each Pouch containing
1 Single Use Syringe, 0.07 fl oz (2 ml)
1 Single Use Applicator Tip
OCULUS
®
SURGICAL
www.oculussurgical.com
Structured Label Content
Section 42229-5 (42229-5)
Drug Facts
Purpose
Ophthalmic Lubricant
Warnings
- For external use only.
- To avoid contamination do not touch tip of container or applicator to any surface.
- Do not reuse. Once opened, discard.
Directions
- Remove cap from syringe and screw on applicator tip.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Indications
- For the temporary relief of burning and irritation due to dryness of the eye.
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Other Information
- Do not use if pouch is damaged or has been previously opened.
- Protect from freezing
- Store at or below 25°C (77°F)
Active Ingredients
Hypromellose (1.7%)
Inactive Ingredients
Calcium chloride dihydrate, hydrochloric acid
Questions Or Comments? (Questions or Comments?)
(855) 734-2466 or (772) 236-2622 or log onto www.oculussurgical.com
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- If you experience eye pain, changes in vision, continued redness or irritation of the eye, or if the condition worsens or persists for more than 72 hours, discontinue use and consult a doctor.
- If solution changes color or becomes cloudy, do not use.
Keep Out of Reach of Children (Keep out of reach of children)
- If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 6 Pouch Carton (PRINCIPAL DISPLAY PANEL - 6 Pouch Carton)
ImproVue ®
Ophthalmic Lubricant Drops
- Sterile
- Preservative-free
- Single use, disposable container
Contains:
6 Pouches, each Pouch containing
1 Single Use Syringe, 0.07 fl oz (2 ml)
1 Single Use Applicator Tip
OCULUS
®
SURGICAL
www.oculussurgical.com
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:24.907116 · Updated: 2026-03-14T23:07:10.032470