These Highlights Do Not Include All The Information Needed To Use Serostim®
62b01d29-90f0-45b2-a0c4-3a750ba36c8a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
Indications and Usage
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
Dosage and Administration
SEROSTIM is administered by subcutaneous injection. SEROSTIM therapy should be carried out under the regular guidance of a physician who is experienced in the diagnosis and management of HIV infection.
Warnings and Precautions
Acute Critical Illness: Increased mortality in patients with acute critical illness following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin ( 5.1 ) Concomitant Antiretroviral Therapy: In vitro experimental systems have demonstrated the potential to potentiate HIV replication. No significant somatropin-associated increase in viral load was observed in clinical trials. HIV patients should be maintained on antiretroviral therapy for the duration of SEROSTIM treatment ( 5.2 ) Neoplasms: Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression, recurrences, or development of a tumor ( 5.3 ) Impaired Glucose Tolerance/Diabetes: May be unmasked. Periodically monitor glucose levels. Dose adjustment of concurrent antihyperglycemic drugs in diabetics may be required ( 5.4 ) Intracranial Hypertension: Exclude preexisting papilledema. May develop and is usually reversible after discontinuation or dose reduction ( 5.5 ) Hypersensitivity: Serious hypersensitivity reactions may occur. In the event of an allergic reaction, seek prompt medical attention ( 5.6 ) Fluid Retention (edema, arthralgia)/Carpal Tunnel Syndrome: May occur frequently. Reduce dose as necessary ( 5.7 ) Pancreatitis: Consider pancreatitis in patients with persistent severe abdominal pain ( 5.9 )
Contraindications
Acute Critical Illness Growth hormone therapy should not be initiated in patients with acute critical illness due to complications following open heart or abdominal surgery, multiple accidental trauma or acute respiratory failure [see Warnings and Precautions (5.1) ]. Active Malignancy In general, somatropin is contraindicated in the presence of active malignancy. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with somatropin. Somatropin should be discontinued if there is evidence of recurrent activity [see Warnings and Precautions (5.3) ] . Hypersensitivity SEROSTIM is contraindicated in patients with a known hypersensitivity to somatropin or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6) ]. Diabetic Retinopathy Somatropin is contraindicated in patients with active proliferative or severe non-proliferative diabetic retinopathy.
Adverse Reactions
The following important adverse reactions are also described elsewhere in the labeling: Acute Critical Illness [see Warnings and Precautions (5.1) ] Neoplasms [see Warnings and Precautions (5.3) ] Impaired glucose tolerance and diabetes mellitus [see Warnings and Precautions (5.4) ] Intracranial hypertension [see Warnings and Precautions (5.5) ] Severe hypersensitivity [see Warnings and Precautions (5.6) ] Fluid retention/Carpal tunnel syndrome [see Warnings and Precautions (5.7) ] Lipoatrophy [see Warnings and Precautions (5.8) ] Pancreatitis [see Warnings and Precautions (5.9) ]
Drug Interactions
Formal drug interaction studies have not been conducted. No data are available on drug interactions between SEROSTIM and HIV protease inhibitors or the non-nucleoside reverse transcriptase inhibitors.
How Supplied
SEROSTIM is available in the following forms: SEROSTIM single-use vials containing 5 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0005-7 SEROSTIM single-use vials containing 6 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0006-7 SEROSTIM multiple-use vials containing 4 mg with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). Package of 7 vials. NDC 44087-0004-7
Medication Information
Warnings and Precautions
Acute Critical Illness: Increased mortality in patients with acute critical illness following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin ( 5.1 ) Concomitant Antiretroviral Therapy: In vitro experimental systems have demonstrated the potential to potentiate HIV replication. No significant somatropin-associated increase in viral load was observed in clinical trials. HIV patients should be maintained on antiretroviral therapy for the duration of SEROSTIM treatment ( 5.2 ) Neoplasms: Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression, recurrences, or development of a tumor ( 5.3 ) Impaired Glucose Tolerance/Diabetes: May be unmasked. Periodically monitor glucose levels. Dose adjustment of concurrent antihyperglycemic drugs in diabetics may be required ( 5.4 ) Intracranial Hypertension: Exclude preexisting papilledema. May develop and is usually reversible after discontinuation or dose reduction ( 5.5 ) Hypersensitivity: Serious hypersensitivity reactions may occur. In the event of an allergic reaction, seek prompt medical attention ( 5.6 ) Fluid Retention (edema, arthralgia)/Carpal Tunnel Syndrome: May occur frequently. Reduce dose as necessary ( 5.7 ) Pancreatitis: Consider pancreatitis in patients with persistent severe abdominal pain ( 5.9 )
Indications and Usage
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
Dosage and Administration
SEROSTIM is administered by subcutaneous injection. SEROSTIM therapy should be carried out under the regular guidance of a physician who is experienced in the diagnosis and management of HIV infection.
Contraindications
Acute Critical Illness Growth hormone therapy should not be initiated in patients with acute critical illness due to complications following open heart or abdominal surgery, multiple accidental trauma or acute respiratory failure [see Warnings and Precautions (5.1) ]. Active Malignancy In general, somatropin is contraindicated in the presence of active malignancy. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with somatropin. Somatropin should be discontinued if there is evidence of recurrent activity [see Warnings and Precautions (5.3) ] . Hypersensitivity SEROSTIM is contraindicated in patients with a known hypersensitivity to somatropin or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6) ]. Diabetic Retinopathy Somatropin is contraindicated in patients with active proliferative or severe non-proliferative diabetic retinopathy.
Adverse Reactions
The following important adverse reactions are also described elsewhere in the labeling: Acute Critical Illness [see Warnings and Precautions (5.1) ] Neoplasms [see Warnings and Precautions (5.3) ] Impaired glucose tolerance and diabetes mellitus [see Warnings and Precautions (5.4) ] Intracranial hypertension [see Warnings and Precautions (5.5) ] Severe hypersensitivity [see Warnings and Precautions (5.6) ] Fluid retention/Carpal tunnel syndrome [see Warnings and Precautions (5.7) ] Lipoatrophy [see Warnings and Precautions (5.8) ] Pancreatitis [see Warnings and Precautions (5.9) ]
Drug Interactions
Formal drug interaction studies have not been conducted. No data are available on drug interactions between SEROSTIM and HIV protease inhibitors or the non-nucleoside reverse transcriptase inhibitors.
How Supplied
SEROSTIM is available in the following forms: SEROSTIM single-use vials containing 5 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0005-7 SEROSTIM single-use vials containing 6 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0006-7 SEROSTIM multiple-use vials containing 4 mg with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). Package of 7 vials. NDC 44087-0004-7
Description
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
Section 42229-5
Clinical trials in HIV-associated wasting or cachexia:
In the 12-week, placebo-controlled Clinical Trial 2, 510 patients were treated with SEROSTIM. The most common adverse reactions judged to be associated with SEROSTIM were musculoskeletal discomfort and increased tissue turgor (swelling, particularly of the hands or feet), and were more frequently observed when SEROSTIM 0.1 mg/kg was administered on a daily basis [Table 1 and Warnings and Precautions (5)]. These symptoms often subsided with continued treatment or dose reduction. Approximately 23% of patients receiving SEROSTIM 0.1 mg/kg daily and 11% of patients receiving 0.1 mg/kg every other day required dose reductions. Discontinuations as a result of adverse reactions occurred in 10.3% of patients receiving SEROSTIM 0.1 mg/kg daily and 6.6% of patients receiving 0.1 mg/kg every other day. The most common reasons for dose reduction and/or drug discontinuation were arthralgia, myalgia, edema, carpal tunnel syndrome, elevated glucose levels, and elevated triglyceride levels.
Clinical adverse reactions which occurred during the first 12 weeks of study in at least 5% of the patients in either active treatment group and at an incidence greater than placebo are listed below, without regard to causality assessment.
| Placebo | 0.1 mg/kg every other day SEROSTIM |
0.1 mg/kg daily SEROSTIM |
|
|---|---|---|---|
| Patients (n=247) |
Patients (n=257) |
Patients (n=253) |
|
| Body System Preferred Term |
% | % | % |
| Musculoskeletal System Disorders | |||
| Arthralgia | 11.3 | 24.5 | 36.4 |
| Myalgia | 11.7 | 17.9 | 30.4 |
| Arthrosis | 3.6 | 7.8 | 10.7 |
| Gastrointestinal System Disorders | |||
| Nausea | 4.9 | 5.4 | 9.1 |
| Body As A Whole - General Disorders | |||
| Edema Peripheral | 2.8 | 11.3 | 26.1 |
| Fatigue | 4.5 | 3.5 | 5.1 |
| Endocrine Disorders | |||
| Gynecomastia | 0.4 | 3.5 | 5.5 |
| Central and Peripheral Nervous System Disorders | |||
| Paresthesia | 4.5 | 7.4 | 7.9 |
| Hypoesthesia | 2.4 | 1.6 | 5.1 |
| Metabolic and Nutritional Disorders | |||
| Edema Generalized | 1.2 | 1.2 | 5.9 |
Adverse reactions that occurred in 1% to less than 5% of trial participants receiving SEROSTIM during the first 12 weeks of Clinical Trial 2 thought to be related to SEROSTIM included dose dependent edema, periorbital edema, carpal tunnel syndrome, hyperglycemia and hypertriglyceridemia.
During the 12-week, placebo-controlled portion of Clinical Trial 2, the incidence of hyperglycemia reported as an adverse reaction was 3.6% for the placebo group, 1.9% for the 0.1 mg/kg every other day group and 3.2% for the 0.1 mg/kg daily group. One case of diabetes mellitus was noted in the 0.1 mg/kg daily group during the first 12-weeks of therapy. In addition, during the extension phase of Clinical Trial 2, two patients converted from placebo to full dose SEROSTIM, and 1 patient converted from placebo to half-dose SEROSTIM, were discontinued because of the development of diabetes mellitus.
The types and incidences of adverse reactions reported during the Clinical Trial 2 extension phase were not different from, or greater in frequency than those observed during the 12-week, placebo-controlled portion of Clinical Trial 2.
5.3 Neoplasms
Because malignancies are more common in HIV positive individuals, the risks and benefits of starting somatropin in HIV positive patients should be carefully considered before initiating SEROSTIM treatment and patients should be monitored carefully for the development of neoplasms if treatment with somatropin is initiated.
Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression or recurrence of the tumor [see Contraindications (4)].
Monitor patients on somatropin therapy carefully for increased growth, or potential malignant changes of preexisting nevi.
8.1 Pregnancy
Reproduction studies have been performed in rats and rabbits. Doses up to 5 to 10 times the human dose, based on body surface area, have revealed no evidence of impaired fertility or harm to the fetus due to SEROSTIM. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, SEROSTIM should be used during pregnancy only if clearly needed.
11 Description
SEROSTIM is a human growth hormone (hGH) produced by recombinant DNA technology. SEROSTIM has 191 amino acid residues and a molecular weight of 22,125 daltons. Its amino acid sequence and structure are identical to the dominant form of human pituitary growth hormone. SEROSTIM is produced by a mammalian cell line (mouse C127) that has been modified by the addition of the hGH gene. SEROSTIM is secreted directly through the cell membrane into the cell-culture medium for collection and purification.
SEROSTIM is a sterile lyophilized powder intended for subcutaneous injection after reconstitution to its liquid form.
Vials of SEROSTIM contain either 4 mg, 5 mg, or 6 mg. Each vial contains the following:
| Vials | |||
|---|---|---|---|
| 4 mg | 5 mg | 6 mg | |
| Component | |||
| Somatropin | 4 mg | 5 mg | 6 mg |
| Sucrose | 27.3 mg | 34.2 mg | 41 mg |
| Phosphoric acid | 0.9 mg | 1.2 mg | 1.4 mg |
Each 4 mg multi-vial is supplied in a combination package with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). The pH is adjusted with sodium hydroxide of phosphoric acid to give a pH of 7.4 to 8.5 after reconstitution.
Each 5 mg single-use vial is supplied in a combination package with Sterile Water for Injection, USP. The pH is adjusted with sodium hydroxide or phosphoric acid to give a pH of 6.5 to 8.5 after reconstitution.
Each 6 mg single-use vial is supplied in a combination package with Sterile Water for Injection, USP. The pH is adjusted with sodium hydroxide of phosphoric acid to give a pH of 7.4 to 8.5 after reconstitution.
5.8 Lipoatrophy
When somatropin is administered subcutaneously at the same site over a long period of time, tissue atrophy may result. This can be avoided by rotating the injection site [see Dosage and Administration (2.2)].
5.9 Pancreatitis
Cases of pancreatitis have been reported rarely in children and adults receiving somatropin treatment, with some evidence supporting a greater risk in children compared with adults. Published literature indicates that girls who have Turner syndrome may be at greater risk than other somatropin-treated children. Pancreatitis should be considered in any somatropin-treated patient, especially a child who develops abdominal pain.
16.1 How Supplied
SEROSTIM is available in the following forms:
- SEROSTIM single-use vials containing 5 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0005-7
- SEROSTIM single-use vials containing 6 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0006-7
- SEROSTIM multiple-use vials containing 4 mg with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). Package of 7 vials. NDC 44087-0004-7
7.3 Oral Estrogen
Because oral estrogens may reduce the serum IGF-1 response to somatropin treatment, girls and women receiving oral estrogen replacement may require greater somatropin dosages [see Dosage and Administration (2)].
8.3 Nursing Women
It is not known whether SEROSTIM is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when SEROSTIM is administered to a nursing woman.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients with HIV have not been established. Available evidence suggests that somatropin clearance is similar in adults and children, but no pharmacokinetic studies have been conducted in children with HIV.
In two small studies, 11 children with HIV-associated failure to thrive were treated subcutaneously with human growth hormone. In one study, five children (age range, 6 to 17 years) were treated with 0.04 mg/kg/day for 26 weeks. In a second study, six children (age range, 8 to 14 years) were treated with 0.07 mg/kg/day for 4 weeks. Treatment appeared to be well tolerated in both studies. The preliminary data collected on a limited number of patients with HIV-associated failure to thrive appear to be consistent with safety observations in growth hormone-treated adults with HIV wasting.
Benzyl alcohol, a component of this product, has been associated with serious adverse events and death, particularly in pediatric patients. The "gasping syndrome," (characterized by central nervous system depression, metabolic acidosis, gasping respirations, and high levels of benzyl alcohol and its metabolites found in the blood and urine) has been associated with benzyl alcohol dosages >99 mg/kg/day in neonates and low-birth weight neonates. Additional symptoms may include gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Practitioners administering this and other medications containing benzyl alcohol should consider the combined daily metabolic load of benzyl alcohol from all sources.
8.5 Geriatric Use
Clinical studies with SEROSTIM did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Elderly patients may be more sensitive to the action of somatropin, and therefore, may be more prone to develop adverse reactions. A lower starting dose and smaller dose increments should be considered for older patients [see Dosage and Administration (2)].
8.8 Gender Effect
Biomedical literature indicates that a gender-related difference in the mean clearance of r-hGH could exist (clearance of r-hGH in males > clearance of r-hGH in females). However, no gender-based analysis is available for SEROSTIM in normal volunteers or patients infected with HIV.
4 Contraindications
-
Acute Critical Illness
Growth hormone therapy should not be initiated in patients with acute critical illness due to complications following open heart or abdominal surgery, multiple accidental trauma or acute respiratory failure [see Warnings and Precautions (5.1)]. -
Active Malignancy
In general, somatropin is contraindicated in the presence of active malignancy. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with somatropin. Somatropin should be discontinued if there is evidence of recurrent activity [see Warnings and Precautions (5.3)]. -
Hypersensitivity
SEROSTIM is contraindicated in patients with a known hypersensitivity to somatropin or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6)]. -
Diabetic Retinopathy
Somatropin is contraindicated in patients with active proliferative or severe non-proliferative diabetic retinopathy.
6 Adverse Reactions
The following important adverse reactions are also described elsewhere in the labeling:
Acute Critical Illness [see Warnings and Precautions (5.1)]
Neoplasms [see Warnings and Precautions (5.3)]
Impaired glucose tolerance and diabetes mellitus [see Warnings and Precautions (5.4)]
Intracranial hypertension [see Warnings and Precautions (5.5)]
Severe hypersensitivity [see Warnings and Precautions (5.6)]
Fluid retention/Carpal tunnel syndrome [see Warnings and Precautions (5.7)]
Lipoatrophy [see Warnings and Precautions (5.8)]
Pancreatitis [see Warnings and Precautions (5.9)]
7 Drug Interactions
Formal drug interaction studies have not been conducted. No data are available on drug interactions between SEROSTIM and HIV protease inhibitors or the non-nucleoside reverse transcriptase inhibitors.
8.7 Renal Impairment
Subjects with chronic renal failure tend to have decreased somatropin clearance compared to those with normal renal function. However, no studies have been conducted for SEROSTIM in patients with renal impairment [see Clinical Pharmacology (12.3)].
8.6 Hepatic Impairment
No studies have been conducted for SEROSTIM in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
12.1 Mechanism of Action
SEROSTIM is an anabolic and anticatabolic agent which exerts its influence by interacting with specific receptors on a variety of cell types including myocytes, hepatocytes, adipocytes, lymphocytes, and hematopoietic cells. Some, but not all of its effects, are mediated by insulin-like growth factor-1 (IGF-1).
5 Warnings and Precautions
- Acute Critical Illness: Increased mortality in patients with acute critical illness following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin (5.1)
- Concomitant Antiretroviral Therapy: In vitro experimental systems have demonstrated the potential to potentiate HIV replication. No significant somatropin-associated increase in viral load was observed in clinical trials. HIV patients should be maintained on antiretroviral therapy for the duration of SEROSTIM treatment (5.2)
- Neoplasms: Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression, recurrences, or development of a tumor (5.3)
- Impaired Glucose Tolerance/Diabetes: May be unmasked. Periodically monitor glucose levels. Dose adjustment of concurrent antihyperglycemic drugs in diabetics may be required (5.4)
- Intracranial Hypertension: Exclude preexisting papilledema. May develop and is usually reversible after discontinuation or dose reduction (5.5)
- Hypersensitivity: Serious hypersensitivity reactions may occur. In the event of an allergic reaction, seek prompt medical attention (5.6)
- Fluid Retention (edema, arthralgia)/Carpal Tunnel Syndrome: May occur frequently. Reduce dose as necessary (5.7)
- Pancreatitis: Consider pancreatitis in patients with persistent severe abdominal pain (5.9)
5.1 Acute Critical Illness
Increased mortality in patients with acute critical illness due to complications following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin. Two placebo-controlled clinical trials in non-growth hormone deficient adult patients (n=522) with these conditions revealed a significant increase in mortality (42% vs. 19%) among somatropin-treated patients (doses 5.3-8 mg/day) compared to those receiving placebo [see Contraindications (4)].
2 Dosage and Administration
SEROSTIM is administered by subcutaneous injection.
SEROSTIM therapy should be carried out under the regular guidance of a physician who is experienced in the diagnosis and management of HIV infection.
5.6 Severe Hypersensitivity
Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropin products. Patients and caregivers should be informed that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs [see Contraindications (4)].
3 Dosage Forms and Strengths
Single-use administration (to be reconstituted with Sterile Water for Injection):
- SEROSTIM 5 mg per vial
- SEROSTIM 6 mg per vial
Multi-use administration (to be reconstituted with Bacteriostatic Water for Injection):
- SEROSTIM 4 mg per vial
5.5 Intracranial Hypertension
Intracranial hypertension (IH) with papilledema, visual changes, headache, nausea, and/or vomiting has been reported in a small number of patients treated with somatropin products. Symptoms usually occurred within the first eight (8) weeks after the initiation of somatropin therapy. In all reported cases, IH-associated signs and symptoms rapidly resolved after cessation of therapy or a reduction of the somatropin dose. Funduscopic examination should be performed routinely before initiating treatment with somatropin to exclude preexisting papilledema, and periodically during the course of somatropin therapy. If papilledema is observed by funduscopy during somatropin treatment, treatment should be stopped. If somatropin-induced IH is diagnosed, treatment with somatropin can be restarted at a lower dose after IH-associated signs and symptoms have resolved.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post approval use of SEROSTIM. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6)].
Endocrine:
- new onset impaired glucose tolerance
- new onset type 2 diabetes mellitus
- exacerbation of preexisting diabetes mellitus
- diabetic ketoacidosis
- diabetic coma
In some patients, these conditions improved when SEROSTIM was discontinued, while in others the glucose intolerance persisted. Some of these patients required initiation or adjustment of antidiabetic treatment while on SEROSTIM [see Warnings and Precautions (5.4)].
Gastrointestinal: Pancreatitis [see Warnings and Precautions (5.9)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Patients being treated with SEROSTIM should be informed of the potential benefits and risks associated with treatment. Patients should be instructed to contact their physician should they experience any side effects or discomfort during treatment with SEROSTIM.
It is recommended that SEROSTIM be administered using sterile, disposable syringes and needles. Patients should be thoroughly instructed in the importance of proper disposal and cautioned against any reuse of needles and syringes. An appropriate container for the disposal of used syringes and needles should be employed.
Patients should be instructed to rotate injection sites to avoid localized tissue atrophy.
2.2 Preparation and Administration
Each vial of SEROSTIM 5 mg or 6 mg is reconstituted with 0.5 to 1 mL Sterile Water for Injection, USP.
Each vial of SEROSTIM 4 mg is reconstituted in 0.5 to 1 mL of Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol preserved). For patients sensitive to Benzyl Alcohol, SEROSTIM may be reconstituted with Sterile Water for Injection, USP [see Pediatric Use (8.4)].
When SEROSTIM is reconstituted with Sterile Water for Injection, USP, the reconstituted solution should be used immediately and any unused portion should be discarded.
When SEROSTIM is reconstituted with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol preserved) the reconstituted solution may be refrigerated (2-8°C/36-46°F) for up to 14 days.
Approximately 10% mechanical loss can be associated with reconstitution and administration from multi-dose vials.
To reconstitute SEROSTIM, inject the diluent into the vial of SEROSTIM aiming the liquid against the glass vial wall. Swirl the vial with a GENTLE rotary motion until contents are dissolved completely. DO NOT SHAKE. Parenteral drug products should always be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. SEROSTIM MUST NOT BE INJECTED if the solution is cloudy or contains particulate matter. Use it only if it is clear and colorless.
SEROSTIM can be administered using (1) a standard sterile, disposable syringe and needle, (2) a compatible SEROSTIM needle-free injection device or (3) a compatible SEROSTIM needle injection device. For proper use, refer to the Instructions for Use provided with the administration device.
Injection sites, which may be located on the thigh, upper arm, abdomen or buttock, should be rotated to avoid local irritation.
7.2 Cytochrome P450 Metabolized Drugs
Limited published data indicate that somatropin treatment increases cytochrome P450 (CYP450)-mediated antipyrine clearance in man. These data suggest that somatropin administration may alter the clearance of compounds metabolized by CYP450 liver enzymes (e.g., corticosteroids, sex steroids, anticonvulsants, cyclosporine). Therefore, careful monitoring is advised when somatropin is administered in combination with drugs metabolized by CYP450 liver enzymes. However, formal drug interaction studies have not been conducted.
2.1 Hiv Associated Wasting Or Cachexia
The usual starting dose of SEROSTIM is 0.1 mg/kg subcutaneously once daily (up to a total dose of 6 mg). SEROSTIM should be administered subcutaneously once daily at bedtime according to the following body weight-based dosage recommendations:
| Weight Range | Dose |
|---|---|
| >55kg (>121 lb) | 6 mg Based on an approximate daily dosage of 0.1 mg/kg. SC daily |
| 45-55 kg (99-121 lb) | 5 mg SC daily |
| 35-45 kg (75-99 lb) | 4 mg SC daily |
| <35 kg (<75 lb) | 0.1 mg/kg SC daily |
Treatment with SEROSTIM 0.1 mg/kg every other day was associated with fewer side effects, and resulted in a similar improvement in work output, as compared with SEROSTIM 0.1 mg/kg daily. Therefore, a starting dose of SEROSTIM 0.1 mg/kg every other day should be considered in patients at increased risk for adverse effects related to recombinant human growth hormone therapy (i.e., glucose intolerance). In general, dose reductions (i.e., reducing the total daily dose or the number of doses per week) should be considered for side effects potentially related to recombinant human growth hormone therapy.
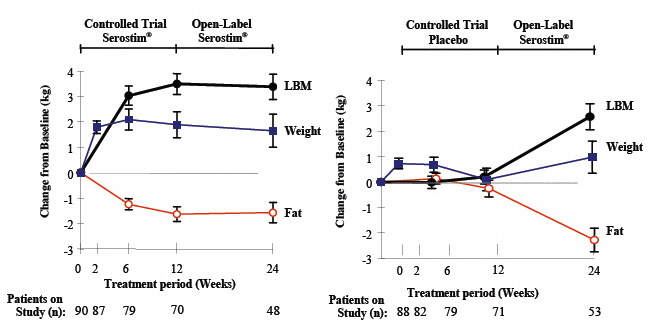
Most of the effect of SEROSTIM on work output and lean body mass was apparent after 12 weeks of treatment. The effect was maintained during an additional 12 weeks of therapy. There are no safety or efficacy data available from controlled studies in which patients were treated with SEROSTIM continuously for more than 48 weeks. There are no safety or efficacy data available from trials in which patients with HIV wasting or cachexia were treated intermittently with SEROSTIM.
5.2 Concomitant Antiretroviral Therapy
In some experimental systems, somatropin has been shown to potentiate HIV replication in vitro at concentrations ranging from 50-250 ng/mL. There was no increase in virus production when the antiretroviral agents, zidovudine, didanosine or lamivudine were added to the culture medium. Additional in vitro studies have shown that somatropin does not interfere with the antiviral activity of zalcitabine or stavudine. In the controlled clinical trials, no significant somatropin-associated increase in viral burden was observed. However, the protocol required all participants to be on concomitant antiretroviral therapy for the duration of the study. In view of the potential for acceleration of virus replication, it is recommended that HIV patients be maintained on antiretroviral therapy for the duration of SEROSTIM treatment.
5.4 Impaired Glucose Tolerance/diabetes
Hyperglycemia may occur in HIV infected individuals due to a variety of reasons. In wasting patients, treatment with SEROSTIM 0.1 mg/kg daily and 0.1 mg/kg every other day for 12 weeks was associated with approximately 10 mg/dL and 6 mg/dL increases in mean fasting blood glucose concentrations, respectively. The increases occurred early in treatment. Patients with other risk factors for glucose intolerance should be monitored closely during SEROSTIM therapy.
During safety surveillance of patients with HIV-associated wasting, cases of new onset impaired glucose tolerance, new onset type 2 diabetes mellitus and exacerbation of preexisting diabetes mellitus have been reported in patients receiving SEROSTIM. Some patients developed diabetic ketoacidosis and diabetic coma. In some patients, these conditions improved when SEROSTIM was discontinued, while in others, the glucose intolerance persisted. Some of these patients required initiation or adjustment of antidiabetic treatment while on SEROSTIM.
In clinical trials of SEROSTIM conducted in HIV patients with lipodystrophy (an unapproved indication), evidence of dose-dependent glucose intolerance and related adverse reaction was observed at doses of 4 mg SEROSTIM daily and 4 mg SEROSTIM every other day for 12 weeks [see Adverse Reactions (6.1)].
Principal Display Panel 4 Mg Kit Carton
Serostim® 4 mg
(somatropin) for injection
4 mg
For subcutaneous injection
Rx Only
7 vials of SEROSTIM
7 vials of Bacteriostatic Water for Injection, USP (0.9 % Benzyl Alcohol)
NDC 44087-0004-7
EMD Serono
Principal Display Panel 5 Mg Kit Carton
Serostim® 5 mg
(somatropin) for injection
5 mg
For subcutaneous injection
Rx Only
7 vials SEROSTIM
7 vials Sterile Diluent
NDC 44087-0005-7
EMD Serono
Principal Display Panel 6 Mg Kit Carton
Serostim® 6 mg
(somatropin) for injection
6 mg
For subcutaneous injection
Rx Only
7 vials SEROSTIM
7 vials Sterile Diluent
NDC 44087-0006-7
EMD Serono
5.7 Fluid Retention/carpal Tunnel Syndrome
Increased tissue turgor (swelling, particularly in the hands and feet) and musculoskeletal discomfort (pain, swelling and/or stiffness) may occur during treatment with SEROSTIM, but may resolve spontaneously, with analgesic therapy, or after reducing the frequency of dosing [see Dosage and Administration (2.1)].
Carpal tunnel syndrome may occur during treatment with SEROSTIM. If the symptoms of carpal tunnel syndrome do not resolve by decreasing the weekly number of doses of SEROSTIM, it is recommended that treatment be discontinued.
7.1 11β Hydroxysteroid Dehydrogenase Type 1
The microsomal enzyme 11β-hydroxysteroid dehydrogenase type 1 (11βHSD-1) is required for conversion of cortisone to its active metabolite, cortisol, in hepatic and adipose tissue. Somatropin inhibits 11βHSD-1. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of somatropin treatment; this may be especially true for patients treated with cortisone acetate and prednisone since conversion of these drugs to their biologically active metabolites is dependent on the activity of 11βHSD-1.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies for carcinogenicity have not been performed with SEROSTIM. There is no evidence from animal studies to date of SEROSTIM-induced mutagenicity or impairment of fertility.
7.4 Insulin And/or Other Oral/injectable Hypoglycemic Agents
Patients with diabetes mellitus who receive concomitant treatment with somatropin may require adjustment of their doses of insulin and/or other hypoglycemic agents [see Warnings and Precautions (5.4)].
Structured Label Content
Section 42229-5 (42229-5)
Clinical trials in HIV-associated wasting or cachexia:
In the 12-week, placebo-controlled Clinical Trial 2, 510 patients were treated with SEROSTIM. The most common adverse reactions judged to be associated with SEROSTIM were musculoskeletal discomfort and increased tissue turgor (swelling, particularly of the hands or feet), and were more frequently observed when SEROSTIM 0.1 mg/kg was administered on a daily basis [Table 1 and Warnings and Precautions (5)]. These symptoms often subsided with continued treatment or dose reduction. Approximately 23% of patients receiving SEROSTIM 0.1 mg/kg daily and 11% of patients receiving 0.1 mg/kg every other day required dose reductions. Discontinuations as a result of adverse reactions occurred in 10.3% of patients receiving SEROSTIM 0.1 mg/kg daily and 6.6% of patients receiving 0.1 mg/kg every other day. The most common reasons for dose reduction and/or drug discontinuation were arthralgia, myalgia, edema, carpal tunnel syndrome, elevated glucose levels, and elevated triglyceride levels.
Clinical adverse reactions which occurred during the first 12 weeks of study in at least 5% of the patients in either active treatment group and at an incidence greater than placebo are listed below, without regard to causality assessment.
| Placebo | 0.1 mg/kg every other day SEROSTIM |
0.1 mg/kg daily SEROSTIM |
|
|---|---|---|---|
| Patients (n=247) |
Patients (n=257) |
Patients (n=253) |
|
| Body System Preferred Term |
% | % | % |
| Musculoskeletal System Disorders | |||
| Arthralgia | 11.3 | 24.5 | 36.4 |
| Myalgia | 11.7 | 17.9 | 30.4 |
| Arthrosis | 3.6 | 7.8 | 10.7 |
| Gastrointestinal System Disorders | |||
| Nausea | 4.9 | 5.4 | 9.1 |
| Body As A Whole - General Disorders | |||
| Edema Peripheral | 2.8 | 11.3 | 26.1 |
| Fatigue | 4.5 | 3.5 | 5.1 |
| Endocrine Disorders | |||
| Gynecomastia | 0.4 | 3.5 | 5.5 |
| Central and Peripheral Nervous System Disorders | |||
| Paresthesia | 4.5 | 7.4 | 7.9 |
| Hypoesthesia | 2.4 | 1.6 | 5.1 |
| Metabolic and Nutritional Disorders | |||
| Edema Generalized | 1.2 | 1.2 | 5.9 |
Adverse reactions that occurred in 1% to less than 5% of trial participants receiving SEROSTIM during the first 12 weeks of Clinical Trial 2 thought to be related to SEROSTIM included dose dependent edema, periorbital edema, carpal tunnel syndrome, hyperglycemia and hypertriglyceridemia.
During the 12-week, placebo-controlled portion of Clinical Trial 2, the incidence of hyperglycemia reported as an adverse reaction was 3.6% for the placebo group, 1.9% for the 0.1 mg/kg every other day group and 3.2% for the 0.1 mg/kg daily group. One case of diabetes mellitus was noted in the 0.1 mg/kg daily group during the first 12-weeks of therapy. In addition, during the extension phase of Clinical Trial 2, two patients converted from placebo to full dose SEROSTIM, and 1 patient converted from placebo to half-dose SEROSTIM, were discontinued because of the development of diabetes mellitus.
The types and incidences of adverse reactions reported during the Clinical Trial 2 extension phase were not different from, or greater in frequency than those observed during the 12-week, placebo-controlled portion of Clinical Trial 2.
5.3 Neoplasms
Because malignancies are more common in HIV positive individuals, the risks and benefits of starting somatropin in HIV positive patients should be carefully considered before initiating SEROSTIM treatment and patients should be monitored carefully for the development of neoplasms if treatment with somatropin is initiated.
Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression or recurrence of the tumor [see Contraindications (4)].
Monitor patients on somatropin therapy carefully for increased growth, or potential malignant changes of preexisting nevi.
8.1 Pregnancy
Reproduction studies have been performed in rats and rabbits. Doses up to 5 to 10 times the human dose, based on body surface area, have revealed no evidence of impaired fertility or harm to the fetus due to SEROSTIM. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, SEROSTIM should be used during pregnancy only if clearly needed.
11 Description (11 DESCRIPTION)
SEROSTIM is a human growth hormone (hGH) produced by recombinant DNA technology. SEROSTIM has 191 amino acid residues and a molecular weight of 22,125 daltons. Its amino acid sequence and structure are identical to the dominant form of human pituitary growth hormone. SEROSTIM is produced by a mammalian cell line (mouse C127) that has been modified by the addition of the hGH gene. SEROSTIM is secreted directly through the cell membrane into the cell-culture medium for collection and purification.
SEROSTIM is a sterile lyophilized powder intended for subcutaneous injection after reconstitution to its liquid form.
Vials of SEROSTIM contain either 4 mg, 5 mg, or 6 mg. Each vial contains the following:
| Vials | |||
|---|---|---|---|
| 4 mg | 5 mg | 6 mg | |
| Component | |||
| Somatropin | 4 mg | 5 mg | 6 mg |
| Sucrose | 27.3 mg | 34.2 mg | 41 mg |
| Phosphoric acid | 0.9 mg | 1.2 mg | 1.4 mg |
Each 4 mg multi-vial is supplied in a combination package with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). The pH is adjusted with sodium hydroxide of phosphoric acid to give a pH of 7.4 to 8.5 after reconstitution.
Each 5 mg single-use vial is supplied in a combination package with Sterile Water for Injection, USP. The pH is adjusted with sodium hydroxide or phosphoric acid to give a pH of 6.5 to 8.5 after reconstitution.
Each 6 mg single-use vial is supplied in a combination package with Sterile Water for Injection, USP. The pH is adjusted with sodium hydroxide of phosphoric acid to give a pH of 7.4 to 8.5 after reconstitution.
5.8 Lipoatrophy
When somatropin is administered subcutaneously at the same site over a long period of time, tissue atrophy may result. This can be avoided by rotating the injection site [see Dosage and Administration (2.2)].
5.9 Pancreatitis
Cases of pancreatitis have been reported rarely in children and adults receiving somatropin treatment, with some evidence supporting a greater risk in children compared with adults. Published literature indicates that girls who have Turner syndrome may be at greater risk than other somatropin-treated children. Pancreatitis should be considered in any somatropin-treated patient, especially a child who develops abdominal pain.
16.1 How Supplied
SEROSTIM is available in the following forms:
- SEROSTIM single-use vials containing 5 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0005-7
- SEROSTIM single-use vials containing 6 mg with Sterile Water for Injection, USP. Package of 7 vials. NDC 44087-0006-7
- SEROSTIM multiple-use vials containing 4 mg with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol). Package of 7 vials. NDC 44087-0004-7
7.3 Oral Estrogen
Because oral estrogens may reduce the serum IGF-1 response to somatropin treatment, girls and women receiving oral estrogen replacement may require greater somatropin dosages [see Dosage and Administration (2)].
8.3 Nursing Women
It is not known whether SEROSTIM is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when SEROSTIM is administered to a nursing woman.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients with HIV have not been established. Available evidence suggests that somatropin clearance is similar in adults and children, but no pharmacokinetic studies have been conducted in children with HIV.
In two small studies, 11 children with HIV-associated failure to thrive were treated subcutaneously with human growth hormone. In one study, five children (age range, 6 to 17 years) were treated with 0.04 mg/kg/day for 26 weeks. In a second study, six children (age range, 8 to 14 years) were treated with 0.07 mg/kg/day for 4 weeks. Treatment appeared to be well tolerated in both studies. The preliminary data collected on a limited number of patients with HIV-associated failure to thrive appear to be consistent with safety observations in growth hormone-treated adults with HIV wasting.
Benzyl alcohol, a component of this product, has been associated with serious adverse events and death, particularly in pediatric patients. The "gasping syndrome," (characterized by central nervous system depression, metabolic acidosis, gasping respirations, and high levels of benzyl alcohol and its metabolites found in the blood and urine) has been associated with benzyl alcohol dosages >99 mg/kg/day in neonates and low-birth weight neonates. Additional symptoms may include gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Practitioners administering this and other medications containing benzyl alcohol should consider the combined daily metabolic load of benzyl alcohol from all sources.
8.5 Geriatric Use
Clinical studies with SEROSTIM did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Elderly patients may be more sensitive to the action of somatropin, and therefore, may be more prone to develop adverse reactions. A lower starting dose and smaller dose increments should be considered for older patients [see Dosage and Administration (2)].
8.8 Gender Effect
Biomedical literature indicates that a gender-related difference in the mean clearance of r-hGH could exist (clearance of r-hGH in males > clearance of r-hGH in females). However, no gender-based analysis is available for SEROSTIM in normal volunteers or patients infected with HIV.
4 Contraindications (4 CONTRAINDICATIONS)
-
Acute Critical Illness
Growth hormone therapy should not be initiated in patients with acute critical illness due to complications following open heart or abdominal surgery, multiple accidental trauma or acute respiratory failure [see Warnings and Precautions (5.1)]. -
Active Malignancy
In general, somatropin is contraindicated in the presence of active malignancy. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with somatropin. Somatropin should be discontinued if there is evidence of recurrent activity [see Warnings and Precautions (5.3)]. -
Hypersensitivity
SEROSTIM is contraindicated in patients with a known hypersensitivity to somatropin or any of its excipients. Systemic hypersensitivity reactions have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6)]. -
Diabetic Retinopathy
Somatropin is contraindicated in patients with active proliferative or severe non-proliferative diabetic retinopathy.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are also described elsewhere in the labeling:
Acute Critical Illness [see Warnings and Precautions (5.1)]
Neoplasms [see Warnings and Precautions (5.3)]
Impaired glucose tolerance and diabetes mellitus [see Warnings and Precautions (5.4)]
Intracranial hypertension [see Warnings and Precautions (5.5)]
Severe hypersensitivity [see Warnings and Precautions (5.6)]
Fluid retention/Carpal tunnel syndrome [see Warnings and Precautions (5.7)]
Lipoatrophy [see Warnings and Precautions (5.8)]
Pancreatitis [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Formal drug interaction studies have not been conducted. No data are available on drug interactions between SEROSTIM and HIV protease inhibitors or the non-nucleoside reverse transcriptase inhibitors.
8.7 Renal Impairment
Subjects with chronic renal failure tend to have decreased somatropin clearance compared to those with normal renal function. However, no studies have been conducted for SEROSTIM in patients with renal impairment [see Clinical Pharmacology (12.3)].
8.6 Hepatic Impairment
No studies have been conducted for SEROSTIM in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
SEROSTIM (somatropin) is indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary.
12.1 Mechanism of Action
SEROSTIM is an anabolic and anticatabolic agent which exerts its influence by interacting with specific receptors on a variety of cell types including myocytes, hepatocytes, adipocytes, lymphocytes, and hematopoietic cells. Some, but not all of its effects, are mediated by insulin-like growth factor-1 (IGF-1).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Acute Critical Illness: Increased mortality in patients with acute critical illness following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin (5.1)
- Concomitant Antiretroviral Therapy: In vitro experimental systems have demonstrated the potential to potentiate HIV replication. No significant somatropin-associated increase in viral load was observed in clinical trials. HIV patients should be maintained on antiretroviral therapy for the duration of SEROSTIM treatment (5.2)
- Neoplasms: Monitor all patients with a history of any neoplasm routinely while on somatropin therapy for progression, recurrences, or development of a tumor (5.3)
- Impaired Glucose Tolerance/Diabetes: May be unmasked. Periodically monitor glucose levels. Dose adjustment of concurrent antihyperglycemic drugs in diabetics may be required (5.4)
- Intracranial Hypertension: Exclude preexisting papilledema. May develop and is usually reversible after discontinuation or dose reduction (5.5)
- Hypersensitivity: Serious hypersensitivity reactions may occur. In the event of an allergic reaction, seek prompt medical attention (5.6)
- Fluid Retention (edema, arthralgia)/Carpal Tunnel Syndrome: May occur frequently. Reduce dose as necessary (5.7)
- Pancreatitis: Consider pancreatitis in patients with persistent severe abdominal pain (5.9)
5.1 Acute Critical Illness
Increased mortality in patients with acute critical illness due to complications following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic amounts of somatropin. Two placebo-controlled clinical trials in non-growth hormone deficient adult patients (n=522) with these conditions revealed a significant increase in mortality (42% vs. 19%) among somatropin-treated patients (doses 5.3-8 mg/day) compared to those receiving placebo [see Contraindications (4)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
SEROSTIM is administered by subcutaneous injection.
SEROSTIM therapy should be carried out under the regular guidance of a physician who is experienced in the diagnosis and management of HIV infection.
5.6 Severe Hypersensitivity
Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropin products. Patients and caregivers should be informed that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs [see Contraindications (4)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Single-use administration (to be reconstituted with Sterile Water for Injection):
- SEROSTIM 5 mg per vial
- SEROSTIM 6 mg per vial
Multi-use administration (to be reconstituted with Bacteriostatic Water for Injection):
- SEROSTIM 4 mg per vial
5.5 Intracranial Hypertension
Intracranial hypertension (IH) with papilledema, visual changes, headache, nausea, and/or vomiting has been reported in a small number of patients treated with somatropin products. Symptoms usually occurred within the first eight (8) weeks after the initiation of somatropin therapy. In all reported cases, IH-associated signs and symptoms rapidly resolved after cessation of therapy or a reduction of the somatropin dose. Funduscopic examination should be performed routinely before initiating treatment with somatropin to exclude preexisting papilledema, and periodically during the course of somatropin therapy. If papilledema is observed by funduscopy during somatropin treatment, treatment should be stopped. If somatropin-induced IH is diagnosed, treatment with somatropin can be restarted at a lower dose after IH-associated signs and symptoms have resolved.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post approval use of SEROSTIM. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity: Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropin products [see Warnings and Precautions (5.6)].
Endocrine:
- new onset impaired glucose tolerance
- new onset type 2 diabetes mellitus
- exacerbation of preexisting diabetes mellitus
- diabetic ketoacidosis
- diabetic coma
In some patients, these conditions improved when SEROSTIM was discontinued, while in others the glucose intolerance persisted. Some of these patients required initiation or adjustment of antidiabetic treatment while on SEROSTIM [see Warnings and Precautions (5.4)].
Gastrointestinal: Pancreatitis [see Warnings and Precautions (5.9)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Patients being treated with SEROSTIM should be informed of the potential benefits and risks associated with treatment. Patients should be instructed to contact their physician should they experience any side effects or discomfort during treatment with SEROSTIM.
It is recommended that SEROSTIM be administered using sterile, disposable syringes and needles. Patients should be thoroughly instructed in the importance of proper disposal and cautioned against any reuse of needles and syringes. An appropriate container for the disposal of used syringes and needles should be employed.
Patients should be instructed to rotate injection sites to avoid localized tissue atrophy.
2.2 Preparation and Administration
Each vial of SEROSTIM 5 mg or 6 mg is reconstituted with 0.5 to 1 mL Sterile Water for Injection, USP.
Each vial of SEROSTIM 4 mg is reconstituted in 0.5 to 1 mL of Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol preserved). For patients sensitive to Benzyl Alcohol, SEROSTIM may be reconstituted with Sterile Water for Injection, USP [see Pediatric Use (8.4)].
When SEROSTIM is reconstituted with Sterile Water for Injection, USP, the reconstituted solution should be used immediately and any unused portion should be discarded.
When SEROSTIM is reconstituted with Bacteriostatic Water for Injection, USP (0.9% Benzyl Alcohol preserved) the reconstituted solution may be refrigerated (2-8°C/36-46°F) for up to 14 days.
Approximately 10% mechanical loss can be associated with reconstitution and administration from multi-dose vials.
To reconstitute SEROSTIM, inject the diluent into the vial of SEROSTIM aiming the liquid against the glass vial wall. Swirl the vial with a GENTLE rotary motion until contents are dissolved completely. DO NOT SHAKE. Parenteral drug products should always be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. SEROSTIM MUST NOT BE INJECTED if the solution is cloudy or contains particulate matter. Use it only if it is clear and colorless.
SEROSTIM can be administered using (1) a standard sterile, disposable syringe and needle, (2) a compatible SEROSTIM needle-free injection device or (3) a compatible SEROSTIM needle injection device. For proper use, refer to the Instructions for Use provided with the administration device.
Injection sites, which may be located on the thigh, upper arm, abdomen or buttock, should be rotated to avoid local irritation.
7.2 Cytochrome P450 Metabolized Drugs (7.2 Cytochrome P450-metabolized drugs)
Limited published data indicate that somatropin treatment increases cytochrome P450 (CYP450)-mediated antipyrine clearance in man. These data suggest that somatropin administration may alter the clearance of compounds metabolized by CYP450 liver enzymes (e.g., corticosteroids, sex steroids, anticonvulsants, cyclosporine). Therefore, careful monitoring is advised when somatropin is administered in combination with drugs metabolized by CYP450 liver enzymes. However, formal drug interaction studies have not been conducted.
2.1 Hiv Associated Wasting Or Cachexia (2.1 HIV-associated wasting or cachexia)
The usual starting dose of SEROSTIM is 0.1 mg/kg subcutaneously once daily (up to a total dose of 6 mg). SEROSTIM should be administered subcutaneously once daily at bedtime according to the following body weight-based dosage recommendations:
| Weight Range | Dose |
|---|---|
| >55kg (>121 lb) | 6 mg Based on an approximate daily dosage of 0.1 mg/kg. SC daily |
| 45-55 kg (99-121 lb) | 5 mg SC daily |
| 35-45 kg (75-99 lb) | 4 mg SC daily |
| <35 kg (<75 lb) | 0.1 mg/kg SC daily |
Treatment with SEROSTIM 0.1 mg/kg every other day was associated with fewer side effects, and resulted in a similar improvement in work output, as compared with SEROSTIM 0.1 mg/kg daily. Therefore, a starting dose of SEROSTIM 0.1 mg/kg every other day should be considered in patients at increased risk for adverse effects related to recombinant human growth hormone therapy (i.e., glucose intolerance). In general, dose reductions (i.e., reducing the total daily dose or the number of doses per week) should be considered for side effects potentially related to recombinant human growth hormone therapy.
Most of the effect of SEROSTIM on work output and lean body mass was apparent after 12 weeks of treatment. The effect was maintained during an additional 12 weeks of therapy. There are no safety or efficacy data available from controlled studies in which patients were treated with SEROSTIM continuously for more than 48 weeks. There are no safety or efficacy data available from trials in which patients with HIV wasting or cachexia were treated intermittently with SEROSTIM.
5.2 Concomitant Antiretroviral Therapy
In some experimental systems, somatropin has been shown to potentiate HIV replication in vitro at concentrations ranging from 50-250 ng/mL. There was no increase in virus production when the antiretroviral agents, zidovudine, didanosine or lamivudine were added to the culture medium. Additional in vitro studies have shown that somatropin does not interfere with the antiviral activity of zalcitabine or stavudine. In the controlled clinical trials, no significant somatropin-associated increase in viral burden was observed. However, the protocol required all participants to be on concomitant antiretroviral therapy for the duration of the study. In view of the potential for acceleration of virus replication, it is recommended that HIV patients be maintained on antiretroviral therapy for the duration of SEROSTIM treatment.
5.4 Impaired Glucose Tolerance/diabetes (5.4 Impaired Glucose Tolerance/Diabetes)
Hyperglycemia may occur in HIV infected individuals due to a variety of reasons. In wasting patients, treatment with SEROSTIM 0.1 mg/kg daily and 0.1 mg/kg every other day for 12 weeks was associated with approximately 10 mg/dL and 6 mg/dL increases in mean fasting blood glucose concentrations, respectively. The increases occurred early in treatment. Patients with other risk factors for glucose intolerance should be monitored closely during SEROSTIM therapy.
During safety surveillance of patients with HIV-associated wasting, cases of new onset impaired glucose tolerance, new onset type 2 diabetes mellitus and exacerbation of preexisting diabetes mellitus have been reported in patients receiving SEROSTIM. Some patients developed diabetic ketoacidosis and diabetic coma. In some patients, these conditions improved when SEROSTIM was discontinued, while in others, the glucose intolerance persisted. Some of these patients required initiation or adjustment of antidiabetic treatment while on SEROSTIM.
In clinical trials of SEROSTIM conducted in HIV patients with lipodystrophy (an unapproved indication), evidence of dose-dependent glucose intolerance and related adverse reaction was observed at doses of 4 mg SEROSTIM daily and 4 mg SEROSTIM every other day for 12 weeks [see Adverse Reactions (6.1)].
Principal Display Panel 4 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 4 mg Kit Carton)
Serostim® 4 mg
(somatropin) for injection
4 mg
For subcutaneous injection
Rx Only
7 vials of SEROSTIM
7 vials of Bacteriostatic Water for Injection, USP (0.9 % Benzyl Alcohol)
NDC 44087-0004-7
EMD Serono
Principal Display Panel 5 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 5 mg Kit Carton)
Serostim® 5 mg
(somatropin) for injection
5 mg
For subcutaneous injection
Rx Only
7 vials SEROSTIM
7 vials Sterile Diluent
NDC 44087-0005-7
EMD Serono
Principal Display Panel 6 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 6 mg Kit Carton)
Serostim® 6 mg
(somatropin) for injection
6 mg
For subcutaneous injection
Rx Only
7 vials SEROSTIM
7 vials Sterile Diluent
NDC 44087-0006-7
EMD Serono
5.7 Fluid Retention/carpal Tunnel Syndrome (5.7 Fluid Retention/Carpal Tunnel Syndrome)
Increased tissue turgor (swelling, particularly in the hands and feet) and musculoskeletal discomfort (pain, swelling and/or stiffness) may occur during treatment with SEROSTIM, but may resolve spontaneously, with analgesic therapy, or after reducing the frequency of dosing [see Dosage and Administration (2.1)].
Carpal tunnel syndrome may occur during treatment with SEROSTIM. If the symptoms of carpal tunnel syndrome do not resolve by decreasing the weekly number of doses of SEROSTIM, it is recommended that treatment be discontinued.
7.1 11β Hydroxysteroid Dehydrogenase Type 1 (7.1 11β-Hydroxysteroid Dehydrogenase Type 1)
The microsomal enzyme 11β-hydroxysteroid dehydrogenase type 1 (11βHSD-1) is required for conversion of cortisone to its active metabolite, cortisol, in hepatic and adipose tissue. Somatropin inhibits 11βHSD-1. Patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of somatropin treatment; this may be especially true for patients treated with cortisone acetate and prednisone since conversion of these drugs to their biologically active metabolites is dependent on the activity of 11βHSD-1.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies for carcinogenicity have not been performed with SEROSTIM. There is no evidence from animal studies to date of SEROSTIM-induced mutagenicity or impairment of fertility.
7.4 Insulin And/or Other Oral/injectable Hypoglycemic Agents (7.4 Insulin and/or Other Oral/Injectable Hypoglycemic Agents)
Patients with diabetes mellitus who receive concomitant treatment with somatropin may require adjustment of their doses of insulin and/or other hypoglycemic agents [see Warnings and Precautions (5.4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:37.678985 · Updated: 2026-03-14T21:47:00.329153