These Highlights Do Not Include All The Information Needed To Use Argatroban Injection Safely And Effectively. See Full Prescribing Information For Argatroban Injection.
629aee90-3bf3-4947-a978-21cbba2e1c41
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Argatroban is a direct thrombin inhibitor indicated: For prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT) ( 1.1 ) As an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI) ( 1.2 )
Indications and Usage
Argatroban is a direct thrombin inhibitor indicated: For prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT) ( 1.1 ) As an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI) ( 1.2 )
Dosage and Administration
Argatroban Injection 50 mg per 50 mL (1 mg per mL) is ready for intravenous infusion. Dilution is not required. ( 2.1 ) Heparin-Induced Thrombocytopenia The dose for heparin-induced thrombocytopenia without hepatic impairment is 2 mcg/kg/min administered as a continuous infusion. ( 2.2 ) Percutaneous Coronary Intervention The dose for patients with or at risk for heparin-induced thrombocytopenia undergoing percutaneous coronary intervention is started at 25 mcg/kg/min and a bolus of 350 mcg/kg administered via a large bore intravenous line over 3 to 5 minutes. ( 2.3 )
Warnings and Precautions
Hemorrhage can occur. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage ( 5.1 ) Hepatic impairment: Adjust starting dose and titrate carefully in patients with HIT who have moderate or severe hepatic impairment. Avoid use in PCI in patients with clinically significant hepatic impairment. ( 5.2 )
Contraindications
Argatroban is contraindicated in: Patients with major bleeding, [see Warnings and Precautions ( 5.1 )] Patients with a history of hypersensitivity to argatroban. Airway, skin, and generalized hypersensitivity reactions have been reported [see Adverse Reactions ( 6.1 )]
Adverse Reactions
The following adverse reaction is also discussed in other sections of the labeling: Risk of Hemorrhage [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
Heparin: Allow sufficient time for heparin's effect on activated partial thromboplastin time (aPTT) to decrease before initiating Argatroban Injection therapy. ( 7.1 ) Warfarin: Concomitant use results in increased prolongation of PT and INR. ( 7.2 ) Thrombolytic agents or glycoprotein IIb/IIIa antagonists: Safety and effectiveness of concomitant use with argatroban have not been established. ( 7.4 , 7.5 )
Storage and Handling
Argatroban Injection is available as follows: NDC Argatroban Injection (1 mg per mL) Package Factor 25021-414-50 50 mg per 50 mL Single-Dose Vial 1 vial per carton Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
How Supplied
Argatroban Injection is available as follows: NDC Argatroban Injection (1 mg per mL) Package Factor 25021-414-50 50 mg per 50 mL Single-Dose Vial 1 vial per carton Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
Medication Information
Warnings and Precautions
Hemorrhage can occur. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage ( 5.1 ) Hepatic impairment: Adjust starting dose and titrate carefully in patients with HIT who have moderate or severe hepatic impairment. Avoid use in PCI in patients with clinically significant hepatic impairment. ( 5.2 )
Indications and Usage
Argatroban is a direct thrombin inhibitor indicated: For prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT) ( 1.1 ) As an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI) ( 1.2 )
Dosage and Administration
Argatroban Injection 50 mg per 50 mL (1 mg per mL) is ready for intravenous infusion. Dilution is not required. ( 2.1 ) Heparin-Induced Thrombocytopenia The dose for heparin-induced thrombocytopenia without hepatic impairment is 2 mcg/kg/min administered as a continuous infusion. ( 2.2 ) Percutaneous Coronary Intervention The dose for patients with or at risk for heparin-induced thrombocytopenia undergoing percutaneous coronary intervention is started at 25 mcg/kg/min and a bolus of 350 mcg/kg administered via a large bore intravenous line over 3 to 5 minutes. ( 2.3 )
Contraindications
Argatroban is contraindicated in: Patients with major bleeding, [see Warnings and Precautions ( 5.1 )] Patients with a history of hypersensitivity to argatroban. Airway, skin, and generalized hypersensitivity reactions have been reported [see Adverse Reactions ( 6.1 )]
Adverse Reactions
The following adverse reaction is also discussed in other sections of the labeling: Risk of Hemorrhage [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
Heparin: Allow sufficient time for heparin's effect on activated partial thromboplastin time (aPTT) to decrease before initiating Argatroban Injection therapy. ( 7.1 ) Warfarin: Concomitant use results in increased prolongation of PT and INR. ( 7.2 ) Thrombolytic agents or glycoprotein IIb/IIIa antagonists: Safety and effectiveness of concomitant use with argatroban have not been established. ( 7.4 , 7.5 )
Storage and Handling
Argatroban Injection is available as follows: NDC Argatroban Injection (1 mg per mL) Package Factor 25021-414-50 50 mg per 50 mL Single-Dose Vial 1 vial per carton Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
How Supplied
Argatroban Injection is available as follows: NDC Argatroban Injection (1 mg per mL) Package Factor 25021-414-50 50 mg per 50 mL Single-Dose Vial 1 vial per carton Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
Description
Argatroban is a direct thrombin inhibitor indicated: For prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT) ( 1.1 ) As an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI) ( 1.2 )
Section 42229-5
Argatroban Injection 50 mg per 50 mL (1 mg per mL)
Each 50 mL glass vial contains 50 mg argatroban (1 mg per mL); and, as supplied, is ready for intravenous infusion. Dilution is not required. Argatroban Injection is a clear, colorless to pale yellow solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Do not use if solution is cloudy, contains precipitates, or if the flip-off seals is not intact.
Vial may be inverted for use with a medical infusion set.
Section 44425-7
Storage Conditions
Store at 20° to 25° C (68° to 77° F). [See USP Controlled Room Temperature.]
Do not freeze.
Protect from light. Retain in carton until time of use.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
If the solution is cloudy, or if an insoluble precipitate is noted, the vial should be discarded.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-414-50
Rx only
Argatroban Injection
50 mg per 50 mL
(1 mg per mL)
For Intravenous Infusion Only
50 mL Single-Dose Vial
Discard Unused Portion
7.1 Heparin
If argatroban is to be initiated after cessation of heparin therapy, allow sufficient time for heparin's effect on the aPTT to decrease prior to initiation of argatroban therapy.
10 Overdosage
Excessive anticoagulation, with or without bleeding, may be controlled by discontinuing argatroban or by decreasing the argatroban dose. In clinical studies, anticoagulation parameters generally returned from therapeutic levels to baseline within 2 to 4 hours after discontinuation of the drug. Reversal of anticoagulant effect may take longer in patients with hepatic impairment. No specific antidote to argatroban is available; if life-threatening bleeding occurs and excessive plasma levels of argatroban are suspected, discontinue argatroban immediately and measure aPTT and other coagulation parameters. When argatroban was administered as a continuous infusion (2 mcg/kg/min) prior to and during a 4-hour hemodialysis session, approximately 20% of argatroban was cleared through dialysis.
Single intravenous doses of argatroban at 200, 124, 150, and 200 mg/kg were lethal to mice, rats, rabbits, and dogs, respectively. The symptoms of acute toxicity were loss of righting reflex, tremors, clonic convulsions, paralysis of hind limbs, and coma.
11 Description
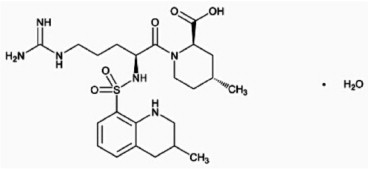
Argatroban is a synthetic direct thrombin inhibitor and the chemical name is 1-[5- [(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-methyl-8-quinolinyl)sulfonyl]amino]pentyl]-4-methyl-2-piperidinecarboxylic acid, monohydrate. Argatroban has 4 asymmetric carbons. One of the asymmetric carbons has an R configuration (stereoisomer Type I) and an S configuration (stereoisomer Type II). Argatroban consists of a mixture of R and S stereoisomers at a ratio of approximately 65:35.
The molecular formula of argatroban is C23H36N6O5S•H2O. Its molecular weight is 526.66 g/mol. The structural formula is shown below:
Argatroban is a white, odorless crystalline powder that is freely soluble in glacial acetic acid, slightly soluble in ethanol, and insoluble in acetone, ethyl acetate, and ether.
Argatroban Injection 50 mg per 50 mL (1 mg per mL) is a sterile clear, colorless to pale yellow, solution. Argatroban 50 mg per 50 mL (1 mg per mL) is available in 50-mg (in 50-mL) single-dose clear vials, with flip-off seals. Each mL of sterile, nonpyrogenic solution contains 1 mg Argatroban. Inert ingredients (per vial): 260 mg Propylene glycol, 152 mg Dehydrated alcohol, and 450 mg Sodium Chloride.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
Argatroban was studied among 18 seriously ill pediatric patients who required an alternative to heparin anticoagulation. Most patients were diagnosed with HIT or suspected HIT. Age ranges of patients were <6 months, n = 8; six months to <8 years, n = 6; 8 to 16 years, n = 4. All patients had serious underlying conditions and were receiving multiple concomitant medications. Thirteen patients received argatroban solely as a continuous infusion (no bolus dose). Dosing was initiated in the majority of these 13 patients at 1 mcg/kg/min. Dosing was titrated as needed to achieve and maintain an aPTT of 1.5 to 3 times the baseline value. Most patients required multiple dose adjustments to maintain anticoagulation parameters within the desired range. During the 30-day study period, thrombotic events occurred during argatroban administration to two patients and following argatroban discontinuation in three other patients. Major bleeding occurred among two patients: one patient experienced an intracranial hemorrhage after 4 days of argatroban therapy in the setting of sepsis and thrombocytopenia and another patient experienced an intracranial hemorrhage after receiving argatroban for greater than 14 days. The study findings did not establish the safe and effective use of argatroban in pediatric patients and the dosing of 1 mcg/kg/min was not supported by the pharmacokinetic data described below.
8.5 Geriatric Use
Of the total number of subjects (1340) in clinical studies of argatroban, 35% were 65 and over. In the clinical studies of adult patients with HIT (with or without thrombosis), the effectiveness of argatroban was not affected by age. No trends were observed across age groups for both aPTT and the ACT. The safety analysis did suggest that older patients had increased underlying conditions, which may predispose them to adverse reactions. The studies were not sized appropriately to detect differences in safety between age groups.
4 Contraindications
6 Adverse Reactions
The following adverse reaction is also discussed in other sections of the labeling:
- Risk of Hemorrhage [see Warnings and Precautions (5.1)].
7 Drug Interactions
- Heparin: Allow sufficient time for heparin's effect on activated partial thromboplastin time (aPTT) to decrease before initiating Argatroban Injection therapy. (7.1)
- Warfarin: Concomitant use results in increased prolongation of PT and INR. (7.2)
- Thrombolytic agents or glycoprotein IIb/IIIa antagonists: Safety and effectiveness of concomitant use with argatroban have not been established. (7.4, 7.5)
5.3 Laboratory Tests
Anticoagulation effects associated with argatroban infusion at doses up to 40 mcg/kg/min correlate with increases of the aPTT. Although other global clot-based tests including prothrombin time (PT), the International Normalized Ratio (INR), and thrombin time (TT) are affected by argatroban, the therapeutic ranges for these tests have not been identified for argatroban therapy. In clinical trials in PCI, the ACT was used for monitoring argatroban anticoagulant activity during the procedure. The concomitant use of argatroban and warfarin results in prolongation of the PT and INR beyond that produced by warfarin alone [see Dosage and Administration (2.5), Clinical Pharmacology (12.2)].
12.2 Pharmacodynamics
When argatroban is administered by continuous infusion, anticoagulant effects and plasma concentrations of argatroban follow similar, predictable temporal response profiles, with low intersubject variability. Immediately upon initiation of argatroban infusion, anticoagulant effects are produced as plasma argatroban concentrations begin to rise. Steady-state levels of both drug and anticoagulant effect are typically attained within 1 to 3 hours and are maintained until the infusion is discontinued or the dosage adjusted. Steady-state plasma argatroban concentrations increase proportionally with dose (for infusion doses up to 40 mcg/kg/min in healthy subjects) and are well correlated with steady-state anticoagulant effects. For infusion doses up to 40 mcg/kg/min, argatroban increases in a dose-dependent fashion, the aPTT, ACT, the prothrombin time (PT), the International Normalized Ratio (INR), and the thrombin time (TT) in healthy volunteers and cardiac patients. Representative steady-state plasma argatroban concentrations and anticoagulant effects are shown below for argatroban infusion doses up to 10 mcg/kg/min (see Figure 1).
| Figure 1. Relationship at Steady State Between Argatroban Dose, Plasma Argatroban Concentration and Anticoagulant Effect |
5.1 Risk of Hemorrhage
Hemorrhage can occur at any site in the body in patients receiving argatroban. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage. Intracranial and retroperitoneal hemorrhage [see Adverse Reactions (6.1)] have been reported. The risk of hemorrhage with argatroban may be increased in severe hypertension; immediately following lumbar puncture, spinal anesthesia, major surgery (especially involving the brain, spinal cord, or eye), hematologic conditions associated with increased bleeding tendencies such as congenital or acquired bleeding disorders, and gastrointestinal lesions such as ulcerations.
Concomitant use of argatroban with antiplatelet agents, thrombolytics, and other anticoagulants may increase the risk of bleeding.
8.6 Hepatic Impairment
1 Indications and Usage
7.4 Thrombolytic Agents
The safety and effectiveness of argatroban with thrombolytic agents have not been established [see Adverse Reactions (6.1)].
12.1 Mechanism of Action
Argatroban is a direct thrombin inhibitor that reversibly binds to the thrombin active site. Argatroban does not require the co-factor antithrombin III for antithrombotic activity. Argatroban exerts its anticoagulant effects by inhibiting thrombin-catalyzed or -induced reactions, including fibrin formation; activation of coagulation factors V, VIII, and XIII; activation of protein C; and platelet aggregation.
Argatroban inhibits thrombin with an inhibition constant (Ki) of 0.04 mcM. At therapeutic concentrations, argatroban has little or no effect on related serine proteases (trypsin, factor Xa, plasmin, and kallikrein).
Argatroban is capable of inhibiting the action of both free and clot-associated thrombin.
7.3 Aspirin/acetaminophen
No drug-drug interactions have been demonstrated between argatroban and concomitantly administered aspirin or acetaminophen [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions
- Hemorrhage can occur. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage (5.1)
- Hepatic impairment: Adjust starting dose and titrate carefully in patients with HIT who have moderate or severe hepatic impairment. Avoid use in PCI in patients with clinically significant hepatic impairment. (5.2)
2 Dosage and Administration
- Argatroban Injection 50 mg per 50 mL (1 mg per mL) is ready for intravenous infusion. Dilution is not required. (2.1)
Heparin-Induced Thrombocytopenia
The dose for heparin-induced thrombocytopenia without hepatic impairment is 2 mcg/kg/min administered as a continuous infusion. (2.2)
Percutaneous Coronary Intervention
The dose for patients with or at risk for heparin-induced thrombocytopenia undergoing percutaneous coronary intervention is started at 25 mcg/kg/min and a bolus of 350 mcg/kg administered via a large bore intravenous line over 3 to 5 minutes. (2.3)
3 Dosage Forms and Strengths
- Injection: 50 mg per 50 mL (1 mg per mL) ready for intravenous infusion single-dose vial
5.2 Use in Hepatic Impairment
When administering argatroban to patients with hepatic impairment, start with a lower dose and carefully titrate until the desired level of anticoagulation is achieved. Achievement of steady state aPTT levels may take longer and require more argatroban dose adjustments in patients with hepatic impairment compared to patients with normal hepatic function [see Use in Specific Populations (8.6)]. Also, upon cessation of argatroban infusion in the hepatically impaired patient, full reversal of anticoagulant effects may require longer than 4 hours due to decreased clearance and increased elimination half-life of argatroban [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)]. Avoid the use of high doses of argatroban in patients undergoing PCI who have clinically significant hepatic disease or AST/ALT levels ≥3 times the upper limit of normal.
7.2 Oral Anticoagulant Agents
Pharmacokinetic drug-drug interactions between argatroban and warfarin (7.5 mg single oral dose) have not been demonstrated. However, the concomitant use of argatroban and warfarin (5 to 7.5 mg initial oral dose, followed by 2.5 to 6 mg/day orally for 6 to 10 days) results in prolongation of the prothrombin time (PT) and International Normalized Ratio (INR) [see Dosage and Administration (2.5) and Clinical Pharmacology (12.2)].
8 Use in Specific Populations
6.1 Clinical Trials Experience
Adverse Reactions in Patients with HIT (With or Without Thrombosis)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following safety information is based on all 568 patients treated with argatroban in Study 1 and Study 2. The safety profile of the patients from these studies is compared with that of 193 historical controls in which the adverse reactions were collected retrospectively. Adverse reactions are separated into hemorrhagic and non-hemorrhagic reactions. Major bleeding was defined as bleeding that was overt and associated with a hemoglobin decrease ≥ 2 g/dL, that led to a transfusion of ≥ 2 units, or that was intracranial, retroperitoneal, or into a major prosthetic joint. Minor bleeding was overt bleeding that did not meet the criteria for major bleeding.
Table 4 gives an overview of the most frequently observed hemorrhagic reactions, presented separately by major and minor bleeding, sorted by decreasing occurrence among argatroban-treated patients with HIT (with or without thrombosis).
|
* with or without thrombosis |
||
|
a) Patients may have experienced more than 1 adverse reaction. |
||
|
b) One patient experienced intracranial hemorrhage 4 days after discontinuation of argatroban and following therapy with urokinase and oral anticoagulation. |
||
|
c) The historical control group consisted of patients with a clinical diagnosis of HIT (with or without thrombosis) that were considered eligible by an independent medical panel. |
||
| Major Hemorrhagic Reactions a | ||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical
Control c (n = 193) % |
|
| Overall bleeding |
5.3 | 6.7 |
| Gastrointestinal | 2.3 | 1.6 |
| Genitourinary and hematuria | 0.9 | 0.5 |
| Decrease in hemoglobin and hematocrit | 0.7 | 0 |
| Multisystem hemorrhage and DIC | 0.5 | 1 |
| Limb and BKA stump | 0.5 | 0 |
| Intracranial hemorrhage | 0b | 0.5 |
| Minor Hemorrhagic Reactions a | ||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical Control
c
(n = 193) % |
|
| Gastrointestinal | 14.4 | 18.1 |
| Genitourinary and hematuria | 11.6 | 0.8 |
| Decrease in hemoglobin and hematocrit | 10.4 | 0 |
| Groin | 5.4 | 3.1 |
| Hemoptysis | 2.9 | 0.8 |
| Brachial | 2.4 | 0.8 |
Table 5 gives an overview of the most frequently observed non-hemorrhagic reactions sorted by decreasing frequency of occurrence (≥2%) among argatroban-treated HIT/HITTS patients.
|
a) Patients may have experienced more than 1 adverse reaction. |
||
|
b) With or without thrombosis |
||
|
c) The historical control group consisted of patients with a clinical diagnosis of HIT (with or without thrombosis) that were considered eligible by an independent medical panel. |
||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical
Control c (n = 193) % |
|
| Dyspnea | 8.1 | 8.8 |
| Hypotension | 7.2 | 2.6 |
| Fever | 6.9 | 2.1 |
| Diarrhea | 6.2 | 1.6 |
| Sepsis | 6.0 | 12.4 |
| Cardiac arrest | 5.8 | 3.1 |
| Nausea | 4.8 | 0.5 |
| Ventricular tachycardia | 4.8 | 3.1 |
| Pain | 4.6 | 3.1 |
| Urinary tract infection | 4.6 | 5.2 |
| Vomiting | 4.2 | 0 |
| Infection | 3.7 | 3.6 |
| Pneumonia | 3.3 | 9.3 |
| Atrial fibrillation | 3.0 | 11.4 |
| Coughing | 2.8 | 1.6 |
| Abnormal renal function | 2.8 | 4.7 |
| Abdominal pain | 2.6 | 1.6 |
| Cerebrovascular disorder | 2.3 | 4.1 |
17 Patient Counseling Information
Inform patients of the risks associated with Argatroban Injection as well as the plan for regular monitoring during administration of the drug. Specifically, inform patients to report:
- the use of any other products known to affect bleeding.
- any medical history that may increase the risk for bleeding, including a history of severe hypertension; recent lumbar puncture or spinal anesthesia; major surgery, especially involving the brain, spinal cord, or eye; hematologic conditions associated with increased bleeding tendencies such as congenital or acquired bleeding disorders and gastrointestinal lesions such as ulcerations.
- any bleeding signs or symptoms
- the occurrence of any signs or symptoms of allergic reactions (e.g., airway reactions, skin reactions and vasodilation reactions)
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2021 Sagent Pharmaceuticals, Inc.
March 2021
SAGENT Pharmaceuticals®
1.1 Heparin Induced Thrombocytopenia
Argatroban Injection is indicated for prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT).
16 How Supplied/storage and Handling
Argatroban Injection is available as follows:
| NDC | Argatroban Injection (1 mg per mL) | Package Factor |
| 25021-414-50 | 50 mg per 50 mL Single-Dose Vial | 1 vial per carton |
Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
14.1 Heparin Induced Thrombocytopenia
The safety and efficacy of argatroban were evaluated in a historically controlled efficacy and safety study (Study 1) and a follow-on efficacy and safety study (Study 2). These studies were comparable with regard to study design, study objectives, dosing regimens as well as study outline, conduct, and monitoring. In these studies, 568 adult patients were treated with argatroban and 193 adult patients made up the historical control group. Patients had a clinical diagnosis of heparin-induced thrombocytopenia, either without thrombosis (HIT) or with thrombosis (HITTS [heparin-induced thrombocytopenia and thrombosis syndrome]) and were males or non-pregnant females between the age of 18 and 80 years old. HIT/HITTS was defined by a fall in platelet count to less than 100,000/mcL or a 50% decrease in platelets after the initiation of heparin therapy with no apparent explanation other than HIT. Patients with HITTS also had an arterial or venous thrombosis documented by appropriate imaging techniques or supported by clinical evidence such as acute myocardial infarction, stroke, pulmonary embolism, or other clinical indications of vascular occlusion. Patients who had documented histories of positive heparin-dependent antibody tests without current thrombocytopenia or heparin challenge (e.g., patients with latent disease) were also included if they required anticoagulation.
These studies did not include patients with documented unexplained aPTT >200% of control at baseline, documented coagulation disorder or bleeding diathesis unrelated to HIT, a lumbar puncture within the past 7 days or a history of previous aneurysm, hemorrhagic stroke, or a thrombotic stroke within the past 6 months unrelated to HIT.
The initial dose of argatroban was 2 mcg/kg/min. Two hours after the start of the argatroban infusion, an aPTT level was obtained and dose adjustments were made (up to a maximum of 10 mcg/kg/min) to achieve a steady-state aPTT value that was 1.5 to 3.0 times the baseline value, not to exceed 100 seconds. Overall the mean aPTT level for HIT and HITTS patients during the argatroban infusion increased from baseline values of 34 and 38 seconds, respectively, to 62.5 and 64.5 seconds, respectively.
The primary efficacy analysis was based on a comparison of event rates for a composite endpoint that included death (all causes), amputation (all causes) or new thrombosis during the treatment and follow-up period (study days 0 to 37). Secondary analyses included evaluation of the event rates for the components of the composite endpoint as well as time-to-event analyses.
In Study 1, a total of 304 patients were enrolled as follows: active HIT (n = 129), active HITTS (n =144), or latent disease (n = 31). Among the 193 historical controls, 139 (72%) had active HIT, 46 (24%) had active HITTS, and 8 (4%) had latent disease. Within each group, those with active HIT and those with latent disease were analyzed together. Positive laboratory confirmation of HIT/HITTS by the heparin-induced platelet aggregation test or serotonin release assay was demonstrated in 174 of 304 (57%) argatroban-treated patients (i.e., in 80 with HIT or latent disease and 94 with HITTS) and in 149 of 193 (77%) historical controls (i.e., in 119 with HIT or latent disease and 30 with HITTS). The test results for the remainder of the patients and controls were either negative or not determined.
There was a significant improvement in the composite outcome in patients with HIT and HITTS treated with argatroban versus those in the historical control group (see Table 9). The components of the composite endpoint are shown in Table 9.
|
a) Death (all cause), amputation (all cause), or new thrombosis within 37-day study period. |
||||||
|
b) Reported as the most severe outcome among the components of composite endpoint (severity ranking: death > amputation > new thrombosis); patients may have had multiple outcomes. |
||||||
|
Parameter, N (%) |
HIT | HITTS | HIT/HITTS | |||
|
Control
n = 147 |
Argatroban
n = 160 |
Control
n = 46 |
Argatroban
n = 144 |
Control
n = 193 |
Argatroban
n = 304 |
|
| Composite Endpoint | 57 (38.8) | 41 (25.6) | 26 (56.5) | 63 (43.8) | 83 (43.0) | 104 (34.2) |
| Individual Components b | ||||||
| Death | 32 (21.8) | 27 (16.9) | 13 (28.3) | 26 (18.1) | 45 (23.3) | 53 (17.4) |
| Amputation | 3 (2.0) | 3 (1.9) | 4 (8.7) | 16 (11.1) | 7 (3.6) | 19 (6.2) |
| New Thrombosis |
22 (15.0) |
11 (6.9) |
9 (19.6) |
21 (14.6) |
31 (16.1) |
32 (10.5) |
Time-to-event analyses showed significant improvements in the time-to-first event in patients with HIT or HITTS treated with argatroban versus those in the historical control group. The between-group differences in the proportion of patients who remained free of death, amputation, or new thrombosis were statistically significant in favor of argatroban by these analyses.
A time-to-event analysis for the composite endpoint is shown in Figure 3 for patients with HIT and Figure 4 for patients with HITTS.
| Figure 3. Time to First Event for the Composite Efficacy Endpoint: HIT Patients |
* Censored indicates no clinical endpoint (defined as death, amputation, or new thrombosis) was observed during the follow-up period (maximum period of follow-up was 37 days).
| Figure 4. Time to First Event for the Composite Efficacy Endpoint: HITTS Patients |
* Censored indicates no clinical endpoint (defined as death, amputation, or new thrombosis) was observed during the follow-up period (maximum period of follow-up was 37 days).
In Study 2, a total of 264 patients were enrolled as follows: HIT (n = 125) or HITTS (n = 139). There was a significant improvement in the composite efficacy outcome for argatroban-treated patients, versus the same historical control group from Study 1, among patients having HIT (25.6% vs. 38.8%), patients having HITTS (41.0% vs. 56.5%), and patients having either HIT or HITTS (33.7% vs. 43.0%). Time-to-event analyses showed significant improvements in the time-to-first event in patients with HIT or HITTS treated with argatroban versus those in the historical control group. The between-group differences in the proportion of patients who remained free of death, amputation, or new thrombosis were statistically significant in favor of argatroban.
7.5 Glycoprotein Iib/iiia Antagonists
The safety and effectiveness of argatroban with glycoprotein IIb/IIIa antagonists have not been established.
1.2 Percutaneous Coronary Intervention
Argatroban Injection is indicated as an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI).
2.1 Preparation for Intravenous Administration
Dilution is not required for Argatroban Injection 50 mg per 50 mL (1 mg per mL).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with argatroban have not been performed.
Argatroban was not genotoxic in the Ames test, the Chinese hamster ovary cell (CHO/HGPRT) forward mutation test, the Chinese hamster lung fibroblast chromosome aberration test, the rat hepatocyte, and WI-38 human fetal lung cell unscheduled DNA synthesis (UDS) tests, or the mouse micronucleus test.
Argatroban at intravenous doses up to 27 mg/kg/day (0.3 times the recommended maximum human dose based on body surface area) had no effect on fertility and reproductive function of male and female rats.
14.2 Percutaneous Coronary Intervention (pci) Patients With Or At Risk for Hit
In 3 similarly designed trials, argatroban was administered to 91 patients with current or previous clinical diagnosis of HIT or heparin-dependent antibodies, who underwent a total of 112 percutaneous coronary interventions (PCIs) including percutaneous transluminal coronary angioplasty (PTCA), coronary stent placement, or atherectomy. Among the 91 patients undergoing their first PCI with argatroban, notable ongoing or recent medical history included myocardial infarction (n = 35), unstable angina (n = 23), and chronic angina (n = 34). There were 33 females and 58 males. The average age was 67.6 years (median 70.7, range 44 to 86), and the average weight was 82.5 kg (median 81.0 kg, range 49 to 141).
Twenty-one of the 91 patients had a repeat PCI using argatroban an average of 150 days after their initial PCI. Seven of 91 patients received glycoprotein IIb/IIIa inhibitors. Safety and efficacy were assessed against historical control populations who had been anticoagulated with heparin.
All patients received oral aspirin (325 mg) 2 to 24 hours prior to the interventional procedure. After venous or arterial sheaths were in place, anticoagulation was initiated with a bolus of argatroban of 350 mcg/kg via a large-bore intravenous line or through the venous sheath over 3 to 5 minutes. Simultaneously, a maintenance infusion of 25 mcg/kg/min was initiated to achieve a therapeutic ACT of 300 to 450 seconds. If necessary to achieve this therapeutic range, the maintenance infusion dose was titrated (15 to 40 mcg/kg/min) and/or an additional bolus dose of 150 mcg/kg could be given. Each patient's ACT was checked 5 to 10 minutes following the bolus dose. The ACT was checked as clinically indicated. Arterial and venous sheaths were removed no sooner than 2 hours after discontinuation of argatroban and when the ACT was less than 160 seconds.
If a patient required anticoagulation after the procedure, argatroban could be continued, but at a lower infusion dose between 2.5 and 5 mcg/kg/min. An aPTT was drawn 2 hours after this dose reduction and the dose of argatroban then was adjusted as clinically indicated (not to exceed 10 mcg/kg/min), to reach an aPTT between 1.5 and 3 times baseline value (not to exceed 100 seconds).
In 92 of the 112 interventions (82%), the patient received the initial bolus of 350 mcg/kg and an initial infusion dose of 25 mcg/kg/min. The majority of patients did not require additional bolus dosing during the PCI procedure. The mean value for the initial ACT measurement after the start of dosing for all interventions was 379 sec (median 338 sec; 5th percentile-95th percentile 238 to 675 sec). The mean ACT value per intervention over all measurements taken during the procedure was 416 sec (median 390 sec; 5th percentile-95th percentile 261 to 698 sec). About 65% of patients had ACTs within the recommended range of 300 to 450 seconds throughout the procedure. The investigators did not achieve anticoagulation within the recommended range in about 23% of patients. However, in this small sample, patients with ACTs below 300 seconds did not have more coronary thrombotic events, and patients with ACTs over 450 seconds did not have higher bleeding rates.
Acute procedural success was defined as lack of death, emergent coronary artery bypass graft (CABG), or Q-wave myocardial infarction. Acute procedural success was reported in 98.2% of patients who underwent PCIs with argatroban anticoagulation compared with 94.3% of historical control patients anticoagulated with heparin (p = NS). Among the 112 interventions, 2 patients had emergency CABGs, 3 had repeat PTCAs, 4 had non-Q-wave myocardial infarctions, 3 had myocardial ischemia, 1 had an abrupt closure, and 1 had an impending closure (some patients may have experienced more than 1 event). No patients died.
Structured Label Content
Section 42229-5 (42229-5)
Argatroban Injection 50 mg per 50 mL (1 mg per mL)
Each 50 mL glass vial contains 50 mg argatroban (1 mg per mL); and, as supplied, is ready for intravenous infusion. Dilution is not required. Argatroban Injection is a clear, colorless to pale yellow solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Do not use if solution is cloudy, contains precipitates, or if the flip-off seals is not intact.
Vial may be inverted for use with a medical infusion set.
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25° C (68° to 77° F). [See USP Controlled Room Temperature.]
Do not freeze.
Protect from light. Retain in carton until time of use.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
If the solution is cloudy, or if an insoluble precipitate is noted, the vial should be discarded.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-414-50
Rx only
Argatroban Injection
50 mg per 50 mL
(1 mg per mL)
For Intravenous Infusion Only
50 mL Single-Dose Vial
Discard Unused Portion
7.1 Heparin
If argatroban is to be initiated after cessation of heparin therapy, allow sufficient time for heparin's effect on the aPTT to decrease prior to initiation of argatroban therapy.
10 Overdosage (10 OVERDOSAGE)
Excessive anticoagulation, with or without bleeding, may be controlled by discontinuing argatroban or by decreasing the argatroban dose. In clinical studies, anticoagulation parameters generally returned from therapeutic levels to baseline within 2 to 4 hours after discontinuation of the drug. Reversal of anticoagulant effect may take longer in patients with hepatic impairment. No specific antidote to argatroban is available; if life-threatening bleeding occurs and excessive plasma levels of argatroban are suspected, discontinue argatroban immediately and measure aPTT and other coagulation parameters. When argatroban was administered as a continuous infusion (2 mcg/kg/min) prior to and during a 4-hour hemodialysis session, approximately 20% of argatroban was cleared through dialysis.
Single intravenous doses of argatroban at 200, 124, 150, and 200 mg/kg were lethal to mice, rats, rabbits, and dogs, respectively. The symptoms of acute toxicity were loss of righting reflex, tremors, clonic convulsions, paralysis of hind limbs, and coma.
11 Description (11 DESCRIPTION)
Argatroban is a synthetic direct thrombin inhibitor and the chemical name is 1-[5- [(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-methyl-8-quinolinyl)sulfonyl]amino]pentyl]-4-methyl-2-piperidinecarboxylic acid, monohydrate. Argatroban has 4 asymmetric carbons. One of the asymmetric carbons has an R configuration (stereoisomer Type I) and an S configuration (stereoisomer Type II). Argatroban consists of a mixture of R and S stereoisomers at a ratio of approximately 65:35.
The molecular formula of argatroban is C23H36N6O5S•H2O. Its molecular weight is 526.66 g/mol. The structural formula is shown below:
Argatroban is a white, odorless crystalline powder that is freely soluble in glacial acetic acid, slightly soluble in ethanol, and insoluble in acetone, ethyl acetate, and ether.
Argatroban Injection 50 mg per 50 mL (1 mg per mL) is a sterile clear, colorless to pale yellow, solution. Argatroban 50 mg per 50 mL (1 mg per mL) is available in 50-mg (in 50-mL) single-dose clear vials, with flip-off seals. Each mL of sterile, nonpyrogenic solution contains 1 mg Argatroban. Inert ingredients (per vial): 260 mg Propylene glycol, 152 mg Dehydrated alcohol, and 450 mg Sodium Chloride.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
Argatroban was studied among 18 seriously ill pediatric patients who required an alternative to heparin anticoagulation. Most patients were diagnosed with HIT or suspected HIT. Age ranges of patients were <6 months, n = 8; six months to <8 years, n = 6; 8 to 16 years, n = 4. All patients had serious underlying conditions and were receiving multiple concomitant medications. Thirteen patients received argatroban solely as a continuous infusion (no bolus dose). Dosing was initiated in the majority of these 13 patients at 1 mcg/kg/min. Dosing was titrated as needed to achieve and maintain an aPTT of 1.5 to 3 times the baseline value. Most patients required multiple dose adjustments to maintain anticoagulation parameters within the desired range. During the 30-day study period, thrombotic events occurred during argatroban administration to two patients and following argatroban discontinuation in three other patients. Major bleeding occurred among two patients: one patient experienced an intracranial hemorrhage after 4 days of argatroban therapy in the setting of sepsis and thrombocytopenia and another patient experienced an intracranial hemorrhage after receiving argatroban for greater than 14 days. The study findings did not establish the safe and effective use of argatroban in pediatric patients and the dosing of 1 mcg/kg/min was not supported by the pharmacokinetic data described below.
8.5 Geriatric Use
Of the total number of subjects (1340) in clinical studies of argatroban, 35% were 65 and over. In the clinical studies of adult patients with HIT (with or without thrombosis), the effectiveness of argatroban was not affected by age. No trends were observed across age groups for both aPTT and the ACT. The safety analysis did suggest that older patients had increased underlying conditions, which may predispose them to adverse reactions. The studies were not sized appropriately to detect differences in safety between age groups.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reaction is also discussed in other sections of the labeling:
- Risk of Hemorrhage [see Warnings and Precautions (5.1)].
7 Drug Interactions (7 DRUG INTERACTIONS)
- Heparin: Allow sufficient time for heparin's effect on activated partial thromboplastin time (aPTT) to decrease before initiating Argatroban Injection therapy. (7.1)
- Warfarin: Concomitant use results in increased prolongation of PT and INR. (7.2)
- Thrombolytic agents or glycoprotein IIb/IIIa antagonists: Safety and effectiveness of concomitant use with argatroban have not been established. (7.4, 7.5)
5.3 Laboratory Tests
Anticoagulation effects associated with argatroban infusion at doses up to 40 mcg/kg/min correlate with increases of the aPTT. Although other global clot-based tests including prothrombin time (PT), the International Normalized Ratio (INR), and thrombin time (TT) are affected by argatroban, the therapeutic ranges for these tests have not been identified for argatroban therapy. In clinical trials in PCI, the ACT was used for monitoring argatroban anticoagulant activity during the procedure. The concomitant use of argatroban and warfarin results in prolongation of the PT and INR beyond that produced by warfarin alone [see Dosage and Administration (2.5), Clinical Pharmacology (12.2)].
12.2 Pharmacodynamics
When argatroban is administered by continuous infusion, anticoagulant effects and plasma concentrations of argatroban follow similar, predictable temporal response profiles, with low intersubject variability. Immediately upon initiation of argatroban infusion, anticoagulant effects are produced as plasma argatroban concentrations begin to rise. Steady-state levels of both drug and anticoagulant effect are typically attained within 1 to 3 hours and are maintained until the infusion is discontinued or the dosage adjusted. Steady-state plasma argatroban concentrations increase proportionally with dose (for infusion doses up to 40 mcg/kg/min in healthy subjects) and are well correlated with steady-state anticoagulant effects. For infusion doses up to 40 mcg/kg/min, argatroban increases in a dose-dependent fashion, the aPTT, ACT, the prothrombin time (PT), the International Normalized Ratio (INR), and the thrombin time (TT) in healthy volunteers and cardiac patients. Representative steady-state plasma argatroban concentrations and anticoagulant effects are shown below for argatroban infusion doses up to 10 mcg/kg/min (see Figure 1).
| Figure 1. Relationship at Steady State Between Argatroban Dose, Plasma Argatroban Concentration and Anticoagulant Effect |
5.1 Risk of Hemorrhage
Hemorrhage can occur at any site in the body in patients receiving argatroban. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage. Intracranial and retroperitoneal hemorrhage [see Adverse Reactions (6.1)] have been reported. The risk of hemorrhage with argatroban may be increased in severe hypertension; immediately following lumbar puncture, spinal anesthesia, major surgery (especially involving the brain, spinal cord, or eye), hematologic conditions associated with increased bleeding tendencies such as congenital or acquired bleeding disorders, and gastrointestinal lesions such as ulcerations.
Concomitant use of argatroban with antiplatelet agents, thrombolytics, and other anticoagulants may increase the risk of bleeding.
8.6 Hepatic Impairment
1 Indications and Usage (1 INDICATIONS AND USAGE)
7.4 Thrombolytic Agents
The safety and effectiveness of argatroban with thrombolytic agents have not been established [see Adverse Reactions (6.1)].
12.1 Mechanism of Action
Argatroban is a direct thrombin inhibitor that reversibly binds to the thrombin active site. Argatroban does not require the co-factor antithrombin III for antithrombotic activity. Argatroban exerts its anticoagulant effects by inhibiting thrombin-catalyzed or -induced reactions, including fibrin formation; activation of coagulation factors V, VIII, and XIII; activation of protein C; and platelet aggregation.
Argatroban inhibits thrombin with an inhibition constant (Ki) of 0.04 mcM. At therapeutic concentrations, argatroban has little or no effect on related serine proteases (trypsin, factor Xa, plasmin, and kallikrein).
Argatroban is capable of inhibiting the action of both free and clot-associated thrombin.
7.3 Aspirin/acetaminophen (7.3 Aspirin/Acetaminophen)
No drug-drug interactions have been demonstrated between argatroban and concomitantly administered aspirin or acetaminophen [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hemorrhage can occur. Unexplained fall in hematocrit or blood pressure may indicate hemorrhage (5.1)
- Hepatic impairment: Adjust starting dose and titrate carefully in patients with HIT who have moderate or severe hepatic impairment. Avoid use in PCI in patients with clinically significant hepatic impairment. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Argatroban Injection 50 mg per 50 mL (1 mg per mL) is ready for intravenous infusion. Dilution is not required. (2.1)
Heparin-Induced Thrombocytopenia
The dose for heparin-induced thrombocytopenia without hepatic impairment is 2 mcg/kg/min administered as a continuous infusion. (2.2)
Percutaneous Coronary Intervention
The dose for patients with or at risk for heparin-induced thrombocytopenia undergoing percutaneous coronary intervention is started at 25 mcg/kg/min and a bolus of 350 mcg/kg administered via a large bore intravenous line over 3 to 5 minutes. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 50 mg per 50 mL (1 mg per mL) ready for intravenous infusion single-dose vial
5.2 Use in Hepatic Impairment
When administering argatroban to patients with hepatic impairment, start with a lower dose and carefully titrate until the desired level of anticoagulation is achieved. Achievement of steady state aPTT levels may take longer and require more argatroban dose adjustments in patients with hepatic impairment compared to patients with normal hepatic function [see Use in Specific Populations (8.6)]. Also, upon cessation of argatroban infusion in the hepatically impaired patient, full reversal of anticoagulant effects may require longer than 4 hours due to decreased clearance and increased elimination half-life of argatroban [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)]. Avoid the use of high doses of argatroban in patients undergoing PCI who have clinically significant hepatic disease or AST/ALT levels ≥3 times the upper limit of normal.
7.2 Oral Anticoagulant Agents
Pharmacokinetic drug-drug interactions between argatroban and warfarin (7.5 mg single oral dose) have not been demonstrated. However, the concomitant use of argatroban and warfarin (5 to 7.5 mg initial oral dose, followed by 2.5 to 6 mg/day orally for 6 to 10 days) results in prolongation of the prothrombin time (PT) and International Normalized Ratio (INR) [see Dosage and Administration (2.5) and Clinical Pharmacology (12.2)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Adverse Reactions in Patients with HIT (With or Without Thrombosis)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following safety information is based on all 568 patients treated with argatroban in Study 1 and Study 2. The safety profile of the patients from these studies is compared with that of 193 historical controls in which the adverse reactions were collected retrospectively. Adverse reactions are separated into hemorrhagic and non-hemorrhagic reactions. Major bleeding was defined as bleeding that was overt and associated with a hemoglobin decrease ≥ 2 g/dL, that led to a transfusion of ≥ 2 units, or that was intracranial, retroperitoneal, or into a major prosthetic joint. Minor bleeding was overt bleeding that did not meet the criteria for major bleeding.
Table 4 gives an overview of the most frequently observed hemorrhagic reactions, presented separately by major and minor bleeding, sorted by decreasing occurrence among argatroban-treated patients with HIT (with or without thrombosis).
|
* with or without thrombosis |
||
|
a) Patients may have experienced more than 1 adverse reaction. |
||
|
b) One patient experienced intracranial hemorrhage 4 days after discontinuation of argatroban and following therapy with urokinase and oral anticoagulation. |
||
|
c) The historical control group consisted of patients with a clinical diagnosis of HIT (with or without thrombosis) that were considered eligible by an independent medical panel. |
||
| Major Hemorrhagic Reactions a | ||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical
Control c (n = 193) % |
|
| Overall bleeding |
5.3 | 6.7 |
| Gastrointestinal | 2.3 | 1.6 |
| Genitourinary and hematuria | 0.9 | 0.5 |
| Decrease in hemoglobin and hematocrit | 0.7 | 0 |
| Multisystem hemorrhage and DIC | 0.5 | 1 |
| Limb and BKA stump | 0.5 | 0 |
| Intracranial hemorrhage | 0b | 0.5 |
| Minor Hemorrhagic Reactions a | ||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical Control
c
(n = 193) % |
|
| Gastrointestinal | 14.4 | 18.1 |
| Genitourinary and hematuria | 11.6 | 0.8 |
| Decrease in hemoglobin and hematocrit | 10.4 | 0 |
| Groin | 5.4 | 3.1 |
| Hemoptysis | 2.9 | 0.8 |
| Brachial | 2.4 | 0.8 |
Table 5 gives an overview of the most frequently observed non-hemorrhagic reactions sorted by decreasing frequency of occurrence (≥2%) among argatroban-treated HIT/HITTS patients.
|
a) Patients may have experienced more than 1 adverse reaction. |
||
|
b) With or without thrombosis |
||
|
c) The historical control group consisted of patients with a clinical diagnosis of HIT (with or without thrombosis) that were considered eligible by an independent medical panel. |
||
|
Argatroban-treated Patients (Study 1 and Study 2)
(n = 568) % |
Historical
Control c (n = 193) % |
|
| Dyspnea | 8.1 | 8.8 |
| Hypotension | 7.2 | 2.6 |
| Fever | 6.9 | 2.1 |
| Diarrhea | 6.2 | 1.6 |
| Sepsis | 6.0 | 12.4 |
| Cardiac arrest | 5.8 | 3.1 |
| Nausea | 4.8 | 0.5 |
| Ventricular tachycardia | 4.8 | 3.1 |
| Pain | 4.6 | 3.1 |
| Urinary tract infection | 4.6 | 5.2 |
| Vomiting | 4.2 | 0 |
| Infection | 3.7 | 3.6 |
| Pneumonia | 3.3 | 9.3 |
| Atrial fibrillation | 3.0 | 11.4 |
| Coughing | 2.8 | 1.6 |
| Abnormal renal function | 2.8 | 4.7 |
| Abdominal pain | 2.6 | 1.6 |
| Cerebrovascular disorder | 2.3 | 4.1 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Inform patients of the risks associated with Argatroban Injection as well as the plan for regular monitoring during administration of the drug. Specifically, inform patients to report:
- the use of any other products known to affect bleeding.
- any medical history that may increase the risk for bleeding, including a history of severe hypertension; recent lumbar puncture or spinal anesthesia; major surgery, especially involving the brain, spinal cord, or eye; hematologic conditions associated with increased bleeding tendencies such as congenital or acquired bleeding disorders and gastrointestinal lesions such as ulcerations.
- any bleeding signs or symptoms
- the occurrence of any signs or symptoms of allergic reactions (e.g., airway reactions, skin reactions and vasodilation reactions)
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2021 Sagent Pharmaceuticals, Inc.
March 2021
SAGENT Pharmaceuticals®
1.1 Heparin Induced Thrombocytopenia (1.1 Heparin-Induced Thrombocytopenia)
Argatroban Injection is indicated for prophylaxis or treatment of thrombosis in adult patients with heparin-induced thrombocytopenia (HIT).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Argatroban Injection is available as follows:
| NDC | Argatroban Injection (1 mg per mL) | Package Factor |
| 25021-414-50 | 50 mg per 50 mL Single-Dose Vial | 1 vial per carton |
Argatroban Injection is a clear colorless to pale yellow color solution, free from visible particulates.
14.1 Heparin Induced Thrombocytopenia (14.1 Heparin-Induced Thrombocytopenia)
The safety and efficacy of argatroban were evaluated in a historically controlled efficacy and safety study (Study 1) and a follow-on efficacy and safety study (Study 2). These studies were comparable with regard to study design, study objectives, dosing regimens as well as study outline, conduct, and monitoring. In these studies, 568 adult patients were treated with argatroban and 193 adult patients made up the historical control group. Patients had a clinical diagnosis of heparin-induced thrombocytopenia, either without thrombosis (HIT) or with thrombosis (HITTS [heparin-induced thrombocytopenia and thrombosis syndrome]) and were males or non-pregnant females between the age of 18 and 80 years old. HIT/HITTS was defined by a fall in platelet count to less than 100,000/mcL or a 50% decrease in platelets after the initiation of heparin therapy with no apparent explanation other than HIT. Patients with HITTS also had an arterial or venous thrombosis documented by appropriate imaging techniques or supported by clinical evidence such as acute myocardial infarction, stroke, pulmonary embolism, or other clinical indications of vascular occlusion. Patients who had documented histories of positive heparin-dependent antibody tests without current thrombocytopenia or heparin challenge (e.g., patients with latent disease) were also included if they required anticoagulation.
These studies did not include patients with documented unexplained aPTT >200% of control at baseline, documented coagulation disorder or bleeding diathesis unrelated to HIT, a lumbar puncture within the past 7 days or a history of previous aneurysm, hemorrhagic stroke, or a thrombotic stroke within the past 6 months unrelated to HIT.
The initial dose of argatroban was 2 mcg/kg/min. Two hours after the start of the argatroban infusion, an aPTT level was obtained and dose adjustments were made (up to a maximum of 10 mcg/kg/min) to achieve a steady-state aPTT value that was 1.5 to 3.0 times the baseline value, not to exceed 100 seconds. Overall the mean aPTT level for HIT and HITTS patients during the argatroban infusion increased from baseline values of 34 and 38 seconds, respectively, to 62.5 and 64.5 seconds, respectively.
The primary efficacy analysis was based on a comparison of event rates for a composite endpoint that included death (all causes), amputation (all causes) or new thrombosis during the treatment and follow-up period (study days 0 to 37). Secondary analyses included evaluation of the event rates for the components of the composite endpoint as well as time-to-event analyses.
In Study 1, a total of 304 patients were enrolled as follows: active HIT (n = 129), active HITTS (n =144), or latent disease (n = 31). Among the 193 historical controls, 139 (72%) had active HIT, 46 (24%) had active HITTS, and 8 (4%) had latent disease. Within each group, those with active HIT and those with latent disease were analyzed together. Positive laboratory confirmation of HIT/HITTS by the heparin-induced platelet aggregation test or serotonin release assay was demonstrated in 174 of 304 (57%) argatroban-treated patients (i.e., in 80 with HIT or latent disease and 94 with HITTS) and in 149 of 193 (77%) historical controls (i.e., in 119 with HIT or latent disease and 30 with HITTS). The test results for the remainder of the patients and controls were either negative or not determined.
There was a significant improvement in the composite outcome in patients with HIT and HITTS treated with argatroban versus those in the historical control group (see Table 9). The components of the composite endpoint are shown in Table 9.
|
a) Death (all cause), amputation (all cause), or new thrombosis within 37-day study period. |
||||||
|
b) Reported as the most severe outcome among the components of composite endpoint (severity ranking: death > amputation > new thrombosis); patients may have had multiple outcomes. |
||||||
|
Parameter, N (%) |
HIT | HITTS | HIT/HITTS | |||
|
Control
n = 147 |
Argatroban
n = 160 |
Control
n = 46 |
Argatroban
n = 144 |
Control
n = 193 |
Argatroban
n = 304 |
|
| Composite Endpoint | 57 (38.8) | 41 (25.6) | 26 (56.5) | 63 (43.8) | 83 (43.0) | 104 (34.2) |
| Individual Components b | ||||||
| Death | 32 (21.8) | 27 (16.9) | 13 (28.3) | 26 (18.1) | 45 (23.3) | 53 (17.4) |
| Amputation | 3 (2.0) | 3 (1.9) | 4 (8.7) | 16 (11.1) | 7 (3.6) | 19 (6.2) |
| New Thrombosis |
22 (15.0) |
11 (6.9) |
9 (19.6) |
21 (14.6) |
31 (16.1) |
32 (10.5) |
Time-to-event analyses showed significant improvements in the time-to-first event in patients with HIT or HITTS treated with argatroban versus those in the historical control group. The between-group differences in the proportion of patients who remained free of death, amputation, or new thrombosis were statistically significant in favor of argatroban by these analyses.
A time-to-event analysis for the composite endpoint is shown in Figure 3 for patients with HIT and Figure 4 for patients with HITTS.
| Figure 3. Time to First Event for the Composite Efficacy Endpoint: HIT Patients |
* Censored indicates no clinical endpoint (defined as death, amputation, or new thrombosis) was observed during the follow-up period (maximum period of follow-up was 37 days).
| Figure 4. Time to First Event for the Composite Efficacy Endpoint: HITTS Patients |
* Censored indicates no clinical endpoint (defined as death, amputation, or new thrombosis) was observed during the follow-up period (maximum period of follow-up was 37 days).
In Study 2, a total of 264 patients were enrolled as follows: HIT (n = 125) or HITTS (n = 139). There was a significant improvement in the composite efficacy outcome for argatroban-treated patients, versus the same historical control group from Study 1, among patients having HIT (25.6% vs. 38.8%), patients having HITTS (41.0% vs. 56.5%), and patients having either HIT or HITTS (33.7% vs. 43.0%). Time-to-event analyses showed significant improvements in the time-to-first event in patients with HIT or HITTS treated with argatroban versus those in the historical control group. The between-group differences in the proportion of patients who remained free of death, amputation, or new thrombosis were statistically significant in favor of argatroban.
7.5 Glycoprotein Iib/iiia Antagonists (7.5 Glycoprotein IIb/IIIa Antagonists)
The safety and effectiveness of argatroban with glycoprotein IIb/IIIa antagonists have not been established.
1.2 Percutaneous Coronary Intervention
Argatroban Injection is indicated as an anticoagulant in adult patients with or at risk for HIT undergoing percutaneous coronary intervention (PCI).
2.1 Preparation for Intravenous Administration
Dilution is not required for Argatroban Injection 50 mg per 50 mL (1 mg per mL).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with argatroban have not been performed.
Argatroban was not genotoxic in the Ames test, the Chinese hamster ovary cell (CHO/HGPRT) forward mutation test, the Chinese hamster lung fibroblast chromosome aberration test, the rat hepatocyte, and WI-38 human fetal lung cell unscheduled DNA synthesis (UDS) tests, or the mouse micronucleus test.
Argatroban at intravenous doses up to 27 mg/kg/day (0.3 times the recommended maximum human dose based on body surface area) had no effect on fertility and reproductive function of male and female rats.
14.2 Percutaneous Coronary Intervention (pci) Patients With Or At Risk for Hit (14.2 Percutaneous Coronary Intervention (PCI) Patients with or at Risk for HIT)
In 3 similarly designed trials, argatroban was administered to 91 patients with current or previous clinical diagnosis of HIT or heparin-dependent antibodies, who underwent a total of 112 percutaneous coronary interventions (PCIs) including percutaneous transluminal coronary angioplasty (PTCA), coronary stent placement, or atherectomy. Among the 91 patients undergoing their first PCI with argatroban, notable ongoing or recent medical history included myocardial infarction (n = 35), unstable angina (n = 23), and chronic angina (n = 34). There were 33 females and 58 males. The average age was 67.6 years (median 70.7, range 44 to 86), and the average weight was 82.5 kg (median 81.0 kg, range 49 to 141).
Twenty-one of the 91 patients had a repeat PCI using argatroban an average of 150 days after their initial PCI. Seven of 91 patients received glycoprotein IIb/IIIa inhibitors. Safety and efficacy were assessed against historical control populations who had been anticoagulated with heparin.
All patients received oral aspirin (325 mg) 2 to 24 hours prior to the interventional procedure. After venous or arterial sheaths were in place, anticoagulation was initiated with a bolus of argatroban of 350 mcg/kg via a large-bore intravenous line or through the venous sheath over 3 to 5 minutes. Simultaneously, a maintenance infusion of 25 mcg/kg/min was initiated to achieve a therapeutic ACT of 300 to 450 seconds. If necessary to achieve this therapeutic range, the maintenance infusion dose was titrated (15 to 40 mcg/kg/min) and/or an additional bolus dose of 150 mcg/kg could be given. Each patient's ACT was checked 5 to 10 minutes following the bolus dose. The ACT was checked as clinically indicated. Arterial and venous sheaths were removed no sooner than 2 hours after discontinuation of argatroban and when the ACT was less than 160 seconds.
If a patient required anticoagulation after the procedure, argatroban could be continued, but at a lower infusion dose between 2.5 and 5 mcg/kg/min. An aPTT was drawn 2 hours after this dose reduction and the dose of argatroban then was adjusted as clinically indicated (not to exceed 10 mcg/kg/min), to reach an aPTT between 1.5 and 3 times baseline value (not to exceed 100 seconds).
In 92 of the 112 interventions (82%), the patient received the initial bolus of 350 mcg/kg and an initial infusion dose of 25 mcg/kg/min. The majority of patients did not require additional bolus dosing during the PCI procedure. The mean value for the initial ACT measurement after the start of dosing for all interventions was 379 sec (median 338 sec; 5th percentile-95th percentile 238 to 675 sec). The mean ACT value per intervention over all measurements taken during the procedure was 416 sec (median 390 sec; 5th percentile-95th percentile 261 to 698 sec). About 65% of patients had ACTs within the recommended range of 300 to 450 seconds throughout the procedure. The investigators did not achieve anticoagulation within the recommended range in about 23% of patients. However, in this small sample, patients with ACTs below 300 seconds did not have more coronary thrombotic events, and patients with ACTs over 450 seconds did not have higher bleeding rates.
Acute procedural success was defined as lack of death, emergent coronary artery bypass graft (CABG), or Q-wave myocardial infarction. Acute procedural success was reported in 98.2% of patients who underwent PCIs with argatroban anticoagulation compared with 94.3% of historical control patients anticoagulated with heparin (p = NS). Among the 112 interventions, 2 patients had emergency CABGs, 3 had repeat PTCAs, 4 had non-Q-wave myocardial infarctions, 3 had myocardial ischemia, 1 had an abrupt closure, and 1 had an impending closure (some patients may have experienced more than 1 event). No patients died.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:19.542609 · Updated: 2026-03-14T21:49:42.549823