Camphor Ointment 11%
62359237-de2d-4497-bfd6-36991cfc8acf
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Camphor, USP 11%
Purpose
External analgesic
Medication Information
Purpose
External analgesic
Description
Camphor, USP 11%
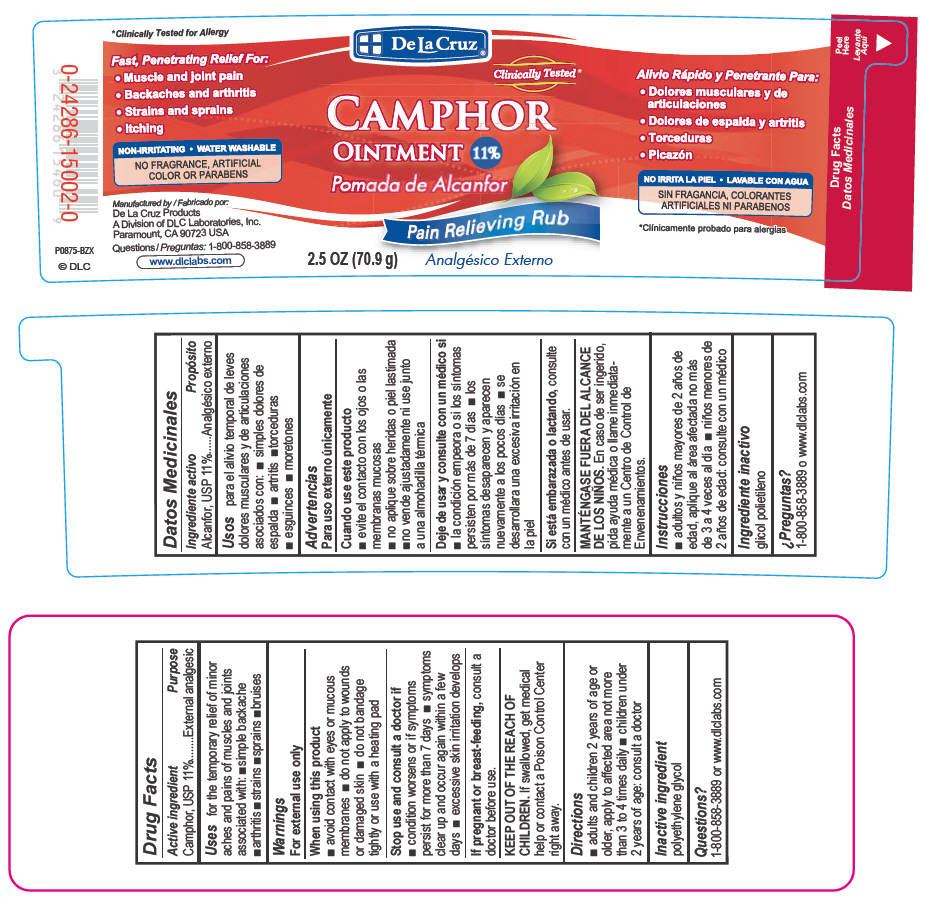
Uses
for the temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises and sprains
Section 42229-5
De La Cruz
CAMPHOR
Ointment 11%
Pain relieving rub
2.5 OZ (70.9g)
FAST, PENETRATING RELIEF FOR:
Muscle and joint pain
Backaches and arthritis
Strains and sprains
Itching
NON-IRRITATING
WATER WASHABLE
NO PARABENS OR ARTIFICIAL FRAGRANCES OR COLORS
Manufactured by:
De La Cruz Products
A Division of DLC Laboratories, Inc.
Paramount, CA 90723 USA
Questions: 1-800-858-3889
www.dlclabs.com (c) DLC
Warnings
For external use only
Directions
adults and children 2 years of age or older, apply to affected area not more than 3 to 4 times daily
children under 2 years of age: consult a doctor
Questions?
1-800-858-3889 or www.dlclabs.com
Active Ingredient
Camphor, USP 11%
Inactive Ingredient
polyethylene glycol
When Using This Product
avoid contact with eyes or mucous membranes
do not apply to wounds or damaged skin
do not bandage tightly or use with a heating pad
If Pregnant Or Breastfeeding,
consult a doctor before use.
Stop Use and Consult A Doctor If
condition worsens or if symptoms persist for more than 7 days
symptoms clear up and occur again within a few days.
excessive skin irritation develops
Keep Out of the Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 70.9 G Jar Label
De La Cruz ®
Clinically Tested*
CAMPHOR
OINTMENT 11%
Pain Relieving Rub
2.5 OZ (70.9 g)
Structured Label Content
Uses
for the temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises and sprains
Section 42229-5 (42229-5)
De La Cruz
CAMPHOR
Ointment 11%
Pain relieving rub
2.5 OZ (70.9g)
FAST, PENETRATING RELIEF FOR:
Muscle and joint pain
Backaches and arthritis
Strains and sprains
Itching
NON-IRRITATING
WATER WASHABLE
NO PARABENS OR ARTIFICIAL FRAGRANCES OR COLORS
Manufactured by:
De La Cruz Products
A Division of DLC Laboratories, Inc.
Paramount, CA 90723 USA
Questions: 1-800-858-3889
www.dlclabs.com (c) DLC
Purpose
External analgesic
Warnings
For external use only
Directions
adults and children 2 years of age or older, apply to affected area not more than 3 to 4 times daily
children under 2 years of age: consult a doctor
Questions?
1-800-858-3889 or www.dlclabs.com
Active Ingredient
Camphor, USP 11%
Inactive Ingredient (Inactive ingredient)
polyethylene glycol
When Using This Product (When using this product)
avoid contact with eyes or mucous membranes
do not apply to wounds or damaged skin
do not bandage tightly or use with a heating pad
If Pregnant Or Breastfeeding, (If pregnant or breastfeeding,)
consult a doctor before use.
Stop Use and Consult A Doctor If (Stop use and consult a doctor if)
condition worsens or if symptoms persist for more than 7 days
symptoms clear up and occur again within a few days.
excessive skin irritation develops
Keep Out of the Reach of Children. (KEEP OUT OF THE REACH OF CHILDREN.)
If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 70.9 G Jar Label (PRINCIPAL DISPLAY PANEL - 70.9 g Jar Label)
De La Cruz ®
Clinically Tested*
CAMPHOR
OINTMENT 11%
Pain Relieving Rub
2.5 OZ (70.9 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:44.966663 · Updated: 2026-03-14T23:03:58.855522