Acetaminophen Injection
621282f6-75b3-401b-8e1a-c5153a0c9ecd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Acetaminophen Injection is indicated for the management of mild to moderate pain in adult and pediatric patients 2 years and older the management of moderate to severe pain with adjunctive opioid analgesics in adult and pediatric patients 2 years and older the reduction of fever in adult and pediatric patients.
Dosage and Administration
Acetaminophen Injection may be given as a single or repeated dose. ( 2.1 ) Acetaminophen Injection should be administered only as a 15-minute intravenous infusion. ( 2.5 ) Adults and Adolescents Weighing 50 kg and Over : 1,000 mg every 6 hours or 650 mg every 4 hours to a maximum of 4,000 mg per day. Minimum dosing interval of 4 hours. ( 2.2 ) Adults and Adolescents Weighing Under 50 kg : 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. ( 2.2 ) Children : Children 2 to 12 years of age: 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. ( 2.3 ) Neonates and Infants : Neonates including premature neonates born at greater than or equal to 32 weeks gestational age to 28 days chronological age, 12.5 mg/kg every 6 hours to a maximum of 50 mg/kg per day. Minimum dosing interval of 6 hours. ( 2.4 ) Infants (29 days to 2 years of age): 15 mg/kg every 6 hours to a maximum of 60 mg/kg per day. Minimum dosing interval of 6 hours. ( 2.4 )
Contraindications
Acetaminophen is contraindicated: in patients with known hypersensitivity to acetaminophen or to any of the excipients in the intravenous formulation. in patients with severe hepatic impairment or severe active liver disease [see Warnings and Precautions ( 5.1 )] .
Warnings and Precautions
Risk of Hepatic Injury: Administration of acetaminophen in doses higher than recommended (by all routes of administration and from all acetaminophen-containing products including combination products) may result in hepatic injury, including the risk of liver failure and death. ( 5.1 ) Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, in cases of alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment (creatinine clearance less than or equal to 30 mL/min). ( 5.1 ) Risk of Serious Skin or Hypersensitivity Reactions: Discontinue Acetaminophen Injection immediately at the first appearance of skin rash and if symptoms associated with allergy or hypersensitivity occur. Do not use in patients with acetaminophen allergy. ( 5.2 , 5.4 ) Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. ( 5.3 )
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Hepatic Injury [see Warnings and Precautions ( 5.1 )] Serious Skin Reactions [see Warnings and Precautions ( 5.2 )] Allergy and Hypersensitivity [see Warnings and Precautions ( 5.4 )]
Drug Interactions
Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. ( 7.1 ) Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. ( 7.2 )
How Supplied
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows: NDC Acetaminophen Injection (10 mg per mL) Package Factor 25021-705-82 500 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-705-87 1,000 mg per 100 mL Single-Dose Bag 20 bags per carton Do not remove unit from overwrap until ready for use. To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
Storage and Handling
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows: NDC Acetaminophen Injection (10 mg per mL) Package Factor 25021-705-82 500 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-705-87 1,000 mg per 100 mL Single-Dose Bag 20 bags per carton Do not remove unit from overwrap until ready for use. To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
Description
Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: the dose in milligrams (mg) and milliliters (mL) is not confused; the dosing is based on weight for patients under 50 kg; infusion pumps are properly programmed; and the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. Acetaminophen Injection contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the maximum daily limits, and often involve more than one acetaminophen-containing product [see Warnings and Precautions ( 5.1 )] .
Medication Information
Warnings and Precautions
Risk of Hepatic Injury: Administration of acetaminophen in doses higher than recommended (by all routes of administration and from all acetaminophen-containing products including combination products) may result in hepatic injury, including the risk of liver failure and death. ( 5.1 ) Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, in cases of alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment (creatinine clearance less than or equal to 30 mL/min). ( 5.1 ) Risk of Serious Skin or Hypersensitivity Reactions: Discontinue Acetaminophen Injection immediately at the first appearance of skin rash and if symptoms associated with allergy or hypersensitivity occur. Do not use in patients with acetaminophen allergy. ( 5.2 , 5.4 ) Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. ( 5.3 )
Indications and Usage
Acetaminophen Injection is indicated for the management of mild to moderate pain in adult and pediatric patients 2 years and older the management of moderate to severe pain with adjunctive opioid analgesics in adult and pediatric patients 2 years and older the reduction of fever in adult and pediatric patients.
Dosage and Administration
Acetaminophen Injection may be given as a single or repeated dose. ( 2.1 ) Acetaminophen Injection should be administered only as a 15-minute intravenous infusion. ( 2.5 ) Adults and Adolescents Weighing 50 kg and Over : 1,000 mg every 6 hours or 650 mg every 4 hours to a maximum of 4,000 mg per day. Minimum dosing interval of 4 hours. ( 2.2 ) Adults and Adolescents Weighing Under 50 kg : 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. ( 2.2 ) Children : Children 2 to 12 years of age: 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. ( 2.3 ) Neonates and Infants : Neonates including premature neonates born at greater than or equal to 32 weeks gestational age to 28 days chronological age, 12.5 mg/kg every 6 hours to a maximum of 50 mg/kg per day. Minimum dosing interval of 6 hours. ( 2.4 ) Infants (29 days to 2 years of age): 15 mg/kg every 6 hours to a maximum of 60 mg/kg per day. Minimum dosing interval of 6 hours. ( 2.4 )
Contraindications
Acetaminophen is contraindicated: in patients with known hypersensitivity to acetaminophen or to any of the excipients in the intravenous formulation. in patients with severe hepatic impairment or severe active liver disease [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Hepatic Injury [see Warnings and Precautions ( 5.1 )] Serious Skin Reactions [see Warnings and Precautions ( 5.2 )] Allergy and Hypersensitivity [see Warnings and Precautions ( 5.4 )]
Drug Interactions
Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. ( 7.1 ) Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. ( 7.2 )
Storage and Handling
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows: NDC Acetaminophen Injection (10 mg per mL) Package Factor 25021-705-82 500 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-705-87 1,000 mg per 100 mL Single-Dose Bag 20 bags per carton Do not remove unit from overwrap until ready for use. To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
How Supplied
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows: NDC Acetaminophen Injection (10 mg per mL) Package Factor 25021-705-82 500 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-705-87 1,000 mg per 100 mL Single-Dose Bag 20 bags per carton Do not remove unit from overwrap until ready for use. To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
Description
Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: the dose in milligrams (mg) and milliliters (mL) is not confused; the dosing is based on weight for patients under 50 kg; infusion pumps are properly programmed; and the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. Acetaminophen Injection contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the maximum daily limits, and often involve more than one acetaminophen-containing product [see Warnings and Precautions ( 5.1 )] .
Section 42229-5
Adult Population
A total of 1,020 adult patients have received Acetaminophen Injection in clinical trials, including 37.3% (n=380) who received 5 or more doses, and 17.0% (n=173) who received more than 10 doses. Most patients were treated with Acetaminophen Injection 1,000 mg every 6 hours. A total of 13.1% (n=134) received Acetaminophen Injection 650 mg every 4 hours.
All adverse reactions that occurred in adult patients treated with either Acetaminophen Injection or placebo in repeated dose, placebo-controlled clinical trials at an incidence greater than or equal to 3% and at a greater frequency than placebo are listed in Table 4. The most common adverse events in adult patients treated with Acetaminophen Injection (incidence greater than or equal to 5% and greater than placebo) were nausea, vomiting, headache, and insomnia.
|
*Pyrexia adverse reaction frequency data is included in order to alert healthcare practitioners that the antipyretic effects of Acetaminophen Injection may mask fever. |
||
| System Organ Class – Preferred Term | Acetaminophen Injection (N=402) n (%) |
Placebo (N=379)
n (%) |
|
Gastrointestinal Disorders
Nausea Vomiting |
138 (34) 62 (15) |
119 (31) 42 (11) |
|
General Disorders and Administration Site Conditions
Pyrexia* |
22 (5) |
52 (14) |
|
Nervous System Disorders
Headache |
39 (10) | 33 (9) |
|
Psychiatric Disorders
Insomnia |
30 (7) |
21 (5) |
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] For single dose only. Use the product within 18 hours after opening.
Do not freeze.
Protect from light. Retain in carton until time of use.
Use only if prepared solution is clear and free from particulate matter.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container closure is not made with natural rubber latex.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Made in India
©2024 Sagent Pharmaceuticals
October 2024
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-705-82
50 mL Single-Dose Container
Acetaminophen Injection
500 mg per 50 mL
(10 mg per mL)
Rx only
For Intravenous Use Only
CAUTIONS: DO NOT ADD SUPPLEMENTARY MEDICATION
11 Description
Acetaminophen is a non-salicylate antipyretic and non-opioid analgesic agent. Its chemical name is N-acetyl-p-aminophenol.
Acetaminophen has a molecular weight of 151.16. Its structural formula is:
Acetaminophen Injection is a sterile, clear, colorless, non pyrogenic, isotonic formulation of acetaminophen intended for intravenous infusion. It has a pH of approximately 4.5 to 5.5 and an osmolality of approximately between 270 mOsm/kg and 310 mOsm/kg.
Each 100 mL contains 1,000 mg Acetaminophen, USP, 3,800 mg Mannitol, USP, 30 mg Sodium Citrate Dihydrate USP, Water for Injection, USP qs.
Each 50 mL contains 500 mg Acetaminophen, USP, 1,900 mg Mannitol, USP, 15 mg Sodium Citrate Dihydrate USP, Water for Injection, USP qs.
pH is adjusted with glacial acetic acid.
14.2 Adult Fever
The efficacy of acetaminophen 1,000 mg in the treatment of adult fever was evaluated in one randomized, double-blind, placebo-controlled clinical trial. The study was a 6-hour, single-dose, endotoxin-induced fever study in 60 healthy adult males. A statistically significant antipyretic effect of acetaminophen was demonstrated through 6 hours in comparison to placebo. The mean temperature over time is shown in Figure 1.
8.5 Geriatric Use
Of the total number of subjects in clinical studies of acetaminophen, 15% were age 65 and over, while 5% were age 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.1 Hepatic Injury
Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death [see Overdosage (10)]. Do not exceed the maximum recommended daily dose of acetaminophen [see Dosage and Administration (2)]. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products.
Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e.g., due to dehydration or blood loss), or severe renal impairment (creatinine clearance less than or equal to 30 mL/min) [see Use In Specific Populations (8.6, 8.7)].
7.2 Anticoagulants
Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. As no studies have been performed evaluating the short-term use of Acetaminophen Injection in patients on oral anticoagulants, more frequent assessment of INR may be appropriate in such circumstances.
4 Contraindications
Acetaminophen is contraindicated:
- in patients with known hypersensitivity to acetaminophen or to any of the excipients in the intravenous formulation.
- in patients with severe hepatic impairment or severe active liver disease [see Warnings and Precautions (5.1)].
6 Adverse Reactions
7 Drug Interactions
- Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. (7.1)
- Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. (7.2)
12.2 Pharmacodynamics
Acetaminophen has been shown to have analgesic and antipyretic activities in animal and human studies. Single doses of acetaminophen up to 3,000 mg and repeated doses of 1,000 mg every 6 hours for 48 hours have not been shown to cause a significant effect on platelet aggregation. Acetaminophen does not have any immediate or delayed effects on small-vessel hemostasis. Clinical studies of both healthy subjects and patients with hemophilia showed no significant changes in bleeding time after receiving multiple doses of oral acetaminophen.
14.1 Adult Acute Pain
The efficacy of Acetaminophen Injection in the treatment of acute pain in adults was evaluated in two randomized, double-blind, placebo-controlled clinical trials in patients with postoperative pain.
1 Indications and Usage
Acetaminophen Injection is indicated for
- the management of mild to moderate pain in adult and pediatric patients 2 years and older
- the management of moderate to severe pain with adjunctive opioid analgesics in adult and pediatric patients 2 years and older
- the reduction of fever in adult and pediatric patients.
12.1 Mechanism of Action
The precise mechanism of the analgesic and antipyretic properties of acetaminophen is not established but is thought to primarily involve central actions.
5 Warnings and Precautions
- Risk of Hepatic Injury: Administration of acetaminophen in doses higher than recommended (by all routes of administration and from all acetaminophen-containing products including combination products) may result in hepatic injury, including the risk of liver failure and death. (5.1)
- Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, in cases of alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment (creatinine clearance less than or equal to 30 mL/min). (5.1)
- Risk of Serious Skin or Hypersensitivity Reactions: Discontinue Acetaminophen Injection immediately at the first appearance of skin rash and if symptoms associated with allergy or hypersensitivity occur. Do not use in patients with acetaminophen allergy. (5.2, 5.4)
- Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. (5.3)
5.2 Serious Skin Reactions
Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. Patients should be informed about the signs of serious skin reactions, and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
2 Dosage and Administration
- Acetaminophen Injection may be given as a single or repeated dose. (2.1)
- Acetaminophen Injection should be administered only as a 15-minute intravenous infusion. (2.5)
Adults and Adolescents Weighing 50 kg and Over:
- 1,000 mg every 6 hours or 650 mg every 4 hours to a maximum of 4,000 mg per day. Minimum dosing interval of 4 hours. (2.2)
Adults and Adolescents Weighing Under 50 kg:
- 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. (2.2)
Children:
- Children 2 to 12 years of age: 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. (2.3)
Neonates and Infants:
- Neonates including premature neonates born at greater than or equal to 32 weeks gestational age to 28 days chronological age, 12.5 mg/kg every 6 hours to a maximum of 50 mg/kg per day. Minimum dosing interval of 6 hours. (2.4)
- Infants (29 days to 2 years of age): 15 mg/kg every 6 hours to a maximum of 60 mg/kg per day. Minimum dosing interval of 6 hours. (2.4)
3 Dosage Forms and Strengths
Acetaminophen Injection is a sterile, clear, colorless, non-pyrogenic, preservative free, isotonic formulation of acetaminophen available as:
- 1,000 mg acetaminophen in 100 mL (10 mg per mL) injection for intravenous infusion
- 500 mg acetaminophen in 50 mL (10 mg per mL) injection for intravenous infusion
5.3 Risk of Medication Errors
Take care when prescribing, preparing, and administering Acetaminophen Injection in order to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that:
- the dose in milligrams (mg) and milliliters (mL) is not confused;
- the dosing is based on weight for patients under 50 kg;
- infusion pumps are properly programmed; and
- the total daily dose of acetaminophen from all sources does not exceed maximum daily limits [see Dosage and Administration (2)].
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in practice.
8 Use in Specific Populations
- Pediatric Use: The effectiveness of acetaminophen for the treatment of acute pain in pediatric patients younger than 2 years of age has not been established. The safety and effectiveness of acetaminophen in pediatric patients is supported by evidence from adequate and well controlled studies in adults with additional safety and pharmacokinetic data for this age group. (8.4)
- Geriatric Use: No overall differences in safety or effectiveness were observed between geriatric and younger subjects. (8.5)
- Hepatic Impairment: Acetaminophen is contraindicated in patients with severe hepatic impairment or severe active liver disease and should be used with caution in patients with hepatic impairment or active liver disease. (4, 5.1, 8.6)
- Renal Impairment: In cases of severe renal impairment, longer dosing intervals and a reduced total daily dose of acetaminophen may be warranted. (5.1, 8.7)
2.3 Recommended Dosage: Children
Children 2 to 12 years of age: the recommended dosage of Acetaminophen Injection is 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours, with a maximum single dose of Acetaminophen Injection of 15 mg/kg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 75 mg/kg per day.
| Age group | Dose given every 4 hours | Dose given every 6 hours | Maximum single dose | Maximum total daily dose of acetaminophen (by all routes) |
| Children 2 to 12 years of age | 12.5 mg/kg | 15 mg/kg | 15 mg/kg (up to 750 mg) | 75 mg/kg in 24 hours (up to 3,750 mg) |
5.4 Allergy and Hypersensitivity
There have been post-marketing reports of hypersensitivity and anaphylaxis associated with the use of acetaminophen. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritus. There were infrequent reports of life-threatening anaphylaxis requiring emergent medical attention. Discontinue Acetaminophen Injection immediately if symptoms associated with allergy or hypersensitivity occur. Do not use Acetaminophen Injection in patients with acetaminophen allergy.
8.7 Patients With Renal Impairment
In cases of severe renal impairment (creatinine clearance less than or equal to 30 mL/min), longer dosing intervals and a reduced total daily dose of acetaminophen may be warranted.
14.3 Pediatric Acute Pain and Fever
Acetaminophen was studied in pediatric patients in three active-controlled trials and three open-label safety and pharmacokinetic trials [see Use in Specific Populations (8.4)].
16 How Supplied/storage and Handling
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows:
| NDC | Acetaminophen Injection (10 mg per mL) | Package Factor |
| 25021-705-82 | 500 mg per 50 mL Single-Dose Bag | 10 bags per carton |
| 25021-705-87 | 1,000 mg per 100 mL Single-Dose Bag | 20 bags per carton |
Do not remove unit from overwrap until ready for use.
To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
8.6 Patients With Hepatic Impairment
Acetaminophen is contraindicated in patients with severe hepatic impairment or severe active liver disease and should be used with caution in patients with hepatic impairment or active liver disease [see Warnings and Precautions (5.1) and Clinical Pharmacology (12)]. A reduced total daily dose of acetaminophen may be warranted.
2.2 Recommended Dosage: Adults and Adolescents
Adults and adolescents weighing 50 kg and over: the recommended dosage of Acetaminophen Injection is 1,000 mg every 6 hours or 650 mg every 4 hours, with a maximum single dose of Acetaminophen Injection of 1,000 mg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 4,000 mg per day (includes all routes of administration and all acetaminophen-containing products including combination products).
Adults and adolescents weighing under 50 kg: the recommended dosage of Acetaminophen Injection is 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours, with a maximum single dose of Acetaminophen Injection of 15 mg/kg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 75 mg/kg per day (includes all routes of administration and all acetaminophen-containing products including combination products).
| Age group | Dose given every 4 hours | Dose given every 6 hours | Maximum single dose | Maximum total daily dose of acetaminophen (by all routes) |
| Adults and adolescents (13 years and older) weighing greater than or equal to 50 kg | 650 mg | 1,000 mg | 1,000 mg | 4,000 mg in 24 hours |
| Adults and adolescents (13 years and older) weighing less than 50 kg | 12.5 mg/kg | 15 mg/kg | 15 mg/kg (up to 750 mg) | 75 mg/kg in 24 hours (up to 3,750 mg) |
8.3 Females and Males of Reproductive Potential
Based on animal data use of acetaminophen may cause reduced fertility in males and females of reproductive potential. It is not known whether these effects on fertility are reversible. Published animal studies reported that oral acetaminophen treatment of male animals at doses that are 1.2 times the MHDD and greater (based on a body surface area comparison) result in decreased testicular weights, reduced spermatogenesis, and reduced fertility. In female animals given the same doses, reduced implantation sites were reported. Additional published animal studies indicate that acetaminophen exposure in utero adversely impacts reproductive capacity of both male and female offspring at clinically relevant exposures [see Nonclinical Toxicology (13.1)].
7.1 Effects of Other Substances On Acetaminophen
Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. The clinical consequences of these effects have not been established. Effects of ethanol are complex, because excessive alcohol usage can induce hepatic cytochromes, but ethanol also acts as a competitive inhibitor of the metabolism of acetaminophen.
2.1 Important Dosing and Administration Information
- DO NOT ADD SUPPLEMENTARY MEDICATION.
- Aseptic technique is required.
- Before use, perform the following checks: Read the label. Ensure solution is the one ordered and is within the expiration date.
- Inspect the solution in good light for cloudiness, haze or particulate matter; check the container for leakage or damage. Any container which is suspect should not be used.
- Use only if solution is clear and container and seals are intact. Single-dose plastic container. Discard unused portion. Consult Package Insert for complete product information.
- DO NOT USE PLASTIC CONTAINER IN SERIES CONNECTION.
Acetaminophen Injection may be given as a single or repeated dose for the treatment of acute pain or fever. No dose adjustment is required when converting between oral acetaminophen and Acetaminophen Injection dosing in adults and adolescents who weigh 50 kg and above. Calculated maximum daily dose of acetaminophen is based on all routes of administration (i.e. intravenous, oral, and rectal) and all products containing acetaminophen. Exceeding the maximum mg/kg daily dose of acetaminophen as described in Tables 1 to 3 may result in hepatic injury, including the risk of liver failure and death. To avoid the risk of overdose, ensure that the total amount of acetaminophen from all routes and from all sources does not exceed the maximum recommended dose.
Warning: Risk of Medication Errors and Hepatotoxicity
Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that:
- the dose in milligrams (mg) and milliliters (mL) is not confused;
- the dosing is based on weight for patients under 50 kg;
- infusion pumps are properly programmed; and
- the total daily dose of acetaminophen from all sources does not exceed maximum daily limits.
Acetaminophen Injection contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the maximum daily limits, and often involve more than one acetaminophen-containing product [see Warnings and Precautions (5.1)] .
2.4 Recommended Dosage for Treatment of Fever in Neonates and Infants
Neonates, including premature neonates born at greater than or equal to 32 weeks gestational age, up to 28 days chronological age: the recommended dosage of Acetaminophen Injection is 12.5 mg /kg every 6 hours, to a maximum daily dose of acetaminophen of 50 mg/kg per day, with a minimum dosing interval of 6 hours.
Infants 29 days to 2 years of age: the recommended dosage of Acetaminophen Injection is 15 mg /kg every 6 hours, to a maximum daily dose of acetaminophen of 60 mg/kg per day, with a minimum dosing interval of 6 hours.
| Age Group | Dose given every 6 hours | Maximum total daily dose of acetaminophen (by all routes) |
| Neonates (birth to 28 days) | 12.5 mg/kg | 50 mg/kg |
| Infants (29 days to 2 years) | 15 mg/kg | 60 mg/kg |
2.5 Instructions for Intravenous Administration From Polypropylene Bag Container
DO NOT ADD SUPPLEMENTARY MEDICATION.
Aseptic technique is required.
Before use, perform the following checks: Read the label. Ensure solution is the one ordered and is within the expiration date.
Inspect the solution in good light for cloudiness, haze or particulate matter; check the container for leakage or damage. Any container which is suspect should not be used.
Use only if solution is clear and container and seals are intact. Single dose plastic container. Discard unused portion. Consult Package Insert for complete product information.
DO NOT USE PLASTIC CONTAINER IN SERIES CONNECTION.
This solution is intended for intravenous administration over 15 minutes using sterile equipment. It is recommended that intravenous administration apparatus be replaced at least once every 24 hours.
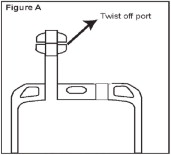
- Identify Twist-off Port (See Figure A).
- To Attach Administration Set
To aseptically remove the top portion of twist off port: hold container below the twist-off port and twist between the thumb and forefinger as shown in Figure B Steps 1 and 2. - Push spike through the diaphragm of the port (See Figure C
). Hang container using hole on the lower flap. Prime set in accordance with the Directions for Use provided with the set in use.
Polypropylene bag container can be safely transported in a standard 6-inch carrier through pneumatic tube system that is well maintained and running properly.
2.6 Instructions for Intravenous Administration of Doses Not Equivalent to 500 Mg Or 1,000 Mg
Doses that are not equivalent to 500 mg or 1,000 mg require aseptic transfer to a separate container prior to dispensing. Discard unused portion. Using aseptic technique, withdraw the appropriate dose (650 mg or weight-based) from an intact sealed polypropylene bag container and place the measured dose in a separate empty, sterile container (e.g., glass bottle, plastic intravenous container, or syringe) for intravenous infusion to avoid the inadvertent delivery and administration of the total volume of the commercially available container. The entire 100 mL container is not intended for use in patients weighing less than 50 kg. Acetaminophen injection is supplied in a single-dose container and the unused portion must be discarded.
Place small volume pediatric doses up to 60 mL in volume in a syringe and administer over 15 minutes using a syringe pump.
Structured Label Content
Section 42229-5 (42229-5)
Adult Population
A total of 1,020 adult patients have received Acetaminophen Injection in clinical trials, including 37.3% (n=380) who received 5 or more doses, and 17.0% (n=173) who received more than 10 doses. Most patients were treated with Acetaminophen Injection 1,000 mg every 6 hours. A total of 13.1% (n=134) received Acetaminophen Injection 650 mg every 4 hours.
All adverse reactions that occurred in adult patients treated with either Acetaminophen Injection or placebo in repeated dose, placebo-controlled clinical trials at an incidence greater than or equal to 3% and at a greater frequency than placebo are listed in Table 4. The most common adverse events in adult patients treated with Acetaminophen Injection (incidence greater than or equal to 5% and greater than placebo) were nausea, vomiting, headache, and insomnia.
|
*Pyrexia adverse reaction frequency data is included in order to alert healthcare practitioners that the antipyretic effects of Acetaminophen Injection may mask fever. |
||
| System Organ Class – Preferred Term | Acetaminophen Injection (N=402) n (%) |
Placebo (N=379)
n (%) |
|
Gastrointestinal Disorders
Nausea Vomiting |
138 (34) 62 (15) |
119 (31) 42 (11) |
|
General Disorders and Administration Site Conditions
Pyrexia* |
22 (5) |
52 (14) |
|
Nervous System Disorders
Headache |
39 (10) | 33 (9) |
|
Psychiatric Disorders
Insomnia |
30 (7) |
21 (5) |
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] For single dose only. Use the product within 18 hours after opening.
Do not freeze.
Protect from light. Retain in carton until time of use.
Use only if prepared solution is clear and free from particulate matter.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container closure is not made with natural rubber latex.
sagent
®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60173 (USA)
Made in India
©2024 Sagent Pharmaceuticals
October 2024
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-705-82
50 mL Single-Dose Container
Acetaminophen Injection
500 mg per 50 mL
(10 mg per mL)
Rx only
For Intravenous Use Only
CAUTIONS: DO NOT ADD SUPPLEMENTARY MEDICATION
11 Description (11 DESCRIPTION)
Acetaminophen is a non-salicylate antipyretic and non-opioid analgesic agent. Its chemical name is N-acetyl-p-aminophenol.
Acetaminophen has a molecular weight of 151.16. Its structural formula is:
Acetaminophen Injection is a sterile, clear, colorless, non pyrogenic, isotonic formulation of acetaminophen intended for intravenous infusion. It has a pH of approximately 4.5 to 5.5 and an osmolality of approximately between 270 mOsm/kg and 310 mOsm/kg.
Each 100 mL contains 1,000 mg Acetaminophen, USP, 3,800 mg Mannitol, USP, 30 mg Sodium Citrate Dihydrate USP, Water for Injection, USP qs.
Each 50 mL contains 500 mg Acetaminophen, USP, 1,900 mg Mannitol, USP, 15 mg Sodium Citrate Dihydrate USP, Water for Injection, USP qs.
pH is adjusted with glacial acetic acid.
14.2 Adult Fever
The efficacy of acetaminophen 1,000 mg in the treatment of adult fever was evaluated in one randomized, double-blind, placebo-controlled clinical trial. The study was a 6-hour, single-dose, endotoxin-induced fever study in 60 healthy adult males. A statistically significant antipyretic effect of acetaminophen was demonstrated through 6 hours in comparison to placebo. The mean temperature over time is shown in Figure 1.
8.5 Geriatric Use
Of the total number of subjects in clinical studies of acetaminophen, 15% were age 65 and over, while 5% were age 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.1 Hepatic Injury
Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death [see Overdosage (10)]. Do not exceed the maximum recommended daily dose of acetaminophen [see Dosage and Administration (2)]. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products.
Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e.g., due to dehydration or blood loss), or severe renal impairment (creatinine clearance less than or equal to 30 mL/min) [see Use In Specific Populations (8.6, 8.7)].
7.2 Anticoagulants
Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. As no studies have been performed evaluating the short-term use of Acetaminophen Injection in patients on oral anticoagulants, more frequent assessment of INR may be appropriate in such circumstances.
4 Contraindications (4 CONTRAINDICATIONS)
Acetaminophen is contraindicated:
- in patients with known hypersensitivity to acetaminophen or to any of the excipients in the intravenous formulation.
- in patients with severe hepatic impairment or severe active liver disease [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
7 Drug Interactions (7 DRUG INTERACTIONS)
- Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. (7.1)
- Chronic oral acetaminophen use at a dose of 4,000 mg/day has been shown to cause an increase in international normalized ratio (INR) in some patients who have been stabilized on sodium warfarin as an anticoagulant. (7.2)
12.2 Pharmacodynamics
Acetaminophen has been shown to have analgesic and antipyretic activities in animal and human studies. Single doses of acetaminophen up to 3,000 mg and repeated doses of 1,000 mg every 6 hours for 48 hours have not been shown to cause a significant effect on platelet aggregation. Acetaminophen does not have any immediate or delayed effects on small-vessel hemostasis. Clinical studies of both healthy subjects and patients with hemophilia showed no significant changes in bleeding time after receiving multiple doses of oral acetaminophen.
14.1 Adult Acute Pain
The efficacy of Acetaminophen Injection in the treatment of acute pain in adults was evaluated in two randomized, double-blind, placebo-controlled clinical trials in patients with postoperative pain.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Acetaminophen Injection is indicated for
- the management of mild to moderate pain in adult and pediatric patients 2 years and older
- the management of moderate to severe pain with adjunctive opioid analgesics in adult and pediatric patients 2 years and older
- the reduction of fever in adult and pediatric patients.
12.1 Mechanism of Action
The precise mechanism of the analgesic and antipyretic properties of acetaminophen is not established but is thought to primarily involve central actions.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Hepatic Injury: Administration of acetaminophen in doses higher than recommended (by all routes of administration and from all acetaminophen-containing products including combination products) may result in hepatic injury, including the risk of liver failure and death. (5.1)
- Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, in cases of alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment (creatinine clearance less than or equal to 30 mL/min). (5.1)
- Risk of Serious Skin or Hypersensitivity Reactions: Discontinue Acetaminophen Injection immediately at the first appearance of skin rash and if symptoms associated with allergy or hypersensitivity occur. Do not use in patients with acetaminophen allergy. (5.2, 5.4)
- Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. (5.3)
5.2 Serious Skin Reactions
Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. Patients should be informed about the signs of serious skin reactions, and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Acetaminophen Injection may be given as a single or repeated dose. (2.1)
- Acetaminophen Injection should be administered only as a 15-minute intravenous infusion. (2.5)
Adults and Adolescents Weighing 50 kg and Over:
- 1,000 mg every 6 hours or 650 mg every 4 hours to a maximum of 4,000 mg per day. Minimum dosing interval of 4 hours. (2.2)
Adults and Adolescents Weighing Under 50 kg:
- 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. (2.2)
Children:
- Children 2 to 12 years of age: 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours to a maximum of 75 mg/kg per day. Minimum dosing interval of 4 hours. (2.3)
Neonates and Infants:
- Neonates including premature neonates born at greater than or equal to 32 weeks gestational age to 28 days chronological age, 12.5 mg/kg every 6 hours to a maximum of 50 mg/kg per day. Minimum dosing interval of 6 hours. (2.4)
- Infants (29 days to 2 years of age): 15 mg/kg every 6 hours to a maximum of 60 mg/kg per day. Minimum dosing interval of 6 hours. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Acetaminophen Injection is a sterile, clear, colorless, non-pyrogenic, preservative free, isotonic formulation of acetaminophen available as:
- 1,000 mg acetaminophen in 100 mL (10 mg per mL) injection for intravenous infusion
- 500 mg acetaminophen in 50 mL (10 mg per mL) injection for intravenous infusion
5.3 Risk of Medication Errors
Take care when prescribing, preparing, and administering Acetaminophen Injection in order to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that:
- the dose in milligrams (mg) and milliliters (mL) is not confused;
- the dosing is based on weight for patients under 50 kg;
- infusion pumps are properly programmed; and
- the total daily dose of acetaminophen from all sources does not exceed maximum daily limits [see Dosage and Administration (2)].
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in practice.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatric Use: The effectiveness of acetaminophen for the treatment of acute pain in pediatric patients younger than 2 years of age has not been established. The safety and effectiveness of acetaminophen in pediatric patients is supported by evidence from adequate and well controlled studies in adults with additional safety and pharmacokinetic data for this age group. (8.4)
- Geriatric Use: No overall differences in safety or effectiveness were observed between geriatric and younger subjects. (8.5)
- Hepatic Impairment: Acetaminophen is contraindicated in patients with severe hepatic impairment or severe active liver disease and should be used with caution in patients with hepatic impairment or active liver disease. (4, 5.1, 8.6)
- Renal Impairment: In cases of severe renal impairment, longer dosing intervals and a reduced total daily dose of acetaminophen may be warranted. (5.1, 8.7)
2.3 Recommended Dosage: Children
Children 2 to 12 years of age: the recommended dosage of Acetaminophen Injection is 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours, with a maximum single dose of Acetaminophen Injection of 15 mg/kg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 75 mg/kg per day.
| Age group | Dose given every 4 hours | Dose given every 6 hours | Maximum single dose | Maximum total daily dose of acetaminophen (by all routes) |
| Children 2 to 12 years of age | 12.5 mg/kg | 15 mg/kg | 15 mg/kg (up to 750 mg) | 75 mg/kg in 24 hours (up to 3,750 mg) |
5.4 Allergy and Hypersensitivity
There have been post-marketing reports of hypersensitivity and anaphylaxis associated with the use of acetaminophen. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritus. There were infrequent reports of life-threatening anaphylaxis requiring emergent medical attention. Discontinue Acetaminophen Injection immediately if symptoms associated with allergy or hypersensitivity occur. Do not use Acetaminophen Injection in patients with acetaminophen allergy.
8.7 Patients With Renal Impairment (8.7 Patients with Renal Impairment)
In cases of severe renal impairment (creatinine clearance less than or equal to 30 mL/min), longer dosing intervals and a reduced total daily dose of acetaminophen may be warranted.
14.3 Pediatric Acute Pain and Fever
Acetaminophen was studied in pediatric patients in three active-controlled trials and three open-label safety and pharmacokinetic trials [see Use in Specific Populations (8.4)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Acetaminophen Injection (10 mg per mL) is a clear, colorless solution and is supplied sterile and nonpyrogenic in 50 mL and 100 mL fill polypropylene bag container as follows:
| NDC | Acetaminophen Injection (10 mg per mL) | Package Factor |
| 25021-705-82 | 500 mg per 50 mL Single-Dose Bag | 10 bags per carton |
| 25021-705-87 | 1,000 mg per 100 mL Single-Dose Bag | 20 bags per carton |
Do not remove unit from overwrap until ready for use.
To open, tear outer wrap at the notch and remove solution bag. After removing the outer wrap, check the container for minute leaks by squeezing the solution bag firmly. If leaks are found, discard the solution because the sterility may be impaired. A small amount of moisture may be present inside the outer wrap.
8.6 Patients With Hepatic Impairment (8.6 Patients with Hepatic Impairment)
Acetaminophen is contraindicated in patients with severe hepatic impairment or severe active liver disease and should be used with caution in patients with hepatic impairment or active liver disease [see Warnings and Precautions (5.1) and Clinical Pharmacology (12)]. A reduced total daily dose of acetaminophen may be warranted.
2.2 Recommended Dosage: Adults and Adolescents
Adults and adolescents weighing 50 kg and over: the recommended dosage of Acetaminophen Injection is 1,000 mg every 6 hours or 650 mg every 4 hours, with a maximum single dose of Acetaminophen Injection of 1,000 mg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 4,000 mg per day (includes all routes of administration and all acetaminophen-containing products including combination products).
Adults and adolescents weighing under 50 kg: the recommended dosage of Acetaminophen Injection is 15 mg/kg every 6 hours or 12.5 mg/kg every 4 hours, with a maximum single dose of Acetaminophen Injection of 15 mg/kg, a minimum dosing interval of 4 hours, and a maximum daily dose of acetaminophen of 75 mg/kg per day (includes all routes of administration and all acetaminophen-containing products including combination products).
| Age group | Dose given every 4 hours | Dose given every 6 hours | Maximum single dose | Maximum total daily dose of acetaminophen (by all routes) |
| Adults and adolescents (13 years and older) weighing greater than or equal to 50 kg | 650 mg | 1,000 mg | 1,000 mg | 4,000 mg in 24 hours |
| Adults and adolescents (13 years and older) weighing less than 50 kg | 12.5 mg/kg | 15 mg/kg | 15 mg/kg (up to 750 mg) | 75 mg/kg in 24 hours (up to 3,750 mg) |
8.3 Females and Males of Reproductive Potential
Based on animal data use of acetaminophen may cause reduced fertility in males and females of reproductive potential. It is not known whether these effects on fertility are reversible. Published animal studies reported that oral acetaminophen treatment of male animals at doses that are 1.2 times the MHDD and greater (based on a body surface area comparison) result in decreased testicular weights, reduced spermatogenesis, and reduced fertility. In female animals given the same doses, reduced implantation sites were reported. Additional published animal studies indicate that acetaminophen exposure in utero adversely impacts reproductive capacity of both male and female offspring at clinically relevant exposures [see Nonclinical Toxicology (13.1)].
7.1 Effects of Other Substances On Acetaminophen (7.1 Effects of Other Substances on Acetaminophen)
Substances that induce or regulate hepatic cytochrome enzyme CYP2E1 may alter the metabolism of acetaminophen and increase its hepatotoxic potential. The clinical consequences of these effects have not been established. Effects of ethanol are complex, because excessive alcohol usage can induce hepatic cytochromes, but ethanol also acts as a competitive inhibitor of the metabolism of acetaminophen.
2.1 Important Dosing and Administration Information
- DO NOT ADD SUPPLEMENTARY MEDICATION.
- Aseptic technique is required.
- Before use, perform the following checks: Read the label. Ensure solution is the one ordered and is within the expiration date.
- Inspect the solution in good light for cloudiness, haze or particulate matter; check the container for leakage or damage. Any container which is suspect should not be used.
- Use only if solution is clear and container and seals are intact. Single-dose plastic container. Discard unused portion. Consult Package Insert for complete product information.
- DO NOT USE PLASTIC CONTAINER IN SERIES CONNECTION.
Acetaminophen Injection may be given as a single or repeated dose for the treatment of acute pain or fever. No dose adjustment is required when converting between oral acetaminophen and Acetaminophen Injection dosing in adults and adolescents who weigh 50 kg and above. Calculated maximum daily dose of acetaminophen is based on all routes of administration (i.e. intravenous, oral, and rectal) and all products containing acetaminophen. Exceeding the maximum mg/kg daily dose of acetaminophen as described in Tables 1 to 3 may result in hepatic injury, including the risk of liver failure and death. To avoid the risk of overdose, ensure that the total amount of acetaminophen from all routes and from all sources does not exceed the maximum recommended dose.
Warning: Risk of Medication Errors and Hepatotoxicity (WARNING: RISK OF MEDICATION ERRORS AND HEPATOTOXICITY)
Take care when prescribing, preparing, and administering Acetaminophen Injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that:
- the dose in milligrams (mg) and milliliters (mL) is not confused;
- the dosing is based on weight for patients under 50 kg;
- infusion pumps are properly programmed; and
- the total daily dose of acetaminophen from all sources does not exceed maximum daily limits.
Acetaminophen Injection contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the maximum daily limits, and often involve more than one acetaminophen-containing product [see Warnings and Precautions (5.1)] .
2.4 Recommended Dosage for Treatment of Fever in Neonates and Infants (2.4 Recommended Dosage For Treatment of Fever in Neonates and Infants)
Neonates, including premature neonates born at greater than or equal to 32 weeks gestational age, up to 28 days chronological age: the recommended dosage of Acetaminophen Injection is 12.5 mg /kg every 6 hours, to a maximum daily dose of acetaminophen of 50 mg/kg per day, with a minimum dosing interval of 6 hours.
Infants 29 days to 2 years of age: the recommended dosage of Acetaminophen Injection is 15 mg /kg every 6 hours, to a maximum daily dose of acetaminophen of 60 mg/kg per day, with a minimum dosing interval of 6 hours.
| Age Group | Dose given every 6 hours | Maximum total daily dose of acetaminophen (by all routes) |
| Neonates (birth to 28 days) | 12.5 mg/kg | 50 mg/kg |
| Infants (29 days to 2 years) | 15 mg/kg | 60 mg/kg |
2.5 Instructions for Intravenous Administration From Polypropylene Bag Container (2.5 Instructions for Intravenous Administration from polypropylene bag container)
DO NOT ADD SUPPLEMENTARY MEDICATION.
Aseptic technique is required.
Before use, perform the following checks: Read the label. Ensure solution is the one ordered and is within the expiration date.
Inspect the solution in good light for cloudiness, haze or particulate matter; check the container for leakage or damage. Any container which is suspect should not be used.
Use only if solution is clear and container and seals are intact. Single dose plastic container. Discard unused portion. Consult Package Insert for complete product information.
DO NOT USE PLASTIC CONTAINER IN SERIES CONNECTION.
This solution is intended for intravenous administration over 15 minutes using sterile equipment. It is recommended that intravenous administration apparatus be replaced at least once every 24 hours.
- Identify Twist-off Port (See Figure A).
- To Attach Administration Set
To aseptically remove the top portion of twist off port: hold container below the twist-off port and twist between the thumb and forefinger as shown in Figure B Steps 1 and 2. - Push spike through the diaphragm of the port (See Figure C
). Hang container using hole on the lower flap. Prime set in accordance with the Directions for Use provided with the set in use.
Polypropylene bag container can be safely transported in a standard 6-inch carrier through pneumatic tube system that is well maintained and running properly.
2.6 Instructions for Intravenous Administration of Doses Not Equivalent to 500 Mg Or 1,000 Mg (2.6 Instructions for Intravenous Administration of Doses Not Equivalent to 500 mg or 1,000 mg)
Doses that are not equivalent to 500 mg or 1,000 mg require aseptic transfer to a separate container prior to dispensing. Discard unused portion. Using aseptic technique, withdraw the appropriate dose (650 mg or weight-based) from an intact sealed polypropylene bag container and place the measured dose in a separate empty, sterile container (e.g., glass bottle, plastic intravenous container, or syringe) for intravenous infusion to avoid the inadvertent delivery and administration of the total volume of the commercially available container. The entire 100 mL container is not intended for use in patients weighing less than 50 kg. Acetaminophen injection is supplied in a single-dose container and the unused portion must be discarded.
Place small volume pediatric doses up to 60 mL in volume in a syringe and administer over 15 minutes using a syringe pump.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:11.307100 · Updated: 2026-03-14T22:29:13.416831