These Highlights Do Not Include All The Information Needed To Use Chemet Safely And Effectively. See Full Prescribing Information For Chemet.
62035612-9505-3a3f-1ac8-e2dbd711d24e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
Indications and Usage
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
Dosage and Administration
See Full Prescribing Information for important pretreatment evaluations. ( 2.1 ) Ensure patients receiving CHEMET are adequately hydrated. ( 2.3 ) Administer CHEMET capsules whole when possible. ( 2.3 ) Pediatric patients who cannot swallow whole capsules: Sprinkle contents of capsule in food (or on a spoon followed by a drink). ( 2.3 ) Recommended Dosage: 10 mg/kg or 350 mg/m 2 orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2 orally every 12 hours for an additional 14 days. ( 2.2 )
Warnings and Precautions
Hypersensitivity and dermatologic reactions: Interrupt treatment if rash or mucocutaneous vesicular eruptions occur. ( 5.1 ) Neutropenia: Monitor complete blood counts, interrupt treatment for ANC below 1200/mcL, and monitor for infection. ( 5.2 ) Hepatic Toxicity: Monitor hepatic transaminases (ALT/AST); interrupt treatment if above 5 times ULN. ( 5.3 ) Embryo-Fetal Toxicity: May cause fetal harm when administered to a pregnant woman. ( 5.5 )
Contraindications
CHEMET is contraindicated in patients with a history of hypersensitivity reaction to succimer. Reactions have included mucocutaneous vesicular eruptions, urticaria, and angioedema [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity and Dermatologic Reactions [see Warnings and Precautions (5.1) ] Neutropenia [see Warnings and Precautions (5.2) ] Hepatic Toxicity [see Warnings and Precautions (5.3) ]
Drug Interactions
CHEMET may interfere with serum and urinary laboratory test. ( 7.1 )
Storage and Handling
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
How Supplied
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
Medication Information
Warnings and Precautions
Hypersensitivity and dermatologic reactions: Interrupt treatment if rash or mucocutaneous vesicular eruptions occur. ( 5.1 ) Neutropenia: Monitor complete blood counts, interrupt treatment for ANC below 1200/mcL, and monitor for infection. ( 5.2 ) Hepatic Toxicity: Monitor hepatic transaminases (ALT/AST); interrupt treatment if above 5 times ULN. ( 5.3 ) Embryo-Fetal Toxicity: May cause fetal harm when administered to a pregnant woman. ( 5.5 )
Indications and Usage
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
Dosage and Administration
See Full Prescribing Information for important pretreatment evaluations. ( 2.1 ) Ensure patients receiving CHEMET are adequately hydrated. ( 2.3 ) Administer CHEMET capsules whole when possible. ( 2.3 ) Pediatric patients who cannot swallow whole capsules: Sprinkle contents of capsule in food (or on a spoon followed by a drink). ( 2.3 ) Recommended Dosage: 10 mg/kg or 350 mg/m 2 orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2 orally every 12 hours for an additional 14 days. ( 2.2 )
Contraindications
CHEMET is contraindicated in patients with a history of hypersensitivity reaction to succimer. Reactions have included mucocutaneous vesicular eruptions, urticaria, and angioedema [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity and Dermatologic Reactions [see Warnings and Precautions (5.1) ] Neutropenia [see Warnings and Precautions (5.2) ] Hepatic Toxicity [see Warnings and Precautions (5.3) ]
Drug Interactions
CHEMET may interfere with serum and urinary laboratory test. ( 7.1 )
Storage and Handling
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
How Supplied
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
Description
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
Section 42229-5
Limitations of Use
- CHEMET is not indicated for prophylaxis of lead poisoning in a lead-containing environment.
- CHEMET does not cross the blood-brain barrier and is not indicated to treat encephalopathy associated with lead toxicity.
Section 44425-7
Store between 15°C and 25°C and avoid excessive heat.
Dispense in tight, light-resistant container.
10 Overdosage
Doses of 2300 mg/kg in the rat and 2400 mg/kg in the mouse produced ataxia, convulsions, labored respiration and frequently death. Limited data indicate that CHEMET is dialyzable. In case of acute overdosage, consider use of induction of vomiting or gastric lavage followed by administration of an activated charcoal slurry and appropriate supportive therapy.
11 Description
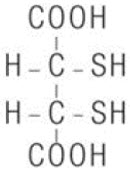
CHEMET (succimer) is an orally active, lead chelating agent. The chemical name for succimer is meso2, 3-dimercaptosuccinic acid (DMSA). Its empirical formula is C 4H 6O 4S 2and molecular weight is 182.2. The meso-structural formula is:
Succimer is a white crystalline powder with an unpleasant, characteristic mercaptan odor and taste.
Each CHEMET opaque white capsule for oral administration contains medicated beads with 100 mg of succimer and the following inactive ingredients: povidone, sodium starch glycolate, and sugar spheres. The capsule shell contains benzyl alcohol, butylparaben, edetate calcium disodium, gelatin, methylparaben, propylparaben, sodium lauryl sulfate, sodium propionate, titanium dioxide, and is imprinted with edible black ink.
5.2 Neutropenia
Iron chelators, including CHEMET, can cause neutropenia. Monitoring of complete blood counts is recommended [see Dosage and Administration (2.2)] . Interrupt treatment if absolute neutrophil count (ANC) is <1200/mcL and interrupt treatment until recovery to above 1500/mcL (or the patient's baseline count). Only rechallenge patients who developed neutropenia with CHEMET therapy if the benefit clearly outweighs the potential risk. If rechallenge is attempted, monitor CBC more frequently.
Monitor for signs and symptoms of infection and immediately discontinue CHEMET if they develop.
8.4 Pediatric Use
The safety and effectiveness of CHEMET for the treatment of lead poisoning in patients with blood levels above 45 mcg/mL have been established in pediatric patients aged 1 year and older. The safety and effectiveness of CHEMET have not been established in pediatric patients younger than 1 year of age.
14 Clinical Studies
The efficacy of CHEMET in the treatment of lead poisoning in pediatric patients was established in a dose-ranging, actively controlled study of 15 pediatric patients aged 2 to 7 years with blood lead levels of 30-49 mcg/dL and positive CaNa 2EDTA lead mobilization tests. Fifteen patients were assigned to a dose of 350 mg/m 2or 233 mg/m 2, or 116 mg/m 2(5 patients per group) orally every 8 hours for 5 days. Six control patients received 1000 mg/m 2/day CaNa 2EDTA intravenously for 5 days. Following therapy, the mean blood lead levels decreased 78, 63, and 42% respectively in the three CHEMET treatment groups. The response of the 350 mg/m 2every 8 hours (10 mg/kg every 8 hours) group was significantly better than that of the other CHEMET dose level groups as well as that of the control group, whose mean blood lead level fell 48%. Although other dosing regimens were used in the study described, only 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days is the recommended dosage.
Patients experienced a rebound in blood lead levels after discontinuation of CHEMET. In these studies, after treatment with a dose of 350 mg/m 2(10 mg/kg) every 8 hours for five days, the mean lead level rebounded and plateaued at 60-85% of pretreatment levels two weeks after therapy.
In an attempt to control rebound of blood lead levels, 19 pediatric patients, ages 1 to 7 years, with blood lead levels of 42-67 mcg/dL, were treated with 350 mg/m 2CHEMET every 8 hours for five days and then divided into three groups. One group was followed for two weeks with no further therapy, the second group was treated for two weeks with 350 mg/m 2daily, and the third with 350 mg/m 2every 12 hours. After the initial 5 days of therapy, the mean blood lead level in all subjects declined 61%. While the untreated group and the group treated with 350 mg/m 2daily experienced rebound during the ensuing two weeks, the group who received the 350 mg/m 2every 12 hours experienced no such rebound during the treatment period and less rebound following cessation of therapy.
In another study, ten pediatric patients, ages 21 to 72 months, with blood lead levels of 30-57 mcg/dL were treated with CHEMET 350 mg/m 2every eight hours for five days followed by an additional 19-22 days of therapy at a dose of 350 mg/m 2every 12 hours. The mean blood lead levels decreased and remained stable at under 15 mcg/dL during the extended dosing period.
In addition to the controlled studies, approximately 250 patients with lead poisoning have been treated with CHEMET either orally or parenterally in open U.S. and foreign studies.
4 Contraindications
CHEMET is contraindicated in patients with a history of hypersensitivity reaction to succimer. Reactions have included mucocutaneous vesicular eruptions, urticaria, and angioedema [see Warnings and Precautions (5.1)] .
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity and Dermatologic Reactions [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Hepatic Toxicity [see Warnings and Precautions (5.3)]
7 Drug Interactions
CHEMET may interfere with serum and urinary laboratory test. ( 7.1)
5.3 Hepatic Toxicity
Elevated transaminases (ALT/AST) occurred in 6-10% of patients treated with CHEMET. Monitor serum AST and ALT at baseline and at least weekly during treatment. Monitor patients with a history of liver disease more frequently. Serum aminotransferase elevations above 5 times the upper limit of normal (if confirmed) should lead to dose reduction or temporary cessation.
8.5 Renal Impairment
Assess renal function prior to and periodically during prolonged therapy. Adequately hydrate patients during therapy. Limited data suggests that succimer is dialyzable, but the lead chelates are not. Monitor patients with a history of renal impairment more frequently.
2.2 Recommended Dosage
The recommended dosage of CHEMET for pediatric patients with lead poisoning is 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days [see Table 1 CHEMET Pediatric Dosing Chart]. Initiation of therapy at higher doses is not recommended. The total treatment course consists of 19 days.
After discontinuation of CHEMET, elevated blood levels and associated symptoms may return rapidly because of redistribution of lead from bone stores to soft tissues and blood. Assess blood lead concentration after the completion of a 19 day course and every week until stable. Repeated courses may be administered after two weeks off treatment if blood lead concentrations remain elevated. A minimum of two weeks between treatment courses is recommended unless blood lead concentrations indicate the need for more prompt treatment.
| Weight in Kilograms (kg) | Dose (mg) | Number of Capsules |
|---|---|---|
| 8 to15 kg | 100 mg | 1 |
| 16 to 23 kg | 200 mg | 2 |
| 24 to 34 kg | 300 mg | 3 |
| 35 to 44 kg | 400 mg | 4 |
| >45 kg | 500 mg | 5 |
The safety of uninterrupted dosing longer than 3 weeks has not been established and is not recommended.
8.6 Hepatic Impairment
Assess hepatic function prior to and periodically during therapy. Monitor patients with a history of liver disease more frequently [see Warnings and Precautions (5.3)] .
1 Indications and Usage
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
12.1 Mechanism of Action
Succimer is a lead chelator; it forms water soluble chelates and, consequently, increases the urinary excretion of lead.
5.4 Embryo Fetal Toxicity
Based on findings from animal reproduction studies, CHEMET may cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use an effective method of contraception during treatment with CHEMET and for 14 days after the final dose [see Use in Specific Populations (8.1, 8.3)] .
5 Warnings and Precautions
- Hypersensitivity and dermatologic reactions: Interrupt treatment if rash or mucocutaneous vesicular eruptions occur. ( 5.1)
- Neutropenia: Monitor complete blood counts, interrupt treatment for ANC below 1200/mcL, and monitor for infection. ( 5.2)
- Hepatic Toxicity: Monitor hepatic transaminases (ALT/AST); interrupt treatment if above 5 times ULN. ( 5.3)
- Embryo-Fetal Toxicity: May cause fetal harm when administered to a pregnant woman. ( 5.5)
2 Dosage and Administration
- See Full Prescribing Information for important pretreatment evaluations. ( 2.1)
- Ensure patients receiving CHEMET are adequately hydrated. ( 2.3)
- Administer CHEMET capsules whole when possible. ( 2.3)
- Pediatric patients who cannot swallow whole capsules: Sprinkle contents of capsule in food (or on a spoon followed by a drink). ( 2.3)
- Recommended Dosage: 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days. ( 2.2)
3 Dosage Forms and Strengths
Capsules: 100 mg, opaque white, imprinted black with "CHEMET 100".
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Table 2 presents the adverse reactions associated with Chemet in pediatric patients.
| Body System: Adverse Reactions | Pediatric Patients
(n=191) |
|---|---|
| Digestive
Nausea, vomiting, diarrhea, appetite loss, hemorrhoidal symptoms, metallic taste in mouth
|
12% |
| Body as a Whole
Back pain, abdominal cramps, stomach pains, head pain, rib pain, chills, flank pain, fever, flu-like symptoms, heavy head/tired, head cold, headache, moniliasis.
|
5% |
| Metabolic
Elevated ALT or AST, alkaline phosphatase, serum cholesterol.
|
4% |
| Respiratory
Sore throat, rhinorrhea, nasal congestion, cough.
|
4% |
| Skin
Papular rash, herpetic rash, rash, mucocutaneous eruptions, pruritis.
|
3% |
| Nervous
Drowsiness, dizziness, sensorimotor neuropathy, sleepiness, paresthesia.
|
1% |
| Special Senses
Cloudy film in eye, ears plugged, otitis media, eyes watery.
|
1% |
| Heme/Lymphatic
Neutropenia, increased platelet count, eosinophilia
|
1% |
5.5 Laboratory Test Interference
CHEMET may interfere with serum and urinary laboratory tests [see Drug Interactions (7.1)] .
7.1 Laboratory Test Interference
CHEMET may interfere with serum and urinary laboratory tests. In vitro studies have shown CHEMET to cause false positive results for ketones in urine using nitroprusside reagents and falsely decreased measurements of serum uric acid and creatinine phosphokinase (CPK).
17 Patient Counseling Information
- Advise caregivers and/or patients to maintain adequate fluid intake [see Dosage and Administration (2.3)].
- Advise caregivers and/or patients to contact their healthcare provider should a rash develop [see Warnings and Precautions (5.1)] .
- Advise caregivers and/or patients to contact their healthcare provider right away if signs/symptoms of infection develop [see Warnings and Precautions (5.2)] .
- Advise caregivers that in pediatric patients who cannot swallow the capsules whole, separate the capsule and sprinkle the medicated beads on a small amount of soft food or put them in a spoon and follow with a fruit drink [see Dosage and Administration (2.3)].
16 How Supplied/storage and Handling
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
2.1 Important Pretreatment Evaluations
- Identify the source of lead in the pediatric patient's environment and eliminate the source prior to beginning treatment with CHEMET.
- Assess the following before initiating treatment with CHEMET:
- Blood lead concentration
- Complete blood count (CBC) with differential and platelets [see Dosage and Administration (2.2), Warnings and Precautions (5.2)]
- Ensure absolute neutrophil count (ANC) > 1500/mcL [see Dosage and Administration (2.2)]
- Transaminases (AST/ALT) [see Warnings and Precautions (5.3)and Use in Specific Populations (8.6)]
- Renal function with blood urea nitrogen (BUN), creatinine, urinary protein [see Use in Specific Populations (8.5)] .
- Patients who have previously received Edetate calcium disodium (CaNa 2EDTA) with or without dimercaprol may receive CHEMET for subsequent treatment after an interval of four weeks.
7.2 Use With Other Chelation Therapies
Concomitant administration of CHEMET with other chelation therapy, such as CaNa 2EDTA is not recommended.
2.3 Preparation and Administration Instructions
Administer CHEMET capsules whole.
In pediatric patients who cannot swallow the capsules whole, separate the capsule and sprinkle the medicated beads on a small amount of soft food or put them in a spoon and follow with a fruit drink.
Ensure that all patients receiving CHEMET are adequately hydrated [see Use in Specific Populations (8.5), Pharmacokinetics (12.3)] .
5.1 Hypersensitivity and Dermatologic Reactions
CHEMET can cause hypersensitivity reactions and dermatologic reactions.
8.3 Females and Males of Reproductive Potential
CHEMET may cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)] .
Principal Display Panel 100 Mg Capsule Bottle Label
NDC 55292-201-11
100 Capsules
Chemet
®
(succimer) Capsules
100 mg
RECORDATI
RARE DISEASES
GROUP
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with CHEMET. Succimer was not mutagenic in the Ames bacterial reverse mutation assay and in the mammalian cell forward gene mutation assay. Succimer did not show any adverse effects on fertility and reproductive performance in rats up to doses of 510 mg/kg/day in males and 100 mg/kg/day in females (7- and 11-times the MRHD based on BSA, respectively).
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- CHEMET is not indicated for prophylaxis of lead poisoning in a lead-containing environment.
- CHEMET does not cross the blood-brain barrier and is not indicated to treat encephalopathy associated with lead toxicity.
Section 44425-7 (44425-7)
Store between 15°C and 25°C and avoid excessive heat.
Dispense in tight, light-resistant container.
10 Overdosage (10 OVERDOSAGE)
Doses of 2300 mg/kg in the rat and 2400 mg/kg in the mouse produced ataxia, convulsions, labored respiration and frequently death. Limited data indicate that CHEMET is dialyzable. In case of acute overdosage, consider use of induction of vomiting or gastric lavage followed by administration of an activated charcoal slurry and appropriate supportive therapy.
11 Description (11 DESCRIPTION)
CHEMET (succimer) is an orally active, lead chelating agent. The chemical name for succimer is meso2, 3-dimercaptosuccinic acid (DMSA). Its empirical formula is C 4H 6O 4S 2and molecular weight is 182.2. The meso-structural formula is:
Succimer is a white crystalline powder with an unpleasant, characteristic mercaptan odor and taste.
Each CHEMET opaque white capsule for oral administration contains medicated beads with 100 mg of succimer and the following inactive ingredients: povidone, sodium starch glycolate, and sugar spheres. The capsule shell contains benzyl alcohol, butylparaben, edetate calcium disodium, gelatin, methylparaben, propylparaben, sodium lauryl sulfate, sodium propionate, titanium dioxide, and is imprinted with edible black ink.
5.2 Neutropenia
Iron chelators, including CHEMET, can cause neutropenia. Monitoring of complete blood counts is recommended [see Dosage and Administration (2.2)] . Interrupt treatment if absolute neutrophil count (ANC) is <1200/mcL and interrupt treatment until recovery to above 1500/mcL (or the patient's baseline count). Only rechallenge patients who developed neutropenia with CHEMET therapy if the benefit clearly outweighs the potential risk. If rechallenge is attempted, monitor CBC more frequently.
Monitor for signs and symptoms of infection and immediately discontinue CHEMET if they develop.
8.4 Pediatric Use
The safety and effectiveness of CHEMET for the treatment of lead poisoning in patients with blood levels above 45 mcg/mL have been established in pediatric patients aged 1 year and older. The safety and effectiveness of CHEMET have not been established in pediatric patients younger than 1 year of age.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of CHEMET in the treatment of lead poisoning in pediatric patients was established in a dose-ranging, actively controlled study of 15 pediatric patients aged 2 to 7 years with blood lead levels of 30-49 mcg/dL and positive CaNa 2EDTA lead mobilization tests. Fifteen patients were assigned to a dose of 350 mg/m 2or 233 mg/m 2, or 116 mg/m 2(5 patients per group) orally every 8 hours for 5 days. Six control patients received 1000 mg/m 2/day CaNa 2EDTA intravenously for 5 days. Following therapy, the mean blood lead levels decreased 78, 63, and 42% respectively in the three CHEMET treatment groups. The response of the 350 mg/m 2every 8 hours (10 mg/kg every 8 hours) group was significantly better than that of the other CHEMET dose level groups as well as that of the control group, whose mean blood lead level fell 48%. Although other dosing regimens were used in the study described, only 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days is the recommended dosage.
Patients experienced a rebound in blood lead levels after discontinuation of CHEMET. In these studies, after treatment with a dose of 350 mg/m 2(10 mg/kg) every 8 hours for five days, the mean lead level rebounded and plateaued at 60-85% of pretreatment levels two weeks after therapy.
In an attempt to control rebound of blood lead levels, 19 pediatric patients, ages 1 to 7 years, with blood lead levels of 42-67 mcg/dL, were treated with 350 mg/m 2CHEMET every 8 hours for five days and then divided into three groups. One group was followed for two weeks with no further therapy, the second group was treated for two weeks with 350 mg/m 2daily, and the third with 350 mg/m 2every 12 hours. After the initial 5 days of therapy, the mean blood lead level in all subjects declined 61%. While the untreated group and the group treated with 350 mg/m 2daily experienced rebound during the ensuing two weeks, the group who received the 350 mg/m 2every 12 hours experienced no such rebound during the treatment period and less rebound following cessation of therapy.
In another study, ten pediatric patients, ages 21 to 72 months, with blood lead levels of 30-57 mcg/dL were treated with CHEMET 350 mg/m 2every eight hours for five days followed by an additional 19-22 days of therapy at a dose of 350 mg/m 2every 12 hours. The mean blood lead levels decreased and remained stable at under 15 mcg/dL during the extended dosing period.
In addition to the controlled studies, approximately 250 patients with lead poisoning have been treated with CHEMET either orally or parenterally in open U.S. and foreign studies.
4 Contraindications (4 CONTRAINDICATIONS)
CHEMET is contraindicated in patients with a history of hypersensitivity reaction to succimer. Reactions have included mucocutaneous vesicular eruptions, urticaria, and angioedema [see Warnings and Precautions (5.1)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity and Dermatologic Reactions [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Hepatic Toxicity [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
CHEMET may interfere with serum and urinary laboratory test. ( 7.1)
5.3 Hepatic Toxicity
Elevated transaminases (ALT/AST) occurred in 6-10% of patients treated with CHEMET. Monitor serum AST and ALT at baseline and at least weekly during treatment. Monitor patients with a history of liver disease more frequently. Serum aminotransferase elevations above 5 times the upper limit of normal (if confirmed) should lead to dose reduction or temporary cessation.
8.5 Renal Impairment
Assess renal function prior to and periodically during prolonged therapy. Adequately hydrate patients during therapy. Limited data suggests that succimer is dialyzable, but the lead chelates are not. Monitor patients with a history of renal impairment more frequently.
2.2 Recommended Dosage
The recommended dosage of CHEMET for pediatric patients with lead poisoning is 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days [see Table 1 CHEMET Pediatric Dosing Chart]. Initiation of therapy at higher doses is not recommended. The total treatment course consists of 19 days.
After discontinuation of CHEMET, elevated blood levels and associated symptoms may return rapidly because of redistribution of lead from bone stores to soft tissues and blood. Assess blood lead concentration after the completion of a 19 day course and every week until stable. Repeated courses may be administered after two weeks off treatment if blood lead concentrations remain elevated. A minimum of two weeks between treatment courses is recommended unless blood lead concentrations indicate the need for more prompt treatment.
| Weight in Kilograms (kg) | Dose (mg) | Number of Capsules |
|---|---|---|
| 8 to15 kg | 100 mg | 1 |
| 16 to 23 kg | 200 mg | 2 |
| 24 to 34 kg | 300 mg | 3 |
| 35 to 44 kg | 400 mg | 4 |
| >45 kg | 500 mg | 5 |
The safety of uninterrupted dosing longer than 3 weeks has not been established and is not recommended.
8.6 Hepatic Impairment
Assess hepatic function prior to and periodically during therapy. Monitor patients with a history of liver disease more frequently [see Warnings and Precautions (5.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
CHEMET is indicated for the treatment of lead poisoning in pediatric patients aged 1 year and older with blood lead levels above 45 mcg/dL.
12.1 Mechanism of Action
Succimer is a lead chelator; it forms water soluble chelates and, consequently, increases the urinary excretion of lead.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on findings from animal reproduction studies, CHEMET may cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use an effective method of contraception during treatment with CHEMET and for 14 days after the final dose [see Use in Specific Populations (8.1, 8.3)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity and dermatologic reactions: Interrupt treatment if rash or mucocutaneous vesicular eruptions occur. ( 5.1)
- Neutropenia: Monitor complete blood counts, interrupt treatment for ANC below 1200/mcL, and monitor for infection. ( 5.2)
- Hepatic Toxicity: Monitor hepatic transaminases (ALT/AST); interrupt treatment if above 5 times ULN. ( 5.3)
- Embryo-Fetal Toxicity: May cause fetal harm when administered to a pregnant woman. ( 5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- See Full Prescribing Information for important pretreatment evaluations. ( 2.1)
- Ensure patients receiving CHEMET are adequately hydrated. ( 2.3)
- Administer CHEMET capsules whole when possible. ( 2.3)
- Pediatric patients who cannot swallow whole capsules: Sprinkle contents of capsule in food (or on a spoon followed by a drink). ( 2.3)
- Recommended Dosage: 10 mg/kg or 350 mg/m 2orally every 8 hours for five days followed by 10 mg/kg or 350 mg/m 2orally every 12 hours for an additional 14 days. ( 2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 100 mg, opaque white, imprinted black with "CHEMET 100".
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Table 2 presents the adverse reactions associated with Chemet in pediatric patients.
| Body System: Adverse Reactions | Pediatric Patients
(n=191) |
|---|---|
| Digestive
Nausea, vomiting, diarrhea, appetite loss, hemorrhoidal symptoms, metallic taste in mouth
|
12% |
| Body as a Whole
Back pain, abdominal cramps, stomach pains, head pain, rib pain, chills, flank pain, fever, flu-like symptoms, heavy head/tired, head cold, headache, moniliasis.
|
5% |
| Metabolic
Elevated ALT or AST, alkaline phosphatase, serum cholesterol.
|
4% |
| Respiratory
Sore throat, rhinorrhea, nasal congestion, cough.
|
4% |
| Skin
Papular rash, herpetic rash, rash, mucocutaneous eruptions, pruritis.
|
3% |
| Nervous
Drowsiness, dizziness, sensorimotor neuropathy, sleepiness, paresthesia.
|
1% |
| Special Senses
Cloudy film in eye, ears plugged, otitis media, eyes watery.
|
1% |
| Heme/Lymphatic
Neutropenia, increased platelet count, eosinophilia
|
1% |
5.5 Laboratory Test Interference
CHEMET may interfere with serum and urinary laboratory tests [see Drug Interactions (7.1)] .
7.1 Laboratory Test Interference
CHEMET may interfere with serum and urinary laboratory tests. In vitro studies have shown CHEMET to cause false positive results for ketones in urine using nitroprusside reagents and falsely decreased measurements of serum uric acid and creatinine phosphokinase (CPK).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Advise caregivers and/or patients to maintain adequate fluid intake [see Dosage and Administration (2.3)].
- Advise caregivers and/or patients to contact their healthcare provider should a rash develop [see Warnings and Precautions (5.1)] .
- Advise caregivers and/or patients to contact their healthcare provider right away if signs/symptoms of infection develop [see Warnings and Precautions (5.2)] .
- Advise caregivers that in pediatric patients who cannot swallow the capsules whole, separate the capsule and sprinkle the medicated beads on a small amount of soft food or put them in a spoon and follow with a fruit drink [see Dosage and Administration (2.3)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Capsules: 100 mg of CHEMET (succimer), imprinted black with "CHEMET 100", in a bottle of 100 (NDC 55292-201-11).
2.1 Important Pretreatment Evaluations
- Identify the source of lead in the pediatric patient's environment and eliminate the source prior to beginning treatment with CHEMET.
- Assess the following before initiating treatment with CHEMET:
- Blood lead concentration
- Complete blood count (CBC) with differential and platelets [see Dosage and Administration (2.2), Warnings and Precautions (5.2)]
- Ensure absolute neutrophil count (ANC) > 1500/mcL [see Dosage and Administration (2.2)]
- Transaminases (AST/ALT) [see Warnings and Precautions (5.3)and Use in Specific Populations (8.6)]
- Renal function with blood urea nitrogen (BUN), creatinine, urinary protein [see Use in Specific Populations (8.5)] .
- Patients who have previously received Edetate calcium disodium (CaNa 2EDTA) with or without dimercaprol may receive CHEMET for subsequent treatment after an interval of four weeks.
7.2 Use With Other Chelation Therapies (7.2 Use with Other Chelation Therapies)
Concomitant administration of CHEMET with other chelation therapy, such as CaNa 2EDTA is not recommended.
2.3 Preparation and Administration Instructions
Administer CHEMET capsules whole.
In pediatric patients who cannot swallow the capsules whole, separate the capsule and sprinkle the medicated beads on a small amount of soft food or put them in a spoon and follow with a fruit drink.
Ensure that all patients receiving CHEMET are adequately hydrated [see Use in Specific Populations (8.5), Pharmacokinetics (12.3)] .
5.1 Hypersensitivity and Dermatologic Reactions
CHEMET can cause hypersensitivity reactions and dermatologic reactions.
8.3 Females and Males of Reproductive Potential
CHEMET may cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)] .
Principal Display Panel 100 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle Label)
NDC 55292-201-11
100 Capsules
Chemet
®
(succimer) Capsules
100 mg
RECORDATI
RARE DISEASES
GROUP
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been conducted with CHEMET. Succimer was not mutagenic in the Ames bacterial reverse mutation assay and in the mammalian cell forward gene mutation assay. Succimer did not show any adverse effects on fertility and reproductive performance in rats up to doses of 510 mg/kg/day in males and 100 mg/kg/day in females (7- and 11-times the MRHD based on BSA, respectively).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:11.110945 · Updated: 2026-03-14T22:29:13.416831