608b568c-5441-405a-b218-51333fe9fd9b
34390-5
HUMAN OTC DRUG LABEL
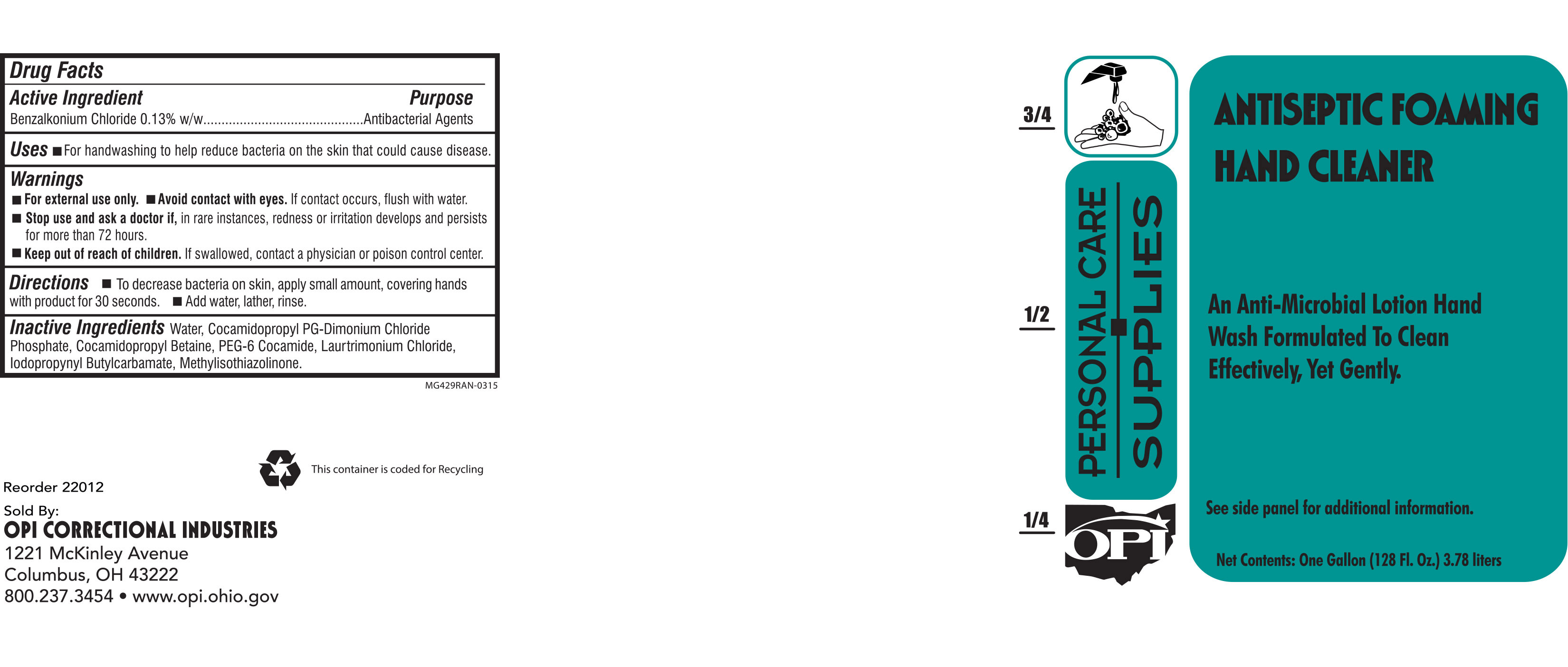
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Benzalkonium Chloride 0.13% w/w
Medication Information
Warnings and Precautions
Warnings
For external use only. Do not use in the eyes. If eye contact occurs, flush eyes with water.
Stop use and ask a doctor if redness and irritation occur and last for more than 72 hours.
Indications and Usage
Uses For handwashing to decrease bacteria on skin
Dosage and Administration
Directions Apply small amount, covering hands with product for 30 seconds. Add water, lather, rinse.
Description
Active Ingredient Benzalkonium Chloride 0.13% w/w
Section 50565-1
Keep out of reach of children. If swallowed, contact a physician or poison control center.
Section 51727-6
Inactive Ingredients
water, cocamidopropyl PG-dimonium chloride phosphate, cocamidopropyl betaine, PEG-6 cocamide, laurtrimonium chloride, iodopropynyl butylcarbamate, methylisothiazolinone .
Section 51945-4
Section 55105-1
Purpose
Antibacterial
Section 55106-9
Active Ingredient
Benzalkonium Chloride 0.13% w/w
Structured Label Content
Indications and Usage (34067-9)
Uses For handwashing to decrease bacteria on skin
Dosage and Administration (34068-7)
Directions Apply small amount, covering hands with product for 30 seconds. Add water, lather, rinse.
Warnings and Precautions (34071-1)
Warnings
For external use only. Do not use in the eyes. If eye contact occurs, flush eyes with water.
Stop use and ask a doctor if redness and irritation occur and last for more than 72 hours.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, contact a physician or poison control center.
Section 51727-6 (51727-6)
Inactive Ingredients
water, cocamidopropyl PG-dimonium chloride phosphate, cocamidopropyl betaine, PEG-6 cocamide, laurtrimonium chloride, iodopropynyl butylcarbamate, methylisothiazolinone .
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Antibacterial
Section 55106-9 (55106-9)
Active Ingredient
Benzalkonium Chloride 0.13% w/w
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:03.046012 · Updated: 2026-03-14T22:54:14.381059