6046a5c6-96b1-4c20-acac-2716025c1665
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Expectorant
Medication Information
Warnings and Precautions
Warnings
Purpose
Expectorant
Description
Drug Facts
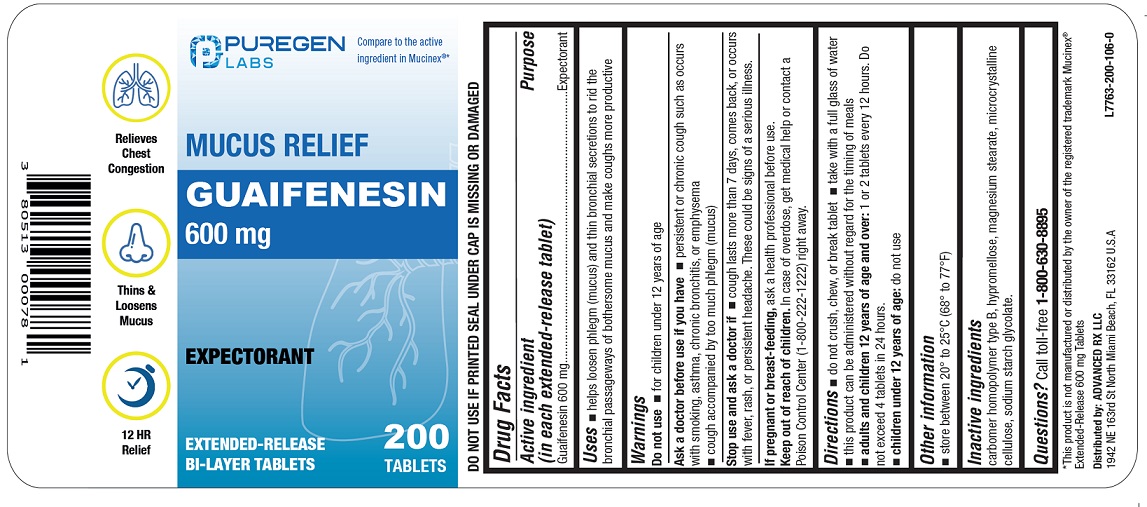
Uses
■ helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if
■ cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Section 50569-3
Ask a doctor before use if you have
■ persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
■ cough accompanied by too much phlegm (mucus)
Section 50570-1
Do not use
■ for children under 12 years of age
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
■ do not crush, chew, or break tablet
■ take with a full glass of water
■ this product can be administered without regard for the timing of meals
■ adults and children 12 years of age and over:1 or 2 tablets every 12 hours. Do not exceed 4 tablets in 24 hours.
■ children under 12 years of age:do not use
Questions?
Call toll-free 1-800-630-8895
Active Ingredient
Guaifenesin 600 mg
Other Information
■ store between 20º to 25ºC (68º to 77ºF)
■ DO NOT USE IF PRINTED SEAL UNDER CAP IS MISSING OR DAMAGED
Inactive Ingredients
carbomer homopolymer type B, hypromellose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate.
Principal Display Panel
NDC 80513-403-20
*Compare to the active ingredient in Mucinex ®
Mucus Relief
Guaifenesin 600 mg
Expectorant
EXTENDED-RELEASE TABLETS BI-LAYER TABLETS
200 Tablets
*This product is not manufactured or distributed by the owner of the registered trademark Mucinex ®Extended-Release 600 mg Tablets
Structured Label Content
Uses
■ helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings and Precautions (34071-1)
Warnings
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Section 50569-3 (50569-3)
Ask a doctor before use if you have
■ persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
■ cough accompanied by too much phlegm (mucus)
Section 50570-1 (50570-1)
Do not use
■ for children under 12 years of age
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Expectorant
Directions
■ do not crush, chew, or break tablet
■ take with a full glass of water
■ this product can be administered without regard for the timing of meals
■ adults and children 12 years of age and over:1 or 2 tablets every 12 hours. Do not exceed 4 tablets in 24 hours.
■ children under 12 years of age:do not use
Questions?
Call toll-free 1-800-630-8895
Active Ingredient (Active ingredient)
Guaifenesin 600 mg
Other Information (Other information)
■ store between 20º to 25ºC (68º to 77ºF)
■ DO NOT USE IF PRINTED SEAL UNDER CAP IS MISSING OR DAMAGED
Inactive Ingredients (Inactive ingredients)
carbomer homopolymer type B, hypromellose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 80513-403-20
*Compare to the active ingredient in Mucinex ®
Mucus Relief
Guaifenesin 600 mg
Expectorant
EXTENDED-RELEASE TABLETS BI-LAYER TABLETS
200 Tablets
*This product is not manufactured or distributed by the owner of the registered trademark Mucinex ®Extended-Release 600 mg Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:30.374074 · Updated: 2026-03-14T23:03:35.185430