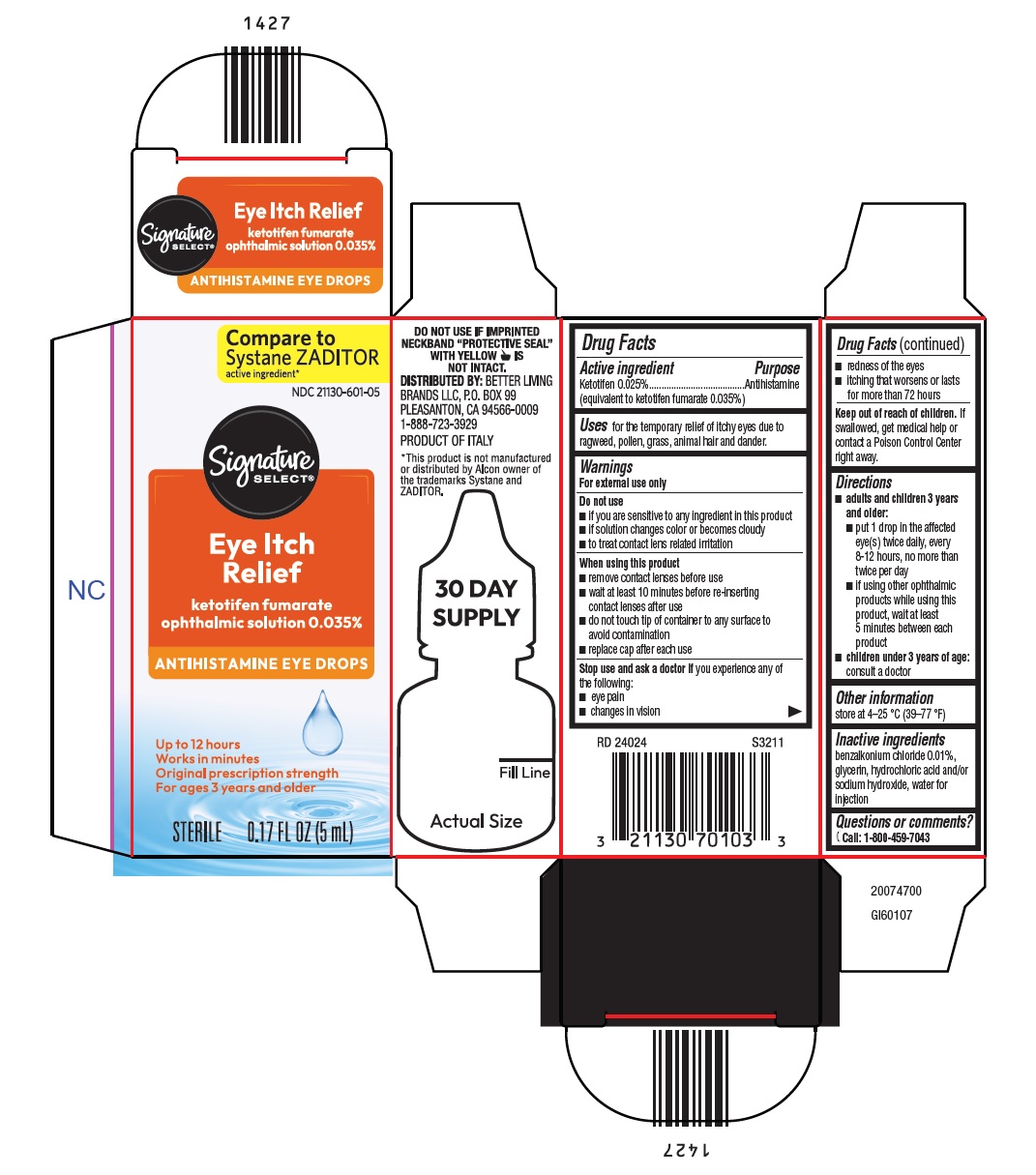
drug facts
5fccbb63-7859-484c-b7b3-9de711364c09
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine (equivalent to ketotifen fumarate 0.035%)
Description
Ketotifen 0.025%
Medication Information
Warnings
Do not use
-
▪if you are sensitive to any ingredient in this product
-
▪if solution changes color or becomes cloudy
-
▪to treat contact lens related irritation
When using this product
-
▪remove contact lenses before use
-
▪wait at least 10 minutes before re-inserting contact lenses after use
-
▪do not touch tip of container to any surface to avoid contamination
-
▪replace cap after each use
Uses
for the temporary relief of itchy eyes due to ragweed, pollen, grass, animal hair and dander.
Purpose
Antihistamine
(equivalent to ketotifen fumarate 0.035%)
Directions
-
▪adults and children 3 years and older:
-
▪put 1 drop in the affected eye(s) twice daily, every 8-12 hours, no more than twice per day
-
▪if using other ophthalmic products while using this product, wait at least 5 minutes between each product
-
▪children under 3 years of age: consult a doctor
Other Information
store at 4-25 °C (39-77 °F)
Inactive Ingredients
benzalkonium chloride 0.01%, glycerin, hydrochloric acid and/or sodium hydroxide, water for injection
Description
Ketotifen 0.025%
Questions?
{Phone icon} Call: 1-800-459-7043
*This product is not manufactured or distributed by Alcon owner of the trademarks Systane and ZADITOR.
Active Ingredient
Ketotifen 0.025%
Stop Use and Ask A Doctor If
you experience any of the following:
-
▪eye pain
-
▪changes in vision
-
▪redness of the eyes
-
▪itching that worsens or lasts for more than 72 hours
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel – 5ml Carton
Compare to
Systane ZADITOR
active ingredient*
NDC 21130-601-05
Signature
SELECT®
Eye Itch Relief
ketotifen fumarate
ophthalmic solution 0.035%
ANTIHISTAMINE EYE DROPS
Up to 12 hours
Works in minutes
Original prescription strength
For ages 3 years and older
STERILE 0.17 FL OZ (5 mL)
20074700
GI60107
Structured Label Content
Warnings
Do not use
-
▪if you are sensitive to any ingredient in this product
-
▪if solution changes color or becomes cloudy
-
▪to treat contact lens related irritation
When using this product
-
▪remove contact lenses before use
-
▪wait at least 10 minutes before re-inserting contact lenses after use
-
▪do not touch tip of container to any surface to avoid contamination
-
▪replace cap after each use
Uses
for the temporary relief of itchy eyes due to ragweed, pollen, grass, animal hair and dander.
Purpose
Antihistamine
(equivalent to ketotifen fumarate 0.035%)
Directions
-
▪adults and children 3 years and older:
-
▪put 1 drop in the affected eye(s) twice daily, every 8-12 hours, no more than twice per day
-
▪if using other ophthalmic products while using this product, wait at least 5 minutes between each product
-
▪children under 3 years of age: consult a doctor
Other Information (Other information)
store at 4-25 °C (39-77 °F)
Inactive Ingredients (Inactive ingredients)
benzalkonium chloride 0.01%, glycerin, hydrochloric acid and/or sodium hydroxide, water for injection
Questions?
{Phone icon} Call: 1-800-459-7043
*This product is not manufactured or distributed by Alcon owner of the trademarks Systane and ZADITOR.
Active Ingredient (Active ingredient)
Ketotifen 0.025%
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
you experience any of the following:
-
▪eye pain
-
▪changes in vision
-
▪redness of the eyes
-
▪itching that worsens or lasts for more than 72 hours
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel – 5ml Carton (Package/Label Principal Display Panel – 5mL carton)
Compare to
Systane ZADITOR
active ingredient*
NDC 21130-601-05
Signature
SELECT®
Eye Itch Relief
ketotifen fumarate
ophthalmic solution 0.035%
ANTIHISTAMINE EYE DROPS
Up to 12 hours
Works in minutes
Original prescription strength
For ages 3 years and older
STERILE 0.17 FL OZ (5 mL)
20074700
GI60107
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:04.918969 · Updated: 2026-03-14T23:02:57.214195