skinuva®
5fcad8c3-a268-4966-b0a9-6cd2c7afd3b7
34390-5
HUMAN OTC DRUG LABEL
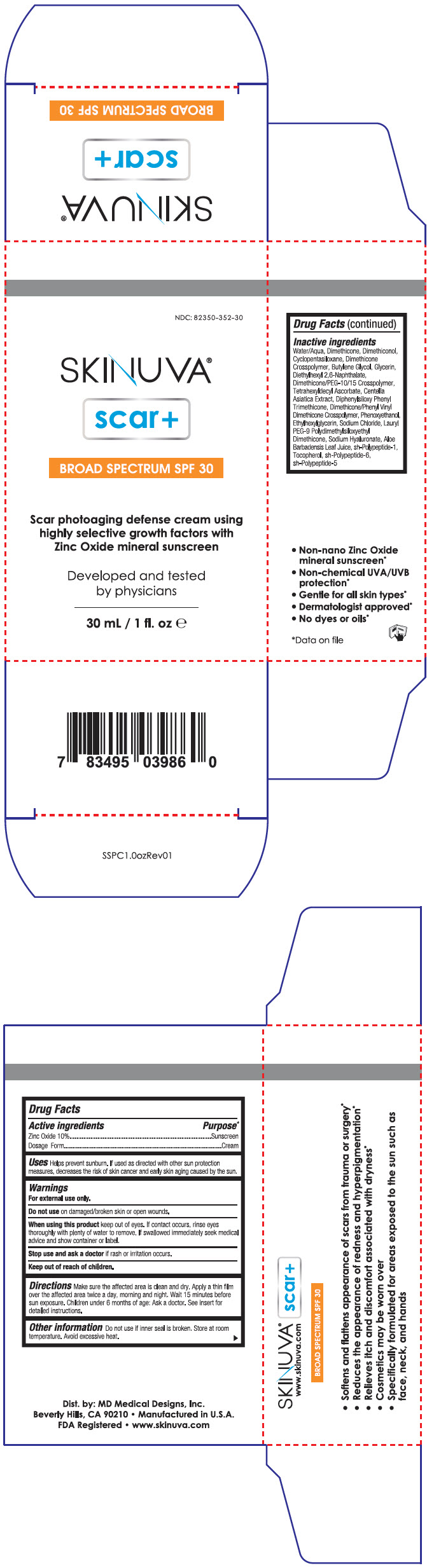
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings
For external use only.
Uses
Helps prevent sunburn. If used as directed with other sun protection measures, decreases the risk of skin cancer and early skin aging caused by the sun.
Directions
Make sure the affected area is clean and dry. Apply a thin film over the affected area twice a day, morning and night. Wait 15 minutes before sun exposure. Children under 6 months of age: Ask a doctor. See insert for detailed instructions.
Other Information
Do not use if inner seal is broken. Store at room temperature. Avoid excessive heat.
Inactive Ingredients
Water/Aqua, Dimethicone, Dimethiconol, Cyclopentasiloxane, Dimethicone Crosspolymer, Butylene Glycol, Glycerin, Diethylhexyl 2,6-Naphthalate, Dimethicone/PEG-10/15 Crosspolymer, Tetrahexyldecyl Ascorbate, Centella Asiatica Extract, Diphenylsiloxy Phenyl Trimethicone, Dimethicone/Phenyl Vinyl Dimethicone Crosspolymer, Phenoxyethanol, Ethylhexylglycerin, Sodium Chloride, Lauryl PEG-9 Polydimethylsiloxyethyl Dimethicone, Sodium Hyaluronate, Aloe Barbadensis Leaf Juice, sh-Polypeptide-1, Tocopherol, sh-Polypeptide-6, sh-Polypeptide-5
Description
Drug Facts
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if rash or irritation occurs.
Section 50567-7
When using this product keep out of eyes. If contact occurs, rinse eyes thoroughly with plenty of water to remove. If swallowed immediately seek medical advice and show container or label.
Section 50570-1
Do not use on damaged/broken skin or open wounds.
Section 55105-1
| Active ingredients |
Purpose Data on file
|
|---|---|
| Zinc Oxide 10% | Sunscreen |
| Dosage Form | Cream |
Principal Display Panel 30 Ml Bottle Carton
NDC: 82350-352-30
SKINUVA®
scar+
BROAD SPECTRUM SPF 30
Scar photoaging defense cream using
highly selective growth factors with
Zinc Oxide mineral sunscreen
Developed and tested
by physicians
30 mL / 1 fl. oz e
Structured Label Content
Warnings
For external use only.
Uses
Helps prevent sunburn. If used as directed with other sun protection measures, decreases the risk of skin cancer and early skin aging caused by the sun.
Directions
Make sure the affected area is clean and dry. Apply a thin film over the affected area twice a day, morning and night. Wait 15 minutes before sun exposure. Children under 6 months of age: Ask a doctor. See insert for detailed instructions.
Other Information (Other information)
Do not use if inner seal is broken. Store at room temperature. Avoid excessive heat.
Inactive Ingredients (Inactive ingredients)
Water/Aqua, Dimethicone, Dimethiconol, Cyclopentasiloxane, Dimethicone Crosspolymer, Butylene Glycol, Glycerin, Diethylhexyl 2,6-Naphthalate, Dimethicone/PEG-10/15 Crosspolymer, Tetrahexyldecyl Ascorbate, Centella Asiatica Extract, Diphenylsiloxy Phenyl Trimethicone, Dimethicone/Phenyl Vinyl Dimethicone Crosspolymer, Phenoxyethanol, Ethylhexylglycerin, Sodium Chloride, Lauryl PEG-9 Polydimethylsiloxyethyl Dimethicone, Sodium Hyaluronate, Aloe Barbadensis Leaf Juice, sh-Polypeptide-1, Tocopherol, sh-Polypeptide-6, sh-Polypeptide-5
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash or irritation occurs.
Section 50567-7 (50567-7)
When using this product keep out of eyes. If contact occurs, rinse eyes thoroughly with plenty of water to remove. If swallowed immediately seek medical advice and show container or label.
Section 50570-1 (50570-1)
Do not use on damaged/broken skin or open wounds.
Section 55105-1 (55105-1)
| Active ingredients |
Purpose Data on file
|
|---|---|
| Zinc Oxide 10% | Sunscreen |
| Dosage Form | Cream |
Principal Display Panel 30 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 30 mL Bottle Carton)
NDC: 82350-352-30
SKINUVA®
scar+
BROAD SPECTRUM SPF 30
Scar photoaging defense cream using
highly selective growth factors with
Zinc Oxide mineral sunscreen
Developed and tested
by physicians
30 mL / 1 fl. oz e
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:37.868974 · Updated: 2026-03-14T23:09:29.869893