5fb07d97-ce03-416f-a4a2-75c23cfebc05
5fb07d97-ce03-416f-a4a2-75c23cfebc05
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
1.1% neutral sodium fluoride
Medication Information
Uses
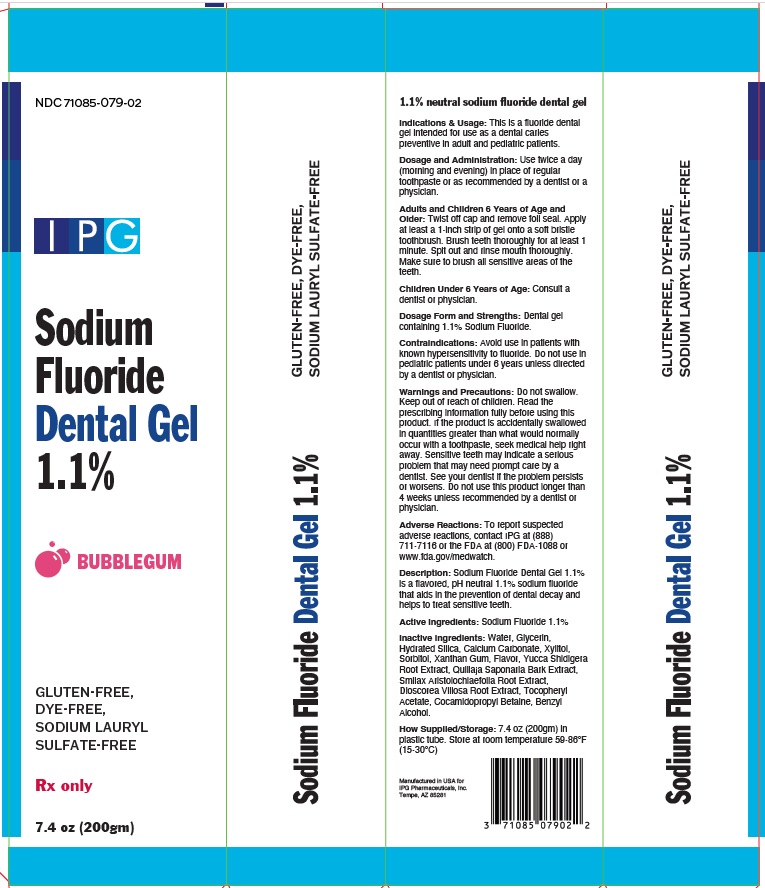
This is a fluoride dental gel intended for use as a dental caries preventive in adults and pediatric patients.
Directions
Use twice a day (morning and evening) in place of regular toothpaste or as recommended by a dentist or a physician.
Adults and Children 6 Years of Age and Older: Twist off cap and remove foil seal. Apply at least a1-inch strip of gel onto a soft bristle toothbrush. Brush teeth thoroughly for at least 1 minute. Spit out and rinse mouth thoroughly. Make sure to brush all
sensitive areas of the teeth.
Dosage form and Strengths: Dental gel containing 1.1% sodium fluoride.
Description
1.1% neutral sodium fluoride
Section 42229-5
How Supplied/Storage: 7.4 oz (200gm) in plastic tube. Store at room temperature 59-86°F (15-30°C)
Section 50565-1
Section 55105-1
Sodium Fluoride Dental Gel 1.1% is a flavored, pH neutral 1.1% sodium fluoride that aids in the prevention of dental decay and helps to treat sensitive teeth.
Warning
Contraindications: Avoid use in patients with known hypersensitivity to fluoride. Do not use in pediatric patients under 6 unless directed by a dentist or physician.
Warnings and Precautions: Do no swallow. Keep out of reach of children. Read the prescribing information fully before using this product. If the product is accidentally swallowed in quantities greater than would normally occur with a toothpaste, seek medical help right away. See your dentist if the problem persists or worsens. Do not use this product longer than 4 weeks unless recommended by a dentist or physician.
Adverse reactions: To report suspected
Adverse reactions, To report suspected adverse reactions, contact IPG Pharmaceuticals at (888) 711-7116 or the FDA at (800) FDA-1088 or www.fda.gov/medwatch.
Product Label
Active Ingredient
1.1% neutral sodium fluoride
Inactive Ingredient
Water, Glycerin, Hydrated Silica, Calcium Carbonate, Xylitol, Sorbitol, Xanthan Gum, Flavor, Yucca Shidigera Root Extract, Quillaja
Saponaria Bark Extract, Smilax Aristolochiaefolia Root Extract, Dioscorea Villosa Root Extract, Tocopheryl Acetate, Cocamidopropyl Betaine,
Benzyl Alcohol.
Structured Label Content
Uses
This is a fluoride dental gel intended for use as a dental caries preventive in adults and pediatric patients.
Directions
Use twice a day (morning and evening) in place of regular toothpaste or as recommended by a dentist or a physician.
Adults and Children 6 Years of Age and Older: Twist off cap and remove foil seal. Apply at least a1-inch strip of gel onto a soft bristle toothbrush. Brush teeth thoroughly for at least 1 minute. Spit out and rinse mouth thoroughly. Make sure to brush all
sensitive areas of the teeth.
Dosage form and Strengths: Dental gel containing 1.1% sodium fluoride.
Section 42229-5 (42229-5)
How Supplied/Storage: 7.4 oz (200gm) in plastic tube. Store at room temperature 59-86°F (15-30°C)
Section 50565-1 (50565-1)
Section 55105-1 (55105-1)
Sodium Fluoride Dental Gel 1.1% is a flavored, pH neutral 1.1% sodium fluoride that aids in the prevention of dental decay and helps to treat sensitive teeth.
Warning
Contraindications: Avoid use in patients with known hypersensitivity to fluoride. Do not use in pediatric patients under 6 unless directed by a dentist or physician.
Warnings and Precautions: Do no swallow. Keep out of reach of children. Read the prescribing information fully before using this product. If the product is accidentally swallowed in quantities greater than would normally occur with a toothpaste, seek medical help right away. See your dentist if the problem persists or worsens. Do not use this product longer than 4 weeks unless recommended by a dentist or physician.
Adverse reactions: To report suspected
Adverse reactions, To report suspected adverse reactions, contact IPG Pharmaceuticals at (888) 711-7116 or the FDA at (800) FDA-1088 or www.fda.gov/medwatch.
Product Label (Product label)
Active Ingredient
1.1% neutral sodium fluoride
Inactive Ingredient (Inactive ingredient )
Water, Glycerin, Hydrated Silica, Calcium Carbonate, Xylitol, Sorbitol, Xanthan Gum, Flavor, Yucca Shidigera Root Extract, Quillaja
Saponaria Bark Extract, Smilax Aristolochiaefolia Root Extract, Dioscorea Villosa Root Extract, Tocopheryl Acetate, Cocamidopropyl Betaine,
Benzyl Alcohol.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:08.359181 · Updated: 2026-03-14T22:29:06.951873