These Highlights Do Not Include All The Information Needed To Use Revlimid®
5fa97bf5-28a2-48f1-8955-f56012d296be
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
REVLIMID is a thalidomide analogue indicated for the treatment of adult patients with: • Multiple myeloma (MM), in combination with dexamethasone ( 1.1 ). • MM, as maintenance following autologous hematopoietic stem cell transplantation (auto-HSCT) ( 1.1 ). • Transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q abnormality with or without additional cytogenetic abnormalities ( 1.2 ). • Mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib ( 1.3 ). • Previously treated follicular lymphoma (FL), in combination with a rituximab product ( 1.4 ). • Previously treated marginal zone lymphoma (MZL), in combination with a rituximab product ( 1.5 ). Limitations of Use: • REVLIMID is not indicated and is not recommended for the treatment of patients with chronic lymphocytic leukemia (CLL) outside of controlled clinical trials ( 1.4 ).
Dosage and Administration
• MM combination therapy: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles. ( 2.1 ). • MM maintenance therapy following auto-HSCT: 10 mg once daily continuously on Days 1-28 of repeated 28-day cycles ( 2.1 ). • MDS: 10 mg once daily ( 2.2 ). • MCL: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles ( 2.3 ). • FL or MZL: 20 mg once daily orally on Days 1-21 of repeated 28-day cycles for up to 12 cycles ( 2.4 ). • Renal impairment: Adjust starting dose based on the creatinine clearance value ( 2.6 ). • For concomitant therapy doses, see Full Prescribing Information ( 2.1 , 2.4 , 14.1 , 14.4 ).
Contraindications
• Pregnancy ( Boxed Warning , 4.1 , 5.1 , 8.1 ). • Demonstrated severe hypersensitivity to lenalidomide ( 4.2 , 5.9 , 5.15 ).
Warnings and Precautions
• Increased Mortality: serious and fatal cardiac adverse reactions occurred in patients with CLL treated with REVLIMID ( 5.5 ). • Second Primary Malignancies (SPM): Higher incidences of SPM were observed in controlled trials of patients with MM receiving REVLIMID ( 5.6 ). • Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue ( 5.7 ). • Hepatotoxicity: Hepatic failure including fatalities; monitor liver function. Stop REVLIMID and evaluate if hepatotoxicity is suspected ( 5.8 ). • Severe Cutaneous Reactions: Discontinue REVLIMID for severe reactions ( 5.9 ). • Tumor lysis syndrome (TLS) including fatalities: Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions ( 5.10 ). • Tumor flare reaction: Serious tumor flare reactions, including fatal reactions, have occurred during investigational use of REVLIMID for chronic lymphocytic leukemia and lymphoma ( 5.11 ). • Impaired Stem Cell mobilization: A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. Consider early referral to transplant center ( 5.12 ). • Early mortality in MCL: Higher rate of early deaths have occurred in patients with MCL ( 5.14 ). • Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue REVLIMID for angioedema and anaphylaxis ( 5.15 ).
Adverse Reactions
The following clinically significant adverse reactions are described in detail in other sections of the prescribing information: o Embryo-Fetal Toxicity [see Boxed Warning , Warnings and Precautions (5.1 , 5.2) ] o Hematologic Toxicity [see Boxed Warning , Warnings and Precautions (5.3) ] o Venous and Arterial Thromboembolism [see Boxed Warning , Warnings and Precautions (5.4) ] o Increased Mortality in Patients with CLL [see Warnings and Precautions (5.5) ] o Second Primary Malignancies [see Warnings and Precautions (5.6) ] o Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.7) ] o Hepatotoxicity [see Warnings and Precautions (5.8) ] o Severe Cutaneous Reactions [see Warnings and Precautions (5.9) ] o Tumor Lysis Syndrome [see Warnings and Precautions (5.10) ] o Tumor Flare Reactions [see Warnings and Precautions (5.11) ] o Impaired Stem Cell Mobilization [see Warnings and Precautions (5.12) ] o Thyroid Disorders [see Warnings and Precautions (5.13) ] o Early Mortality in Patients with MCL [see Warnings and Precautions (5.14) ] o Hypersensitivity [see Warnings and Precautions (5.15) ]
Drug Interactions
• Digoxin: Monitor digoxin plasma levels periodically due to increased C max and AUC with concomitant REVLIMID therapy ( 7.1 ). • Concomitant use of erythropoietin stimulating agents or estrogen containing therapies with REVLIMID may increase the risk of thrombosis ( 7.2 ).
How Supplied
White and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink: 2.5 mg bottles of 28 (NDC 59572-402-28) 2.5 mg bottles of 100 (NDC 59572-402-00) White opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink: 5 mg bottles of 28 (NDC 59572-405-28) 5 mg bottles of 100 (NDC 59572-405-00) Blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink: 10 mg bottles of 28 (NDC 59572-410-28) 10 mg bottles of 100 (NDC 59572-410-00) Powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink: 15 mg bottles of 21 (NDC 59572-415-21) 15 mg bottles of 100 (NDC 59572-415-00) Powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink. 20 mg bottles of 21 (NDC 59572-420-21) 20 mg bottles of 100 (NDC 59572-420-00) White opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink: 25 mg bottles of 21 (NDC 59572-425-21) 25 mg bottles of 100 (NDC 59572-425-00)
Description
Warnings and Precautions ( 5.1 , 5.2 ) 8/2021 Warnings and Precautions ( 5.1 , 5.11 ) 5/2022
Medication Information
Warnings and Precautions
• Increased Mortality: serious and fatal cardiac adverse reactions occurred in patients with CLL treated with REVLIMID ( 5.5 ). • Second Primary Malignancies (SPM): Higher incidences of SPM were observed in controlled trials of patients with MM receiving REVLIMID ( 5.6 ). • Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue ( 5.7 ). • Hepatotoxicity: Hepatic failure including fatalities; monitor liver function. Stop REVLIMID and evaluate if hepatotoxicity is suspected ( 5.8 ). • Severe Cutaneous Reactions: Discontinue REVLIMID for severe reactions ( 5.9 ). • Tumor lysis syndrome (TLS) including fatalities: Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions ( 5.10 ). • Tumor flare reaction: Serious tumor flare reactions, including fatal reactions, have occurred during investigational use of REVLIMID for chronic lymphocytic leukemia and lymphoma ( 5.11 ). • Impaired Stem Cell mobilization: A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. Consider early referral to transplant center ( 5.12 ). • Early mortality in MCL: Higher rate of early deaths have occurred in patients with MCL ( 5.14 ). • Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue REVLIMID for angioedema and anaphylaxis ( 5.15 ).
Indications and Usage
REVLIMID is a thalidomide analogue indicated for the treatment of adult patients with: • Multiple myeloma (MM), in combination with dexamethasone ( 1.1 ). • MM, as maintenance following autologous hematopoietic stem cell transplantation (auto-HSCT) ( 1.1 ). • Transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q abnormality with or without additional cytogenetic abnormalities ( 1.2 ). • Mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib ( 1.3 ). • Previously treated follicular lymphoma (FL), in combination with a rituximab product ( 1.4 ). • Previously treated marginal zone lymphoma (MZL), in combination with a rituximab product ( 1.5 ). Limitations of Use: • REVLIMID is not indicated and is not recommended for the treatment of patients with chronic lymphocytic leukemia (CLL) outside of controlled clinical trials ( 1.4 ).
Dosage and Administration
• MM combination therapy: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles. ( 2.1 ). • MM maintenance therapy following auto-HSCT: 10 mg once daily continuously on Days 1-28 of repeated 28-day cycles ( 2.1 ). • MDS: 10 mg once daily ( 2.2 ). • MCL: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles ( 2.3 ). • FL or MZL: 20 mg once daily orally on Days 1-21 of repeated 28-day cycles for up to 12 cycles ( 2.4 ). • Renal impairment: Adjust starting dose based on the creatinine clearance value ( 2.6 ). • For concomitant therapy doses, see Full Prescribing Information ( 2.1 , 2.4 , 14.1 , 14.4 ).
Contraindications
• Pregnancy ( Boxed Warning , 4.1 , 5.1 , 8.1 ). • Demonstrated severe hypersensitivity to lenalidomide ( 4.2 , 5.9 , 5.15 ).
Adverse Reactions
The following clinically significant adverse reactions are described in detail in other sections of the prescribing information: o Embryo-Fetal Toxicity [see Boxed Warning , Warnings and Precautions (5.1 , 5.2) ] o Hematologic Toxicity [see Boxed Warning , Warnings and Precautions (5.3) ] o Venous and Arterial Thromboembolism [see Boxed Warning , Warnings and Precautions (5.4) ] o Increased Mortality in Patients with CLL [see Warnings and Precautions (5.5) ] o Second Primary Malignancies [see Warnings and Precautions (5.6) ] o Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.7) ] o Hepatotoxicity [see Warnings and Precautions (5.8) ] o Severe Cutaneous Reactions [see Warnings and Precautions (5.9) ] o Tumor Lysis Syndrome [see Warnings and Precautions (5.10) ] o Tumor Flare Reactions [see Warnings and Precautions (5.11) ] o Impaired Stem Cell Mobilization [see Warnings and Precautions (5.12) ] o Thyroid Disorders [see Warnings and Precautions (5.13) ] o Early Mortality in Patients with MCL [see Warnings and Precautions (5.14) ] o Hypersensitivity [see Warnings and Precautions (5.15) ]
Drug Interactions
• Digoxin: Monitor digoxin plasma levels periodically due to increased C max and AUC with concomitant REVLIMID therapy ( 7.1 ). • Concomitant use of erythropoietin stimulating agents or estrogen containing therapies with REVLIMID may increase the risk of thrombosis ( 7.2 ).
How Supplied
White and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink: 2.5 mg bottles of 28 (NDC 59572-402-28) 2.5 mg bottles of 100 (NDC 59572-402-00) White opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink: 5 mg bottles of 28 (NDC 59572-405-28) 5 mg bottles of 100 (NDC 59572-405-00) Blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink: 10 mg bottles of 28 (NDC 59572-410-28) 10 mg bottles of 100 (NDC 59572-410-00) Powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink: 15 mg bottles of 21 (NDC 59572-415-21) 15 mg bottles of 100 (NDC 59572-415-00) Powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink. 20 mg bottles of 21 (NDC 59572-420-21) 20 mg bottles of 100 (NDC 59572-420-00) White opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink: 25 mg bottles of 21 (NDC 59572-425-21) 25 mg bottles of 100 (NDC 59572-425-00)
Description
Warnings and Precautions ( 5.1 , 5.2 ) 8/2021 Warnings and Precautions ( 5.1 , 5.11 ) 5/2022
Data
Animal data
In an embryo-fetal developmental toxicity study in monkeys, teratogenicity, including thalidomide-like limb defects, occurred in offspring when pregnant monkeys received oral lenalidomide during organogenesis. Exposure (AUC) in monkeys at the lowest dose was 0.17 times the human exposure at the maximum recommended human dose (MRHD) of 25 mg. Similar studies in pregnant rabbits and rats at 20 times and 200 times the MRHD respectively, produced embryo lethality in rabbits and no adverse reproductive effects in rats.
In a pre- and post-natal development study in rats, animals received lenalidomide from organogenesis through lactation. The study revealed a few adverse effects on the offspring of female rats treated with lenalidomide at doses up to 500 mg/kg (approximately 200 times the human dose of 25 mg based on body surface area). The male offspring exhibited slightly delayed sexual maturation and the female offspring had slightly lower body weight gains during gestation when bred to male offspring. As with thalidomide, the rat model may not adequately address the full spectrum of potential human embryo-fetal developmental effects for lenalidomide.
Following daily oral administration of lenalidomide from Gestation Day 7 through Gestation Day 20 in pregnant rabbits, fetal plasma lenalidomide concentrations were approximately 20-40% of the maternal Cmax. Following a single oral dose to pregnant rats, lenalidomide was detected in fetal plasma and tissues; concentrations of radioactivity in fetal tissues were generally lower than those in maternal tissues. These data indicated that lenalidomide crossed the placenta.
Section 42229-5
REVLIMID Combination Therapy
The recommended starting dose of REVLIMID is 25 mg orally once daily on Days 1-21 of repeated 28-day cycles in combination with dexamethasone. Refer to Section 14.1 for specific dexamethasone dosing. For patients greater than 75 years old, the starting dose of dexamethasone may be reduced [see Clinical Studies (14.1)]. Treatment should be continued until disease progression or unacceptable toxicity.
In patients who are not eligible for auto-HSCT, treatment should continue until disease progression or unacceptable toxicity. For patients who are auto-HSCT-eligible, hematopoietic stem cell mobilization should occur within 4 cycles of a REVLIMID-containing therapy [see Warnings and Precautions (5.12)].
Section 43683-2
7.1 Digoxin
When digoxin was co-administered with multiple doses of REVLIMID (10 mg/day) the digoxin Cmax and AUCinf were increased by 14%. Periodically monitor digoxin plasma levels, in accordance with clinical judgment and based on standard clinical practice in patients receiving this medication, during administration of REVLIMID.
16.2 Storage
Store at 20°C - 25°C (68°F - 77°F); excursions permitted to 15°C - 30°C (59°F - 86°F) [See USP Controlled Room Temperature].
7.3 Warfarin
Co-administration of multiple doses of REVLIMID (10 mg/day) with a single dose of warfarin (25 mg) had no effect on the pharmacokinetics of lenalidomide or R- and S-warfarin. Expected changes in laboratory assessments of PT and INR were observed after warfarin administration, but these changes were not affected by concomitant REVLIMID administration. It is not known whether there is an interaction between dexamethasone and warfarin. Close monitoring of PT and INR is recommended in patients with MM taking concomitant warfarin.
10 Overdosage
There is no specific experience in the management of REVLIMID overdose in patients with MM, MDS, MCL, FL, or MZL. In dose-ranging studies in healthy subjects, some were exposed to up to 200 mg (administered 100 mg BID) and in single-dose studies, some subjects were exposed to up to 400 mg. Pruritus, urticaria, rash, and elevated liver transaminases were the primary reported AEs. In clinical trials, the dose-limiting toxicity was neutropenia and thrombocytopenia.
15 References
1. OSHA Hazardous Drugs. OSHA [Accessed on 29 January 2013, from http://www.osha.gov/SLTC/hazardousdrugs/index.html]
4.1 Pregnancy
REVLIMID can cause fetal harm when administered to a pregnant female. Limb abnormalities were seen in the offspring of monkeys that were dosed with lenalidomide during organogenesis. This effect was seen at all doses tested. Due to the results of this developmental monkey study, and lenalidomide's structural similarities to thalidomide, a known human teratogen, lenalidomide is contraindicated in females who are pregnant [see Boxed Warning]. If this drug is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus [see Warnings and Precautions (5.1, 5.2), Use in Special Populations (8.1, 8.3)].
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in females exposed to REVLIMID during pregnancy as well as female partners of male patients who are exposed to REVLIMID. This registry is also used to understand the root cause for the pregnancy. Report any suspected fetal exposure to REVLIMID to the FDA via the MedWatch program at 1-800-FDA-1088 and also to the REMS Call Center at 1-888-423-5436.
Risk Summary
Based on the mechanism of action [see Clinical Pharmacology (12.1)] and findings from animal studies [see Data], REVLIMID can cause embryo-fetal harm when administered to a pregnant female and is contraindicated during pregnancy [see Boxed Warning, Contraindications (4.1), and Use in Specific Populations (5.1)].
REVLIMID is a thalidomide analogue. Thalidomide is a human teratogen, inducing a high frequency of severe and life-threatening birth defects such as amelia (absence of limbs), phocomelia (short limbs), hypoplasticity of the bones, absence of bones, external ear abnormalities (including anotia, micropinna, small or absent external auditory canals), facial palsy, eye abnormalities (anophthalmos, microphthalmos), and congenital heart defects. Alimentary tract, urinary tract, and genital malformations have also been documented and mortality at or shortly after birth has been reported in about 40% of infants.
Lenalidomide caused thalidomide-type limb defects in monkey offspring. Lenalidomide crossed the placenta after administration to pregnant rabbits and pregnant rats [see Data]. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus.
If pregnancy does occur during treatment, immediately discontinue the drug. Under these conditions, refer patient to an obstetrician/gynecologist experienced in reproductive toxicity for further evaluation and counseling. Report any suspected fetal exposure to REVLIMID to the FDA via the MedWatch program at 1-800-FDA-1088 and also to the REMS Call Center at 1-888-423-5436.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk in the U.S. general population of major birth defects is 2%-4% and of miscarriage is 15%-20% of clinically recognized pregnancies.
11 Description
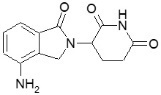
REVLIMID, a thalidomide analogue, is an immunomodulatory agent with antiangiogenic and antineoplastic properties. The chemical name is 3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione and it has the following chemical structure:
3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione
The empirical formula for lenalidomide is C13H13N3O3, and the gram molecular weight is 259.3.
Lenalidomide is an off-white to pale-yellow solid powder. It is soluble in organic solvent/water mixtures, and buffered aqueous solvents. Lenalidomide is more soluble in organic solvents and low pH solutions. Solubility was significantly lower in less acidic buffers, ranging from about 0.4 to 0.5 mg/ml. Lenalidomide has an asymmetric carbon atom and can exist as the optically active forms S(-) and R(+), and is produced as a racemic mixture with a net optical rotation of zero.
REVLIMID is available in 2.5 mg, 5 mg, 10 mg, 15 mg, 20 mg and 25 mg capsules for oral administration. Each capsule contains lenalidomide as the active ingredient and the following inactive ingredients: lactose anhydrous, microcrystalline cellulose, croscarmellose sodium, and magnesium stearate. The 5 mg and 25 mg capsule shell contains gelatin, titanium dioxide and black ink. The 2.5 mg and 10 mg capsule shell contains gelatin, FD&C blue #2, yellow iron oxide, titanium dioxide and black ink. The 15 mg capsule shell contains gelatin, FD&C blue #2, titanium dioxide and black ink. The 20 mg capsule shell contains gelatin, FD&C blue #2, yellow iron oxide, titanium dioxide and black ink.
Medication Guide
REVLIMID® (rev-li-mid)
(lenalidomide) capsules
|
What is the most important information I should know about REVLIMID?
Females must not get pregnant:
Females who can become pregnant:
If you become pregnant while taking REVLIMID, stop taking it right away and call your healthcare provider. If your healthcare provider is not available, you can call the REMS Call Center at 1-888-423-5436. Healthcare providers and patients should report all cases of pregnancy to:
There is a pregnancy exposure registry that monitors the outcomes of females who take REVLIMID during pregnancy, or if their male partner takes REVLIMID and they are exposed during pregnancy. You can enroll in this registry by calling the REMS Call Center at the phone number listed above. REVLIMID can pass into human semen:
Men, if your female partner becomes pregnant, you should call your healthcare provider right away.
|
||
|
What is REVLIMID?
REVLIMID should not be used to treat people who have chronic lymphocytic leukemia (CLL) unless they are participants in a controlled clinical trial. |
||
|
Who should not take REVLIMID?
|
||
|
What should I tell my healthcare provider before taking REVLIMID?
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. REVLIMID and other medicines may affect each other, causing serious side effects. Talk with your healthcare provider before taking any new medicines. |
||
|
How should I take REVLIMID?
|
||
|
What should I avoid while taking REVLIMID?
|
||
|
What are the possible side effects of REVLIMID?
|
||
|
|
|
Call your healthcare provider right away if you develop any of the following signs or symptoms during treatment with REVLIMID: |
||
|
|
|
|
Get emergency medical help right away if you develop any of the following signs or symptoms during treatment with REVLIMID: |
||
|
|
|
Your healthcare provider may tell you to decrease your dose, temporarily stop or permanently stop taking REVLIMID if you develop certain serious side effects during treatment with REVLIMID.
The most common side effects of REVLIMID include: |
||
|
|
|
|
These are not all the possible side effects of REVLIMID. |
||
|
How should I store REVLIMID?
Keep REVLIMID and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of REVLIMID.
For more information, call 1-888-423-5436 or go to www.lenalidomiderems.com. |
||
|
What are the ingredients in REVLIMID?
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: MAR 2023
16.1 How Supplied
White and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink:
|
2.5 mg bottles of 28 |
(NDC 59572-402-28) |
|
|
2.5 mg bottles of 100 |
(NDC 59572-402-00) |
White opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink:
|
5 mg bottles of 28 |
(NDC 59572-405-28) |
|
|
5 mg bottles of 100 |
(NDC 59572-405-00) |
Blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink:
|
10 mg bottles of 28 |
(NDC 59572-410-28) |
|
|
10 mg bottles of 100 |
(NDC 59572-410-00) |
Powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink:
|
15 mg bottles of 21 |
(NDC 59572-415-21) |
|
|
15 mg bottles of 100 |
(NDC 59572-415-00) |
Powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink.
|
20 mg bottles of 21 |
(NDC 59572-420-21) |
|
|
20 mg bottles of 100 |
(NDC 59572-420-00) |
White opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink:
|
25 mg bottles of 21 |
(NDC 59572-425-21) |
|
|
25 mg bottles of 100 |
(NDC 59572-425-00) |
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
2.7 Administration
Advise patients to take REVLIMID orally at about the same time each day, either with or without food. Advise patients to swallow REVLIMID capsules whole with water and not to open, break, or chew them.
5.8 Hepatotoxicity
Hepatic failure, including fatal cases, has occurred in patients treated with REVLIMID in combination with dexamethasone. In clinical trials, 15% of patients experienced hepatotoxicity (with hepatocellular, cholestatic and mixed characteristics); 2% of patients with MM and 1% of patients with myelodysplasia had serious hepatotoxicity events. The mechanism of drug-induced hepatotoxicity is unknown. Pre-existing viral liver disease, elevated baseline liver enzymes, and concomitant medications may be risk factors. Monitor liver enzymes periodically. Stop REVLIMID upon elevation of liver enzymes. After return to baseline values, treatment at a lower dose may be considered.
4 Contraindications
6 Adverse Reactions
The following clinically significant adverse reactions are described in detail in other sections of the prescribing information:
-
oEmbryo-Fetal Toxicity [see Boxed Warning, Warnings and Precautions (5.1, 5.2)]
-
oHematologic Toxicity [see Boxed Warning, Warnings and Precautions (5.3)]
-
oVenous and Arterial Thromboembolism [see Boxed Warning, Warnings and Precautions (5.4)]
-
oIncreased Mortality in Patients with CLL [see Warnings and Precautions (5.5)]
-
oSecond Primary Malignancies [see Warnings and Precautions (5.6)]
-
oIncreased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.7)]
-
oHepatotoxicity [see Warnings and Precautions (5.8)]
-
oSevere Cutaneous Reactions[see Warnings and Precautions (5.9)]
-
oTumor Lysis Syndrome [see Warnings and Precautions (5.10)]
-
oTumor Flare Reactions [see Warnings and Precautions (5.11)]
-
oImpaired Stem Cell Mobilization [see Warnings and Precautions (5.12)]
-
oThyroid Disorders [see Warnings and Precautions (5.13)]
-
oEarly Mortality in Patients with MCL [see Warnings and Precautions (5.14)]
-
oHypersensitivity [see Warnings and Precautions (5.15)]
7 Drug Interactions
1.1 Multiple Myeloma
REVLIMID in combination with dexamethasone is indicated for the treatment of adult patients with multiple myeloma (MM).
REVLIMID is indicated as maintenance therapy in adult patients with MM following autologous hematopoietic stem cell transplantation (auto-HSCT).
8.6 Renal Impairment
Adjust the starting dose of REVLIMID based on the creatinine clearance value and for patients on dialysis [see Dosage and Administration (2.6)].
5.15 Hypersensitivity
Hypersensitivity, including angioedema, anaphylaxis, and anaphylactic reactions to REVLIMID has been reported. Permanently discontinue REVLIMID for angioedema and anaphylaxis [see Dosage and Administration (2.2)].
1.6 Limitations of Use
REVLIMID is not indicated and is not recommended for the treatment of patients with CLL outside of controlled clinical trials [see Warnings and Precautions (5.5)].
5.13 Thyroid Disorders
Both hypothyroidism and hyperthyroidism have been reported [see Adverse Reactions (6.2)]. Measure thyroid function before start of REVLIMID treatment and during therapy.
1 Indications and Usage
REVLIMID is a thalidomide analogue indicated for the treatment of adult patients with:
-
•Multiple myeloma (MM), in combination with dexamethasone (1.1).
-
•MM, as maintenance following autologous hematopoietic stem cell transplantation (auto-HSCT) (1.1).
-
•Transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q abnormality with or without additional cytogenetic abnormalities (1.2).
-
•Mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib (1.3).
-
•Previously treated follicular lymphoma (FL), in combination with a rituximab product (1.4).
-
•Previously treated marginal zone lymphoma (MZL), in combination with a rituximab product (1.5).
Limitations of Use:
-
•REVLIMID is not indicated and is not recommended for the treatment of patients with chronic lymphocytic leukemia (CLL) outside of controlled clinical trials (1.4).
1.4 Follicular Lymphoma
REVLIMID in combination with a rituximab product, is indicated for the treatment of adult patients with previously treated follicular lymphoma (FL).
1.3 Mantle Cell Lymphoma
REVLIMID is indicated for the treatment of adult patients with mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib.
12.1 Mechanism of Action
Lenalidomide is an analogue of thalidomide with immunomodulatory, antiangiogenic, and antineoplastic properties. Cellular activities of lenalidomide are mediated through its target cereblon, a component of a cullin ring E3 ubiquitin ligase enzyme complex. In vitro, in the presence of drug, substrate proteins (including Aiolos, Ikaros, and CK1α) are targeted for ubiquitination and subsequent degradation leading to direct cytotoxic and immunomodulatory effects. Lenalidomide inhibits proliferation and induces apoptosis of certain hematopoietic tumor cells including MM, mantle cell lymphoma, and del (5q) myelodysplastic syndromes, follicular lymphoma and marginal zone lymphoma in vitro. Lenalidomide causes a delay in tumor growth in some in vivo nonclinical hematopoietic tumor models including MM. Immunomodulatory properties of lenalidomide include increased number and activation of T cells and natural killer (NK) cells leading to direct and enhanced antibody-dependent cell-mediated cytotoxicity (ADCC) via increased secretion of interleukin-2 and interferon-gamma, increased numbers of NKT cells, and inhibition of pro-inflammatory cytokines (e.g., TNF-α and IL-6) by monocytes. In MM cells, the combination of lenalidomide and dexamethasone synergizes the inhibition of cell proliferation and the induction of apoptosis. The combination of lenalidomide and rituximab increases ADCC and direct tumor apoptosis in follicular lymphoma cells and increases ADCC in marginal zone lymphoma cells compared to rituximab alone in vitro.
5.3 Hematologic Toxicity
REVLIMID can cause significant neutropenia and thrombocytopenia. Monitor patients with neutropenia for signs of infection. Advise patients to observe for bleeding or bruising, especially with use of concomitant medication that may increase risk of bleeding. Patients taking REVLIMID should have their complete blood counts assessed periodically as described below [see Dosage and Administration (2.1, 2.2, 2.3)].
Monitor complete blood counts (CBC) in patients taking REVLIMID in combination with dexamethasone or as REVLIMID maintenance therapy for MM every 7 days (weekly) for the first 2 cycles, on Days 1 and 15 of Cycle 3, and every 28 days (4 weeks) thereafter. A dose interruption and/or dose reduction may be required [see Dosage and Administration (2.1)]. In the MM maintenance therapy trials, Grade 3 or 4 neutropenia was reported in up to 59% of REVLIMID-treated patients and Grade 3 or 4 thrombocytopenia in up to 38% of REVLIMID-treated patients [see Adverse Reactions (6.1)].
Monitor complete blood counts (CBC) in patients taking REVLIMID for MDS weekly for the first 8 weeks and at least monthly thereafter. Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the MDS study. In the 48% of patients who developed Grade 3 or 4 neutropenia, the median time to onset was 42 days (range, 14-411 days), and the median time to documented recovery was 17 days (range, 2-170 days). In the 54% of patients who developed Grade 3 or 4 thrombocytopenia, the median time to onset was 28 days (range, 8-290 days), and the median time to documented recovery was 22 days (range, 5-224 days) [see Boxed Warning and Dosage and Administration (2.2)].
Monitor complete blood counts (CBC) in patients taking REVLIMID for MCL weekly for the first cycle (28 days), every 2 weeks during cycles 2-4, and then monthly thereafter. Patients may require dose interruption and/or dose reduction. In the MCL trial, Grade 3 or 4 neutropenia was reported in 43% of the patients. Grade 3 or 4 thrombocytopenia was reported in 28% of the patients.
Monitor complete blood counts (CBC) in patients taking REVLIMID for FL or MZL weekly for the first 3 weeks of Cycle 1 (28 days), every 2 weeks during Cycles 2-4, and then monthly thereafter. Patients may require dose interruption and/or dose reduction. In the AUGMENT and MAGNIFY trials, Grade 3 or 4 neutropenia was reported in 50% and 33%, respectively, of patients in the REVLIMID/rituximab arm. Grade 3 or 4 thrombocytopenia was reported in 2% and 8%, respectively, of patients in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)].
14.3 Mantle Cell Lymphoma
A multicenter, single-arm, open-label trial of single-agent REVLIMID was conducted to evaluate the safety and efficacy of REVLIMID in patients with mantle cell lymphoma who have relapsed after or were refractory to bortezomib or a bortezomib-containing regimen. Patients with a creatinine clearance ≥60 mL/min were given REVLIMID at a dose of 25 mg once daily for 21 days every 28 days. Patients with a creatinine clearance ≥30 mL/min and <60 mL/min were given REVLIMID at a dose of 10 mg once daily for 21 days every 28 days. Treatment was continued until disease progression, unacceptable toxicity, or withdrawal of consent.
The trial included patients who were at least 18 years of age with biopsy-proven MCL with measurable disease by CT scan. Patients were required to have received prior treatment with an anthracycline or mitoxantrone, cyclophosphamide, rituximab, and bortezomib, alone or in combination. Patients were required to have documented refractory disease (defined as without any response of PR or better during treatment with bortezomib or a bortezomib-containing regimen), or relapsed disease (defined as progression within one year after treatment with bortezomib or a bortezomib-containing regimen). At enrollment patients were to have an absolute neutrophil counts (ANC) ≥1500/ mm3, platelet counts ≥ 60,000/mm3, serum SGOT/AST or SGPT/ALT ≤3 x upper limit of normal (ULN) unless there was documented evidence of liver involvement by lymphoma, serum total bilirubin ≤1.5 x ULN except in cases of Gilbert's syndrome or documented liver involvement by lymphoma, and calculated creatinine clearance (Cockcroft-Gault formula) ≥30 mL/min.
The median age was 67 years (43-83), 81% were male and 96% were Caucasian. The table below summarizes the baseline disease-related characteristics and prior anti-lymphoma therapy in the Mantle Cell Lymphoma trial.
|
a ECOG = Eastern Cooperative Oncology Group. b MIPI = MCL International Prognostic Index. c High tumor burden is defined as at least one lesion that is ≥5 cm in diameter or 3 lesions that are ≥3 cm in diameter. d Bulky disease is defined as at least one lesion that is ≥7cm in the longest diameter. |
|
|
Baseline Disease Characteristics and Prior Anti -Lymphoma Treatment |
Total Patients
|
|
ECOG Performance Statusa n (%) |
|
|
0 |
43 (32) |
|
1 |
73 (54) |
|
2 |
17 (13) |
|
3 |
1 (<1) |
|
Advanced MCL Stage, n (%) |
|
|
III |
27 (20) |
|
IV |
97 (72) |
|
High or Intermediate MIPI Score b, n (%) |
90 (67) |
|
High Tumor Burdenc, n (%) |
77 (57) |
|
Bulky Diseased, n (%) |
44 (33) |
|
Extranodal Disease, n (%) |
101 (75) |
|
Number of Prior Systemic Anti-Lymphoma Therapies, n (%) |
|
|
Median (range) |
4 (2, 10) |
|
1 |
0 (0) |
|
2 |
29 (22) |
|
3 |
34 (25) |
|
≥ 4 |
71 (53) |
|
Number of Subjects Who Received Prior Regimen Containing, n (%): |
|
|
Anthracycline/mitoxantrone |
133 (99) |
|
Cyclophosphamide |
133 (99) |
|
Rituximab |
134 (100) |
|
Bortezomib |
134 (100) |
|
Refractory to Prior Bortezomib, n (%) |
81 (60) |
|
Refractory to Last Prior Therapy, n (%) |
74 (55) |
|
Prior Autologous Bone Marrow or Stem Cell Transplant, n (%) |
39 (29) |
The efficacy endpoints in the MCL trial were overall response rate (ORR) and duration of response (DOR). Response was determined based on review of radiographic scans by an independent review committee according to a modified version of the International Workshop Lymphoma Response Criteria (Cheson, 1999). The DOR is defined as the time from the initial response (at least PR) to documented disease progression. The efficacy results for the MCL population were based on all evaluable patients who received at least one dose of study drug and are presented in Table 20. The median time to response was 2.2 months (range 1.8 to 13 months).
|
Response Analyses (N = 133) |
N (%) |
95% CI |
|
|
Overall Response Rate (IWRC) (CR + CRu +PR) |
34 (26) |
(18.4, 33.9) |
|
|
Complete Response (CR + CRu) |
9 (7) |
(3.1, 12.5) |
|
|
CR |
1 (1) |
||
|
CRu |
8 (6) |
||
|
Partial Response (PR) |
25 (19) |
||
|
Duration of Response (months) |
Median |
95% CI |
|
|
Duration of Overall Response (CR + CRu + PR) |
(N = 34) |
16.6 |
(7.7, 26.7) |
5.1 Embryo Fetal Toxicity
REVLIMID is a thalidomide analogue and is contraindicated for use during pregnancy. Thalidomide is a known human teratogen that causes life-threatening human birth defects or embryo-fetal death [see Use in Specific Populations (8.1)]. An embryo-fetal development study in monkeys indicates that lenalidomide produced malformations in the offspring of female monkeys who received the drug during pregnancy, similar to birth defects observed in humans following exposure to thalidomide during pregnancy.
REVLIMID is only available through the Lenalidomide REMS program [see Warnings and Precautions (5.2)].
5.10 Tumor Lysis Syndrome
Fatal instances of tumor lysis syndrome (TLS) have been reported during treatment with REVLIMID. The patients at risk of TLS are those with high tumor burden prior to treatment. Monitor patients at risk closely and take appropriate preventive approaches. In the AUGMENT trial in FL or MZL patients, TLS occurred in 2 patients (1.1%) in the REVLIMID/rituximab arm. TLS occurred in 1 patient (0.5%) in the MAGNIFY trial during the REVLIMID/rituximab induction period; the event was a serious, Grade 3 adverse reaction.
5.11 Tumor Flare Reaction
Tumor flare reaction (TFR), including fatal reactions, have occurred during investigational use of REVLIMID for CLL and lymphoma, and is characterized by tender lymph node swelling, low grade fever, pain and rash. REVLIMID is not indicated and not recommended for use in CLL outside of controlled clinical trials.
Monitoring and evaluation for TFR is recommended in patients with MCL, FL, or MZL. Tumor flare reaction may mimic progression of disease (PD).
In the MCL trial, 13/134 (10%) of subjects experienced TFR; all reports were Grade 1 or 2 in severity. All of the events occurred in Cycle 1 and one patient developed TFR again in Cycle 11. In the AUGMENT trial in FL or MZL patients, TFR was reported in 19/176 (10.8%) of patients in REVLIMID with rituximab arm; one patient in the REVLIMID/rituximab arm experienced a Grade 3 TFR. In the MAGNIFY trial, 9/222 (4.1%) of patients experienced TFR; all reports were Grade 1 or 2 in severity and 1 event was considered as serious. In a separate MCL phase 2 trial, one case of TFR resulted in a fatal outcome.
REVLIMID may be continued in patients with Grade 1 and 2 TFR without interruption or modification, at the physician's discretion. Patients with Grade 1 and 2 TFR may also be treated with corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs) and/or narcotic analgesics for management of TFR symptoms. In patients with Grade 3 or 4 TFR, it is recommended to withhold treatment with REVLIMID until TFR resolves to ≤ Grade 1. Patients with Grade 3 or 4 TFR may be treated for management of symptoms per the guidance for treatment of Grade 1 and 2 TFR.
1.5 Marginal Zone Lymphoma
REVLIMID in combination with a rituximab product, is indicated for the treatment of adult patients with previously treated marginal zone lymphoma (MZL).
16.3 Handling and Disposal
Care should be exercised in the handling of REVLIMID. REVLIMID capsules should not be opened or broken. If powder from REVLIMID contacts the skin, wash the skin immediately and thoroughly with soap and water. If REVLIMID contacts the mucous membranes, flush thoroughly with water.
Procedures for the proper handling and disposal of anticancer drugs should be considered. Several guidelines on the subject have been published.1
Dispense no more than a 28-day supply.
5 Warnings and Precautions
-
•Increased Mortality: serious and fatal cardiac adverse reactions occurred in patients with CLL treated with REVLIMID (5.5).
-
•Second Primary Malignancies (SPM): Higher incidences of SPM were observed in controlled trials of patients with MM receiving REVLIMID (5.6).
-
•Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue (5.7).
-
•Hepatotoxicity: Hepatic failure including fatalities; monitor liver function. Stop REVLIMID and evaluate if hepatotoxicity is suspected (5.8).
-
•Severe Cutaneous Reactions: Discontinue REVLIMID for severe reactions (5.9).
-
•Tumor lysis syndrome (TLS) including fatalities: Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions (5.10).
-
•Tumor flare reaction: Serious tumor flare reactions, including fatal reactions, have occurred during investigational use of REVLIMID for chronic lymphocytic leukemia and lymphoma (5.11).
-
•Impaired Stem Cell mobilization: A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. Consider early referral to transplant center (5.12).
-
•Early mortality in MCL: Higher rate of early deaths have occurred in patients with MCL (5.14).
-
•Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue REVLIMID for angioedema and anaphylaxis (5.15).
2 Dosage and Administration
-
•MM combination therapy: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles. (2.1).
-
•MM maintenance therapy following auto-HSCT: 10 mg once daily continuously on Days 1-28 of repeated 28-day cycles (2.1).
-
•MDS: 10 mg once daily (2.2).
-
•MCL: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles (2.3).
-
•FL or MZL: 20 mg once daily orally on Days 1-21 of repeated 28-day cycles for up to 12 cycles (2.4).
-
•Renal impairment: Adjust starting dose based on the creatinine clearance value (2.6).
-
•For concomitant therapy doses, see Full Prescribing Information (2.1, 2.4, 14.1, 14.4).
3 Dosage Forms and Strengths
Capsules:
-
•2.5 mg, white and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink
-
•5 mg, white opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink
-
•10 mg, blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink
-
•15 mg, powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink
-
•20 mg, powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink
-
•25 mg, white opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink
6.2 Postmarketing Experience
The following adverse drug reactions have been identified from the worldwide post-marketing experience with REVLIMID. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure [see Warnings and Precautions Section (5.8 to 5.11, and 5.13)]
Endocrine disorders: Hypothyroidism, hyperthyroidism
Hepatobiliary disorders: Hepatic failure (including fatality), toxic hepatitis, cytolytic hepatitis, cholestatic hepatitis, mixed cytolytic/cholestatic hepatitis, transient abnormal liver laboratory tests
Immune system disorders: Angioedema, anaphylaxis, acute graft-versus-host disease (following allogeneic hematopoietic transplant), solid organ transplant rejection
Infections and infestations: Viral reactivation (such as hepatitis B virus and herpes zoster), progressive multifocal leukoencephalopathy (PML)
Neoplasms benign, malignant and unspecified (including cysts and polyps): Tumor lysis syndrome, tumor flare reaction
Respiratory, thoracic and mediastinal disorders: Pneumonitis
Skin and subcutaneous tissue disorders: Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS)
1.2 Myelodysplastic Syndromes
REVLIMID is indicated for the treatment of adult patients with transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q cytogenetic abnormality with or without additional cytogenetic abnormalities.
5.2 Lenalidomide Rems Program
Because of the embryo-fetal risk [see Warnings and Precautions (5.1)], REVLIMID is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), the Lenalidomide REMS program.
Required components of the Lenalidomide REMS program include the following:
-
•Prescribers must be certified with the Lenalidomide REMS program by enrolling and complying with the REMS requirements.
-
•Patients must sign a Patient-Physician agreement form and comply with the REMS requirements. In particular, female patients of reproductive potential who are not pregnant must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)] and males must comply with contraception requirements [see Use in Specific Populations (8.3)].
-
•Pharmacies must be certified with the Lenalidomide REMS program, must only dispense to patients who are authorized to receive REVLIMID and comply with REMS requirements.
Further information about the Lenalidomide REMS program is available at www.lenalidomiderems.com or by telephone at 1-888-423-5436.
8 Use in Specific Populations
-
•Lactation: Advise not to breastfeed (8.2).
5.9 Severe Cutaneous Reactions
Severe cutaneous reactions including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. These events can be fatal. Patients with a prior history of Grade 4 rash associated with thalidomide treatment should not receive REVLIMID. Consider REVLIMID interruption or discontinuation for Grade 2-3 skin rash. Permanently discontinue REVLIMID for Grade 4 rash, exfoliative or bullous rash, or for other severe cutaneous reactions such as SJS, TEN or DRESS [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Second Primary Malignancies
In clinical trials in patients with MM receiving REVLIMID, an increase of hematologic plus solid tumor second primary malignancies (SPM) notably AML and MDS have been observed. An increase in hematologic SPM including AML and MDS occurred in 5.3% of patients with NDMM receiving REVLIMID in combination with oral melphalan compared with 1.3% of patients receiving melphalan without REVLIMID. The frequency of AML and MDS cases in patients with NDMM treated with REVLIMID in combination with dexamethasone without melphalan was 0.4%.
In patients receiving REVLIMID maintenance therapy following high dose intravenous melphalan and auto-HSCT, hematologic SPM occurred in 7.5% of patients compared to 3.3% in patients receiving placebo. The incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 14.9%, compared to 8.8% in patients receiving placebo with a median follow-up of 91.5 months. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.9% of patients receiving REVLIMID maintenance, compared to 2.6% in the placebo arm.
In patients with relapsed or refractory MM treated with REVLIMID/dexamethasone, the incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 2.3% versus 0.6% in the dexamethasone alone arm. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.1% of patients receiving REVLIMID/dexamethasone, compared to 0.6% in the dexamethasone alone arm.
Patients who received REVLIMID-containing therapy until disease progression did not show a higher incidence of invasive SPM than patients treated in the fixed duration REVLIMID-containing arms. Monitor patients for the development of second primary malignancies. Take into account both the potential benefit of REVLIMID and the risk of second primary malignancies when considering treatment with REVLIMID.
In the AUGMENT trial with FL or MZL patients receiving REVLIMID/rituximab therapy, hematologic plus solid tumor SPMs, notably AML, have been observed. In the AUGMENT trial, hematologic SPM of AML occurred in 0.6% of patients with FL or MZL receiving REVLIMID/rituximab therapy. The incidence of hematologic plus solid tumor SPMs (excluding nonmelanoma skin cancers) was 1.7% in the REVLIMID/rituximab arm with a median follow-up of 29.8 months (range 0.5 to 51.3 months) [see Adverse Reactions (6.1)]. Monitor patients for the development of second primary malignancies. Take into account both the potential benefit of REVLIMID and the risk of second primary malignancies when considering treatment with REVLIMID.
17 Patient Counseling Information
Advise the patient to read the FDA-approved Patient labeling (Medication Guide)
5.12 Impaired Stem Cell Mobilization
A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. In patients who are auto-HSCT candidates, referral to a transplant center should occur early in treatment to optimize the timing of the stem cell collection. In patients who received more than 4 cycles of a REVLIMID-containing treatment or for whom inadequate numbers of CD 34+ cells have been collected with G-CSF alone, G-CSF with cyclophosphamide or the combination of G-CSF with a CXCR4 inhibitor may be considered.
4.2 Severe Hypersensitivity Reactions
REVLIMID is contraindicated in patients who have demonstrated severe hypersensitivity (e.g., angioedema, Stevens-Johnson syndrome, toxic epidermal necrolysis) to lenalidomide [see Warnings and Precautions (5.9, 5.15)].
Package/label Principal Display Panel
NDC 59572-402-28
Revlimid
(lenalidomide) Capsules
2.5 mg
WARNING: POTENTIAL FOR HUMAN BIRTH DEFECTS.
Rx Only
28 Capsules
5.4 Venous and Arterial Thromboembolism
Venous thromboembolic events (VTE [DVT and PE]) and arterial thromboembolic events (ATE, myocardial infarction and stroke) are increased in patients treated with REVLIMID.
A significantly increased risk of DVT (7.4%) and of PE (3.7%) occurred in patients with MM after at least one prior therapy who were treated with REVLIMID and dexamethasone therapy compared to patients treated in the placebo and dexamethasone group (3.1% and 0.9%) in clinical trials with varying use of anticoagulant therapies. In the newly diagnosed multiple myeloma (NDMM) study in which nearly all patients received antithrombotic prophylaxis, DVT was reported as a serious adverse reaction (3.6%, 2.0%, and 1.7%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of PE was similar between the Rd Continuous, Rd18, and MPT Arms (3.8%, 2.8%, and 3.7%, respectively) [see Boxed Warning and Adverse Reactions (6.1)].
Myocardial infarction (1.7%) and stroke (CVA) (2.3%) are increased in patients with MM after at least one prior therapy who were treated with REVLIMID and dexamethasone therapy compared to patients treated with placebo and dexamethasone (0.6%, and 0.9%) in clinical trials. In the NDMM study, myocardial infarction (including acute) was reported as a serious adverse reaction (2.3%, 0.6%, and 1.1%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of CVA was similar between the Rd Continuous, Rd18, and MPT Arms (0.8%, 0.6 %, and 0.6%, respectively) [see Adverse Reactions (6.1)].
Patients with known risk factors, including prior thrombosis, may be at greater risk and actions should be taken to try to minimize all modifiable factors (e.g. hyperlipidemia, hypertension, smoking).
In controlled clinical trials that did not use concomitant thromboprophylaxis, 21.5% overall thrombotic events (Standardized MedDRA Query Embolic and Thrombotic events) occurred in patients with refractory and relapsed MM who were treated with REVLIMID and dexamethasone compared to 8.3% thrombosis in patients treated with placebo and dexamethasone. The median time to first thrombosis event was 2.8 months. In the NDMM study in which nearly all patients received antithrombotic prophylaxis, the overall frequency of thrombotic events was 17.4% in patients in the combined Rd Continuous and Rd18 Arms, and was 11.6% in the MPT Arm. The median time to first thrombosis event was 4.3 months in the combined Rd Continuous and Rd18 Arms.
In the AUGMENT trial, the incidence of VTE (including DVT and PE) in FL or MZL patients was 3.4% in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)]. In the AUGMENT trial, the incidence of ATE (including MI) in FL or MZL patients was 0.6% in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)].
Thromboprophylaxis is recommended. The regimen of thromboprophylaxis should be based on an assessment of the patient's underlying risks. Instruct patients to report immediately any signs and symptoms suggestive of thrombotic events. ESAs and estrogens may further increase the risk of thrombosis and their use should be based on a benefit-risk decision in patients receiving REVLIMID [see Drug Interactions (7.2)].
5.14 Early Mortality in Patients With Mcl
In another MCL study, there was an increase in early deaths (within 20 weeks), 12.9% in the REVLIMID arm versus 7.1% in the control arm. On exploratory multivariate analysis, risk factors for early deaths include high tumor burden, MIPI score at diagnosis, and high WBC at baseline (≥ 10 x 109/L).
14.4 Follicular and Marginal Zone Lymphoma
The efficacy of REVLIMID with rituximab in patients with relapsed or refractory follicular and marginal zone lymphoma was evaluated in the AUGMENT (NCT01938001) and MAGNIFY (NCT01996865) trials.
AUGMENT is a randomized, double-blind, multicenter trial (n=358) in which patients with relapsed or refractory follicular or marginal zone lymphoma were randomized 1:1 to receive REVLIMID and rituximab or rituximab and placebo. AUGMENT included patients diagnosed with Grade 1, 2, or 3a follicular lymphoma, who received at least 1 prior systemic therapy, were refractory or relapsed, not rituximab-refractory, had at least one measurable nodal or extranodal lesion by CT or MRI scan, and had adequate bone marrow, liver, and renal function. Randomization was stratified by follicular versus marginal zone lymphoma, previous rituximab therapy, and time since other anti-lymphoma therapy. In AUGMENT, REVLIMID was administered orally 20 mg once daily for Days 1 to 21 of repeating 28-day cycles for a maximum of 12 cycles or until unacceptable toxicity. The dose of rituximab was 375 mg/m2 every week in Cycle 1 (Days 1, 8, 15, and 22) and on Day 1 of every 28-day cycle from Cycles 2 through 5. All dosage calculations for rituximab were based on the patient's body surface area (BSA), using actual patient weight. Dose adjustments for REVLIMID were allowed based on clinical and laboratory findings. A patient with moderate renal insufficiency (≥30 to <60 mL/minute) received a lower REVLIMID starting dose of 10 mg daily on the same schedule. After 2 cycles, the REVLIMID dose could be increased to 15 mg once daily on Days 1 to 21 of each 28-day cycle if the patient tolerated the medication.
MAGNIFY is an open-label, multicenter trial (n=232) in which patients with relapsed or refractory follicular, marginal zone, or mantle cell lymphoma received 12 induction cycles of REVLIMID and rituximab. MAGNIFY included patients diagnosed with Grade 1, 2,3a, 3b follicular (including transformed), marginal zone, or mantle cell lymphoma Stage I to IV who were previously treated for their lymphoma, had been refractory or had a relapse after their last treatment, had at least one measurable nodal or extranodal lesion by CT or MRI scan, and had adequate bone marrow, liver, and renal function. Patients refractory to rituximab were also included. The information from the subjects who received at least 1 dose of initial therapy in the first 12 induction cycles (n=222) in the MAGNIFY trial was included in the evaluation of the efficacy of REVLIMID/rituximab in patients with relapsed or refractory follicular and marginal zone lymphoma. In MAGNIFY, REVLIMID 20 mg was given on Days 1-21 of repeated 28-day cycles for up to 12 cycles or until unacceptable toxicity, progression, or withdrawal of consent. The dose of rituximab was 375 mg/m2 every week in Cycle 1 (Days 1, 8, 15, and 22) and on Day 1 of every other 28-day cycle (Cycles 3,5,7,9, and 11) up to 12 cycles therapy. All dosage calculations for rituximab were based on the patient BSA and actual weight. Dose adjustments were allowed based on clinical and laboratory findings.
The demographic and disease-related baseline characteristics in the AUGMENT and MAGNIFY trials are shown in the following table.
| Data Cutoff: 22 June 2018 (AUGMENT) and 1 May 2017 (MAGNIFY). a Defined by GELF criteria. b Patient had either 0 (n=2) or 1 prior systemic therapy. ECOG = Eastern Cooperative Oncology Group; FLIPI = follicular lymphoma international prognostic index |
|||
|
Parameter |
AUGMENT Trial |
MAGNIFY Trial |
|
|
REVLIMID + Rituximab
|
Rituximab + Placebo
|
REVLIMID + Rituximab
|
|
|
Age (years) |
|||
|
Median (Max, Min) |
64 (26, 86) |
62 (35, 88) |
65 (35, 91) |
|
Age distribution, n (%) |
|||
|
<65 years |
96 (54) |
107 (59) |
103 (46) |
|
≥65 years |
82 (46) |
73 (41) |
119 (54) |
|
Sex, n (%) |
|||
|
Male |
75 (42) |
97 (54) |
122 (55) |
|
Female |
103 (58) |
83 (46) |
100 (45) |
|
Race |
|||
|
White |
118 (66) |
115 (64) |
206 (93) |
|
Other races |
54 (30) |
64 (36) |
14 (6) |
|
Not collected or reported |
6 (3) |
1 (0.6) |
2 (1) |
|
Body Surface Area (BSA, m2) |
|||
|
Median (Max, Min) |
1.8 (1.4, 3.1) |
1.8 (1.3, 2.7) |
2 (1.3, 2.6) |
|
Disease Type FL or MZL |
|||
|
Follicular lymphoma |
147 (83) |
148 (82) |
177 (80) |
|
Marginal zone lymphoma |
31 (17) |
32 (18) |
45 (20) |
|
MZL subtype at diagnosis (investigator), n (%) |
|||
|
MALT |
14 (45) |
16 (50) |
10 (22) |
|
Nodal |
8 (26) |
10 (31) |
25 (56) |
|
Splenic |
9 (29) |
6 (19) |
10 (22) |
|
FL stage at diagnosis (investigator), n (%) |
|||
|
FL Grade 1-2 |
125 (85) |
123 (83) |
149 (84) |
|
FL Grade 3a |
22 (15) |
25 (17) |
28 (16) |
|
FLIPI score at baseline (calculated), n (%) |
Not Collected |
||
|
Low risk (0,1) |
52 (29) |
67 (37) |
|
|
Intermediate risk (2) |
55 (31) |
58 (32) |
|
|
High risk (≥3) |
69 (39) |
54 (30) |
|
|
Missing |
2 (1) |
1 (0.6) |
|
|
ECOG score at baseline, n (%) |
|||
|
0 |
116 (65) |
128 (71) |
102 (46) |
|
1 |
60 (34) |
50 (28) |
113 (51) |
|
2 |
2 (1) |
2 (1) |
7 (3) |
|
High tumor burdena at baseline, n (%) |
|||
|
Yes |
97 (54) |
86 (48) |
148 (67) |
|
No |
81 (46) |
94 (52) |
74 (33) |
|
Number of prior systemic antilymphoma therapies |
|||
|
1 |
102 (57) |
97 (54) |
94 (42)b |
|
>1 |
76 (43) |
83 (46) |
128 (58) |
In AUGMENT, efficacy was established in the intent-to-treat (ITT) population based on progression-free survival by Independent Review Committee using modified 2007 International Working Group response criteria. Efficacy results are summarized in Table 22.
|
a Median estimate is from Kaplan-Meier analysis. b hazard ratio and its CI were estimated from Cox proportional hazard model adjusting for the stratification 3: previous rituximab treatment (yes, no), time since last antilymphoma therapy (≤ 2, > 2 years), and disease histology (FL, MZL). c p-value from log-rank test stratified by 3 factors noted above: previous rituximab treatment (yes, no), time since last antilymphoma therapy (≤ 2, > 2 years), and disease histology (FL, MZL). d Exact confidence interval for binomial distribution. |
||
|
Parameter |
REVLIMID + Rituximab
|
Rituximab + Placebo
|
|
PFS |
||
|
Patients with event, n (%) |
68 (38.2) |
115 (63.9) |
|
Death |
6 (8.8) |
2 (1.7) |
|
Progression of disease |
62 (91.2) |
113 (98.3) |
|
PFS, median a [95% CI] (months) |
39.4 [ 22.9, NE] |
14.1 [11.4, 16.7] |
|
HR b [95% CI] |
0.46 [ 0.34, 0.62] |
|
|
p-value c |
<0.0001 |
|
|
Objective response (CR+PR) , n(%) [95% CI] d |
138 (77.5) [70.7, 83.4] |
96 (53.3) [45.8, 60.8] |
Kaplan-Meier Curves of Progression-free Survival by IRC Assessment Between Arms in AUGMENT Trial (ITT FL and MZL Population)
a = Stratification factors included: previous rituximab treatment (y/n), time since last anti-lymphoma therapy (≤2 years, >2years), and disease histology (FL or MZL). CI = confidence interval; HR = hazard ratio; KM = Kaplan-Meier; PFS = progression-free survival
5.5 Increased Mortality in Patients With Cll
In a prospective randomized (1:1) clinical trial in the first line treatment of patients with chronic lymphocytic leukemia, single agent REVLIMID therapy increased the risk of death as compared to single agent chlorambucil. In an interim analysis, there were 34 deaths among 210 patients on the REVLIMID treatment arm compared to 18 deaths among 211 patients in the chlorambucil treatment arm, and hazard ratio for overall survival was 1.92 [95% CI: 1.08 – 3.41], consistent with a 92% increase in the risk of death. The trial was halted for safety in July 2013.
Serious adverse cardiovascular reactions, including atrial fibrillation, myocardial infarction, and cardiac failure occurred more frequently in the REVLIMID treatment arm. REVLIMID is not indicated and not recommended for use in CLL outside of controlled clinical trials.
2.3 Recommended Dosage for Mantle Cell Lymphoma
The recommended starting dose of REVLIMID is 25 mg/day orally on Days 1-21 of repeated 28-day cycles for relapsed or refractory mantle cell lymphoma. Treatment should be continued until disease progression or unacceptable toxicity.
Treatment is continued, modified or discontinued based upon clinical and laboratory findings.
2.2 Recommended Dosage for Myelodysplastic Syndromes
The recommended starting dose of REVLIMID is 10 mg daily. Treatment is continued or modified based upon clinical and laboratory findings. Continue treatment until disease progression or unacceptable toxicity.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with lenalidomide have not been conducted.
Lenalidomide was not mutagenic in the bacterial reverse mutation assay (Ames test) and did not induce chromosome aberrations in cultured human peripheral blood lymphocytes, or mutations at the thymidine kinase (tk) locus of mouse lymphoma L5178Y cells. Lenalidomide did not increase morphological transformation in Syrian Hamster Embryo assay or induce micronuclei in the polychromatic erythrocytes of the bone marrow of male rats.
A fertility and early embryonic development study in rats, with administration of lenalidomide up to 500 mg/kg (approximately 200 times the human dose of 25 mg, based on body surface area) produced no parental toxicity and no adverse effects on fertility.
2.6 Recommended Dosage for Patients With Renal Impairment
The recommendations for dosing patients with renal impairment are shown in the following table [see Clinical Pharmacology (12.3)].
|
Renal Function
|
Dose in REVLIMID Combination Therapy for MM and MCL |
Dose in REVLIMID Combination Therapy for FL and MZL |
Dose in REVLIMID Maintenance Therapy Following Auto-HSCT for MM and for MDS |
|
CLcr 30 to 60 mL/min |
10 mg once daily |
10 mg once daily |
5 mg once daily |
|
CLcr below 30 mL/min (not requiring dialysis) |
15 mg every other day |
5 mg once daily |
2.5 mg once daily |
|
CLcr below 30 mL/min (requiring dialysis) |
5 mg once daily. On dialysis days, administer the dose following dialysis. |
5 mg once daily. On dialysis days, administer the dose following dialysis. |
2.5 mg once daily. On dialysis days, administer the dose following dialysis. |
2.5 Dosage Modifications for Non Hematologic Adverse Reactions
For non-hematologic Grade 3/4 toxicities judged to be related to REVLIMID, hold treatment and restart at the physician's discretion at next lower dose level when toxicity has resolved to Grade 2 or below.
Permanently discontinue REVLIMID for angioedema, anaphylaxis, Grade 4 rash, skin exfoliation, bullae, or any other severe dermatologic reactions [see Warnings and Precautions (5.9, 5.15)].
7.2 Concomitant Therapies That May Increase the Risk of Thrombosis
Erythropoietic agents, or other agents that may increase the risk of thrombosis, such as estrogen containing therapies, should be used with caution after making a benefit-risk assessment in patients receiving REVLIMID [see Warnings and Precautions (5.4)].
2.4 Recommended Dosage for Follicular Lymphoma Or Marginal Zone Lymphoma
The recommended starting dose of REVLIMID is 20 mg orally once daily on Days 1-21 of repeated 28-day cycles for up to 12 cycles of treatment in combination with a rituximab-product. Refer to Section 14.4 for specific rituximab dosing from the AUGMENT trial. For dose adjustments due to toxicity with rituximab, refer to the product prescribing information.
14.2 Myelodysplastic Syndromes (mds) With A Deletion 5q Cytogenetic Abnormality
The efficacy and safety of REVLIMID were evaluated in patients with transfusion-dependent anemia in low- or intermediate-1- risk MDS with a 5q (q31-33) cytogenetic abnormality in isolation or with additional cytogenetic abnormalities, at a dose of 10 mg once daily or 10 mg once daily for 21 days every 28 days in an open-label, single-arm, multi-center study. The major study was not designed nor powered to prospectively compare the efficacy of the 2 dosing regimens. Sequential dose reductions to 5 mg daily and 5 mg every other day, as well as dose delays, were allowed for toxicity [Dosage and Administration (2.2)].
This major study enrolled 148 patients who had RBC transfusion dependent anemia. RBC transfusion dependence was defined as having received ≥ 2 units of RBCs within 8 weeks prior to study treatment. The study enrolled patients with absolute neutrophil counts (ANC) ≥ 500/mm3, platelet counts ≥ 50,000/mm3, serum creatinine ≤ 2.5 mg/dL, serum SGOT/AST or SGPT/ALT ≤ 3 x upper limit of normal (ULN), and serum direct bilirubin ≤ 2 mg/dL. Granulocyte colony-stimulating factor was permitted for patients who developed neutropenia or fever in association with neutropenia. Baseline patient and disease-related characteristics are summarized in Table 18.
|
a IPSS Risk Category: Low (combined score = 0), Intermediate-1 (combined score = 0.5 to 1), Intermediate-2 (combined score = 1.5 to 2.0), High (combined score ≥ 2.5); Combined score = (Marrow blast score + Karyotype score + Cytopenia score). b French-American-British (FAB) classification of MDS. |
||
|
Overall
|
||
|
Age (years) |
||
|
Median |
71 |
|
|
Min, Max |
37, 95 |
|
|
Gender |
n |
(%) |
|
Male |
51 |
(34.5) |
|
Female |
97 |
(65.5) |
|
Race |
n |
(%) |
|
White |
143 |
(96.6) |
|
Other |
5 |
( 3.4) |
|
Duration of MDS (years) |
||
|
Median |
2.5 |
|
|
Min, Max |
0.1, 20.7 |
|
|
Del 5 (q31-33) Cytogenetic Abnormality |
n |
(%) |
|
Yes |
148 |
(100) |
|
Other cytogenetic abnormalities |
37 |
(25.2) |
|
IPSS Score a |
n |
(%) |
|
Low (0) |
55 |
(37.2) |
|
Intermediate-1 (0.5-1.0) |
65 |
(43.9) |
|
Intermediate-2 (1.5-2.0) |
6 |
( 4.1) |
|
High (≥2.5) |
2 |
( 1.4) |
|
Missing |
20 |
(13.5) |
|
FAB Classification b from central review |
n |
(%) |
|
RA |
77 |
(52) |
|
RARS |
16 |
(10.8) |
|
RAEB |
30 |
(20.3) |
|
CMML |
3 |
( 2) |
The frequency of RBC transfusion independence was assessed using criteria modified from the International Working Group (IWG) response criteria for MDS. RBC transfusion independence was defined as the absence of any RBC transfusion during any consecutive "rolling" 56 days (8 weeks) during the treatment period.
Transfusion independence was seen in 99/148 (67%) patients (95% CI [59, 74]). The median duration from the date when RBC transfusion independence was first declared (i.e., the last day of the 56-day RBC transfusion-free period) to the date when an additional transfusion was received after the 56-day transfusion-free period among the 99 responders was 44 weeks (range of 0 to >67 weeks). Ninety percent of patients who achieved a transfusion benefit did so by completion of three months in the study.
RBC transfusion independence rates were unaffected by age or gender.
The dose of REVLIMID was reduced or interrupted at least once due to an adverse event in 118 (79.7%) of the 148 patients; the median time to the first dose reduction or interruption was 21 days (mean, 35.1 days; range, 2-253 days), and the median duration of the first dose interruption was 22 days (mean, 28.5 days; range, 2-265 days). A second dose reduction or interruption due to adverse events was required in 50 (33.8%) of the 148 patients. The median interval between the first and second dose reduction or interruption was 51 days (mean, 59.7 days; range, 15-205 days) and the median duration of the second dose interruption was 21 days (mean, 26 days; range, 2-148 days).
Warning: Embryo Fetal Toxicity, Hematologic Toxicity, and Venous and Arterial Thromboembolism
Embryo-Fetal Toxicity
Do not use REVLIMID during pregnancy. Lenalidomide, a thalidomide analogue, caused limb abnormalities in a developmental monkey study. Thalidomide is a known human teratogen that causes severe life-threatening human birth defects. If lenalidomide is used during pregnancy, it may cause birth defects or embryo-fetal death. In females of reproductive potential, obtain 2 negative pregnancy tests before starting REVLIMID® treatment. Females of reproductive potential must use 2 forms of contraception or continuously abstain from heterosexual sex during and for 4 weeks after REVLIMID treatment [see Warnings and Precautions (5.1), and Medication Guide (17)]. To avoid embryo-fetal exposure to lenalidomide, REVLIMID is only available through a restricted distribution program, the Lenalidomide REMS program (5.2).
Information about the Lenalidomide REMS program is available at www.lenalidomiderems.com or by calling the REMS Call Center at 1-888-423-5436.
Hematologic Toxicity (Neutropenia and Thrombocytopenia)
REVLIMID can cause significant neutropenia and thrombocytopenia. Eighty percent of patients with del 5q myelodysplastic syndromes had to have a dose delay/reduction during the major study. Thirty-four percent of patients had to have a second dose delay/reduction. Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the study. Patients on therapy for del 5q myelodysplastic syndromes should have their complete blood counts monitored weekly for the first 8 weeks of therapy and at least monthly thereafter. Patients may require dose interruption and/or reduction. Patients may require use of blood product support and/or growth factors [see Dosage and Administration (2.2)].
Venous and Arterial Thromboembolism
REVLIMID has demonstrated a significantly increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as risk of myocardial infarction and stroke in patients with multiple myeloma who were treated with REVLIMID and dexamethasone therapy. Monitor for and advise patients about signs and symptoms of thromboembolism. Advise patients to seek immediate medical care if they develop symptoms such as shortness of breath, chest pain, or arm or leg swelling. Thromboprophylaxis is recommended and the choice of regimen should be based on an assessment of the patient’s underlying risks [see Warnings and Precautions (5.4)].
5.7 Increased Mortality in Patients With Mm When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone
In two randomized clinical trials in patients with MM, the addition of pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with MM with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
Structured Label Content
Data
Animal data
In an embryo-fetal developmental toxicity study in monkeys, teratogenicity, including thalidomide-like limb defects, occurred in offspring when pregnant monkeys received oral lenalidomide during organogenesis. Exposure (AUC) in monkeys at the lowest dose was 0.17 times the human exposure at the maximum recommended human dose (MRHD) of 25 mg. Similar studies in pregnant rabbits and rats at 20 times and 200 times the MRHD respectively, produced embryo lethality in rabbits and no adverse reproductive effects in rats.
In a pre- and post-natal development study in rats, animals received lenalidomide from organogenesis through lactation. The study revealed a few adverse effects on the offspring of female rats treated with lenalidomide at doses up to 500 mg/kg (approximately 200 times the human dose of 25 mg based on body surface area). The male offspring exhibited slightly delayed sexual maturation and the female offspring had slightly lower body weight gains during gestation when bred to male offspring. As with thalidomide, the rat model may not adequately address the full spectrum of potential human embryo-fetal developmental effects for lenalidomide.
Following daily oral administration of lenalidomide from Gestation Day 7 through Gestation Day 20 in pregnant rabbits, fetal plasma lenalidomide concentrations were approximately 20-40% of the maternal Cmax. Following a single oral dose to pregnant rats, lenalidomide was detected in fetal plasma and tissues; concentrations of radioactivity in fetal tissues were generally lower than those in maternal tissues. These data indicated that lenalidomide crossed the placenta.
Section 42229-5 (42229-5)
REVLIMID Combination Therapy
The recommended starting dose of REVLIMID is 25 mg orally once daily on Days 1-21 of repeated 28-day cycles in combination with dexamethasone. Refer to Section 14.1 for specific dexamethasone dosing. For patients greater than 75 years old, the starting dose of dexamethasone may be reduced [see Clinical Studies (14.1)]. Treatment should be continued until disease progression or unacceptable toxicity.
In patients who are not eligible for auto-HSCT, treatment should continue until disease progression or unacceptable toxicity. For patients who are auto-HSCT-eligible, hematopoietic stem cell mobilization should occur within 4 cycles of a REVLIMID-containing therapy [see Warnings and Precautions (5.12)].
Section 43683-2 (43683-2)
7.1 Digoxin
When digoxin was co-administered with multiple doses of REVLIMID (10 mg/day) the digoxin Cmax and AUCinf were increased by 14%. Periodically monitor digoxin plasma levels, in accordance with clinical judgment and based on standard clinical practice in patients receiving this medication, during administration of REVLIMID.
16.2 Storage
Store at 20°C - 25°C (68°F - 77°F); excursions permitted to 15°C - 30°C (59°F - 86°F) [See USP Controlled Room Temperature].
7.3 Warfarin
Co-administration of multiple doses of REVLIMID (10 mg/day) with a single dose of warfarin (25 mg) had no effect on the pharmacokinetics of lenalidomide or R- and S-warfarin. Expected changes in laboratory assessments of PT and INR were observed after warfarin administration, but these changes were not affected by concomitant REVLIMID administration. It is not known whether there is an interaction between dexamethasone and warfarin. Close monitoring of PT and INR is recommended in patients with MM taking concomitant warfarin.
10 Overdosage (10 OVERDOSAGE)
There is no specific experience in the management of REVLIMID overdose in patients with MM, MDS, MCL, FL, or MZL. In dose-ranging studies in healthy subjects, some were exposed to up to 200 mg (administered 100 mg BID) and in single-dose studies, some subjects were exposed to up to 400 mg. Pruritus, urticaria, rash, and elevated liver transaminases were the primary reported AEs. In clinical trials, the dose-limiting toxicity was neutropenia and thrombocytopenia.
15 References (15 REFERENCES)
1. OSHA Hazardous Drugs. OSHA [Accessed on 29 January 2013, from http://www.osha.gov/SLTC/hazardousdrugs/index.html]
4.1 Pregnancy
REVLIMID can cause fetal harm when administered to a pregnant female. Limb abnormalities were seen in the offspring of monkeys that were dosed with lenalidomide during organogenesis. This effect was seen at all doses tested. Due to the results of this developmental monkey study, and lenalidomide's structural similarities to thalidomide, a known human teratogen, lenalidomide is contraindicated in females who are pregnant [see Boxed Warning]. If this drug is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus [see Warnings and Precautions (5.1, 5.2), Use in Special Populations (8.1, 8.3)].
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in females exposed to REVLIMID during pregnancy as well as female partners of male patients who are exposed to REVLIMID. This registry is also used to understand the root cause for the pregnancy. Report any suspected fetal exposure to REVLIMID to the FDA via the MedWatch program at 1-800-FDA-1088 and also to the REMS Call Center at 1-888-423-5436.
Risk Summary
Based on the mechanism of action [see Clinical Pharmacology (12.1)] and findings from animal studies [see Data], REVLIMID can cause embryo-fetal harm when administered to a pregnant female and is contraindicated during pregnancy [see Boxed Warning, Contraindications (4.1), and Use in Specific Populations (5.1)].
REVLIMID is a thalidomide analogue. Thalidomide is a human teratogen, inducing a high frequency of severe and life-threatening birth defects such as amelia (absence of limbs), phocomelia (short limbs), hypoplasticity of the bones, absence of bones, external ear abnormalities (including anotia, micropinna, small or absent external auditory canals), facial palsy, eye abnormalities (anophthalmos, microphthalmos), and congenital heart defects. Alimentary tract, urinary tract, and genital malformations have also been documented and mortality at or shortly after birth has been reported in about 40% of infants.
Lenalidomide caused thalidomide-type limb defects in monkey offspring. Lenalidomide crossed the placenta after administration to pregnant rabbits and pregnant rats [see Data]. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus.
If pregnancy does occur during treatment, immediately discontinue the drug. Under these conditions, refer patient to an obstetrician/gynecologist experienced in reproductive toxicity for further evaluation and counseling. Report any suspected fetal exposure to REVLIMID to the FDA via the MedWatch program at 1-800-FDA-1088 and also to the REMS Call Center at 1-888-423-5436.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk in the U.S. general population of major birth defects is 2%-4% and of miscarriage is 15%-20% of clinically recognized pregnancies.
11 Description (11 DESCRIPTION)
REVLIMID, a thalidomide analogue, is an immunomodulatory agent with antiangiogenic and antineoplastic properties. The chemical name is 3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione and it has the following chemical structure:
3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione
The empirical formula for lenalidomide is C13H13N3O3, and the gram molecular weight is 259.3.
Lenalidomide is an off-white to pale-yellow solid powder. It is soluble in organic solvent/water mixtures, and buffered aqueous solvents. Lenalidomide is more soluble in organic solvents and low pH solutions. Solubility was significantly lower in less acidic buffers, ranging from about 0.4 to 0.5 mg/ml. Lenalidomide has an asymmetric carbon atom and can exist as the optically active forms S(-) and R(+), and is produced as a racemic mixture with a net optical rotation of zero.
REVLIMID is available in 2.5 mg, 5 mg, 10 mg, 15 mg, 20 mg and 25 mg capsules for oral administration. Each capsule contains lenalidomide as the active ingredient and the following inactive ingredients: lactose anhydrous, microcrystalline cellulose, croscarmellose sodium, and magnesium stearate. The 5 mg and 25 mg capsule shell contains gelatin, titanium dioxide and black ink. The 2.5 mg and 10 mg capsule shell contains gelatin, FD&C blue #2, yellow iron oxide, titanium dioxide and black ink. The 15 mg capsule shell contains gelatin, FD&C blue #2, titanium dioxide and black ink. The 20 mg capsule shell contains gelatin, FD&C blue #2, yellow iron oxide, titanium dioxide and black ink.
Medication Guide
REVLIMID® (rev-li-mid)
(lenalidomide) capsules
|
What is the most important information I should know about REVLIMID?
Females must not get pregnant:
Females who can become pregnant:
If you become pregnant while taking REVLIMID, stop taking it right away and call your healthcare provider. If your healthcare provider is not available, you can call the REMS Call Center at 1-888-423-5436. Healthcare providers and patients should report all cases of pregnancy to:
There is a pregnancy exposure registry that monitors the outcomes of females who take REVLIMID during pregnancy, or if their male partner takes REVLIMID and they are exposed during pregnancy. You can enroll in this registry by calling the REMS Call Center at the phone number listed above. REVLIMID can pass into human semen:
Men, if your female partner becomes pregnant, you should call your healthcare provider right away.
|
||
|
What is REVLIMID?
REVLIMID should not be used to treat people who have chronic lymphocytic leukemia (CLL) unless they are participants in a controlled clinical trial. |
||
|
Who should not take REVLIMID?
|
||
|
What should I tell my healthcare provider before taking REVLIMID?
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. REVLIMID and other medicines may affect each other, causing serious side effects. Talk with your healthcare provider before taking any new medicines. |
||
|
How should I take REVLIMID?
|
||
|
What should I avoid while taking REVLIMID?
|
||
|
What are the possible side effects of REVLIMID?
|
||
|
|
|
Call your healthcare provider right away if you develop any of the following signs or symptoms during treatment with REVLIMID: |
||
|
|
|
|
Get emergency medical help right away if you develop any of the following signs or symptoms during treatment with REVLIMID: |
||
|
|
|
Your healthcare provider may tell you to decrease your dose, temporarily stop or permanently stop taking REVLIMID if you develop certain serious side effects during treatment with REVLIMID.
The most common side effects of REVLIMID include: |
||
|
|
|
|
These are not all the possible side effects of REVLIMID. |
||
|
How should I store REVLIMID?
Keep REVLIMID and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of REVLIMID.
For more information, call 1-888-423-5436 or go to www.lenalidomiderems.com. |
||
|
What are the ingredients in REVLIMID?
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: MAR 2023
16.1 How Supplied
White and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink:
|
2.5 mg bottles of 28 |
(NDC 59572-402-28) |
|
|
2.5 mg bottles of 100 |
(NDC 59572-402-00) |
White opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink:
|
5 mg bottles of 28 |
(NDC 59572-405-28) |
|
|
5 mg bottles of 100 |
(NDC 59572-405-00) |
Blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink:
|
10 mg bottles of 28 |
(NDC 59572-410-28) |
|
|
10 mg bottles of 100 |
(NDC 59572-410-00) |
Powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink:
|
15 mg bottles of 21 |
(NDC 59572-415-21) |
|
|
15 mg bottles of 100 |
(NDC 59572-415-00) |
Powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink.
|
20 mg bottles of 21 |
(NDC 59572-420-21) |
|
|
20 mg bottles of 100 |
(NDC 59572-420-00) |
White opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink:
|
25 mg bottles of 21 |
(NDC 59572-425-21) |
|
|
25 mg bottles of 100 |
(NDC 59572-425-00) |
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients.
2.7 Administration
Advise patients to take REVLIMID orally at about the same time each day, either with or without food. Advise patients to swallow REVLIMID capsules whole with water and not to open, break, or chew them.
5.8 Hepatotoxicity
Hepatic failure, including fatal cases, has occurred in patients treated with REVLIMID in combination with dexamethasone. In clinical trials, 15% of patients experienced hepatotoxicity (with hepatocellular, cholestatic and mixed characteristics); 2% of patients with MM and 1% of patients with myelodysplasia had serious hepatotoxicity events. The mechanism of drug-induced hepatotoxicity is unknown. Pre-existing viral liver disease, elevated baseline liver enzymes, and concomitant medications may be risk factors. Monitor liver enzymes periodically. Stop REVLIMID upon elevation of liver enzymes. After return to baseline values, treatment at a lower dose may be considered.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described in detail in other sections of the prescribing information:
-
oEmbryo-Fetal Toxicity [see Boxed Warning, Warnings and Precautions (5.1, 5.2)]
-
oHematologic Toxicity [see Boxed Warning, Warnings and Precautions (5.3)]
-
oVenous and Arterial Thromboembolism [see Boxed Warning, Warnings and Precautions (5.4)]
-
oIncreased Mortality in Patients with CLL [see Warnings and Precautions (5.5)]
-
oSecond Primary Malignancies [see Warnings and Precautions (5.6)]
-
oIncreased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.7)]
-
oHepatotoxicity [see Warnings and Precautions (5.8)]
-
oSevere Cutaneous Reactions[see Warnings and Precautions (5.9)]
-
oTumor Lysis Syndrome [see Warnings and Precautions (5.10)]
-
oTumor Flare Reactions [see Warnings and Precautions (5.11)]
-
oImpaired Stem Cell Mobilization [see Warnings and Precautions (5.12)]
-
oThyroid Disorders [see Warnings and Precautions (5.13)]
-
oEarly Mortality in Patients with MCL [see Warnings and Precautions (5.14)]
-
oHypersensitivity [see Warnings and Precautions (5.15)]
7 Drug Interactions (7 DRUG INTERACTIONS)
1.1 Multiple Myeloma
REVLIMID in combination with dexamethasone is indicated for the treatment of adult patients with multiple myeloma (MM).
REVLIMID is indicated as maintenance therapy in adult patients with MM following autologous hematopoietic stem cell transplantation (auto-HSCT).
8.6 Renal Impairment
Adjust the starting dose of REVLIMID based on the creatinine clearance value and for patients on dialysis [see Dosage and Administration (2.6)].
5.15 Hypersensitivity
Hypersensitivity, including angioedema, anaphylaxis, and anaphylactic reactions to REVLIMID has been reported. Permanently discontinue REVLIMID for angioedema and anaphylaxis [see Dosage and Administration (2.2)].
1.6 Limitations of Use
REVLIMID is not indicated and is not recommended for the treatment of patients with CLL outside of controlled clinical trials [see Warnings and Precautions (5.5)].
5.13 Thyroid Disorders
Both hypothyroidism and hyperthyroidism have been reported [see Adverse Reactions (6.2)]. Measure thyroid function before start of REVLIMID treatment and during therapy.
1 Indications and Usage (1 INDICATIONS AND USAGE)
REVLIMID is a thalidomide analogue indicated for the treatment of adult patients with:
-
•Multiple myeloma (MM), in combination with dexamethasone (1.1).
-
•MM, as maintenance following autologous hematopoietic stem cell transplantation (auto-HSCT) (1.1).
-
•Transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q abnormality with or without additional cytogenetic abnormalities (1.2).
-
•Mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib (1.3).
-
•Previously treated follicular lymphoma (FL), in combination with a rituximab product (1.4).
-
•Previously treated marginal zone lymphoma (MZL), in combination with a rituximab product (1.5).
Limitations of Use:
-
•REVLIMID is not indicated and is not recommended for the treatment of patients with chronic lymphocytic leukemia (CLL) outside of controlled clinical trials (1.4).
1.4 Follicular Lymphoma
REVLIMID in combination with a rituximab product, is indicated for the treatment of adult patients with previously treated follicular lymphoma (FL).
1.3 Mantle Cell Lymphoma
REVLIMID is indicated for the treatment of adult patients with mantle cell lymphoma (MCL) whose disease has relapsed or progressed after two prior therapies, one of which included bortezomib.
12.1 Mechanism of Action
Lenalidomide is an analogue of thalidomide with immunomodulatory, antiangiogenic, and antineoplastic properties. Cellular activities of lenalidomide are mediated through its target cereblon, a component of a cullin ring E3 ubiquitin ligase enzyme complex. In vitro, in the presence of drug, substrate proteins (including Aiolos, Ikaros, and CK1α) are targeted for ubiquitination and subsequent degradation leading to direct cytotoxic and immunomodulatory effects. Lenalidomide inhibits proliferation and induces apoptosis of certain hematopoietic tumor cells including MM, mantle cell lymphoma, and del (5q) myelodysplastic syndromes, follicular lymphoma and marginal zone lymphoma in vitro. Lenalidomide causes a delay in tumor growth in some in vivo nonclinical hematopoietic tumor models including MM. Immunomodulatory properties of lenalidomide include increased number and activation of T cells and natural killer (NK) cells leading to direct and enhanced antibody-dependent cell-mediated cytotoxicity (ADCC) via increased secretion of interleukin-2 and interferon-gamma, increased numbers of NKT cells, and inhibition of pro-inflammatory cytokines (e.g., TNF-α and IL-6) by monocytes. In MM cells, the combination of lenalidomide and dexamethasone synergizes the inhibition of cell proliferation and the induction of apoptosis. The combination of lenalidomide and rituximab increases ADCC and direct tumor apoptosis in follicular lymphoma cells and increases ADCC in marginal zone lymphoma cells compared to rituximab alone in vitro.
5.3 Hematologic Toxicity
REVLIMID can cause significant neutropenia and thrombocytopenia. Monitor patients with neutropenia for signs of infection. Advise patients to observe for bleeding or bruising, especially with use of concomitant medication that may increase risk of bleeding. Patients taking REVLIMID should have their complete blood counts assessed periodically as described below [see Dosage and Administration (2.1, 2.2, 2.3)].
Monitor complete blood counts (CBC) in patients taking REVLIMID in combination with dexamethasone or as REVLIMID maintenance therapy for MM every 7 days (weekly) for the first 2 cycles, on Days 1 and 15 of Cycle 3, and every 28 days (4 weeks) thereafter. A dose interruption and/or dose reduction may be required [see Dosage and Administration (2.1)]. In the MM maintenance therapy trials, Grade 3 or 4 neutropenia was reported in up to 59% of REVLIMID-treated patients and Grade 3 or 4 thrombocytopenia in up to 38% of REVLIMID-treated patients [see Adverse Reactions (6.1)].
Monitor complete blood counts (CBC) in patients taking REVLIMID for MDS weekly for the first 8 weeks and at least monthly thereafter. Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the MDS study. In the 48% of patients who developed Grade 3 or 4 neutropenia, the median time to onset was 42 days (range, 14-411 days), and the median time to documented recovery was 17 days (range, 2-170 days). In the 54% of patients who developed Grade 3 or 4 thrombocytopenia, the median time to onset was 28 days (range, 8-290 days), and the median time to documented recovery was 22 days (range, 5-224 days) [see Boxed Warning and Dosage and Administration (2.2)].
Monitor complete blood counts (CBC) in patients taking REVLIMID for MCL weekly for the first cycle (28 days), every 2 weeks during cycles 2-4, and then monthly thereafter. Patients may require dose interruption and/or dose reduction. In the MCL trial, Grade 3 or 4 neutropenia was reported in 43% of the patients. Grade 3 or 4 thrombocytopenia was reported in 28% of the patients.
Monitor complete blood counts (CBC) in patients taking REVLIMID for FL or MZL weekly for the first 3 weeks of Cycle 1 (28 days), every 2 weeks during Cycles 2-4, and then monthly thereafter. Patients may require dose interruption and/or dose reduction. In the AUGMENT and MAGNIFY trials, Grade 3 or 4 neutropenia was reported in 50% and 33%, respectively, of patients in the REVLIMID/rituximab arm. Grade 3 or 4 thrombocytopenia was reported in 2% and 8%, respectively, of patients in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)].
14.3 Mantle Cell Lymphoma
A multicenter, single-arm, open-label trial of single-agent REVLIMID was conducted to evaluate the safety and efficacy of REVLIMID in patients with mantle cell lymphoma who have relapsed after or were refractory to bortezomib or a bortezomib-containing regimen. Patients with a creatinine clearance ≥60 mL/min were given REVLIMID at a dose of 25 mg once daily for 21 days every 28 days. Patients with a creatinine clearance ≥30 mL/min and <60 mL/min were given REVLIMID at a dose of 10 mg once daily for 21 days every 28 days. Treatment was continued until disease progression, unacceptable toxicity, or withdrawal of consent.
The trial included patients who were at least 18 years of age with biopsy-proven MCL with measurable disease by CT scan. Patients were required to have received prior treatment with an anthracycline or mitoxantrone, cyclophosphamide, rituximab, and bortezomib, alone or in combination. Patients were required to have documented refractory disease (defined as without any response of PR or better during treatment with bortezomib or a bortezomib-containing regimen), or relapsed disease (defined as progression within one year after treatment with bortezomib or a bortezomib-containing regimen). At enrollment patients were to have an absolute neutrophil counts (ANC) ≥1500/ mm3, platelet counts ≥ 60,000/mm3, serum SGOT/AST or SGPT/ALT ≤3 x upper limit of normal (ULN) unless there was documented evidence of liver involvement by lymphoma, serum total bilirubin ≤1.5 x ULN except in cases of Gilbert's syndrome or documented liver involvement by lymphoma, and calculated creatinine clearance (Cockcroft-Gault formula) ≥30 mL/min.
The median age was 67 years (43-83), 81% were male and 96% were Caucasian. The table below summarizes the baseline disease-related characteristics and prior anti-lymphoma therapy in the Mantle Cell Lymphoma trial.
|
a ECOG = Eastern Cooperative Oncology Group. b MIPI = MCL International Prognostic Index. c High tumor burden is defined as at least one lesion that is ≥5 cm in diameter or 3 lesions that are ≥3 cm in diameter. d Bulky disease is defined as at least one lesion that is ≥7cm in the longest diameter. |
|
|
Baseline Disease Characteristics and Prior Anti -Lymphoma Treatment |
Total Patients
|
|
ECOG Performance Statusa n (%) |
|
|
0 |
43 (32) |
|
1 |
73 (54) |
|
2 |
17 (13) |
|
3 |
1 (<1) |
|
Advanced MCL Stage, n (%) |
|
|
III |
27 (20) |
|
IV |
97 (72) |
|
High or Intermediate MIPI Score b, n (%) |
90 (67) |
|
High Tumor Burdenc, n (%) |
77 (57) |
|
Bulky Diseased, n (%) |
44 (33) |
|
Extranodal Disease, n (%) |
101 (75) |
|
Number of Prior Systemic Anti-Lymphoma Therapies, n (%) |
|
|
Median (range) |
4 (2, 10) |
|
1 |
0 (0) |
|
2 |
29 (22) |
|
3 |
34 (25) |
|
≥ 4 |
71 (53) |
|
Number of Subjects Who Received Prior Regimen Containing, n (%): |
|
|
Anthracycline/mitoxantrone |
133 (99) |
|
Cyclophosphamide |
133 (99) |
|
Rituximab |
134 (100) |
|
Bortezomib |
134 (100) |
|
Refractory to Prior Bortezomib, n (%) |
81 (60) |
|
Refractory to Last Prior Therapy, n (%) |
74 (55) |
|
Prior Autologous Bone Marrow or Stem Cell Transplant, n (%) |
39 (29) |
The efficacy endpoints in the MCL trial were overall response rate (ORR) and duration of response (DOR). Response was determined based on review of radiographic scans by an independent review committee according to a modified version of the International Workshop Lymphoma Response Criteria (Cheson, 1999). The DOR is defined as the time from the initial response (at least PR) to documented disease progression. The efficacy results for the MCL population were based on all evaluable patients who received at least one dose of study drug and are presented in Table 20. The median time to response was 2.2 months (range 1.8 to 13 months).
|
Response Analyses (N = 133) |
N (%) |
95% CI |
|
|
Overall Response Rate (IWRC) (CR + CRu +PR) |
34 (26) |
(18.4, 33.9) |
|
|
Complete Response (CR + CRu) |
9 (7) |
(3.1, 12.5) |
|
|
CR |
1 (1) |
||
|
CRu |
8 (6) |
||
|
Partial Response (PR) |
25 (19) |
||
|
Duration of Response (months) |
Median |
95% CI |
|
|
Duration of Overall Response (CR + CRu + PR) |
(N = 34) |
16.6 |
(7.7, 26.7) |
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
REVLIMID is a thalidomide analogue and is contraindicated for use during pregnancy. Thalidomide is a known human teratogen that causes life-threatening human birth defects or embryo-fetal death [see Use in Specific Populations (8.1)]. An embryo-fetal development study in monkeys indicates that lenalidomide produced malformations in the offspring of female monkeys who received the drug during pregnancy, similar to birth defects observed in humans following exposure to thalidomide during pregnancy.
REVLIMID is only available through the Lenalidomide REMS program [see Warnings and Precautions (5.2)].
5.10 Tumor Lysis Syndrome
Fatal instances of tumor lysis syndrome (TLS) have been reported during treatment with REVLIMID. The patients at risk of TLS are those with high tumor burden prior to treatment. Monitor patients at risk closely and take appropriate preventive approaches. In the AUGMENT trial in FL or MZL patients, TLS occurred in 2 patients (1.1%) in the REVLIMID/rituximab arm. TLS occurred in 1 patient (0.5%) in the MAGNIFY trial during the REVLIMID/rituximab induction period; the event was a serious, Grade 3 adverse reaction.
5.11 Tumor Flare Reaction
Tumor flare reaction (TFR), including fatal reactions, have occurred during investigational use of REVLIMID for CLL and lymphoma, and is characterized by tender lymph node swelling, low grade fever, pain and rash. REVLIMID is not indicated and not recommended for use in CLL outside of controlled clinical trials.
Monitoring and evaluation for TFR is recommended in patients with MCL, FL, or MZL. Tumor flare reaction may mimic progression of disease (PD).
In the MCL trial, 13/134 (10%) of subjects experienced TFR; all reports were Grade 1 or 2 in severity. All of the events occurred in Cycle 1 and one patient developed TFR again in Cycle 11. In the AUGMENT trial in FL or MZL patients, TFR was reported in 19/176 (10.8%) of patients in REVLIMID with rituximab arm; one patient in the REVLIMID/rituximab arm experienced a Grade 3 TFR. In the MAGNIFY trial, 9/222 (4.1%) of patients experienced TFR; all reports were Grade 1 or 2 in severity and 1 event was considered as serious. In a separate MCL phase 2 trial, one case of TFR resulted in a fatal outcome.
REVLIMID may be continued in patients with Grade 1 and 2 TFR without interruption or modification, at the physician's discretion. Patients with Grade 1 and 2 TFR may also be treated with corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs) and/or narcotic analgesics for management of TFR symptoms. In patients with Grade 3 or 4 TFR, it is recommended to withhold treatment with REVLIMID until TFR resolves to ≤ Grade 1. Patients with Grade 3 or 4 TFR may be treated for management of symptoms per the guidance for treatment of Grade 1 and 2 TFR.
1.5 Marginal Zone Lymphoma
REVLIMID in combination with a rituximab product, is indicated for the treatment of adult patients with previously treated marginal zone lymphoma (MZL).
16.3 Handling and Disposal
Care should be exercised in the handling of REVLIMID. REVLIMID capsules should not be opened or broken. If powder from REVLIMID contacts the skin, wash the skin immediately and thoroughly with soap and water. If REVLIMID contacts the mucous membranes, flush thoroughly with water.
Procedures for the proper handling and disposal of anticancer drugs should be considered. Several guidelines on the subject have been published.1
Dispense no more than a 28-day supply.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Increased Mortality: serious and fatal cardiac adverse reactions occurred in patients with CLL treated with REVLIMID (5.5).
-
•Second Primary Malignancies (SPM): Higher incidences of SPM were observed in controlled trials of patients with MM receiving REVLIMID (5.6).
-
•Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue (5.7).
-
•Hepatotoxicity: Hepatic failure including fatalities; monitor liver function. Stop REVLIMID and evaluate if hepatotoxicity is suspected (5.8).
-
•Severe Cutaneous Reactions: Discontinue REVLIMID for severe reactions (5.9).
-
•Tumor lysis syndrome (TLS) including fatalities: Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions (5.10).
-
•Tumor flare reaction: Serious tumor flare reactions, including fatal reactions, have occurred during investigational use of REVLIMID for chronic lymphocytic leukemia and lymphoma (5.11).
-
•Impaired Stem Cell mobilization: A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. Consider early referral to transplant center (5.12).
-
•Early mortality in MCL: Higher rate of early deaths have occurred in patients with MCL (5.14).
-
•Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue REVLIMID for angioedema and anaphylaxis (5.15).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•MM combination therapy: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles. (2.1).
-
•MM maintenance therapy following auto-HSCT: 10 mg once daily continuously on Days 1-28 of repeated 28-day cycles (2.1).
-
•MDS: 10 mg once daily (2.2).
-
•MCL: 25 mg once daily orally on Days 1-21 of repeated 28-day cycles (2.3).
-
•FL or MZL: 20 mg once daily orally on Days 1-21 of repeated 28-day cycles for up to 12 cycles (2.4).
-
•Renal impairment: Adjust starting dose based on the creatinine clearance value (2.6).
-
•For concomitant therapy doses, see Full Prescribing Information (2.1, 2.4, 14.1, 14.4).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules:
-
•2.5 mg, white and blue-green opaque hard capsules imprinted "REV" on one half and "2.5 mg" on the other half in black ink
-
•5 mg, white opaque capsules imprinted "REV" on one half and "5 mg" on the other half in black ink
-
•10 mg, blue/green and pale yellow opaque capsules imprinted "REV" on one half and "10 mg" on the other half in black ink
-
•15 mg, powder blue and white opaque capsules imprinted "REV" on one half and "15 mg" on the other half in black ink
-
•20 mg, powder blue and blue-green opaque hard capsules imprinted "REV" on one half and "20 mg" on the other half in black ink
-
•25 mg, white opaque capsules imprinted "REV" on one half and "25 mg" on the other half in black ink
6.2 Postmarketing Experience
The following adverse drug reactions have been identified from the worldwide post-marketing experience with REVLIMID. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure [see Warnings and Precautions Section (5.8 to 5.11, and 5.13)]
Endocrine disorders: Hypothyroidism, hyperthyroidism
Hepatobiliary disorders: Hepatic failure (including fatality), toxic hepatitis, cytolytic hepatitis, cholestatic hepatitis, mixed cytolytic/cholestatic hepatitis, transient abnormal liver laboratory tests
Immune system disorders: Angioedema, anaphylaxis, acute graft-versus-host disease (following allogeneic hematopoietic transplant), solid organ transplant rejection
Infections and infestations: Viral reactivation (such as hepatitis B virus and herpes zoster), progressive multifocal leukoencephalopathy (PML)
Neoplasms benign, malignant and unspecified (including cysts and polyps): Tumor lysis syndrome, tumor flare reaction
Respiratory, thoracic and mediastinal disorders: Pneumonitis
Skin and subcutaneous tissue disorders: Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS)
1.2 Myelodysplastic Syndromes
REVLIMID is indicated for the treatment of adult patients with transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q cytogenetic abnormality with or without additional cytogenetic abnormalities.
5.2 Lenalidomide Rems Program (5.2 Lenalidomide REMS Program)
Because of the embryo-fetal risk [see Warnings and Precautions (5.1)], REVLIMID is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), the Lenalidomide REMS program.
Required components of the Lenalidomide REMS program include the following:
-
•Prescribers must be certified with the Lenalidomide REMS program by enrolling and complying with the REMS requirements.
-
•Patients must sign a Patient-Physician agreement form and comply with the REMS requirements. In particular, female patients of reproductive potential who are not pregnant must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)] and males must comply with contraception requirements [see Use in Specific Populations (8.3)].
-
•Pharmacies must be certified with the Lenalidomide REMS program, must only dispense to patients who are authorized to receive REVLIMID and comply with REMS requirements.
Further information about the Lenalidomide REMS program is available at www.lenalidomiderems.com or by telephone at 1-888-423-5436.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Lactation: Advise not to breastfeed (8.2).
5.9 Severe Cutaneous Reactions
Severe cutaneous reactions including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. These events can be fatal. Patients with a prior history of Grade 4 rash associated with thalidomide treatment should not receive REVLIMID. Consider REVLIMID interruption or discontinuation for Grade 2-3 skin rash. Permanently discontinue REVLIMID for Grade 4 rash, exfoliative or bullous rash, or for other severe cutaneous reactions such as SJS, TEN or DRESS [see Dosage and Administration (2.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.6 Second Primary Malignancies
In clinical trials in patients with MM receiving REVLIMID, an increase of hematologic plus solid tumor second primary malignancies (SPM) notably AML and MDS have been observed. An increase in hematologic SPM including AML and MDS occurred in 5.3% of patients with NDMM receiving REVLIMID in combination with oral melphalan compared with 1.3% of patients receiving melphalan without REVLIMID. The frequency of AML and MDS cases in patients with NDMM treated with REVLIMID in combination with dexamethasone without melphalan was 0.4%.
In patients receiving REVLIMID maintenance therapy following high dose intravenous melphalan and auto-HSCT, hematologic SPM occurred in 7.5% of patients compared to 3.3% in patients receiving placebo. The incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 14.9%, compared to 8.8% in patients receiving placebo with a median follow-up of 91.5 months. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.9% of patients receiving REVLIMID maintenance, compared to 2.6% in the placebo arm.
In patients with relapsed or refractory MM treated with REVLIMID/dexamethasone, the incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 2.3% versus 0.6% in the dexamethasone alone arm. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.1% of patients receiving REVLIMID/dexamethasone, compared to 0.6% in the dexamethasone alone arm.
Patients who received REVLIMID-containing therapy until disease progression did not show a higher incidence of invasive SPM than patients treated in the fixed duration REVLIMID-containing arms. Monitor patients for the development of second primary malignancies. Take into account both the potential benefit of REVLIMID and the risk of second primary malignancies when considering treatment with REVLIMID.
In the AUGMENT trial with FL or MZL patients receiving REVLIMID/rituximab therapy, hematologic plus solid tumor SPMs, notably AML, have been observed. In the AUGMENT trial, hematologic SPM of AML occurred in 0.6% of patients with FL or MZL receiving REVLIMID/rituximab therapy. The incidence of hematologic plus solid tumor SPMs (excluding nonmelanoma skin cancers) was 1.7% in the REVLIMID/rituximab arm with a median follow-up of 29.8 months (range 0.5 to 51.3 months) [see Adverse Reactions (6.1)]. Monitor patients for the development of second primary malignancies. Take into account both the potential benefit of REVLIMID and the risk of second primary malignancies when considering treatment with REVLIMID.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved Patient labeling (Medication Guide)
5.12 Impaired Stem Cell Mobilization
A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with REVLIMID has been reported. In patients who are auto-HSCT candidates, referral to a transplant center should occur early in treatment to optimize the timing of the stem cell collection. In patients who received more than 4 cycles of a REVLIMID-containing treatment or for whom inadequate numbers of CD 34+ cells have been collected with G-CSF alone, G-CSF with cyclophosphamide or the combination of G-CSF with a CXCR4 inhibitor may be considered.
4.2 Severe Hypersensitivity Reactions
REVLIMID is contraindicated in patients who have demonstrated severe hypersensitivity (e.g., angioedema, Stevens-Johnson syndrome, toxic epidermal necrolysis) to lenalidomide [see Warnings and Precautions (5.9, 5.15)].
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
NDC 59572-402-28
Revlimid
(lenalidomide) Capsules
2.5 mg
WARNING: POTENTIAL FOR HUMAN BIRTH DEFECTS.
Rx Only
28 Capsules
5.4 Venous and Arterial Thromboembolism
Venous thromboembolic events (VTE [DVT and PE]) and arterial thromboembolic events (ATE, myocardial infarction and stroke) are increased in patients treated with REVLIMID.
A significantly increased risk of DVT (7.4%) and of PE (3.7%) occurred in patients with MM after at least one prior therapy who were treated with REVLIMID and dexamethasone therapy compared to patients treated in the placebo and dexamethasone group (3.1% and 0.9%) in clinical trials with varying use of anticoagulant therapies. In the newly diagnosed multiple myeloma (NDMM) study in which nearly all patients received antithrombotic prophylaxis, DVT was reported as a serious adverse reaction (3.6%, 2.0%, and 1.7%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of PE was similar between the Rd Continuous, Rd18, and MPT Arms (3.8%, 2.8%, and 3.7%, respectively) [see Boxed Warning and Adverse Reactions (6.1)].
Myocardial infarction (1.7%) and stroke (CVA) (2.3%) are increased in patients with MM after at least one prior therapy who were treated with REVLIMID and dexamethasone therapy compared to patients treated with placebo and dexamethasone (0.6%, and 0.9%) in clinical trials. In the NDMM study, myocardial infarction (including acute) was reported as a serious adverse reaction (2.3%, 0.6%, and 1.1%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of CVA was similar between the Rd Continuous, Rd18, and MPT Arms (0.8%, 0.6 %, and 0.6%, respectively) [see Adverse Reactions (6.1)].
Patients with known risk factors, including prior thrombosis, may be at greater risk and actions should be taken to try to minimize all modifiable factors (e.g. hyperlipidemia, hypertension, smoking).
In controlled clinical trials that did not use concomitant thromboprophylaxis, 21.5% overall thrombotic events (Standardized MedDRA Query Embolic and Thrombotic events) occurred in patients with refractory and relapsed MM who were treated with REVLIMID and dexamethasone compared to 8.3% thrombosis in patients treated with placebo and dexamethasone. The median time to first thrombosis event was 2.8 months. In the NDMM study in which nearly all patients received antithrombotic prophylaxis, the overall frequency of thrombotic events was 17.4% in patients in the combined Rd Continuous and Rd18 Arms, and was 11.6% in the MPT Arm. The median time to first thrombosis event was 4.3 months in the combined Rd Continuous and Rd18 Arms.
In the AUGMENT trial, the incidence of VTE (including DVT and PE) in FL or MZL patients was 3.4% in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)]. In the AUGMENT trial, the incidence of ATE (including MI) in FL or MZL patients was 0.6% in the REVLIMID/rituximab arm [see Adverse Reactions (6.1)].
Thromboprophylaxis is recommended. The regimen of thromboprophylaxis should be based on an assessment of the patient's underlying risks. Instruct patients to report immediately any signs and symptoms suggestive of thrombotic events. ESAs and estrogens may further increase the risk of thrombosis and their use should be based on a benefit-risk decision in patients receiving REVLIMID [see Drug Interactions (7.2)].
5.14 Early Mortality in Patients With Mcl (5.14 Early Mortality in Patients with MCL)
In another MCL study, there was an increase in early deaths (within 20 weeks), 12.9% in the REVLIMID arm versus 7.1% in the control arm. On exploratory multivariate analysis, risk factors for early deaths include high tumor burden, MIPI score at diagnosis, and high WBC at baseline (≥ 10 x 109/L).
14.4 Follicular and Marginal Zone Lymphoma
The efficacy of REVLIMID with rituximab in patients with relapsed or refractory follicular and marginal zone lymphoma was evaluated in the AUGMENT (NCT01938001) and MAGNIFY (NCT01996865) trials.
AUGMENT is a randomized, double-blind, multicenter trial (n=358) in which patients with relapsed or refractory follicular or marginal zone lymphoma were randomized 1:1 to receive REVLIMID and rituximab or rituximab and placebo. AUGMENT included patients diagnosed with Grade 1, 2, or 3a follicular lymphoma, who received at least 1 prior systemic therapy, were refractory or relapsed, not rituximab-refractory, had at least one measurable nodal or extranodal lesion by CT or MRI scan, and had adequate bone marrow, liver, and renal function. Randomization was stratified by follicular versus marginal zone lymphoma, previous rituximab therapy, and time since other anti-lymphoma therapy. In AUGMENT, REVLIMID was administered orally 20 mg once daily for Days 1 to 21 of repeating 28-day cycles for a maximum of 12 cycles or until unacceptable toxicity. The dose of rituximab was 375 mg/m2 every week in Cycle 1 (Days 1, 8, 15, and 22) and on Day 1 of every 28-day cycle from Cycles 2 through 5. All dosage calculations for rituximab were based on the patient's body surface area (BSA), using actual patient weight. Dose adjustments for REVLIMID were allowed based on clinical and laboratory findings. A patient with moderate renal insufficiency (≥30 to <60 mL/minute) received a lower REVLIMID starting dose of 10 mg daily on the same schedule. After 2 cycles, the REVLIMID dose could be increased to 15 mg once daily on Days 1 to 21 of each 28-day cycle if the patient tolerated the medication.
MAGNIFY is an open-label, multicenter trial (n=232) in which patients with relapsed or refractory follicular, marginal zone, or mantle cell lymphoma received 12 induction cycles of REVLIMID and rituximab. MAGNIFY included patients diagnosed with Grade 1, 2,3a, 3b follicular (including transformed), marginal zone, or mantle cell lymphoma Stage I to IV who were previously treated for their lymphoma, had been refractory or had a relapse after their last treatment, had at least one measurable nodal or extranodal lesion by CT or MRI scan, and had adequate bone marrow, liver, and renal function. Patients refractory to rituximab were also included. The information from the subjects who received at least 1 dose of initial therapy in the first 12 induction cycles (n=222) in the MAGNIFY trial was included in the evaluation of the efficacy of REVLIMID/rituximab in patients with relapsed or refractory follicular and marginal zone lymphoma. In MAGNIFY, REVLIMID 20 mg was given on Days 1-21 of repeated 28-day cycles for up to 12 cycles or until unacceptable toxicity, progression, or withdrawal of consent. The dose of rituximab was 375 mg/m2 every week in Cycle 1 (Days 1, 8, 15, and 22) and on Day 1 of every other 28-day cycle (Cycles 3,5,7,9, and 11) up to 12 cycles therapy. All dosage calculations for rituximab were based on the patient BSA and actual weight. Dose adjustments were allowed based on clinical and laboratory findings.
The demographic and disease-related baseline characteristics in the AUGMENT and MAGNIFY trials are shown in the following table.
| Data Cutoff: 22 June 2018 (AUGMENT) and 1 May 2017 (MAGNIFY). a Defined by GELF criteria. b Patient had either 0 (n=2) or 1 prior systemic therapy. ECOG = Eastern Cooperative Oncology Group; FLIPI = follicular lymphoma international prognostic index |
|||
|
Parameter |
AUGMENT Trial |
MAGNIFY Trial |
|
|
REVLIMID + Rituximab
|
Rituximab + Placebo
|
REVLIMID + Rituximab
|
|
|
Age (years) |
|||
|
Median (Max, Min) |
64 (26, 86) |
62 (35, 88) |
65 (35, 91) |
|
Age distribution, n (%) |
|||
|
<65 years |
96 (54) |
107 (59) |
103 (46) |
|
≥65 years |
82 (46) |
73 (41) |
119 (54) |
|
Sex, n (%) |
|||
|
Male |
75 (42) |
97 (54) |
122 (55) |
|
Female |
103 (58) |
83 (46) |
100 (45) |
|
Race |
|||
|
White |
118 (66) |
115 (64) |
206 (93) |
|
Other races |
54 (30) |
64 (36) |
14 (6) |
|
Not collected or reported |
6 (3) |
1 (0.6) |
2 (1) |
|
Body Surface Area (BSA, m2) |
|||
|
Median (Max, Min) |
1.8 (1.4, 3.1) |
1.8 (1.3, 2.7) |
2 (1.3, 2.6) |
|
Disease Type FL or MZL |
|||
|
Follicular lymphoma |
147 (83) |
148 (82) |
177 (80) |
|
Marginal zone lymphoma |
31 (17) |
32 (18) |
45 (20) |
|
MZL subtype at diagnosis (investigator), n (%) |
|||
|
MALT |
14 (45) |
16 (50) |
10 (22) |
|
Nodal |
8 (26) |
10 (31) |
25 (56) |
|
Splenic |
9 (29) |
6 (19) |
10 (22) |
|
FL stage at diagnosis (investigator), n (%) |
|||
|
FL Grade 1-2 |
125 (85) |
123 (83) |
149 (84) |
|
FL Grade 3a |
22 (15) |
25 (17) |
28 (16) |
|
FLIPI score at baseline (calculated), n (%) |
Not Collected |
||
|
Low risk (0,1) |
52 (29) |
67 (37) |
|
|
Intermediate risk (2) |
55 (31) |
58 (32) |
|
|
High risk (≥3) |
69 (39) |
54 (30) |
|
|
Missing |
2 (1) |
1 (0.6) |
|
|
ECOG score at baseline, n (%) |
|||
|
0 |
116 (65) |
128 (71) |
102 (46) |
|
1 |
60 (34) |
50 (28) |
113 (51) |
|
2 |
2 (1) |
2 (1) |
7 (3) |
|
High tumor burdena at baseline, n (%) |
|||
|
Yes |
97 (54) |
86 (48) |
148 (67) |
|
No |
81 (46) |
94 (52) |
74 (33) |
|
Number of prior systemic antilymphoma therapies |
|||
|
1 |
102 (57) |
97 (54) |
94 (42)b |
|
>1 |
76 (43) |
83 (46) |
128 (58) |
In AUGMENT, efficacy was established in the intent-to-treat (ITT) population based on progression-free survival by Independent Review Committee using modified 2007 International Working Group response criteria. Efficacy results are summarized in Table 22.
|
a Median estimate is from Kaplan-Meier analysis. b hazard ratio and its CI were estimated from Cox proportional hazard model adjusting for the stratification 3: previous rituximab treatment (yes, no), time since last antilymphoma therapy (≤ 2, > 2 years), and disease histology (FL, MZL). c p-value from log-rank test stratified by 3 factors noted above: previous rituximab treatment (yes, no), time since last antilymphoma therapy (≤ 2, > 2 years), and disease histology (FL, MZL). d Exact confidence interval for binomial distribution. |
||
|
Parameter |
REVLIMID + Rituximab
|
Rituximab + Placebo
|
|
PFS |
||
|
Patients with event, n (%) |
68 (38.2) |
115 (63.9) |
|
Death |
6 (8.8) |
2 (1.7) |
|
Progression of disease |
62 (91.2) |
113 (98.3) |
|
PFS, median a [95% CI] (months) |
39.4 [ 22.9, NE] |
14.1 [11.4, 16.7] |
|
HR b [95% CI] |
0.46 [ 0.34, 0.62] |
|
|
p-value c |
<0.0001 |
|
|
Objective response (CR+PR) , n(%) [95% CI] d |
138 (77.5) [70.7, 83.4] |
96 (53.3) [45.8, 60.8] |
Kaplan-Meier Curves of Progression-free Survival by IRC Assessment Between Arms in AUGMENT Trial (ITT FL and MZL Population)
a = Stratification factors included: previous rituximab treatment (y/n), time since last anti-lymphoma therapy (≤2 years, >2years), and disease histology (FL or MZL). CI = confidence interval; HR = hazard ratio; KM = Kaplan-Meier; PFS = progression-free survival
5.5 Increased Mortality in Patients With Cll (5.5 Increased Mortality in Patients with CLL)
In a prospective randomized (1:1) clinical trial in the first line treatment of patients with chronic lymphocytic leukemia, single agent REVLIMID therapy increased the risk of death as compared to single agent chlorambucil. In an interim analysis, there were 34 deaths among 210 patients on the REVLIMID treatment arm compared to 18 deaths among 211 patients in the chlorambucil treatment arm, and hazard ratio for overall survival was 1.92 [95% CI: 1.08 – 3.41], consistent with a 92% increase in the risk of death. The trial was halted for safety in July 2013.
Serious adverse cardiovascular reactions, including atrial fibrillation, myocardial infarction, and cardiac failure occurred more frequently in the REVLIMID treatment arm. REVLIMID is not indicated and not recommended for use in CLL outside of controlled clinical trials.
2.3 Recommended Dosage for Mantle Cell Lymphoma
The recommended starting dose of REVLIMID is 25 mg/day orally on Days 1-21 of repeated 28-day cycles for relapsed or refractory mantle cell lymphoma. Treatment should be continued until disease progression or unacceptable toxicity.
Treatment is continued, modified or discontinued based upon clinical and laboratory findings.
2.2 Recommended Dosage for Myelodysplastic Syndromes
The recommended starting dose of REVLIMID is 10 mg daily. Treatment is continued or modified based upon clinical and laboratory findings. Continue treatment until disease progression or unacceptable toxicity.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with lenalidomide have not been conducted.
Lenalidomide was not mutagenic in the bacterial reverse mutation assay (Ames test) and did not induce chromosome aberrations in cultured human peripheral blood lymphocytes, or mutations at the thymidine kinase (tk) locus of mouse lymphoma L5178Y cells. Lenalidomide did not increase morphological transformation in Syrian Hamster Embryo assay or induce micronuclei in the polychromatic erythrocytes of the bone marrow of male rats.
A fertility and early embryonic development study in rats, with administration of lenalidomide up to 500 mg/kg (approximately 200 times the human dose of 25 mg, based on body surface area) produced no parental toxicity and no adverse effects on fertility.
2.6 Recommended Dosage for Patients With Renal Impairment (2.6 Recommended Dosage for Patients with Renal Impairment)
The recommendations for dosing patients with renal impairment are shown in the following table [see Clinical Pharmacology (12.3)].
|
Renal Function
|
Dose in REVLIMID Combination Therapy for MM and MCL |
Dose in REVLIMID Combination Therapy for FL and MZL |
Dose in REVLIMID Maintenance Therapy Following Auto-HSCT for MM and for MDS |
|
CLcr 30 to 60 mL/min |
10 mg once daily |
10 mg once daily |
5 mg once daily |
|
CLcr below 30 mL/min (not requiring dialysis) |
15 mg every other day |
5 mg once daily |
2.5 mg once daily |
|
CLcr below 30 mL/min (requiring dialysis) |
5 mg once daily. On dialysis days, administer the dose following dialysis. |
5 mg once daily. On dialysis days, administer the dose following dialysis. |
2.5 mg once daily. On dialysis days, administer the dose following dialysis. |
2.5 Dosage Modifications for Non Hematologic Adverse Reactions (2.5 Dosage Modifications for Non-Hematologic Adverse Reactions)
For non-hematologic Grade 3/4 toxicities judged to be related to REVLIMID, hold treatment and restart at the physician's discretion at next lower dose level when toxicity has resolved to Grade 2 or below.
Permanently discontinue REVLIMID for angioedema, anaphylaxis, Grade 4 rash, skin exfoliation, bullae, or any other severe dermatologic reactions [see Warnings and Precautions (5.9, 5.15)].
7.2 Concomitant Therapies That May Increase the Risk of Thrombosis
Erythropoietic agents, or other agents that may increase the risk of thrombosis, such as estrogen containing therapies, should be used with caution after making a benefit-risk assessment in patients receiving REVLIMID [see Warnings and Precautions (5.4)].
2.4 Recommended Dosage for Follicular Lymphoma Or Marginal Zone Lymphoma (2.4 Recommended Dosage for Follicular Lymphoma or Marginal Zone Lymphoma)
The recommended starting dose of REVLIMID is 20 mg orally once daily on Days 1-21 of repeated 28-day cycles for up to 12 cycles of treatment in combination with a rituximab-product. Refer to Section 14.4 for specific rituximab dosing from the AUGMENT trial. For dose adjustments due to toxicity with rituximab, refer to the product prescribing information.
14.2 Myelodysplastic Syndromes (mds) With A Deletion 5q Cytogenetic Abnormality (14.2 Myelodysplastic Syndromes (MDS) with a Deletion 5q Cytogenetic Abnormality)
The efficacy and safety of REVLIMID were evaluated in patients with transfusion-dependent anemia in low- or intermediate-1- risk MDS with a 5q (q31-33) cytogenetic abnormality in isolation or with additional cytogenetic abnormalities, at a dose of 10 mg once daily or 10 mg once daily for 21 days every 28 days in an open-label, single-arm, multi-center study. The major study was not designed nor powered to prospectively compare the efficacy of the 2 dosing regimens. Sequential dose reductions to 5 mg daily and 5 mg every other day, as well as dose delays, were allowed for toxicity [Dosage and Administration (2.2)].
This major study enrolled 148 patients who had RBC transfusion dependent anemia. RBC transfusion dependence was defined as having received ≥ 2 units of RBCs within 8 weeks prior to study treatment. The study enrolled patients with absolute neutrophil counts (ANC) ≥ 500/mm3, platelet counts ≥ 50,000/mm3, serum creatinine ≤ 2.5 mg/dL, serum SGOT/AST or SGPT/ALT ≤ 3 x upper limit of normal (ULN), and serum direct bilirubin ≤ 2 mg/dL. Granulocyte colony-stimulating factor was permitted for patients who developed neutropenia or fever in association with neutropenia. Baseline patient and disease-related characteristics are summarized in Table 18.
|
a IPSS Risk Category: Low (combined score = 0), Intermediate-1 (combined score = 0.5 to 1), Intermediate-2 (combined score = 1.5 to 2.0), High (combined score ≥ 2.5); Combined score = (Marrow blast score + Karyotype score + Cytopenia score). b French-American-British (FAB) classification of MDS. |
||
|
Overall
|
||
|
Age (years) |
||
|
Median |
71 |
|
|
Min, Max |
37, 95 |
|
|
Gender |
n |
(%) |
|
Male |
51 |
(34.5) |
|
Female |
97 |
(65.5) |
|
Race |
n |
(%) |
|
White |
143 |
(96.6) |
|
Other |
5 |
( 3.4) |
|
Duration of MDS (years) |
||
|
Median |
2.5 |
|
|
Min, Max |
0.1, 20.7 |
|
|
Del 5 (q31-33) Cytogenetic Abnormality |
n |
(%) |
|
Yes |
148 |
(100) |
|
Other cytogenetic abnormalities |
37 |
(25.2) |
|
IPSS Score a |
n |
(%) |
|
Low (0) |
55 |
(37.2) |
|
Intermediate-1 (0.5-1.0) |
65 |
(43.9) |
|
Intermediate-2 (1.5-2.0) |
6 |
( 4.1) |
|
High (≥2.5) |
2 |
( 1.4) |
|
Missing |
20 |
(13.5) |
|
FAB Classification b from central review |
n |
(%) |
|
RA |
77 |
(52) |
|
RARS |
16 |
(10.8) |
|
RAEB |
30 |
(20.3) |
|
CMML |
3 |
( 2) |
The frequency of RBC transfusion independence was assessed using criteria modified from the International Working Group (IWG) response criteria for MDS. RBC transfusion independence was defined as the absence of any RBC transfusion during any consecutive "rolling" 56 days (8 weeks) during the treatment period.
Transfusion independence was seen in 99/148 (67%) patients (95% CI [59, 74]). The median duration from the date when RBC transfusion independence was first declared (i.e., the last day of the 56-day RBC transfusion-free period) to the date when an additional transfusion was received after the 56-day transfusion-free period among the 99 responders was 44 weeks (range of 0 to >67 weeks). Ninety percent of patients who achieved a transfusion benefit did so by completion of three months in the study.
RBC transfusion independence rates were unaffected by age or gender.
The dose of REVLIMID was reduced or interrupted at least once due to an adverse event in 118 (79.7%) of the 148 patients; the median time to the first dose reduction or interruption was 21 days (mean, 35.1 days; range, 2-253 days), and the median duration of the first dose interruption was 22 days (mean, 28.5 days; range, 2-265 days). A second dose reduction or interruption due to adverse events was required in 50 (33.8%) of the 148 patients. The median interval between the first and second dose reduction or interruption was 51 days (mean, 59.7 days; range, 15-205 days) and the median duration of the second dose interruption was 21 days (mean, 26 days; range, 2-148 days).
Warning: Embryo Fetal Toxicity, Hematologic Toxicity, and Venous and Arterial Thromboembolism (WARNING: EMBRYO-FETAL TOXICITY, HEMATOLOGIC TOXICITY, and VENOUS and ARTERIAL THROMBOEMBOLISM)
Embryo-Fetal Toxicity
Do not use REVLIMID during pregnancy. Lenalidomide, a thalidomide analogue, caused limb abnormalities in a developmental monkey study. Thalidomide is a known human teratogen that causes severe life-threatening human birth defects. If lenalidomide is used during pregnancy, it may cause birth defects or embryo-fetal death. In females of reproductive potential, obtain 2 negative pregnancy tests before starting REVLIMID® treatment. Females of reproductive potential must use 2 forms of contraception or continuously abstain from heterosexual sex during and for 4 weeks after REVLIMID treatment [see Warnings and Precautions (5.1), and Medication Guide (17)]. To avoid embryo-fetal exposure to lenalidomide, REVLIMID is only available through a restricted distribution program, the Lenalidomide REMS program (5.2).
Information about the Lenalidomide REMS program is available at www.lenalidomiderems.com or by calling the REMS Call Center at 1-888-423-5436.
Hematologic Toxicity (Neutropenia and Thrombocytopenia)
REVLIMID can cause significant neutropenia and thrombocytopenia. Eighty percent of patients with del 5q myelodysplastic syndromes had to have a dose delay/reduction during the major study. Thirty-four percent of patients had to have a second dose delay/reduction. Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the study. Patients on therapy for del 5q myelodysplastic syndromes should have their complete blood counts monitored weekly for the first 8 weeks of therapy and at least monthly thereafter. Patients may require dose interruption and/or reduction. Patients may require use of blood product support and/or growth factors [see Dosage and Administration (2.2)].
Venous and Arterial Thromboembolism
REVLIMID has demonstrated a significantly increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as risk of myocardial infarction and stroke in patients with multiple myeloma who were treated with REVLIMID and dexamethasone therapy. Monitor for and advise patients about signs and symptoms of thromboembolism. Advise patients to seek immediate medical care if they develop symptoms such as shortness of breath, chest pain, or arm or leg swelling. Thromboprophylaxis is recommended and the choice of regimen should be based on an assessment of the patient’s underlying risks [see Warnings and Precautions (5.4)].
5.7 Increased Mortality in Patients With Mm When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone (5.7 Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone)
In two randomized clinical trials in patients with MM, the addition of pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with MM with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:04.865739 · Updated: 2026-03-14T21:53:19.065561