These Highlights Do Not Include All The Information Needed To Use Fenofibrate Tablets, Usp Safely And Effectively. See Full Prescribing Information For Fenofibrate Tablets, Usp.
5e7b79ab-9df0-4bbf-b7a4-a44aec203555
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 6/2025 Dosage and Administration ( 2 ) 6/2025 Warnings and Precautions, Mortality and Coronary Heart Disease Morbidity ( 5.1 ) 6/2025
Indications and Usage
Fenofibrate tablets are indicated as adjunctive therapy to diet: to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL). to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
Dosage and Administration
Severe hypertriglyceridemia: The recommended dosage of fenofibrate tablets is 54 mg or 160 mg orally once daily. Dosage should be individualized according to patient response, and should be adjusted if necessary following repeat lipid determinations at 4 to 8 week intervals. Primary hyperlipidemia : The recommended dosage of fenofibrate tablets is 160 mg orally once daily. Administer fenofibrate tablets as a single dose at any time of day, with or without food. Advise patients to swallow fenofibrate tablets whole. Do not crush, break, dissolve, or chew tablets. Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment. If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose. Advise patients to take fenofibrate tablets at least 1 hour before or 4 hours to 6 hours after a bile acid binding resin to avoid impeding its absorption.
Warnings and Precautions
Hepatotoxicity: Serious drug-induced liver injury, including liver transplantation and death, has been reported with fenofibrates, including fenofibrate tablets. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy. Discontinue if signs or symptoms of liver injury develop or if elevated enzyme levels persist ( 5.2 ). Myopathy and Rhabdomyolysis : Have been reported in patients taking fenofibrates. Risks are increased during co-administration with a statin, in geriatric patients, and in patients with renal impairment or hypothyroidism. Discontinue fenofibrate tablets if markedly elevated CK levels occur or if myopathy is either diagnosed or suspected. Temporarily discontinue fenofibrate tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the fenofibrate tablets dosage. Instruct patients to promptly report any unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever ( 5.3 ). Increases in Serum Creatinine : Monitor renal function in patients with renal impairment taking fenofibrate tablets. Consider monitoring renal function in patients at risk for renal impairment ( 5.4 ). Cholelithiasis : Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated ( 5.5 ). Hypersensitivity Reactions: Acute hypersensitivity reactions, including anaphylaxis and angioedema, and delayed hypersensitivity reactions, including severe cutaneous adverse drug reactions have been reported postmarketing. Some cases were life-threatening and required emergency treatment. Discontinue fenofibrate tablets and treat appropriately if reactions occur ( 5.9 ).
Contraindications
Fenofibrate tablets are contraindicated in patients with: Severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Clinical Pharmacology (12.3) ] . Active liver disease, including those with unexplained persistent liver function abnormalities [see Warnings and Precautions (5.2) ] . Pre-existing gallbladder disease [see Warnings and Precautions (5.5) ] . Hypersensitivity to fenofibrate, fenofibric acid, or any of the excipients in fenofibrate tablets. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with fenofibrate [see Warnings and Precautions (5.9) ] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Mortality and Coronary Heart Disease Morbidity [see Warnings and Precautions (5.1) ] Hepatoxicity [see Warnings and Precautions (5.2) ] Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.3) ] Increases in Serum Creatinine [see Warnings and Precautions (5.4) ] Cholelithiasis [see Warnings and Precautions (5.5) ] Increased Bleeding Risk with Coumarin Anticoagulants [see Warnings and Precautions (5.6) ] Pancreatitis [see Warnings and Precautions (5.7) ] Hematologic Changes [see Warnings and Precautions (5.8) ] Hypersensitivity Reactions [see Warnings and Precautions (5.9) ] Venothromboembolic Disease [see Warnings and Precautions (5.10) ] Paradoxical Decreases in HDL Cholesterol Levels [see Warnings and Precautions (5.11) ]
Drug Interactions
Table 2 presents clinically important drug interactions with fenofibrate tablets. Table 2. Clinically Important Drug Interactions with Fenofibrate Tablets Statins Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins. Intervention: Consider if the benefit of using fenofibrate tablets concomitantly with statin therapy outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of statin therapy. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with fenofibrates. Intervention: Consider if the benefit of using colchicine concomitantly with fenofibrate tablets outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of colchicine. Coumarin Anticoagulants Clinical Impact: Fibrates may cause potentiation of coumarin-type anticoagulant effects with prolongation of the PT/INR. Intervention: Caution should be exercised when coumarin anticoagulants are given in conjunction with fenofibrate tablets. The dosage of the anticoagulants should be reduced to maintain the PT/INR at the desired level to prevent bleeding complications. Frequent PT/INR determinations are advisable until it has been definitely determined that the PT/INR has stabilized Immunosuppressants Clinical Impact: Immunosuppressants such as cyclosporine and tacrolimus can produce nephrotoxicity with decreases in creatinine clearance and rises in serum creatinine, and because renal excretion is the primary elimination route of fibrate drugs including fenofibrate tablets, there is a risk that an interaction will lead to deterioration of renal function. Intervention: The benefits and risks of using fenofibrate tablets with immunosuppressants and other potentially nephrotoxic agents should be carefully considered, and the lowest effective dosage employed and renal function monitored. Bile-Acid Binding Resins Clinical Impact: Bile-acid binding resins may bind other drugs given concurrently. Intervention: In patients taking a bile acid resin, administer fenofibrate tablets at least 1 hour before or 4 to 6 hours after the bile acid resin to avoid impeding its absorption.
Storage and Handling
Fenofibrate Tablets, USP are supplied as follows: 54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45). 160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
How Supplied
Fenofibrate Tablets, USP are supplied as follows: 54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45). 160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
Medication Information
Warnings and Precautions
Hepatotoxicity: Serious drug-induced liver injury, including liver transplantation and death, has been reported with fenofibrates, including fenofibrate tablets. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy. Discontinue if signs or symptoms of liver injury develop or if elevated enzyme levels persist ( 5.2 ). Myopathy and Rhabdomyolysis : Have been reported in patients taking fenofibrates. Risks are increased during co-administration with a statin, in geriatric patients, and in patients with renal impairment or hypothyroidism. Discontinue fenofibrate tablets if markedly elevated CK levels occur or if myopathy is either diagnosed or suspected. Temporarily discontinue fenofibrate tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the fenofibrate tablets dosage. Instruct patients to promptly report any unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever ( 5.3 ). Increases in Serum Creatinine : Monitor renal function in patients with renal impairment taking fenofibrate tablets. Consider monitoring renal function in patients at risk for renal impairment ( 5.4 ). Cholelithiasis : Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated ( 5.5 ). Hypersensitivity Reactions: Acute hypersensitivity reactions, including anaphylaxis and angioedema, and delayed hypersensitivity reactions, including severe cutaneous adverse drug reactions have been reported postmarketing. Some cases were life-threatening and required emergency treatment. Discontinue fenofibrate tablets and treat appropriately if reactions occur ( 5.9 ).
Indications and Usage
Fenofibrate tablets are indicated as adjunctive therapy to diet: to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL). to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
Dosage and Administration
Severe hypertriglyceridemia: The recommended dosage of fenofibrate tablets is 54 mg or 160 mg orally once daily. Dosage should be individualized according to patient response, and should be adjusted if necessary following repeat lipid determinations at 4 to 8 week intervals. Primary hyperlipidemia : The recommended dosage of fenofibrate tablets is 160 mg orally once daily. Administer fenofibrate tablets as a single dose at any time of day, with or without food. Advise patients to swallow fenofibrate tablets whole. Do not crush, break, dissolve, or chew tablets. Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment. If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose. Advise patients to take fenofibrate tablets at least 1 hour before or 4 hours to 6 hours after a bile acid binding resin to avoid impeding its absorption.
Contraindications
Fenofibrate tablets are contraindicated in patients with: Severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Clinical Pharmacology (12.3) ] . Active liver disease, including those with unexplained persistent liver function abnormalities [see Warnings and Precautions (5.2) ] . Pre-existing gallbladder disease [see Warnings and Precautions (5.5) ] . Hypersensitivity to fenofibrate, fenofibric acid, or any of the excipients in fenofibrate tablets. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with fenofibrate [see Warnings and Precautions (5.9) ] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Mortality and Coronary Heart Disease Morbidity [see Warnings and Precautions (5.1) ] Hepatoxicity [see Warnings and Precautions (5.2) ] Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.3) ] Increases in Serum Creatinine [see Warnings and Precautions (5.4) ] Cholelithiasis [see Warnings and Precautions (5.5) ] Increased Bleeding Risk with Coumarin Anticoagulants [see Warnings and Precautions (5.6) ] Pancreatitis [see Warnings and Precautions (5.7) ] Hematologic Changes [see Warnings and Precautions (5.8) ] Hypersensitivity Reactions [see Warnings and Precautions (5.9) ] Venothromboembolic Disease [see Warnings and Precautions (5.10) ] Paradoxical Decreases in HDL Cholesterol Levels [see Warnings and Precautions (5.11) ]
Drug Interactions
Table 2 presents clinically important drug interactions with fenofibrate tablets. Table 2. Clinically Important Drug Interactions with Fenofibrate Tablets Statins Clinical Impact: Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins. Intervention: Consider if the benefit of using fenofibrate tablets concomitantly with statin therapy outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of statin therapy. Colchicine Clinical Impact: Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with fenofibrates. Intervention: Consider if the benefit of using colchicine concomitantly with fenofibrate tablets outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of colchicine. Coumarin Anticoagulants Clinical Impact: Fibrates may cause potentiation of coumarin-type anticoagulant effects with prolongation of the PT/INR. Intervention: Caution should be exercised when coumarin anticoagulants are given in conjunction with fenofibrate tablets. The dosage of the anticoagulants should be reduced to maintain the PT/INR at the desired level to prevent bleeding complications. Frequent PT/INR determinations are advisable until it has been definitely determined that the PT/INR has stabilized Immunosuppressants Clinical Impact: Immunosuppressants such as cyclosporine and tacrolimus can produce nephrotoxicity with decreases in creatinine clearance and rises in serum creatinine, and because renal excretion is the primary elimination route of fibrate drugs including fenofibrate tablets, there is a risk that an interaction will lead to deterioration of renal function. Intervention: The benefits and risks of using fenofibrate tablets with immunosuppressants and other potentially nephrotoxic agents should be carefully considered, and the lowest effective dosage employed and renal function monitored. Bile-Acid Binding Resins Clinical Impact: Bile-acid binding resins may bind other drugs given concurrently. Intervention: In patients taking a bile acid resin, administer fenofibrate tablets at least 1 hour before or 4 to 6 hours after the bile acid resin to avoid impeding its absorption.
Storage and Handling
Fenofibrate Tablets, USP are supplied as follows: 54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45). 160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
How Supplied
Fenofibrate Tablets, USP are supplied as follows: 54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45). 160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
Description
Indications and Usage ( 1 ) 6/2025 Dosage and Administration ( 2 ) 6/2025 Warnings and Precautions, Mortality and Coronary Heart Disease Morbidity ( 5.1 ) 6/2025
Section 42229-5
Limitations of Use
- Markedly elevated levels of serum TG (e.g. > 2,000 mg/dL) may increase the risk of developing pancreatitis. The effect of fenofibrate therapy on reducing this risk has not been determined [see Warnings and Precautions (5.7)].
- Fenofibrate did not reduce coronary heart disease morbidity and mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus [see Warnings and Precautions (5.1) and Clinical Studies (14.4)].
Section 43683-2
Section 44425-7
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
10 Overdosage
In the event of an overdose of fenofibrate tablets, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. There is no specific treatment for overdose with fenofibrate tablets. General supportive care of the patient is indicated, including monitoring of vital signs and observation of clinical status, should an overdose occur. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage; usual precautions should be observed to maintain the airway. Because fenofibric acid is highly bound to plasma proteins, hemodialysis should not be considered.
11 Description
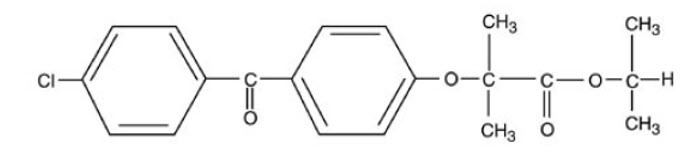
Fenofibrate Tablets, USP are a peroxisome proliferator-activated receptor (PPAR) alpha agonist available as tablets for oral administration. Each tablet contains 54 mg or 160 mg of fenofibrate, USP. The chemical name for fenofibrate, USP is 2-[4-(4-chlorobenzoyl) phenoxy]-2-methyl-propanoic acid, 1-methylethyl ester with the following structural formula:
The empirical formula is C20H21O4Cl and the molecular weight is 360.83; fenofibrate, USP is insoluble in water. The melting point is 79 to 82°C. Fenofibrate, USP is a white solid which is stable under ordinary conditions.
5.7 Pancreatitis
Pancreatitis has been reported in patients taking fenofibrates. This occurrence may represent a failure of efficacy in patients with severe hypertriglyceridemia, a direct drug effect, or a secondary phenomenon mediated through biliary tract stone or sludge formation with obstruction of the common bile duct.
8.4 Pediatric Use
The safety and effectiveness of fenofibrate tablets have not been established in pediatric patients with severe hypertriglyceridemia or primary hyperlipidemia.
8.5 Geriatric Use
Assess renal function in geriatric patients and follow contraindications and dosing recommendations for patients with renal impairment [see Contraindications (4), Warnings and Precautions (5.3, 5.4), and Use in Specific Populations (8.6)]. While fenofibric acid exposure is not influenced by age, geriatric patients are more likely to have renal impairment, and fenofibric acid is substantially excreted by the kidney [see Clinical Pharmacology (12.3)]. Consider monitoring renal function in geriatric patients taking fenofibrate tablets.
5.2 Hepatotoxicity
Serious drug-induced liver injury (DILI), including liver transplantation and death, has been reported with postmarketing use of fenofibrates, including fenofibrate tablets. DILI has been reported within the first few weeks of treatment or after several months of therapy and in some cases has reversed with discontinuation of fenofibrate tablets treatment. Patients with DILI have experienced signs and symptoms including dark urine, abnormal stool, jaundice, malaise, abdominal pain, myalgia, weight loss, pruritus, and nausea. Many patients had concurrent elevations of total bilirubin, serum alanine transaminase (ALT), and aspartate transaminase (AST). DILI has been characterized as hepatocellular, chronic active, and cholestatic hepatitis, and cirrhosis has occurred in association with chronic active hepatitis.
In clinical trials, an intermediate daily dosage or the maximum recommended daily dosage of fenofibrate have been associated with increases in serum AST or ALT. The incidence of increases in transaminases may be dose related [see Adverse Reactions (6.1)].
Fenofibrate tablets are contraindicated in patients with active liver disease, including those with unexplained persistent liver function abnormalities. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy with fenofibrate tablets. Discontinue fenofibrate tablets if signs or symptoms of liver injury develop or if elevated enzyme levels persist (ALT or AST > 3 times the upper limit of normal, or if accompanied by elevation of bilirubin). Do not restart fenofibrate tablets in these patients if there is no alternative explanation for the liver injury.
5.5 Cholelithiasis
Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated. Fenofibrate tablet therapy should be discontinued if gallstones are found. Fenofibrate tablets are contraindicated in patients with pre-existing gallbladder disease.
4 Contraindications
Fenofibrate tablets are contraindicated in patients with:
- Severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Clinical Pharmacology (12.3)].
- Active liver disease, including those with unexplained persistent liver function abnormalities [see Warnings and Precautions (5.2)].
- Pre-existing gallbladder disease [see Warnings and Precautions (5.5)].
- Hypersensitivity to fenofibrate, fenofibric acid, or any of the excipients in fenofibrate tablets. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with fenofibrate [see Warnings and Precautions (5.9)].
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
- Mortality and Coronary Heart Disease Morbidity [see Warnings and Precautions (5.1)]
- Hepatoxicity [see Warnings and Precautions (5.2)]
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.3)]
- Increases in Serum Creatinine [see Warnings and Precautions (5.4)]
- Cholelithiasis [see Warnings and Precautions (5.5)]
- Increased Bleeding Risk with Coumarin Anticoagulants [see Warnings and Precautions (5.6)]
- Pancreatitis [see Warnings and Precautions (5.7)]
- Hematologic Changes [see Warnings and Precautions (5.8)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.9)]
- Venothromboembolic Disease [see Warnings and Precautions (5.10)]
- Paradoxical Decreases in HDL Cholesterol Levels [see Warnings and Precautions (5.11)]
7 Drug Interactions
Table 2 presents clinically important drug interactions with fenofibrate tablets.
| Statins | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins. |
| Intervention: | Consider if the benefit of using fenofibrate tablets concomitantly with statin therapy outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of statin therapy. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with fenofibrates. |
| Intervention: | Consider if the benefit of using colchicine concomitantly with fenofibrate tablets outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of colchicine. |
| Coumarin Anticoagulants | |
| Clinical Impact: | Fibrates may cause potentiation of coumarin-type anticoagulant effects with prolongation of the PT/INR. |
| Intervention: | Caution should be exercised when coumarin anticoagulants are given in conjunction with fenofibrate tablets. The dosage of the anticoagulants should be reduced to maintain the PT/INR at the desired level to prevent bleeding complications. Frequent PT/INR determinations are advisable until it has been definitely determined that the PT/INR has stabilized |
| Immunosuppressants | |
| Clinical Impact: | Immunosuppressants such as cyclosporine and tacrolimus can produce nephrotoxicity with decreases in creatinine clearance and rises in serum creatinine, and because renal excretion is the primary elimination route of fibrate drugs including fenofibrate tablets, there is a risk that an interaction will lead to deterioration of renal function. |
| Intervention: | The benefits and risks of using fenofibrate tablets with immunosuppressants and other potentially nephrotoxic agents should be carefully considered, and the lowest effective dosage employed and renal function monitored. |
| Bile-Acid Binding Resins | |
| Clinical Impact: | Bile-acid binding resins may bind other drugs given concurrently. |
| Intervention: | In patients taking a bile acid resin, administer fenofibrate tablets at least 1 hour before or 4 to 6 hours after the bile acid resin to avoid impeding its absorption. |
8.6 Renal Impairment
Fenofibrate tablets are contraindicated in patients with severe renal impairment (eGFR <30 mL/min/1.73m2), including those with end-stage renal disease (ESRD) and those receiving dialysis. Dosage reduction is required in patients with mild to moderate renal impairment [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)]. Patients with severe renal impairment have 2.7-fold higher exposure of fenofibric acid and increased accumulation of fenofibric acid during chronic dosing compared with healthy volunteers. Renal impairment is a risk factor for myopathy and rhabdomyolysis [see Warnings and Precautions (5.3, 5.4) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Fenofibric acid, the active metabolite of fenofibrate, produces reductions in total cholesterol (Total-C), total triglycerides, and triglyceride rich lipoprotein (VLDL) in treated patients with severe hypertriglyceridemia.
12.3 Pharmacokinetics
Fenofibrate is a pro-drug of the active chemical moiety fenofibric acid. Fenofibrate is converted by ester hydrolysis in the body to fenofibric acid which is the active constituent measurable in the circulation.
8.7 Hepatic Impairment
The use of fenofibrate tablets has not been evaluated in patients with hepatic impairment. Fenofibrate tablets are contraindicated in patients with active liver disease, including those with unexplained persistent liver function abnormalities [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Fenofibrate tablets are indicated as adjunctive therapy to diet:
- to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL).
- to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
5.8 Hematologic Changes
Mild to moderate hemoglobin, hematocrit, and white blood cell decreases have been observed in patients following initiation of therapy with fenofibrates. However, these levels stabilize during long-term administration. Thrombocytopenia and agranulocytosis have been reported in individuals treated with fenofibrate tablets. Periodic monitoring of red and white blood cell counts is recommended during the first 12 months of fenofibrate tablets administration.
12.1 Mechanism of Action
The active moiety of fenofibrate tablets is fenofibric acid. The pharmacological effects of fenofibric acid in both animals and humans have been studied through oral administration of fenofibrate.
The lipid-modifying effects of fenofibric acid seen in clinical practice have been explained in vivo in transgenic mice and in vitro in human hepatocyte cultures by the activation of PPAR alpha receptor. Through this mechanism, fenofibric acid increases lipolysis and elimination of triglyceride-rich particles from plasma by activating lipoprotein lipase and reducing production of apoprotein C-III (an inhibitor of lipoprotein lipase activity).
5 Warnings and Precautions
- Hepatotoxicity: Serious drug-induced liver injury, including liver transplantation and death, has been reported with fenofibrates, including fenofibrate tablets. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy. Discontinue if signs or symptoms of liver injury develop or if elevated enzyme levels persist (5.2).
- Myopathy and Rhabdomyolysis: Have been reported in patients taking fenofibrates. Risks are increased during co-administration with a statin, in geriatric patients, and in patients with renal impairment or hypothyroidism. Discontinue fenofibrate tablets if markedly elevated CK levels occur or if myopathy is either diagnosed or suspected. Temporarily discontinue fenofibrate tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the fenofibrate tablets dosage. Instruct patients to promptly report any unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever (5.3).
- Increases in Serum Creatinine: Monitor renal function in patients with renal impairment taking fenofibrate tablets. Consider monitoring renal function in patients at risk for renal impairment (5.4).
- Cholelithiasis: Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated (5.5).
- Hypersensitivity Reactions: Acute hypersensitivity reactions, including anaphylaxis and angioedema, and delayed hypersensitivity reactions, including severe cutaneous adverse drug reactions have been reported postmarketing. Some cases were life-threatening and required emergency treatment. Discontinue fenofibrate tablets and treat appropriately if reactions occur (5.9).
2 Dosage and Administration
- Severe hypertriglyceridemia: 54 to 160 mg orally once daily; the dosage should be adjusted according to patient response (2.2).
- Primary hyperlipidemia:160 mg orally once daily (2.2).
- Administer as a single dose, at any time of day, with or without food (2.2).
- Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment (2.2).
- Renal impairment: Initial dosage of 54 mg orally once daily (2.3).
- Geriatric patients: Select the dosage on the basis of renal function (2.4).
3 Dosage Forms and Strengths
- 54 mg: Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side.
- 160 mg: White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of fenofibrate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood: Anemia, white blood cell decreases
Gastrointestinal: Pancreatitis
General: Asthenia
Hepatobiliary: Increased total bilirubin, hepatitis, cirrhosis
Immune System: Anaphylaxis, angioedema
Lipid Disorders: Severely depressed HDL-cholesterol levels
Musculoskeletal: Myalgia, muscle spasms, rhabdomyolysis, arthralgia
Renal and Urinary: Acute renal failure
Respiratory: Interstitial lung disease
Skin and Subcutaneous Tissue: Photosensitivity reactions days to months after initiation. This may occur in patients who report a prior photosensitivity reaction to ketoprofen.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of fenofibrate tablets has been established in adults with hypertriglyceridemia or primary hyperlipidemia based on adequate and well-controlled trials of other formulations of fenofibrate, referenced below as "fenofibrate" [see Clinical Studies (14)]. Dosages of fenofibrate used in these trials were comparable to fenofibrate tablets 145 mg per day [see Clinical Pharmacology (12.3)].
Adverse reactions reported by 2% or more of patients treated with fenofibrate (and greater than placebo) during the double-blind, placebo-controlled trials are listed in Table 1. Adverse reactions led to discontinuation of treatment in 5% of patients treated with fenofibrate and in 3% treated with placebo. Increases in liver function tests were the most frequent events, causing discontinuation of fenofibrate treatment in 1.6% of patients in double-blind trials.
| Adverse Reaction | Placebo (N = 365) | Fenofibrate (N = 439) |
|---|---|---|
| Abnormal Liver Tests | 1% | 8% |
| Abdominal Pain | 4% | 5% |
| Increased ALT | 2% | 3% |
| Increased AST | 1% | 3% |
| Increased Creatine Phosphokinase | 1% | 3% |
| Constipation | 1% | 2% |
| Rhinitis | 1% | 2% |
5.10 Venothromboembolic Disease
In the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) trial, pulmonary embolus (PE) and deep vein thrombosis (DVT) were observed at higher rates in the fenofibrate- than the placebo-treated group. Of 9,795 patients enrolled in FIELD, there were 4,900 in the placebo group and 4,895 in the fenofibrate group. For DVT, there were 48 events (1%) in the placebo group and 67 (1.4%) in the fenofibrate group (p = 0.074); and for PE, there were 32 (0.7%) events in the placebo group and 53 (1.1%) in the fenofibrate group (p = 0.022).
In the Coronary Drug Project, a higher proportion of the clofibrate group experienced definite or suspected fatal or nonfatal pulmonary embolism or thrombophlebitis than the placebo group (5.2% vs. 3.3% at five years; p < 0.01).
In the cardiovascular outcome trial with pemafibrate, pulmonary embolism was reported for 37 (0.7%) subjects in the pemafibrate group and 16 (0.3%) subjects in the placebo group. Deep vein thrombosis was reported for 36 (0.7%) subjects in the pemafibrate group and 13 (0.2%) subjects in the placebo group.
5.3 Myopathy and Rhabdomyolysis
Fenofibrate tablets may cause myopathy [muscle pain, tenderness, or weakness associated with elevated creatine kinase (CK)] and rhabdomyolysis.
14.1 Overview of Clinical Trials
The effectiveness of fenofibrate tablets has been established in adults with hypertriglyceridemia or primary hyperlipidemia based on adequate and well-controlled trials of other formulations of fenofibrate, referenced below as "fenofibrate." Dosages of fenofibrate used in these trials were comparable to fenofibrate tablets 145 mg per day [see Clinical Pharmacology (12.3)].
5.4 Increases in Serum Creatinine
Increases in serum creatinine have been reported in patients receiving fenofibrates. These increases tend to return to baseline following discontinuation of fenofibrate tablets. The clinical significance of this finding is unknown. Monitor renal function in patients with renal impairment taking fenofibrate tablets. Renal monitoring should also be considered for patients taking fenofibrate tablets at risk for renal insufficiency such as geriatric patients and patients with diabetes. Fenofibrate tablets are contraindicated in patients with severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Dosage and Administration (2.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling
Fenofibrate Tablets, USP are supplied as follows:
54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45).
160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
2.2 Recommended Dosage and Administration
-
Severe hypertriglyceridemia:
- The recommended dosage of fenofibrate tablets is 54 mg or 160 mg orally once daily.
- Dosage should be individualized according to patient response, and should be adjusted if necessary following repeat lipid determinations at 4 to 8 week intervals.
-
Primary hyperlipidemia:
- The recommended dosage of fenofibrate tablets is 160 mg orally once daily.
- Administer fenofibrate tablets as a single dose at any time of day, with or without food.
- Advise patients to swallow fenofibrate tablets whole. Do not crush, break, dissolve, or chew tablets.
- Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment.
- If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose.
- Advise patients to take fenofibrate tablets at least 1 hour before or 4 hours to 6 hours after a bile acid binding resin to avoid impeding its absorption.
2.4 Recommended Dosage in Geriatric Patients
Dosage selection for geriatric patients should be made on the basis of renal function [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.1 Prior to Initiation of Fenofibrate Tablets
- Assess lipid levels before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, or medications) of high TG levels and manage as appropriate.
- Patients should be placed on an appropriate lipid-lowering diet before receiving fenofibrate tablets, and should continue this diet during treatment with fenofibrate tablets.
- In patients with diabetes and fasting chylomicronemia, improve glycemic control prior to considering starting fenofibrate tablets.
5.1 Mortality and Coronary Heart Disease Morbidity
Fenofibrate did not reduce cardiovascular disease morbidity or mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus [see Clinical Studies (14.4)].
Because of chemical, pharmacological, and clinical similarities between fenofibrates, including fenofibrate tablets; pemafibrate; clofibrate; and gemfibrozil; the findings in 5 large randomized, placebo-controlled clinical trials with these other fibrate drugs may also apply to fenofibrate tablets.
Pemafibrate did not reduce cardiovascular disease morbidity or mortality in a large, randomized, placebo-controlled trial of patients with type 2 diabetes mellitus on background statin therapy [see Clinical Studies (14.4)].
In the Coronary Drug Project, a large trial conducted from 1965 to 1985 in men post myocardial infarction, there was no difference in mortality or nonfatal myocardial infarction between the clofibrate group and the placebo group after 5 years of treatment (NCT00000482).
In a trial conducted by the World Health Organization (WHO) from 1965 to 1976, men without known coronary artery disease were treated with placebo or clofibrate for 5 years and followed for an additional 1 year. There was a statistically significant, higher age-adjusted all-cause mortality in the clofibrate group compared with the placebo group (5.70% vs. 3.96%, p ≤ 0.01). Excess mortality was due to a 33% increase in non-cardiovascular causes, including malignancy, post-cholecystectomy complications, and pancreatitis.
The Helsinki Heart Study, conducted from 1982 to 1987, was a large (n=4,081) trial of middle-aged men without a history of coronary artery disease. Subjects received either placebo or gemfibrozil for 5 years, with a 3.5-year open extension afterward. Total mortality was numerically but not statistically higher in the gemfibrozil randomization group versus placebo [95% confidence interval (CI) of the hazard ratio (HR) 0.91 to 1.64].
A secondary prevention component of the Helsinki Heart Study treated middle-aged men with gemfibrozil or placebo for 5 years. The HR for cardiac deaths was 2.2, 95% CI, 0.94 to 5.05.
Principal Display Panel 54 Mg Tablet Bottle Label
NDC 42858-454-45
Fenofibrate
Tablets, USP
54 mg
Rx only
KEEP THIS AND ALL MEDICATION
OUT OF THE REACH OF CHILDREN.
Rhodes
90 Tablets
5.11 Paradoxical Decreases in Hdl Cholesterol Levels
There have been postmarketing and clinical trial reports of severe decreases in high-density lipoprotein cholesterol (HDL-C) levels (as low as 2 mg/dL) occurring in patients with and without diabetes initiated on fibrate therapy, including fenofibrate. The decrease in HDL-C is mirrored by a decrease in apolipoprotein A1. This decrease has been reported to occur within 2 weeks to years after initiation of fibrate therapy. The HDL-C levels remain depressed until fibrate therapy has been withdrawn; the response to withdrawal of fibrate therapy is rapid and sustained. The clinical significance of this decrease in HDL-C is unknown. Check HDL-C levels within the first few months after initiation of fenofibrate tablets. If a severely depressed HDL-C level is detected, discontinue fenofibrate tablets and monitor HDL-C until it has returned to baseline. Fenofibrate tablets should not be re-initiated.
Principal Display Panel 160 Mg Tablet Bottle Label
NDC 42858-660-45
Fenofibrate
Tablets, USP
160 mg
Rx only
KEEP THIS AND ALL MEDICATION
OUT OF THE REACH OF CHILDREN.
Rhodes
90 Tablets
14.4 Lack of Efficacy in Cardiovascular Outcomes Trials
Fenofibrate did not reduce cardiovascular disease morbidity or mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus.
The Action to Control Cardiovascular Risk in Diabetes Lipid (ACCORD Lipid) (NCT00000620) trial was a randomized placebo-controlled trial of 5,518 patients (2,765 assigned to receive fenofibrate) with type 2 diabetes mellitus on background statin therapy treated with fenofibrate. The mean age at baseline was 62 years and 31% were female. Overall, 68% were White, 15% were Black or African American; 7% identified as Hispanic or Latino ethnicity. The mean duration of follow-up was 4.7 years. The primary outcome of major adverse cardiovascular events (MACE), a composite of non-fatal myocardial infarction, non-fatal stroke, and cardiovascular disease death was a HR of 0.92 (95% CI, 0.79 to 1.08) for fenofibrate plus statin combination therapy as compared to statin monotherapy.
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) trial was a 5-year randomized, placebo-controlled trial of 9,795 patients (4,895 assigned to receive fenofibrate) with type 2 diabetes mellitus treated with fenofibrate. The mean age at baseline was 62 years, 37% were female, and 93% were White. The primary outcome of coronary heart disease events was a HR of 0.89 (95% CI, 0.75 to 1.05) with fenofibrate compared to placebo. The HR for total and coronary heart disease mortality, respectively, was 1.11 (95% CI, 0.95 to 1.29) and 1.19 (95% CI, 0.90 to 1.57) with fenofibrate as compared to placebo.
Because of chemical, pharmacological, and clinical similarities between fenofibrate and pemafibrate, findings in a large randomized, placebo-controlled clinical trial with pemafibrate are relevant for fenofibrate tablets.
Pemafibrate did not reduce cardiovascular disease morbidity or mortality in a large, randomized, placebo-controlled trial of patients with type 2 diabetes mellitus (TG levels of 200 to 499 mg per deciliter and HDL-C levels of 40 mg per deciliter or lower), on background statin therapy (NCT03071692). The trial was a randomized placebo-controlled trial of 10,497 patients (5,240 assigned to receive pemafibrate) with type 2 diabetes mellitus on background lipid-lowering therapy. The median age at baseline was 64 years and 28% were female. Overall, 86% were White, 5% were Asian, 3% were Black or African American; 19% identified as Hispanic or Latino ethnicity. The median duration of follow-up was 3.4 years. The primary outcome of major adverse cardiovascular events (MACE), a composite of non-fatal myocardial infarction, non-fatal ischemic stroke, coronary revascularization, and death from cardiovascular causes, was a HR of 1.03 (95% CI, 0.91 to 1.15) for pemafibrate plus statin combination therapy as compared to statin monotherapy.
14.2 Clinical Trials in Adults With Hypertriglyceridemia
The effects of fenofibrate on serum TG were studied in two randomized, double-blind, placebo-controlled clinical trials of 147 patients with hypertriglyceridemia. Patients were treated for 8 weeks under protocols that differed only in that one entered patients with baseline TG levels of 500 to 1,500 mg/dL, and the other TG levels of 350 to 499 mg/dL. In patients with hypertriglyceridemia and normal cholesterolemia with or without hyperchylomicronemia, treatment with fenofibrate decreased primarily very low-density lipoprotein (VLDL) TG and VLDL cholesterol (VLDL-C). Treatment of patients with elevated TG often results in an increase of LDL-C (see Table 5).
| Trial 1 | Placebo | Fenofibrate Tablets | ||||||
| Baseline TG levels 350 to 499 mg/dL | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change |
| TG | 28 | 449 | 450 | -0.5 | 27 | 432 | 223 | -46.2 =p < 0.05 vs. Placebo
|
| VLDL-TG | 19 | 367 | 350 | 2.7 | 19 | 350 | 178 | -44.1 |
| Total-C | 28 | 255 | 261 | 2.8 | 27 | 252 | 227 | -9.1 |
| HDL-C | 28 | 35 | 36 | 4 | 27 | 34 | 40 | 19.6 |
| LDL-C | 28 | 120 | 129 | 12 | 27 | 128 | 137 | 14.5 |
| VLDL-C | 27 | 99 | 99 | 5.8 | 27 | 92 | 46 | -44.7 |
| Trial 2 | Placebo | Fenofibrate Tablets | ||||||
| Baseline TG levels 500 to 1500 mg/dL | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change |
| TG | 44 | 710 | 750 | 7.2 | 48 | 726 | 308 | -54.5 |
| VLDL-TG | 29 | 537 | 571 | 18.7 | 33 | 543 | 205 | -50.6 |
| Total-C | 44 | 272 | 271 | 0.4 | 48 | 261 | 223 | -13.8 |
| HDL-C | 44 | 27 | 28 | 5.0 | 48 | 30 | 36 | 22.9 |
| LDL-C | 42 | 100 | 90 | -4.2 | 45 | 103 | 131 | 45.0 |
| VLDL-C | 42 | 137 | 142 | 11.0 | 45 | 126 | 54 | -49.4 |
2.3 Recommended Dosage in Patients With Renal Impairment
- Assess renal function prior to initiation of fenofibrate tablets and periodically thereafter [see Warnings and Precautions (5.4)].
- Treatment with fenofibrate tablets should be initiated at a dosage of 54 mg orally once daily in patients with mild to moderately impaired renal function (eGFR 30 to <60 mL/min/1.73m2), and increased only after evaluation of the effects on renal function and TG levels at this dosage.
- Fenofibrate tablets are contraindicated in patients with severe renal impairment (eGFR <30 mL/min/1.73m2), including those with end-stage renal disease (ESRD) and those receiving dialysis [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.6 Increased Bleeding Risk With Coumarin Anticoagulants
Exercise caution when co-administering anticoagulants with fenofibrate tablets because of the potentiation of coumarin-type anticoagulant effects in prolonging the Prothrombin Time/International Normalized Ratio (PT/INR). To prevent bleeding complications, monitor the PT/INR frequently and adjust the dosage of the anticoagulant until the PT/INR has stabilized [see Drug Interactions (7)].
14.3 Clinical Trials in Adults With Primary Hyperlipidemia
The effects of fenofibrate were assessed in four randomized, placebo-controlled, double-blind, parallel-group trials in patients with hyperlipidemia and mixed dyslipidemia. Fenofibrate tablets therapy reduced LDL-C, Total-C, and TG, and increased HDL-C (see Table 6).
| Treatment Group | Total-C | LDL-C | HDL-C | TG |
|---|---|---|---|---|
| Mean baseline lipid values (n=646) | 306.9 mg/dL | 213.8 mg/dL | 52.3 mg/dL | 191.0 mg/dL |
| All fenofibrate (n=361) | -18.7% p = < 0.05 vs. Placebo
|
-20.6% | +11.0% | -28.9% |
| Placebo (n=285) | -0.4% | -2.2% | +0.7% | +7.7% |
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- Markedly elevated levels of serum TG (e.g. > 2,000 mg/dL) may increase the risk of developing pancreatitis. The effect of fenofibrate therapy on reducing this risk has not been determined [see Warnings and Precautions (5.7)].
- Fenofibrate did not reduce coronary heart disease morbidity and mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus [see Warnings and Precautions (5.1) and Clinical Studies (14.4)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose of fenofibrate tablets, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. There is no specific treatment for overdose with fenofibrate tablets. General supportive care of the patient is indicated, including monitoring of vital signs and observation of clinical status, should an overdose occur. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage; usual precautions should be observed to maintain the airway. Because fenofibric acid is highly bound to plasma proteins, hemodialysis should not be considered.
11 Description (11 DESCRIPTION)
Fenofibrate Tablets, USP are a peroxisome proliferator-activated receptor (PPAR) alpha agonist available as tablets for oral administration. Each tablet contains 54 mg or 160 mg of fenofibrate, USP. The chemical name for fenofibrate, USP is 2-[4-(4-chlorobenzoyl) phenoxy]-2-methyl-propanoic acid, 1-methylethyl ester with the following structural formula:
The empirical formula is C20H21O4Cl and the molecular weight is 360.83; fenofibrate, USP is insoluble in water. The melting point is 79 to 82°C. Fenofibrate, USP is a white solid which is stable under ordinary conditions.
5.7 Pancreatitis
Pancreatitis has been reported in patients taking fenofibrates. This occurrence may represent a failure of efficacy in patients with severe hypertriglyceridemia, a direct drug effect, or a secondary phenomenon mediated through biliary tract stone or sludge formation with obstruction of the common bile duct.
8.4 Pediatric Use
The safety and effectiveness of fenofibrate tablets have not been established in pediatric patients with severe hypertriglyceridemia or primary hyperlipidemia.
8.5 Geriatric Use
Assess renal function in geriatric patients and follow contraindications and dosing recommendations for patients with renal impairment [see Contraindications (4), Warnings and Precautions (5.3, 5.4), and Use in Specific Populations (8.6)]. While fenofibric acid exposure is not influenced by age, geriatric patients are more likely to have renal impairment, and fenofibric acid is substantially excreted by the kidney [see Clinical Pharmacology (12.3)]. Consider monitoring renal function in geriatric patients taking fenofibrate tablets.
5.2 Hepatotoxicity
Serious drug-induced liver injury (DILI), including liver transplantation and death, has been reported with postmarketing use of fenofibrates, including fenofibrate tablets. DILI has been reported within the first few weeks of treatment or after several months of therapy and in some cases has reversed with discontinuation of fenofibrate tablets treatment. Patients with DILI have experienced signs and symptoms including dark urine, abnormal stool, jaundice, malaise, abdominal pain, myalgia, weight loss, pruritus, and nausea. Many patients had concurrent elevations of total bilirubin, serum alanine transaminase (ALT), and aspartate transaminase (AST). DILI has been characterized as hepatocellular, chronic active, and cholestatic hepatitis, and cirrhosis has occurred in association with chronic active hepatitis.
In clinical trials, an intermediate daily dosage or the maximum recommended daily dosage of fenofibrate have been associated with increases in serum AST or ALT. The incidence of increases in transaminases may be dose related [see Adverse Reactions (6.1)].
Fenofibrate tablets are contraindicated in patients with active liver disease, including those with unexplained persistent liver function abnormalities. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy with fenofibrate tablets. Discontinue fenofibrate tablets if signs or symptoms of liver injury develop or if elevated enzyme levels persist (ALT or AST > 3 times the upper limit of normal, or if accompanied by elevation of bilirubin). Do not restart fenofibrate tablets in these patients if there is no alternative explanation for the liver injury.
5.5 Cholelithiasis
Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated. Fenofibrate tablet therapy should be discontinued if gallstones are found. Fenofibrate tablets are contraindicated in patients with pre-existing gallbladder disease.
4 Contraindications (4 CONTRAINDICATIONS)
Fenofibrate tablets are contraindicated in patients with:
- Severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Clinical Pharmacology (12.3)].
- Active liver disease, including those with unexplained persistent liver function abnormalities [see Warnings and Precautions (5.2)].
- Pre-existing gallbladder disease [see Warnings and Precautions (5.5)].
- Hypersensitivity to fenofibrate, fenofibric acid, or any of the excipients in fenofibrate tablets. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with fenofibrate [see Warnings and Precautions (5.9)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
- Mortality and Coronary Heart Disease Morbidity [see Warnings and Precautions (5.1)]
- Hepatoxicity [see Warnings and Precautions (5.2)]
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.3)]
- Increases in Serum Creatinine [see Warnings and Precautions (5.4)]
- Cholelithiasis [see Warnings and Precautions (5.5)]
- Increased Bleeding Risk with Coumarin Anticoagulants [see Warnings and Precautions (5.6)]
- Pancreatitis [see Warnings and Precautions (5.7)]
- Hematologic Changes [see Warnings and Precautions (5.8)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.9)]
- Venothromboembolic Disease [see Warnings and Precautions (5.10)]
- Paradoxical Decreases in HDL Cholesterol Levels [see Warnings and Precautions (5.11)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 2 presents clinically important drug interactions with fenofibrate tablets.
| Statins | |
| Clinical Impact: | Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with statins. |
| Intervention: | Consider if the benefit of using fenofibrate tablets concomitantly with statin therapy outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of statin therapy. |
| Colchicine | |
| Clinical Impact: | Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with fenofibrates. |
| Intervention: | Consider if the benefit of using colchicine concomitantly with fenofibrate tablets outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dosage titration of colchicine. |
| Coumarin Anticoagulants | |
| Clinical Impact: | Fibrates may cause potentiation of coumarin-type anticoagulant effects with prolongation of the PT/INR. |
| Intervention: | Caution should be exercised when coumarin anticoagulants are given in conjunction with fenofibrate tablets. The dosage of the anticoagulants should be reduced to maintain the PT/INR at the desired level to prevent bleeding complications. Frequent PT/INR determinations are advisable until it has been definitely determined that the PT/INR has stabilized |
| Immunosuppressants | |
| Clinical Impact: | Immunosuppressants such as cyclosporine and tacrolimus can produce nephrotoxicity with decreases in creatinine clearance and rises in serum creatinine, and because renal excretion is the primary elimination route of fibrate drugs including fenofibrate tablets, there is a risk that an interaction will lead to deterioration of renal function. |
| Intervention: | The benefits and risks of using fenofibrate tablets with immunosuppressants and other potentially nephrotoxic agents should be carefully considered, and the lowest effective dosage employed and renal function monitored. |
| Bile-Acid Binding Resins | |
| Clinical Impact: | Bile-acid binding resins may bind other drugs given concurrently. |
| Intervention: | In patients taking a bile acid resin, administer fenofibrate tablets at least 1 hour before or 4 to 6 hours after the bile acid resin to avoid impeding its absorption. |
8.6 Renal Impairment
Fenofibrate tablets are contraindicated in patients with severe renal impairment (eGFR <30 mL/min/1.73m2), including those with end-stage renal disease (ESRD) and those receiving dialysis. Dosage reduction is required in patients with mild to moderate renal impairment [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)]. Patients with severe renal impairment have 2.7-fold higher exposure of fenofibric acid and increased accumulation of fenofibric acid during chronic dosing compared with healthy volunteers. Renal impairment is a risk factor for myopathy and rhabdomyolysis [see Warnings and Precautions (5.3, 5.4) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Fenofibric acid, the active metabolite of fenofibrate, produces reductions in total cholesterol (Total-C), total triglycerides, and triglyceride rich lipoprotein (VLDL) in treated patients with severe hypertriglyceridemia.
12.3 Pharmacokinetics
Fenofibrate is a pro-drug of the active chemical moiety fenofibric acid. Fenofibrate is converted by ester hydrolysis in the body to fenofibric acid which is the active constituent measurable in the circulation.
8.7 Hepatic Impairment
The use of fenofibrate tablets has not been evaluated in patients with hepatic impairment. Fenofibrate tablets are contraindicated in patients with active liver disease, including those with unexplained persistent liver function abnormalities [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fenofibrate tablets are indicated as adjunctive therapy to diet:
- to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL).
- to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
5.8 Hematologic Changes
Mild to moderate hemoglobin, hematocrit, and white blood cell decreases have been observed in patients following initiation of therapy with fenofibrates. However, these levels stabilize during long-term administration. Thrombocytopenia and agranulocytosis have been reported in individuals treated with fenofibrate tablets. Periodic monitoring of red and white blood cell counts is recommended during the first 12 months of fenofibrate tablets administration.
12.1 Mechanism of Action
The active moiety of fenofibrate tablets is fenofibric acid. The pharmacological effects of fenofibric acid in both animals and humans have been studied through oral administration of fenofibrate.
The lipid-modifying effects of fenofibric acid seen in clinical practice have been explained in vivo in transgenic mice and in vitro in human hepatocyte cultures by the activation of PPAR alpha receptor. Through this mechanism, fenofibric acid increases lipolysis and elimination of triglyceride-rich particles from plasma by activating lipoprotein lipase and reducing production of apoprotein C-III (an inhibitor of lipoprotein lipase activity).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatotoxicity: Serious drug-induced liver injury, including liver transplantation and death, has been reported with fenofibrates, including fenofibrate tablets. Monitor patient's liver function, including serum ALT, AST, and total bilirubin, at baseline and periodically for the duration of therapy. Discontinue if signs or symptoms of liver injury develop or if elevated enzyme levels persist (5.2).
- Myopathy and Rhabdomyolysis: Have been reported in patients taking fenofibrates. Risks are increased during co-administration with a statin, in geriatric patients, and in patients with renal impairment or hypothyroidism. Discontinue fenofibrate tablets if markedly elevated CK levels occur or if myopathy is either diagnosed or suspected. Temporarily discontinue fenofibrate tablets in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis. Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the fenofibrate tablets dosage. Instruct patients to promptly report any unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever (5.3).
- Increases in Serum Creatinine: Monitor renal function in patients with renal impairment taking fenofibrate tablets. Consider monitoring renal function in patients at risk for renal impairment (5.4).
- Cholelithiasis: Fenofibrate may increase cholesterol excretion into the bile, leading to cholelithiasis. If cholelithiasis is suspected, gallbladder studies are indicated (5.5).
- Hypersensitivity Reactions: Acute hypersensitivity reactions, including anaphylaxis and angioedema, and delayed hypersensitivity reactions, including severe cutaneous adverse drug reactions have been reported postmarketing. Some cases were life-threatening and required emergency treatment. Discontinue fenofibrate tablets and treat appropriately if reactions occur (5.9).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Severe hypertriglyceridemia: 54 to 160 mg orally once daily; the dosage should be adjusted according to patient response (2.2).
- Primary hyperlipidemia:160 mg orally once daily (2.2).
- Administer as a single dose, at any time of day, with or without food (2.2).
- Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment (2.2).
- Renal impairment: Initial dosage of 54 mg orally once daily (2.3).
- Geriatric patients: Select the dosage on the basis of renal function (2.4).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 54 mg: Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side.
- 160 mg: White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of fenofibrate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood: Anemia, white blood cell decreases
Gastrointestinal: Pancreatitis
General: Asthenia
Hepatobiliary: Increased total bilirubin, hepatitis, cirrhosis
Immune System: Anaphylaxis, angioedema
Lipid Disorders: Severely depressed HDL-cholesterol levels
Musculoskeletal: Myalgia, muscle spasms, rhabdomyolysis, arthralgia
Renal and Urinary: Acute renal failure
Respiratory: Interstitial lung disease
Skin and Subcutaneous Tissue: Photosensitivity reactions days to months after initiation. This may occur in patients who report a prior photosensitivity reaction to ketoprofen.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of fenofibrate tablets has been established in adults with hypertriglyceridemia or primary hyperlipidemia based on adequate and well-controlled trials of other formulations of fenofibrate, referenced below as "fenofibrate" [see Clinical Studies (14)]. Dosages of fenofibrate used in these trials were comparable to fenofibrate tablets 145 mg per day [see Clinical Pharmacology (12.3)].
Adverse reactions reported by 2% or more of patients treated with fenofibrate (and greater than placebo) during the double-blind, placebo-controlled trials are listed in Table 1. Adverse reactions led to discontinuation of treatment in 5% of patients treated with fenofibrate and in 3% treated with placebo. Increases in liver function tests were the most frequent events, causing discontinuation of fenofibrate treatment in 1.6% of patients in double-blind trials.
| Adverse Reaction | Placebo (N = 365) | Fenofibrate (N = 439) |
|---|---|---|
| Abnormal Liver Tests | 1% | 8% |
| Abdominal Pain | 4% | 5% |
| Increased ALT | 2% | 3% |
| Increased AST | 1% | 3% |
| Increased Creatine Phosphokinase | 1% | 3% |
| Constipation | 1% | 2% |
| Rhinitis | 1% | 2% |
5.10 Venothromboembolic Disease
In the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) trial, pulmonary embolus (PE) and deep vein thrombosis (DVT) were observed at higher rates in the fenofibrate- than the placebo-treated group. Of 9,795 patients enrolled in FIELD, there were 4,900 in the placebo group and 4,895 in the fenofibrate group. For DVT, there were 48 events (1%) in the placebo group and 67 (1.4%) in the fenofibrate group (p = 0.074); and for PE, there were 32 (0.7%) events in the placebo group and 53 (1.1%) in the fenofibrate group (p = 0.022).
In the Coronary Drug Project, a higher proportion of the clofibrate group experienced definite or suspected fatal or nonfatal pulmonary embolism or thrombophlebitis than the placebo group (5.2% vs. 3.3% at five years; p < 0.01).
In the cardiovascular outcome trial with pemafibrate, pulmonary embolism was reported for 37 (0.7%) subjects in the pemafibrate group and 16 (0.3%) subjects in the placebo group. Deep vein thrombosis was reported for 36 (0.7%) subjects in the pemafibrate group and 13 (0.2%) subjects in the placebo group.
5.3 Myopathy and Rhabdomyolysis
Fenofibrate tablets may cause myopathy [muscle pain, tenderness, or weakness associated with elevated creatine kinase (CK)] and rhabdomyolysis.
14.1 Overview of Clinical Trials
The effectiveness of fenofibrate tablets has been established in adults with hypertriglyceridemia or primary hyperlipidemia based on adequate and well-controlled trials of other formulations of fenofibrate, referenced below as "fenofibrate." Dosages of fenofibrate used in these trials were comparable to fenofibrate tablets 145 mg per day [see Clinical Pharmacology (12.3)].
5.4 Increases in Serum Creatinine
Increases in serum creatinine have been reported in patients receiving fenofibrates. These increases tend to return to baseline following discontinuation of fenofibrate tablets. The clinical significance of this finding is unknown. Monitor renal function in patients with renal impairment taking fenofibrate tablets. Renal monitoring should also be considered for patients taking fenofibrate tablets at risk for renal insufficiency such as geriatric patients and patients with diabetes. Fenofibrate tablets are contraindicated in patients with severe renal impairment, including those with end-stage renal disease (ESRD) and those receiving dialysis [see Dosage and Administration (2.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Fenofibrate Tablets, USP are supplied as follows:
54 mg - Yellow, round-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7330" on the other side. Available in bottles of 90 (NDC 42858-454-45).
160 mg - White to off white, oval-shaped, film-coated, unscored tablet, debossed with "93" on one side and "7331" on the other side. Available in bottles of 90 (NDC 42858-660-45).
2.2 Recommended Dosage and Administration
-
Severe hypertriglyceridemia:
- The recommended dosage of fenofibrate tablets is 54 mg or 160 mg orally once daily.
- Dosage should be individualized according to patient response, and should be adjusted if necessary following repeat lipid determinations at 4 to 8 week intervals.
-
Primary hyperlipidemia:
- The recommended dosage of fenofibrate tablets is 160 mg orally once daily.
- Administer fenofibrate tablets as a single dose at any time of day, with or without food.
- Advise patients to swallow fenofibrate tablets whole. Do not crush, break, dissolve, or chew tablets.
- Assess TG when clinically appropriate, as early as 4 to 8 weeks after initiating fenofibrate tablets. Discontinue fenofibrate tablets in patients who do not have an adequate response after 2 months of treatment.
- If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose.
- Advise patients to take fenofibrate tablets at least 1 hour before or 4 hours to 6 hours after a bile acid binding resin to avoid impeding its absorption.
2.4 Recommended Dosage in Geriatric Patients
Dosage selection for geriatric patients should be made on the basis of renal function [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
2.1 Prior to Initiation of Fenofibrate Tablets
- Assess lipid levels before initiating therapy. Identify other causes (e.g., diabetes mellitus, hypothyroidism, or medications) of high TG levels and manage as appropriate.
- Patients should be placed on an appropriate lipid-lowering diet before receiving fenofibrate tablets, and should continue this diet during treatment with fenofibrate tablets.
- In patients with diabetes and fasting chylomicronemia, improve glycemic control prior to considering starting fenofibrate tablets.
5.1 Mortality and Coronary Heart Disease Morbidity
Fenofibrate did not reduce cardiovascular disease morbidity or mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus [see Clinical Studies (14.4)].
Because of chemical, pharmacological, and clinical similarities between fenofibrates, including fenofibrate tablets; pemafibrate; clofibrate; and gemfibrozil; the findings in 5 large randomized, placebo-controlled clinical trials with these other fibrate drugs may also apply to fenofibrate tablets.
Pemafibrate did not reduce cardiovascular disease morbidity or mortality in a large, randomized, placebo-controlled trial of patients with type 2 diabetes mellitus on background statin therapy [see Clinical Studies (14.4)].
In the Coronary Drug Project, a large trial conducted from 1965 to 1985 in men post myocardial infarction, there was no difference in mortality or nonfatal myocardial infarction between the clofibrate group and the placebo group after 5 years of treatment (NCT00000482).
In a trial conducted by the World Health Organization (WHO) from 1965 to 1976, men without known coronary artery disease were treated with placebo or clofibrate for 5 years and followed for an additional 1 year. There was a statistically significant, higher age-adjusted all-cause mortality in the clofibrate group compared with the placebo group (5.70% vs. 3.96%, p ≤ 0.01). Excess mortality was due to a 33% increase in non-cardiovascular causes, including malignancy, post-cholecystectomy complications, and pancreatitis.
The Helsinki Heart Study, conducted from 1982 to 1987, was a large (n=4,081) trial of middle-aged men without a history of coronary artery disease. Subjects received either placebo or gemfibrozil for 5 years, with a 3.5-year open extension afterward. Total mortality was numerically but not statistically higher in the gemfibrozil randomization group versus placebo [95% confidence interval (CI) of the hazard ratio (HR) 0.91 to 1.64].
A secondary prevention component of the Helsinki Heart Study treated middle-aged men with gemfibrozil or placebo for 5 years. The HR for cardiac deaths was 2.2, 95% CI, 0.94 to 5.05.
Principal Display Panel 54 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 54 mg Tablet Bottle Label)
NDC 42858-454-45
Fenofibrate
Tablets, USP
54 mg
Rx only
KEEP THIS AND ALL MEDICATION
OUT OF THE REACH OF CHILDREN.
Rhodes
90 Tablets
5.11 Paradoxical Decreases in Hdl Cholesterol Levels (5.11 Paradoxical Decreases in HDL Cholesterol Levels)
There have been postmarketing and clinical trial reports of severe decreases in high-density lipoprotein cholesterol (HDL-C) levels (as low as 2 mg/dL) occurring in patients with and without diabetes initiated on fibrate therapy, including fenofibrate. The decrease in HDL-C is mirrored by a decrease in apolipoprotein A1. This decrease has been reported to occur within 2 weeks to years after initiation of fibrate therapy. The HDL-C levels remain depressed until fibrate therapy has been withdrawn; the response to withdrawal of fibrate therapy is rapid and sustained. The clinical significance of this decrease in HDL-C is unknown. Check HDL-C levels within the first few months after initiation of fenofibrate tablets. If a severely depressed HDL-C level is detected, discontinue fenofibrate tablets and monitor HDL-C until it has returned to baseline. Fenofibrate tablets should not be re-initiated.
Principal Display Panel 160 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 160 mg Tablet Bottle Label)
NDC 42858-660-45
Fenofibrate
Tablets, USP
160 mg
Rx only
KEEP THIS AND ALL MEDICATION
OUT OF THE REACH OF CHILDREN.
Rhodes
90 Tablets
14.4 Lack of Efficacy in Cardiovascular Outcomes Trials
Fenofibrate did not reduce cardiovascular disease morbidity or mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus.
The Action to Control Cardiovascular Risk in Diabetes Lipid (ACCORD Lipid) (NCT00000620) trial was a randomized placebo-controlled trial of 5,518 patients (2,765 assigned to receive fenofibrate) with type 2 diabetes mellitus on background statin therapy treated with fenofibrate. The mean age at baseline was 62 years and 31% were female. Overall, 68% were White, 15% were Black or African American; 7% identified as Hispanic or Latino ethnicity. The mean duration of follow-up was 4.7 years. The primary outcome of major adverse cardiovascular events (MACE), a composite of non-fatal myocardial infarction, non-fatal stroke, and cardiovascular disease death was a HR of 0.92 (95% CI, 0.79 to 1.08) for fenofibrate plus statin combination therapy as compared to statin monotherapy.
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) trial was a 5-year randomized, placebo-controlled trial of 9,795 patients (4,895 assigned to receive fenofibrate) with type 2 diabetes mellitus treated with fenofibrate. The mean age at baseline was 62 years, 37% were female, and 93% were White. The primary outcome of coronary heart disease events was a HR of 0.89 (95% CI, 0.75 to 1.05) with fenofibrate compared to placebo. The HR for total and coronary heart disease mortality, respectively, was 1.11 (95% CI, 0.95 to 1.29) and 1.19 (95% CI, 0.90 to 1.57) with fenofibrate as compared to placebo.
Because of chemical, pharmacological, and clinical similarities between fenofibrate and pemafibrate, findings in a large randomized, placebo-controlled clinical trial with pemafibrate are relevant for fenofibrate tablets.
Pemafibrate did not reduce cardiovascular disease morbidity or mortality in a large, randomized, placebo-controlled trial of patients with type 2 diabetes mellitus (TG levels of 200 to 499 mg per deciliter and HDL-C levels of 40 mg per deciliter or lower), on background statin therapy (NCT03071692). The trial was a randomized placebo-controlled trial of 10,497 patients (5,240 assigned to receive pemafibrate) with type 2 diabetes mellitus on background lipid-lowering therapy. The median age at baseline was 64 years and 28% were female. Overall, 86% were White, 5% were Asian, 3% were Black or African American; 19% identified as Hispanic or Latino ethnicity. The median duration of follow-up was 3.4 years. The primary outcome of major adverse cardiovascular events (MACE), a composite of non-fatal myocardial infarction, non-fatal ischemic stroke, coronary revascularization, and death from cardiovascular causes, was a HR of 1.03 (95% CI, 0.91 to 1.15) for pemafibrate plus statin combination therapy as compared to statin monotherapy.
14.2 Clinical Trials in Adults With Hypertriglyceridemia (14.2 Clinical Trials in Adults with Hypertriglyceridemia)
The effects of fenofibrate on serum TG were studied in two randomized, double-blind, placebo-controlled clinical trials of 147 patients with hypertriglyceridemia. Patients were treated for 8 weeks under protocols that differed only in that one entered patients with baseline TG levels of 500 to 1,500 mg/dL, and the other TG levels of 350 to 499 mg/dL. In patients with hypertriglyceridemia and normal cholesterolemia with or without hyperchylomicronemia, treatment with fenofibrate decreased primarily very low-density lipoprotein (VLDL) TG and VLDL cholesterol (VLDL-C). Treatment of patients with elevated TG often results in an increase of LDL-C (see Table 5).
| Trial 1 | Placebo | Fenofibrate Tablets | ||||||
| Baseline TG levels 350 to 499 mg/dL | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change |
| TG | 28 | 449 | 450 | -0.5 | 27 | 432 | 223 | -46.2 =p < 0.05 vs. Placebo
|
| VLDL-TG | 19 | 367 | 350 | 2.7 | 19 | 350 | 178 | -44.1 |
| Total-C | 28 | 255 | 261 | 2.8 | 27 | 252 | 227 | -9.1 |
| HDL-C | 28 | 35 | 36 | 4 | 27 | 34 | 40 | 19.6 |
| LDL-C | 28 | 120 | 129 | 12 | 27 | 128 | 137 | 14.5 |
| VLDL-C | 27 | 99 | 99 | 5.8 | 27 | 92 | 46 | -44.7 |
| Trial 2 | Placebo | Fenofibrate Tablets | ||||||
| Baseline TG levels 500 to 1500 mg/dL | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change | N | Baseline Mean (mg/dL) | Endpoint Mean (mg/dL) | Mean % Change |
| TG | 44 | 710 | 750 | 7.2 | 48 | 726 | 308 | -54.5 |
| VLDL-TG | 29 | 537 | 571 | 18.7 | 33 | 543 | 205 | -50.6 |
| Total-C | 44 | 272 | 271 | 0.4 | 48 | 261 | 223 | -13.8 |
| HDL-C | 44 | 27 | 28 | 5.0 | 48 | 30 | 36 | 22.9 |
| LDL-C | 42 | 100 | 90 | -4.2 | 45 | 103 | 131 | 45.0 |
| VLDL-C | 42 | 137 | 142 | 11.0 | 45 | 126 | 54 | -49.4 |
2.3 Recommended Dosage in Patients With Renal Impairment (2.3 Recommended Dosage in Patients with Renal Impairment)
- Assess renal function prior to initiation of fenofibrate tablets and periodically thereafter [see Warnings and Precautions (5.4)].
- Treatment with fenofibrate tablets should be initiated at a dosage of 54 mg orally once daily in patients with mild to moderately impaired renal function (eGFR 30 to <60 mL/min/1.73m2), and increased only after evaluation of the effects on renal function and TG levels at this dosage.
- Fenofibrate tablets are contraindicated in patients with severe renal impairment (eGFR <30 mL/min/1.73m2), including those with end-stage renal disease (ESRD) and those receiving dialysis [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.6 Increased Bleeding Risk With Coumarin Anticoagulants (5.6 Increased Bleeding Risk with Coumarin Anticoagulants)
Exercise caution when co-administering anticoagulants with fenofibrate tablets because of the potentiation of coumarin-type anticoagulant effects in prolonging the Prothrombin Time/International Normalized Ratio (PT/INR). To prevent bleeding complications, monitor the PT/INR frequently and adjust the dosage of the anticoagulant until the PT/INR has stabilized [see Drug Interactions (7)].
14.3 Clinical Trials in Adults With Primary Hyperlipidemia (14.3 Clinical Trials in Adults with Primary Hyperlipidemia)
The effects of fenofibrate were assessed in four randomized, placebo-controlled, double-blind, parallel-group trials in patients with hyperlipidemia and mixed dyslipidemia. Fenofibrate tablets therapy reduced LDL-C, Total-C, and TG, and increased HDL-C (see Table 6).
| Treatment Group | Total-C | LDL-C | HDL-C | TG |
|---|---|---|---|---|
| Mean baseline lipid values (n=646) | 306.9 mg/dL | 213.8 mg/dL | 52.3 mg/dL | 191.0 mg/dL |
| All fenofibrate (n=361) | -18.7% p = < 0.05 vs. Placebo
|
-20.6% | +11.0% | -28.9% |
| Placebo (n=285) | -0.4% | -2.2% | +0.7% | +7.7% |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:32.799986 · Updated: 2026-03-14T22:30:10.662825