These Highlights Do Not Include All The Information Needed To Use Kimyrsa Safely And Effectively. See Full Prescribing Information For Kimyrsa.
5e755c40-6e73-4572-b474-4d8a131693d1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.2 )
Indications and Usage
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.2 )
Dosage and Administration
There are two oritavancin products (KIMYRSA and ORBACTIV ® , another oritavancin product) that: Are supplied in different dose strengths of oritavancin [see Dosage Forms and Strengths (3) ] . Have different recommended durations of infusion [see Dosage and Administration (2.2) ]. Have different preparation instructions, including differences in reconstitution, dilution, and compatible diluents [see Dosage and Administration (2.3 , 2.4) ] . Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in this prescribing information (PI) [see Dosage and Administration (2.1 , 2.2 , 2.3 , 2.4) ]. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
Warnings and Precautions
Coagulation test interference: Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, and may prolong PT and INR for up to 12 hours and ACT for up to 24 hours. For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, consider a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT. ( 5.1 , 7.2 ) Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. Discontinue infusion if signs of acute hypersensitivity occur. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.2 ) Infusion Related Reactions: Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA). Stopping or slowing the infusion may result in cessation of these reactions. ( 5.3 ) Clostridioides difficile -Associated Diarrhea: Evaluate patients if diarrhea occurs. ( 5.4 ) Concomitant warfarin use: Oritavancin has been shown to artificially prolong PT/INR for up to 12 hours ( 5.1 ). Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin. ( 5.5 ) Osteomyelitis: Institute appropriate alternate antibacterial therapy in patients with confirmed or suspected osteomyelitis. ( 5.6 )
Contraindications
Use of intravenous unfractionated heparin sodium is contraindicated for 120 hours (5 days) after KIMYRSA administration. ( 4.1 , 5.1 ) Known hypersensitivity to oritavancin products. ( 4.2 , 5.2 )
Adverse Reactions
The following adverse reactions are also discussed in the Warnings and Precautions section of labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion Related Reactions [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ] Osteomyelitis [see Warnings and Precautions (5.6) ]
How Supplied
KIMYRSA is supplied as a sterile white to off-white or pink lyophilized powder in single-dose clear glass vials containing 1,200 mg of oritavancin. One vial is packaged in a carton to supply a single 1,200 mg dose treatment (NDC 70842-225-01). KIMYRSA vials should be stored at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP, Controlled Room Temperature (CRT)].
Medication Information
Warnings and Precautions
Coagulation test interference: Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, and may prolong PT and INR for up to 12 hours and ACT for up to 24 hours. For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, consider a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT. ( 5.1 , 7.2 ) Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. Discontinue infusion if signs of acute hypersensitivity occur. Carefully monitor patients with known hypersensitivity to glycopeptides. ( 5.2 ) Infusion Related Reactions: Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA). Stopping or slowing the infusion may result in cessation of these reactions. ( 5.3 ) Clostridioides difficile -Associated Diarrhea: Evaluate patients if diarrhea occurs. ( 5.4 ) Concomitant warfarin use: Oritavancin has been shown to artificially prolong PT/INR for up to 12 hours ( 5.1 ). Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin. ( 5.5 ) Osteomyelitis: Institute appropriate alternate antibacterial therapy in patients with confirmed or suspected osteomyelitis. ( 5.6 )
Indications and Usage
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.2 )
Dosage and Administration
There are two oritavancin products (KIMYRSA and ORBACTIV ® , another oritavancin product) that: Are supplied in different dose strengths of oritavancin [see Dosage Forms and Strengths (3) ] . Have different recommended durations of infusion [see Dosage and Administration (2.2) ]. Have different preparation instructions, including differences in reconstitution, dilution, and compatible diluents [see Dosage and Administration (2.3 , 2.4) ] . Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in this prescribing information (PI) [see Dosage and Administration (2.1 , 2.2 , 2.3 , 2.4) ]. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
Contraindications
Use of intravenous unfractionated heparin sodium is contraindicated for 120 hours (5 days) after KIMYRSA administration. ( 4.1 , 5.1 ) Known hypersensitivity to oritavancin products. ( 4.2 , 5.2 )
Adverse Reactions
The following adverse reactions are also discussed in the Warnings and Precautions section of labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion Related Reactions [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions (5.4) ] Osteomyelitis [see Warnings and Precautions (5.6) ]
How Supplied
KIMYRSA is supplied as a sterile white to off-white or pink lyophilized powder in single-dose clear glass vials containing 1,200 mg of oritavancin. One vial is packaged in a carton to supply a single 1,200 mg dose treatment (NDC 70842-225-01). KIMYRSA vials should be stored at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP, Controlled Room Temperature (CRT)].
Description
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.2 )
Section 42229-5
Reconstitution: Aseptic technique should be used to reconstitute one KIMYRSA 1,200 mg vial.
- Add 40 mL of sterile water for injection (SWFI) to reconstitute the vial to provide a 30 mg/mL solution.
- Gently swirl the contents to avoid foaming and ensure that all KIMYRSA powder is completely dissolved to form a reconstituted solution.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The reconstituted vial should appear to be a clear, colorless to pink solution, free of visible particles.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
Based on an in vitro hemodialysis study, KIMYRSA is unlikely to be removed from blood by hemodialysis. In the event of overdose, supportive measures should be taken.
11 Description
KIMYRSA (oritavancin) for injection contains oritavancin diphosphate, a semisynthetic lipoglycopeptide antibacterial drug for intravenous infusion.
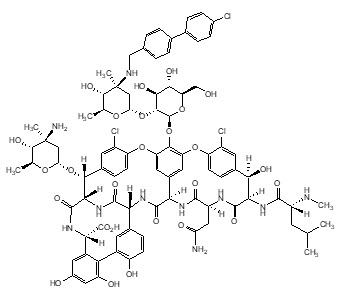
The chemical name for oritavancin is [4"R]-22-O-(3-amino-2,3,6-trideoxy-3-C-methyl-α-L-arabino-hexopyranosyl)-N3''-[(4'-chloro[1,1'-biphenyl]-4-yl)methyl] vancomycin phosphate [1:2] [salt]. The empirical formula of oritavancin diphosphate is C86H97N10O26Cl3•2H3PO4 and the molecular weight is 1989.09. The chemical structure is represented below:
KIMYRSA for injection is supplied as a sterile white to off-white or pink lyophilized powder in a single-dose clear glass vial that contains 1,200 mg of oritavancin (equivalent to 1331.16 mg oritavancin diphosphate) and the following inactive ingredients: hydroxypropyl-β-cyclodextrin (HPβCD) (2400 mg), mannitol (800 mg) and phosphoric acid or sodium hydroxide (to adjust pH 4.0 to 6.0).
The vial is reconstituted with sterile water for injection and further diluted with 0.9% sodium chloride injection or 5% dextrose in sterile water (D5W) for intravenous infusion. Both the reconstituted solution and the diluted solution for infusion should be a clear, colorless to pink solution, free of visible particles [see Dosage and Administration (2.3)].
12.4 Microbiology
KIMYRSA is a semi-synthetic, lipoglycopeptide antibacterial drug. KIMYRSA exerts a concentration-dependent bactericidal activity in vitro against S. aureus, S. pyogenes, and E. faecalis.
5.6 Osteomyelitis
In Phase 3 ABSSSI clinical trials, more cases of osteomyelitis were reported in the oritavancin treated arm than in the vancomycin-treated arm. Monitor patients treated with KIMYRSA for signs and symptoms of osteomyelitis. If osteomyelitis is suspected or diagnosed, institute appropriate alternate antibacterial therapy [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Safety and effectiveness of KIMYRSA in pediatric patients (younger than 18 years of age) have not been established.
8.5 Geriatric Use
The pooled Phase 3 ABSSSI clinical trials of oritavancin did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
12.6 Immunogenicity
Positive indirect and direct antiglobulin tests (IAT/DAT) have been noted with the administration of KIMYRSA and oritavancin in studies with healthy subjects and patients with ABSSSI. The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of KIMYRSA or other oritavancin products.
In a randomized, open-label, multi-center ABSSSI study, positive IAT/DAT were reported in 9.6% (5/52) of subjects who received ORBACTIV, another oritavancin product, and 2% (1/50) of subjects who received KIMYRSA. Oritavancin-dependent red blood cell (RBC) antibodies were detected when tested in the presence of drug for three subjects in the oritavancin group 14 days after dosing.
In a multiple-dose study with oritavancin in healthy volunteers, 90% (9/10) of subjects had a positive IAT 14 days after the second infusion.
In a healthy volunteer study, 66% (22/32) of subjects receiving KIMYRSA had a positive IAT 15 days after receiving dosing and one subject had a positive DAT at 8 days after dosing.
There were no reports of hemolysis in subjects who had positive IAT/DAT in any of the above referenced studies.
An in vitro study further evaluated 403 stored plasma samples from 12 healthy subjects and from 101 patients with ABSSSI from previously conducted clinical studies at multiple timepoints in healthy subjects and in patients with ABSSSI. In samples collected during the 15- and 8-day windows following single-dose treatment in healthy volunteers (Study MDCO-ORI-15-01) and in patients with ABSSSI (Study ML-ORI-201), respectively, low level (<8) direct agglutinating (IgM) anti-oritavancin antibodies were detected in 19 of 113 (17%) subjects. There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of single-dose oritavancin. No hematologic adverse events were reported for subjects with a positive titer.
There were no identified clinically significant effects of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of KIMYRSA over the treatment duration of 14 days.
Positive IAT may interfere with cross-matching before blood transfusion [see Drug Interactions (7.2)].
2.4 Compatibilities
KIMYRSA solution for administration by 1-hour infusion is compatible with:
- 0.9% sodium chloride injection
- 5% dextrose in sterile water (D5W)
4 Contraindications
6 Adverse Reactions
The following adverse reactions are also discussed in the Warnings and Precautions section of labeling:
Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
Infusion Related Reactions [see Warnings and Precautions (5.3)]
Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
Osteomyelitis [see Warnings and Precautions (5.6)]
4.2 Hypersensitivity
KIMYRSA is contraindicated in patients with known hypersensitivity to oritavancin products.
5.2 Hypersensitivity
Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. If an acute hypersensitivity reaction occurs during KIMYRSA infusion, discontinue KIMYRSA immediately and institute appropriate supportive care. Before using KIMYRSA, inquire carefully about previous hypersensitivity reactions to glycopeptides. Due to the possibility of cross-sensitivity, carefully monitor for signs of hypersensitivity during KIMYRSA infusion in patients with a history of glycopeptide allergy. In the Phase 3 ABSSSI clinical trials, the median onset of hypersensitivity reactions in oritavancin-treated patients was 1.2 days and the median duration of these reactions was 2.4 days [see Adverse Reactions (6.1)].
8.6 Renal Impairment
No dosage adjustment of KIMYRSA is needed in patients with mild or moderate renal impairment [see Clinical Pharmacology (12.3)]. The pharmacokinetics of KIMYRSA in severe renal impairment have not been evaluated. Oritavancin is not removed from blood by hemodialysis.
12.2 Pharmacodynamics
The antimicrobial activity of oritavancin appears to correlate with the ratio of area under the concentration-time curve to minimal inhibitory concentration (AUC/MIC) based on animal models of infection. The AUC from time zero to 72 hours correlates with antimicrobial activity in both preclinical and clinical studies.
Exposure-response analyses from both preclinical and clinical studies support the treatment of clinically relevant gram-positive microorganisms (e.g. S. aureus and S. pyogenes) causative of ABSSSI with a single 1,200 mg dose of oritavancin.
12.3 Pharmacokinetics
The mean (±SD) pharmacokinetic parameters of oritavancin products (KIMYRSA and oritavancin) in patients with ABSSSI are presented in Table 3.
| Pharmacokinetic Parameter | KIMYRSA (1 hour) Mean (± SD) |
Oritavancin (3 hour) Mean (± SD) |
|---|---|---|
| Cmax, Maximum plasma concentration; AUC0-72, Area under the plasma concentration-time curve from time zero to 72 hours; SD, Standard deviation. | ||
| Cmax (µg/mL) | 148 (±43.0) | 112 (±34.5) |
| AUC0-72 (h∙µg /mL) | 1460 (±511) | 1470 (±582) |
Oritavancin exhibits linear pharmacokinetics at a dose up to 1,200 mg. The mean, population-predicted oritavancin concentration-time profile displays a multi-exponential decline with a long terminal plasma half-life.
2.5 Incompatibilities
Drugs formulated at a basic or neutral pH may be incompatible with KIMYRSA. KIMYRSA should not be administered simultaneously with commonly used intravenous drugs through a common intravenous port. If the same intravenous line is used for sequential infusion of additional medications, the line should be flushed before and after infusion of KIMYRSA with 0.9% sodium chloride injection or D5W.
2.2 Recommended Dosage
The recommended dosage of KIMYRSA is 1,200 mg administered as a single dose by intravenous infusion over 1 hour in patients 18 years and older [see Warnings and Precautions (5.3)].
8.7 Hepatic Impairment
No dosage adjustment of KIMYRSA is needed in patients with mild or moderate hepatic impairment. The pharmacokinetics of KIMYRSA in patients with severe hepatic insufficiency has not been studied [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
1 Indications and Usage
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.2)
12.1 Mechanism of Action
Oritavancin is an antibacterial drug [see Microbiology (12.4)].
16.1 How Supplied/storage
KIMYRSA is supplied as a sterile white to off-white or pink lyophilized powder in single-dose clear glass vials containing 1,200 mg of oritavancin. One vial is packaged in a carton to supply a single 1,200 mg dose treatment (NDC 70842-225-01).
KIMYRSA vials should be stored at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP, Controlled Room Temperature (CRT)].
5 Warnings and Precautions
- Coagulation test interference: Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, and may prolong PT and INR for up to 12 hours and ACT for up to 24 hours. For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, consider a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT. (5.1, 7.2)
- Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. Discontinue infusion if signs of acute hypersensitivity occur. Carefully monitor patients with known hypersensitivity to glycopeptides. (5.2)
- Infusion Related Reactions: Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA). Stopping or slowing the infusion may result in cessation of these reactions. (5.3)
- Clostridioides difficile-Associated Diarrhea: Evaluate patients if diarrhea occurs. (5.4)
- Concomitant warfarin use: Oritavancin has been shown to artificially prolong PT/INR for up to 12 hours (5.1). Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin. (5.5)
- Osteomyelitis: Institute appropriate alternate antibacterial therapy in patients with confirmed or suspected osteomyelitis. (5.6)
2 Dosage and Administration
- There are two oritavancin products (KIMYRSA and ORBACTIV®, another oritavancin product) that have differences in dose strength, duration of infusion and preparation instructions, including reconstitution and dilution instructions and compatible diluents (2.1, 2.2, 2.3, 2.4)
- Administer 1,200 mg of KIMYRSA as a single dose by intravenous infusion over 1 hour. (2.1, 5.3)
- Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in the full prescribing information. (2.1, 2.2, 2.3)
3 Dosage Forms and Strengths
KIMYRSA is supplied as sterile, white to off-white or pink lyophilized powder containing 1,200 mg of oritavancin (as oritavancin diphosphate) in a single-dose clear glass vial, which must be reconstituted and further diluted prior to intravenous administration.
5.4 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial drugs, including oritavancin products (e.g. KIMYRSA), and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.3 Infusion Related Reactions
Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA), including flushing of the upper body, urticaria, pruritus and/or rash [see Adverse Reactions (6.1)]. Infusion reactions characterized by chest pain, back pain, chills and tremor have been observed with the use of oritavancin, including after the administration of more than one dose of oritavancin during a single course of therapy.
Stopping or slowing the infusion may result in cessation of these reactions. The safety and effectiveness of more than one dose of KIMYRSA during a single course of therapy have not been established [see Dosage and Administration (2.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of oritavancin products cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of KIMYRSA has been established from adequate and well-controlled trials of another oritavancin product, ORBACTIV (hereinafter referred to as oritavancin), in patients with ABSSSI, and a study of KIMYRSA in patients with ABSSSI.
Oritavancin has been evaluated in two, double-blind, controlled ABSSSI clinical trials, which included 976 adult patients treated with a single 1,200 mg intravenous dose of oritavancin and 983 patients treated with intravenous vancomycin for 7 to 10 days. The median age of patients treated with oritavancin was 45.6 years, ranging between 18 and 89 years of age with 8.8% ≥65 years of age. Patients treated with oritavancin were predominantly male (65.4%), 64.4% were Caucasian, 5.8% were African American, and 28.1% were Asian. Safety was evaluated for up to 60 days after dosing.
In the pooled ABSSSI clinical trials, serious adverse reactions were reported in 57/976 (5.8%) patients treated with oritavancin and 58/983 (5.9%) treated with vancomycin. The most commonly reported serious adverse reaction was cellulitis in both treatment groups: 11/976 (1.1%) in oritavancin and 12/983 (1.2%) in the vancomycin arms, respectively.
The most commonly reported adverse reactions (≥3%) in patients receiving a single 1,200 mg dose of oritavancin in the pooled ABSSSI clinical trials were: headache, nausea, vomiting, limb and subcutaneous abscesses, and diarrhea.
In the pooled ABSSSI clinical trials, oritavancin was discontinued due to adverse reactions in 36/976 (3.7%) of patients; the most common reported reactions leading to discontinuation were cellulitis (4/976, 0.4%) and osteomyelitis (3/976, 0.3%).
Table 1 provides selected adverse reactions occurring in ≥1.5% of patients receiving oritavancin in the pooled ABSSSI clinical trials. There were 540 (55.3%) patients in the oritavancin arm and 559 (56.9%) patients in the vancomycin arm, who reported ≥1 adverse reaction.
| Adverse Reactions | Oritavancin N=976 (%) |
Vancomycin N=983 (%) |
|---|---|---|
| Gastrointestinal disorders | ||
| Diarrhea | 36 (3.7) | 32 (3.4) |
| Nausea | 97 (9.9) | 103 (10.5) |
| Vomiting | 45 (4.6) | 46 (4.7) |
| Nervous system disorders | ||
| Dizziness | 26 (2.7) | 26 (2.6) |
| Headache | 69 (7.1) | 66 (6.7) |
| General disorders and administration | ||
| Infusion site phlebitis | 24 (2.5) | 15 (1.5) |
| Infusion site reaction | 19 (1.9) | 34 (3.5) |
| Infections and infestations | ||
| Abscess (limb and subcutaneous) | 37 (3.8) | 23 (2.3) |
| Investigations | ||
| Alanine aminotransferase increased | 27 (2.8) | 15 (1.5) |
| Aspartate aminotransferase increased | 18 (1.8) | 15 (1.5) |
| Cardiac disorders | ||
| Tachycardia | 24 (2.5) | 11 (1.1) |
The following selected adverse reactions were reported in oritavancin-treated patients at a rate of less than 1.5%:
Blood and lymphatic system disorders: anemia, eosinophilia
General disorders and administration site conditions: infusion site erythema, extravasation, induration, pruritus, rash, edema peripheral
Immune system disorders: hypersensitivity
Infections and infestations: osteomyelitis
Investigations: total bilirubin increased, hyperuricemia
Metabolism and nutrition disorders: hypoglycemia
Musculoskeletal and connective tissue disorders: tenosynovitis, myalgia
Respiratory, thoracic and mediastinal disorders: bronchospasm, wheezing
Skin and subcutaneous tissue disorders: urticaria, angioedema, erythema multiforme, pruritus, leucocytoclastic vasculitis, rash.
KIMYRSA has been evaluated in a randomized, open-label, multi-center ABSSSI study which included 50 adult patients treated with a single 1,200 mg intravenous dose of KIMYRSA administered by intravenous infusion over 1 hour, and 52 patients treated with a single 1,200 mg intravenous dose of oritavancin administered by intravenous infusion over 3 hours.
Selected adverse reactions occurring in ≥2 patients receiving either KIMYRSA or oritavancin in the open-label, multi-center ABSSSI study were diarrhea, nausea, vomiting, hypersensitivity, pruritus, chills, headache and pyrexia.
5.1 Coagulation Test Interference
Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, PT and INR for up to 12 hours, and activated clotting time (ACT) for up to 24 hours following administration of a single 1,200 mg dose by binding to and preventing action of the phospholipid reagents commonly used in laboratory coagulation tests. Oritavancin has also been shown to elevate D-dimer concentrations up to 72 hours after oritavancin administration.
For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT monitoring may be considered [see Contraindications (4.1) and Drug Interactions (7.2)].
Oritavancin has no effect on the coagulation system in vivo.
2.1 Dosage and Administration Overview
There are two oritavancin products (KIMYRSA and ORBACTIV®, another oritavancin product) that:
- Are supplied in different dose strengths of oritavancin [see Dosage Forms and Strengths (3)].
- Have different recommended durations of infusion [see Dosage and Administration (2.2)].
- Have different preparation instructions, including differences in reconstitution, dilution, and compatible diluents [see Dosage and Administration (2.3, 2.4)].
Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in this prescribing information (PI) [see Dosage and Administration (2.1, 2.2, 2.3, 2.4)]. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
7.1 Effect of Kimyrsa On Cyp Substrates
A screening drug-drug interaction study indicated that oritavancin is a nonspecific, weak inhibitor (CYP2C9 and CYP2C19) or inducer (CYP3A4 and CYP2D6) of several CYP isoforms [see Clinical Pharmacology (12.3)]. A drug-drug interaction study that assessed the interaction potential of a single 1,200 mg dose of oritavancin on the pharmacokinetics of S-warfarin (CYP2C9 probe substrate) showed no effect of oritavancin on S-warfarin Cmax or AUC.
Avoid administering KIMYRSA concomitantly with drugs that are predominantly metabolized by one of the affected CYP450 enzymes, as co-administration may increase or decrease concentrations of those drugs. Patients should be closely monitored for signs of toxicity or lack of efficacy if they have been given KIMYRSA while on a potentially affected compound (e.g., patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin).
5.7 Development of Drug Resistant Bacteria
Prescribing KIMYRSA in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria [see Patient Counseling Information (17)].
4.1 Intravenous Unfractionated Heparin Sodium
Use of intravenous unfractionated heparin sodium is contraindicated for 120 hours (5 days) after KIMYRSA administration because the activated partial thromboplastin time (aPTT) test results may remain falsely elevated for up to 120 hours (5 days) after KIMYRSA administration [see Warnings and Precautions (5.1) and Drug Interactions (7.2)].
Principal Display Panel 1,200 Mg Vial Label
Rx only
NDC 70842-225-01
Kimyrsa®
(oritavancin) for injection
1,200 mg per vial
Must be reconstituted and
further diluted to a final
volume of 250 mL
For Intravenous Infusion Only
Single-dose Vial
Discard Unused Portion
Principal Display Panel 1,200 Mg Vial Carton
Rx only
NDC 70842-225-01
Kimyrsa®
(oritavancin)
for injection
1,200 mg per vial
Must be reconstituted
and further diluted
to a final volume
of 250 mL
For Intravenous Infusion Only
Single-dose Vial
Discard Unused Portion
2.3 Preparation of Kimyrsa for Intravenous Infusion
There are two oritavancin products (KIMYRSA and ORBACTIV, another oritavancin product) that have differences in dose strengths, duration of infusion, reconstitution and dilution instructions, and compatible diluents. Carefully follow the reconstitution, and dilution instructions with the appropriate compatible diluent for KIMYRSA specified in this prescribing information. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
KIMYRSA is intended for intravenous infusion, only after reconstitution and dilution.
One KIMYRSA 1,200 mg single-dose vial needs to be reconstituted and diluted to prepare a single 1,200 mg intravenous dose.
1.1 Acute Bacterial Skin and Skin Structure Infections
KIMYRSA® is indicated for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following Gram-positive microorganisms:
Staphylococcus aureus (including methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus anginosus group (includes S. anginosus, S. intermedius, and S. constellatus), and Enterococcus faecalis (vancomycin-susceptible isolates only).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals have not been conducted to determine the carcinogenic potential of oritavancin.
No mutagenic or clastogenic potential of oritavancin was found in a battery of tests, including an Ames assay, in vitro chromosome aberration assay in Chinese hamster ovary cells, in vitro forward mutation assay in mouse lymphoma cells and an in vivo mouse micronucleus assay.
Oritavancin did not affect the fertility or reproductive performance of male rats (exposed to daily doses up to 30 mg/kg for at least 4 weeks) and female rats (exposed to daily doses up to 30 mg/kg for at least 2 weeks prior to mating). Those daily doses would be equivalent to a human dose of 300 mg, or 25% of clinical dose. Higher doses were not evaluated in nonclinical fertility studies.
5.5 Potential Risk of Bleeding With Concomitant Use of Warfarin
Oritavancin has been shown to artificially prolong prothrombin time (PT) and international normalized ratio (INR) for up to 12 hours, making the monitoring of the anticoagulation effect of warfarin unreliable up to 12 hours after an oritavancin dose [see Warnings and Precautions (5.1)].
Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin [see Drug Interactions (7.1)].
14.1 Acute Bacterial Skin and Skin Structure Infections (absssi)
A total of 1987 adults with clinically documented ABSSSI suspected or proven to be due to Gram-positive pathogens were randomized into two identically designed, randomized, double-blind, multi-center, multinational, non-inferiority trials (Trial 1 and Trial 2) comparing a single 1,200 mg intravenous dose of oritavancin to intravenous vancomycin (1 g or 15 mg/kg every 12 hours) for 7 to 10 days. The primary analysis population (modified intent to treat, mITT) included all randomized patients who received any study drug. Patients could receive concomitant aztreonam or metronidazole for suspected Gram-negative and anaerobic infection, respectively. Patient demographic and baseline characteristics were balanced between treatment groups. Approximately 64% of patients were Caucasian and 65% were males. The mean age was 45 years and the mean body mass index was 27 kg/m2. Across both trials, approximately 60% of patients were enrolled from the United States and 27% of patients from Asia. A history of diabetes was present in 14% of patients. The types of ABSSSI across both trials included cellulitis/erysipelas (40%), wound infection (29%), and major cutaneous abscesses (31%). Median infection area at baseline across both trials was 266.6 cm2. The primary endpoint in both trials was early clinical response (responder), defined as cessation of spread or reduction in size of baseline lesion, absence of fever, and no rescue antibacterial drug at 48 to 72 hours after initiation of therapy.
Table 4 provides the efficacy results for the primary endpoint in Trial 1 and Trial 2 in the primary analysis population.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | 391/475 (82.3) | 378/479 (78.9) | 3.4 (-1.6, 8.4) |
| Trial 2 | 403/503 (80.1) | 416/502 (82.9) | -2.7 (-7.5, 2.0) |
A key secondary endpoint in these two ABSSSI trials evaluated the percentage of patients achieving a 20% or greater reduction in lesion area from baseline at 48-72 hours after initiation of therapy. Table 5 summarizes the findings for this endpoint in the two ABSSSI trials.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | 413/475 (86.9) | 397/479 (82.9) | 4.1 (-0.5, 8.6) |
| Trial 2 | 432/503 (85.9) | 428/502 (85.3) | 0.6 (-3.7, 5.0) |
Another secondary efficacy endpoint in the two trials was investigator-assessed clinical success at post therapy evaluation at Day 14 to 24 (7 to 14 days from end of blinded therapy). A patient was categorized as a clinical success if the patient experienced a complete or nearly complete resolution of baseline signs and symptoms related to primary ABSSSI site (erythema, induration/edema, purulent drainage, fluctuance, pain, tenderness, local increase in heat/warmth) such that no further treatment with antibacterial drugs was needed.
Table 6 summarizes the findings for this endpoint in the mITT and clinically evaluable population in these two ABSSSI trials. Note that there are insufficient historical data to establish the magnitude of drug effect for antibacterial drugs compared with placebo at the post therapy visits. Therefore, comparisons of oritavancin to vancomycin based on clinical success rates at these visits cannot be utilized to establish non-inferiority conclusions.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | |||
|
mITT
mITT population consisted of all randomized patients who received study drug; CE population consisted of all mITT patients who did not have violations of inclusion and exclusion criteria, completed treatment and had investigator assessment at the Follow-Up Visit.
|
378/475 (79.6) | 383/479 (80.0) | -0.4 (-5.5, 4.7) |
| CE | 362/394 (91.9) | 370/397 (93.2) | -1.3 (-5.0,2.3) |
| Trial 2 | |||
| mITT | 416/503 (82.7) | 404/502 (80.5) | 2.2 (-2.6, 7.0) |
| CE | 398/427 (93.2) | 387/408 (94.9) | -1.6 (-4.9,1.6) |
Structured Label Content
Section 42229-5 (42229-5)
Reconstitution: Aseptic technique should be used to reconstitute one KIMYRSA 1,200 mg vial.
- Add 40 mL of sterile water for injection (SWFI) to reconstitute the vial to provide a 30 mg/mL solution.
- Gently swirl the contents to avoid foaming and ensure that all KIMYRSA powder is completely dissolved to form a reconstituted solution.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The reconstituted vial should appear to be a clear, colorless to pink solution, free of visible particles.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
Based on an in vitro hemodialysis study, KIMYRSA is unlikely to be removed from blood by hemodialysis. In the event of overdose, supportive measures should be taken.
11 Description (11 DESCRIPTION)
KIMYRSA (oritavancin) for injection contains oritavancin diphosphate, a semisynthetic lipoglycopeptide antibacterial drug for intravenous infusion.
The chemical name for oritavancin is [4"R]-22-O-(3-amino-2,3,6-trideoxy-3-C-methyl-α-L-arabino-hexopyranosyl)-N3''-[(4'-chloro[1,1'-biphenyl]-4-yl)methyl] vancomycin phosphate [1:2] [salt]. The empirical formula of oritavancin diphosphate is C86H97N10O26Cl3•2H3PO4 and the molecular weight is 1989.09. The chemical structure is represented below:
KIMYRSA for injection is supplied as a sterile white to off-white or pink lyophilized powder in a single-dose clear glass vial that contains 1,200 mg of oritavancin (equivalent to 1331.16 mg oritavancin diphosphate) and the following inactive ingredients: hydroxypropyl-β-cyclodextrin (HPβCD) (2400 mg), mannitol (800 mg) and phosphoric acid or sodium hydroxide (to adjust pH 4.0 to 6.0).
The vial is reconstituted with sterile water for injection and further diluted with 0.9% sodium chloride injection or 5% dextrose in sterile water (D5W) for intravenous infusion. Both the reconstituted solution and the diluted solution for infusion should be a clear, colorless to pink solution, free of visible particles [see Dosage and Administration (2.3)].
12.4 Microbiology
KIMYRSA is a semi-synthetic, lipoglycopeptide antibacterial drug. KIMYRSA exerts a concentration-dependent bactericidal activity in vitro against S. aureus, S. pyogenes, and E. faecalis.
5.6 Osteomyelitis
In Phase 3 ABSSSI clinical trials, more cases of osteomyelitis were reported in the oritavancin treated arm than in the vancomycin-treated arm. Monitor patients treated with KIMYRSA for signs and symptoms of osteomyelitis. If osteomyelitis is suspected or diagnosed, institute appropriate alternate antibacterial therapy [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Safety and effectiveness of KIMYRSA in pediatric patients (younger than 18 years of age) have not been established.
8.5 Geriatric Use
The pooled Phase 3 ABSSSI clinical trials of oritavancin did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
12.6 Immunogenicity
Positive indirect and direct antiglobulin tests (IAT/DAT) have been noted with the administration of KIMYRSA and oritavancin in studies with healthy subjects and patients with ABSSSI. The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of KIMYRSA or other oritavancin products.
In a randomized, open-label, multi-center ABSSSI study, positive IAT/DAT were reported in 9.6% (5/52) of subjects who received ORBACTIV, another oritavancin product, and 2% (1/50) of subjects who received KIMYRSA. Oritavancin-dependent red blood cell (RBC) antibodies were detected when tested in the presence of drug for three subjects in the oritavancin group 14 days after dosing.
In a multiple-dose study with oritavancin in healthy volunteers, 90% (9/10) of subjects had a positive IAT 14 days after the second infusion.
In a healthy volunteer study, 66% (22/32) of subjects receiving KIMYRSA had a positive IAT 15 days after receiving dosing and one subject had a positive DAT at 8 days after dosing.
There were no reports of hemolysis in subjects who had positive IAT/DAT in any of the above referenced studies.
An in vitro study further evaluated 403 stored plasma samples from 12 healthy subjects and from 101 patients with ABSSSI from previously conducted clinical studies at multiple timepoints in healthy subjects and in patients with ABSSSI. In samples collected during the 15- and 8-day windows following single-dose treatment in healthy volunteers (Study MDCO-ORI-15-01) and in patients with ABSSSI (Study ML-ORI-201), respectively, low level (<8) direct agglutinating (IgM) anti-oritavancin antibodies were detected in 19 of 113 (17%) subjects. There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of single-dose oritavancin. No hematologic adverse events were reported for subjects with a positive titer.
There were no identified clinically significant effects of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of KIMYRSA over the treatment duration of 14 days.
Positive IAT may interfere with cross-matching before blood transfusion [see Drug Interactions (7.2)].
2.4 Compatibilities
KIMYRSA solution for administration by 1-hour infusion is compatible with:
- 0.9% sodium chloride injection
- 5% dextrose in sterile water (D5W)
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are also discussed in the Warnings and Precautions section of labeling:
Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
Infusion Related Reactions [see Warnings and Precautions (5.3)]
Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
Osteomyelitis [see Warnings and Precautions (5.6)]
4.2 Hypersensitivity
KIMYRSA is contraindicated in patients with known hypersensitivity to oritavancin products.
5.2 Hypersensitivity
Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. If an acute hypersensitivity reaction occurs during KIMYRSA infusion, discontinue KIMYRSA immediately and institute appropriate supportive care. Before using KIMYRSA, inquire carefully about previous hypersensitivity reactions to glycopeptides. Due to the possibility of cross-sensitivity, carefully monitor for signs of hypersensitivity during KIMYRSA infusion in patients with a history of glycopeptide allergy. In the Phase 3 ABSSSI clinical trials, the median onset of hypersensitivity reactions in oritavancin-treated patients was 1.2 days and the median duration of these reactions was 2.4 days [see Adverse Reactions (6.1)].
8.6 Renal Impairment
No dosage adjustment of KIMYRSA is needed in patients with mild or moderate renal impairment [see Clinical Pharmacology (12.3)]. The pharmacokinetics of KIMYRSA in severe renal impairment have not been evaluated. Oritavancin is not removed from blood by hemodialysis.
12.2 Pharmacodynamics
The antimicrobial activity of oritavancin appears to correlate with the ratio of area under the concentration-time curve to minimal inhibitory concentration (AUC/MIC) based on animal models of infection. The AUC from time zero to 72 hours correlates with antimicrobial activity in both preclinical and clinical studies.
Exposure-response analyses from both preclinical and clinical studies support the treatment of clinically relevant gram-positive microorganisms (e.g. S. aureus and S. pyogenes) causative of ABSSSI with a single 1,200 mg dose of oritavancin.
12.3 Pharmacokinetics
The mean (±SD) pharmacokinetic parameters of oritavancin products (KIMYRSA and oritavancin) in patients with ABSSSI are presented in Table 3.
| Pharmacokinetic Parameter | KIMYRSA (1 hour) Mean (± SD) |
Oritavancin (3 hour) Mean (± SD) |
|---|---|---|
| Cmax, Maximum plasma concentration; AUC0-72, Area under the plasma concentration-time curve from time zero to 72 hours; SD, Standard deviation. | ||
| Cmax (µg/mL) | 148 (±43.0) | 112 (±34.5) |
| AUC0-72 (h∙µg /mL) | 1460 (±511) | 1470 (±582) |
Oritavancin exhibits linear pharmacokinetics at a dose up to 1,200 mg. The mean, population-predicted oritavancin concentration-time profile displays a multi-exponential decline with a long terminal plasma half-life.
2.5 Incompatibilities
Drugs formulated at a basic or neutral pH may be incompatible with KIMYRSA. KIMYRSA should not be administered simultaneously with commonly used intravenous drugs through a common intravenous port. If the same intravenous line is used for sequential infusion of additional medications, the line should be flushed before and after infusion of KIMYRSA with 0.9% sodium chloride injection or D5W.
2.2 Recommended Dosage
The recommended dosage of KIMYRSA is 1,200 mg administered as a single dose by intravenous infusion over 1 hour in patients 18 years and older [see Warnings and Precautions (5.3)].
8.7 Hepatic Impairment
No dosage adjustment of KIMYRSA is needed in patients with mild or moderate hepatic impairment. The pharmacokinetics of KIMYRSA in patients with severe hepatic insufficiency has not been studied [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
KIMYRSA is a lipoglycopeptide antibacterial drug indicated for the treatment of adult patients with acute bacterial skin and skin structure infections caused or suspected to be caused by susceptible isolates of designated Gram-positive microorganisms. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of KIMYRSA and other antibacterial drugs, KIMYRSA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.2)
12.1 Mechanism of Action
Oritavancin is an antibacterial drug [see Microbiology (12.4)].
16.1 How Supplied/storage (16.1 How Supplied/Storage)
KIMYRSA is supplied as a sterile white to off-white or pink lyophilized powder in single-dose clear glass vials containing 1,200 mg of oritavancin. One vial is packaged in a carton to supply a single 1,200 mg dose treatment (NDC 70842-225-01).
KIMYRSA vials should be stored at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP, Controlled Room Temperature (CRT)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Coagulation test interference: Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, and may prolong PT and INR for up to 12 hours and ACT for up to 24 hours. For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, consider a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT. (5.1, 7.2)
- Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of oritavancin products, including KIMYRSA. Discontinue infusion if signs of acute hypersensitivity occur. Carefully monitor patients with known hypersensitivity to glycopeptides. (5.2)
- Infusion Related Reactions: Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA). Stopping or slowing the infusion may result in cessation of these reactions. (5.3)
- Clostridioides difficile-Associated Diarrhea: Evaluate patients if diarrhea occurs. (5.4)
- Concomitant warfarin use: Oritavancin has been shown to artificially prolong PT/INR for up to 12 hours (5.1). Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin. (5.5)
- Osteomyelitis: Institute appropriate alternate antibacterial therapy in patients with confirmed or suspected osteomyelitis. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- There are two oritavancin products (KIMYRSA and ORBACTIV®, another oritavancin product) that have differences in dose strength, duration of infusion and preparation instructions, including reconstitution and dilution instructions and compatible diluents (2.1, 2.2, 2.3, 2.4)
- Administer 1,200 mg of KIMYRSA as a single dose by intravenous infusion over 1 hour. (2.1, 5.3)
- Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in the full prescribing information. (2.1, 2.2, 2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
KIMYRSA is supplied as sterile, white to off-white or pink lyophilized powder containing 1,200 mg of oritavancin (as oritavancin diphosphate) in a single-dose clear glass vial, which must be reconstituted and further diluted prior to intravenous administration.
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial drugs, including oritavancin products (e.g. KIMYRSA), and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.3 Infusion Related Reactions
Infusion related reactions have been reported with the glycopeptide class of antimicrobial agents, including oritavancin products (e.g. KIMYRSA), including flushing of the upper body, urticaria, pruritus and/or rash [see Adverse Reactions (6.1)]. Infusion reactions characterized by chest pain, back pain, chills and tremor have been observed with the use of oritavancin, including after the administration of more than one dose of oritavancin during a single course of therapy.
Stopping or slowing the infusion may result in cessation of these reactions. The safety and effectiveness of more than one dose of KIMYRSA during a single course of therapy have not been established [see Dosage and Administration (2.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of oritavancin products cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of KIMYRSA has been established from adequate and well-controlled trials of another oritavancin product, ORBACTIV (hereinafter referred to as oritavancin), in patients with ABSSSI, and a study of KIMYRSA in patients with ABSSSI.
Oritavancin has been evaluated in two, double-blind, controlled ABSSSI clinical trials, which included 976 adult patients treated with a single 1,200 mg intravenous dose of oritavancin and 983 patients treated with intravenous vancomycin for 7 to 10 days. The median age of patients treated with oritavancin was 45.6 years, ranging between 18 and 89 years of age with 8.8% ≥65 years of age. Patients treated with oritavancin were predominantly male (65.4%), 64.4% were Caucasian, 5.8% were African American, and 28.1% were Asian. Safety was evaluated for up to 60 days after dosing.
In the pooled ABSSSI clinical trials, serious adverse reactions were reported in 57/976 (5.8%) patients treated with oritavancin and 58/983 (5.9%) treated with vancomycin. The most commonly reported serious adverse reaction was cellulitis in both treatment groups: 11/976 (1.1%) in oritavancin and 12/983 (1.2%) in the vancomycin arms, respectively.
The most commonly reported adverse reactions (≥3%) in patients receiving a single 1,200 mg dose of oritavancin in the pooled ABSSSI clinical trials were: headache, nausea, vomiting, limb and subcutaneous abscesses, and diarrhea.
In the pooled ABSSSI clinical trials, oritavancin was discontinued due to adverse reactions in 36/976 (3.7%) of patients; the most common reported reactions leading to discontinuation were cellulitis (4/976, 0.4%) and osteomyelitis (3/976, 0.3%).
Table 1 provides selected adverse reactions occurring in ≥1.5% of patients receiving oritavancin in the pooled ABSSSI clinical trials. There were 540 (55.3%) patients in the oritavancin arm and 559 (56.9%) patients in the vancomycin arm, who reported ≥1 adverse reaction.
| Adverse Reactions | Oritavancin N=976 (%) |
Vancomycin N=983 (%) |
|---|---|---|
| Gastrointestinal disorders | ||
| Diarrhea | 36 (3.7) | 32 (3.4) |
| Nausea | 97 (9.9) | 103 (10.5) |
| Vomiting | 45 (4.6) | 46 (4.7) |
| Nervous system disorders | ||
| Dizziness | 26 (2.7) | 26 (2.6) |
| Headache | 69 (7.1) | 66 (6.7) |
| General disorders and administration | ||
| Infusion site phlebitis | 24 (2.5) | 15 (1.5) |
| Infusion site reaction | 19 (1.9) | 34 (3.5) |
| Infections and infestations | ||
| Abscess (limb and subcutaneous) | 37 (3.8) | 23 (2.3) |
| Investigations | ||
| Alanine aminotransferase increased | 27 (2.8) | 15 (1.5) |
| Aspartate aminotransferase increased | 18 (1.8) | 15 (1.5) |
| Cardiac disorders | ||
| Tachycardia | 24 (2.5) | 11 (1.1) |
The following selected adverse reactions were reported in oritavancin-treated patients at a rate of less than 1.5%:
Blood and lymphatic system disorders: anemia, eosinophilia
General disorders and administration site conditions: infusion site erythema, extravasation, induration, pruritus, rash, edema peripheral
Immune system disorders: hypersensitivity
Infections and infestations: osteomyelitis
Investigations: total bilirubin increased, hyperuricemia
Metabolism and nutrition disorders: hypoglycemia
Musculoskeletal and connective tissue disorders: tenosynovitis, myalgia
Respiratory, thoracic and mediastinal disorders: bronchospasm, wheezing
Skin and subcutaneous tissue disorders: urticaria, angioedema, erythema multiforme, pruritus, leucocytoclastic vasculitis, rash.
KIMYRSA has been evaluated in a randomized, open-label, multi-center ABSSSI study which included 50 adult patients treated with a single 1,200 mg intravenous dose of KIMYRSA administered by intravenous infusion over 1 hour, and 52 patients treated with a single 1,200 mg intravenous dose of oritavancin administered by intravenous infusion over 3 hours.
Selected adverse reactions occurring in ≥2 patients receiving either KIMYRSA or oritavancin in the open-label, multi-center ABSSSI study were diarrhea, nausea, vomiting, hypersensitivity, pruritus, chills, headache and pyrexia.
5.1 Coagulation Test Interference
Oritavancin has been shown to artificially prolong aPTT for up to 120 hours, PT and INR for up to 12 hours, and activated clotting time (ACT) for up to 24 hours following administration of a single 1,200 mg dose by binding to and preventing action of the phospholipid reagents commonly used in laboratory coagulation tests. Oritavancin has also been shown to elevate D-dimer concentrations up to 72 hours after oritavancin administration.
For patients who require aPTT monitoring within 120 hours of KIMYRSA dosing, a non-phospholipid dependent coagulation test such as a Factor Xa (chromogenic) assay or an alternative anticoagulant not requiring aPTT monitoring may be considered [see Contraindications (4.1) and Drug Interactions (7.2)].
Oritavancin has no effect on the coagulation system in vivo.
2.1 Dosage and Administration Overview
There are two oritavancin products (KIMYRSA and ORBACTIV®, another oritavancin product) that:
- Are supplied in different dose strengths of oritavancin [see Dosage Forms and Strengths (3)].
- Have different recommended durations of infusion [see Dosage and Administration (2.2)].
- Have different preparation instructions, including differences in reconstitution, dilution, and compatible diluents [see Dosage and Administration (2.3, 2.4)].
Carefully follow the recommended dosage and dose preparation instructions for KIMYRSA in this prescribing information (PI) [see Dosage and Administration (2.1, 2.2, 2.3, 2.4)]. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
7.1 Effect of Kimyrsa On Cyp Substrates (7.1 Effect of KIMYRSA on CYP Substrates)
A screening drug-drug interaction study indicated that oritavancin is a nonspecific, weak inhibitor (CYP2C9 and CYP2C19) or inducer (CYP3A4 and CYP2D6) of several CYP isoforms [see Clinical Pharmacology (12.3)]. A drug-drug interaction study that assessed the interaction potential of a single 1,200 mg dose of oritavancin on the pharmacokinetics of S-warfarin (CYP2C9 probe substrate) showed no effect of oritavancin on S-warfarin Cmax or AUC.
Avoid administering KIMYRSA concomitantly with drugs that are predominantly metabolized by one of the affected CYP450 enzymes, as co-administration may increase or decrease concentrations of those drugs. Patients should be closely monitored for signs of toxicity or lack of efficacy if they have been given KIMYRSA while on a potentially affected compound (e.g., patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin).
5.7 Development of Drug Resistant Bacteria
Prescribing KIMYRSA in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria [see Patient Counseling Information (17)].
4.1 Intravenous Unfractionated Heparin Sodium
Use of intravenous unfractionated heparin sodium is contraindicated for 120 hours (5 days) after KIMYRSA administration because the activated partial thromboplastin time (aPTT) test results may remain falsely elevated for up to 120 hours (5 days) after KIMYRSA administration [see Warnings and Precautions (5.1) and Drug Interactions (7.2)].
Principal Display Panel 1,200 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 1,200 mg Vial Label)
Rx only
NDC 70842-225-01
Kimyrsa®
(oritavancin) for injection
1,200 mg per vial
Must be reconstituted and
further diluted to a final
volume of 250 mL
For Intravenous Infusion Only
Single-dose Vial
Discard Unused Portion
Principal Display Panel 1,200 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 1,200 mg Vial Carton)
Rx only
NDC 70842-225-01
Kimyrsa®
(oritavancin)
for injection
1,200 mg per vial
Must be reconstituted
and further diluted
to a final volume
of 250 mL
For Intravenous Infusion Only
Single-dose Vial
Discard Unused Portion
2.3 Preparation of Kimyrsa for Intravenous Infusion (2.3 Preparation of KIMYRSA for Intravenous Infusion)
There are two oritavancin products (KIMYRSA and ORBACTIV, another oritavancin product) that have differences in dose strengths, duration of infusion, reconstitution and dilution instructions, and compatible diluents. Carefully follow the reconstitution, and dilution instructions with the appropriate compatible diluent for KIMYRSA specified in this prescribing information. Refer to the ORBACTIV prescribing information for relevant information of the other oritavancin product.
KIMYRSA is intended for intravenous infusion, only after reconstitution and dilution.
One KIMYRSA 1,200 mg single-dose vial needs to be reconstituted and diluted to prepare a single 1,200 mg intravenous dose.
1.1 Acute Bacterial Skin and Skin Structure Infections
KIMYRSA® is indicated for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following Gram-positive microorganisms:
Staphylococcus aureus (including methicillin-susceptible and methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus anginosus group (includes S. anginosus, S. intermedius, and S. constellatus), and Enterococcus faecalis (vancomycin-susceptible isolates only).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals have not been conducted to determine the carcinogenic potential of oritavancin.
No mutagenic or clastogenic potential of oritavancin was found in a battery of tests, including an Ames assay, in vitro chromosome aberration assay in Chinese hamster ovary cells, in vitro forward mutation assay in mouse lymphoma cells and an in vivo mouse micronucleus assay.
Oritavancin did not affect the fertility or reproductive performance of male rats (exposed to daily doses up to 30 mg/kg for at least 4 weeks) and female rats (exposed to daily doses up to 30 mg/kg for at least 2 weeks prior to mating). Those daily doses would be equivalent to a human dose of 300 mg, or 25% of clinical dose. Higher doses were not evaluated in nonclinical fertility studies.
5.5 Potential Risk of Bleeding With Concomitant Use of Warfarin (5.5 Potential Risk of Bleeding with Concomitant Use of Warfarin)
Oritavancin has been shown to artificially prolong prothrombin time (PT) and international normalized ratio (INR) for up to 12 hours, making the monitoring of the anticoagulation effect of warfarin unreliable up to 12 hours after an oritavancin dose [see Warnings and Precautions (5.1)].
Patients should be monitored for bleeding if concomitantly receiving KIMYRSA and warfarin [see Drug Interactions (7.1)].
14.1 Acute Bacterial Skin and Skin Structure Infections (absssi) (14.1 Acute Bacterial Skin and Skin Structure Infections (ABSSSI))
A total of 1987 adults with clinically documented ABSSSI suspected or proven to be due to Gram-positive pathogens were randomized into two identically designed, randomized, double-blind, multi-center, multinational, non-inferiority trials (Trial 1 and Trial 2) comparing a single 1,200 mg intravenous dose of oritavancin to intravenous vancomycin (1 g or 15 mg/kg every 12 hours) for 7 to 10 days. The primary analysis population (modified intent to treat, mITT) included all randomized patients who received any study drug. Patients could receive concomitant aztreonam or metronidazole for suspected Gram-negative and anaerobic infection, respectively. Patient demographic and baseline characteristics were balanced between treatment groups. Approximately 64% of patients were Caucasian and 65% were males. The mean age was 45 years and the mean body mass index was 27 kg/m2. Across both trials, approximately 60% of patients were enrolled from the United States and 27% of patients from Asia. A history of diabetes was present in 14% of patients. The types of ABSSSI across both trials included cellulitis/erysipelas (40%), wound infection (29%), and major cutaneous abscesses (31%). Median infection area at baseline across both trials was 266.6 cm2. The primary endpoint in both trials was early clinical response (responder), defined as cessation of spread or reduction in size of baseline lesion, absence of fever, and no rescue antibacterial drug at 48 to 72 hours after initiation of therapy.
Table 4 provides the efficacy results for the primary endpoint in Trial 1 and Trial 2 in the primary analysis population.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | 391/475 (82.3) | 378/479 (78.9) | 3.4 (-1.6, 8.4) |
| Trial 2 | 403/503 (80.1) | 416/502 (82.9) | -2.7 (-7.5, 2.0) |
A key secondary endpoint in these two ABSSSI trials evaluated the percentage of patients achieving a 20% or greater reduction in lesion area from baseline at 48-72 hours after initiation of therapy. Table 5 summarizes the findings for this endpoint in the two ABSSSI trials.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | 413/475 (86.9) | 397/479 (82.9) | 4.1 (-0.5, 8.6) |
| Trial 2 | 432/503 (85.9) | 428/502 (85.3) | 0.6 (-3.7, 5.0) |
Another secondary efficacy endpoint in the two trials was investigator-assessed clinical success at post therapy evaluation at Day 14 to 24 (7 to 14 days from end of blinded therapy). A patient was categorized as a clinical success if the patient experienced a complete or nearly complete resolution of baseline signs and symptoms related to primary ABSSSI site (erythema, induration/edema, purulent drainage, fluctuance, pain, tenderness, local increase in heat/warmth) such that no further treatment with antibacterial drugs was needed.
Table 6 summarizes the findings for this endpoint in the mITT and clinically evaluable population in these two ABSSSI trials. Note that there are insufficient historical data to establish the magnitude of drug effect for antibacterial drugs compared with placebo at the post therapy visits. Therefore, comparisons of oritavancin to vancomycin based on clinical success rates at these visits cannot be utilized to establish non-inferiority conclusions.
| Oritavancin n /N (%) |
Vancomycin n /N (%) |
Difference (95% CI) 95% CI based on the Normal approximation to Binomial distribution.
|
|
|---|---|---|---|
| Trial 1 | |||
|
mITT
mITT population consisted of all randomized patients who received study drug; CE population consisted of all mITT patients who did not have violations of inclusion and exclusion criteria, completed treatment and had investigator assessment at the Follow-Up Visit.
|
378/475 (79.6) | 383/479 (80.0) | -0.4 (-5.5, 4.7) |
| CE | 362/394 (91.9) | 370/397 (93.2) | -1.3 (-5.0,2.3) |
| Trial 2 | |||
| mITT | 416/503 (82.7) | 404/502 (80.5) | 2.2 (-2.6, 7.0) |
| CE | 398/427 (93.2) | 387/408 (94.9) | -1.6 (-4.9,1.6) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:25.939540 · Updated: 2026-03-14T22:40:22.461215