These Highlights Do Not Include All The Information Needed To Use Itovebi Safely And Effectively. See Full Prescribing Information For Itovebi.
5de59f5b-e5db-410f-b692-658686ef4107
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Hyperglycemia ( 5.1 ) 09/2025
Indications and Usage
ITOVEBI, in combination with palbociclib and fulvestrant, is indicated for the treatment of adults with endocrine-resistant, PIK3CA -mutated, hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy [see Clinical Studies (14.1) ] .
Dosage and Administration
Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimen. ( 2.1 ) Recommended dosage: 9 mg orally once daily with or without food. ( 2.3 ) See Full Prescribing Information for dosage modifications of ITOVEBI due to adverse reactions. ( 2.4 ) Reduce the starting dose in patients with moderate renal impairment. ( 2.5 )
Warnings and Precautions
Hyperglycemia : ITOVEBI can cause severe or fatal hyperglycemia including ketoacidosis. Before initiating treatment with ITOVEBI, test fasting plasma glucose (FPG), HbA1c, and optimize blood glucose. Initiate or optimize anti-hyperglycemic medications as clinically indicated. Interrupt, reduce dose, or discontinue ITOVEBI if severe hyperglycemia occurs. ( 2.4 , 5.1 ) Stomatitis : ITOVEBI can cause severe stomatitis. Consider treating with a corticosteroid-containing mouthwash if stomatitis occurs. Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity. ( 2.4 , 5.2 ) Diarrhea : ITOVEBI can cause diarrhea, which may be severe, and result in dehydration and acute kidney injury. Advise patients to start anti-diarrheal treatment, increase oral fluids, and notify their healthcare provider if severe diarrhea occurs. Interrupt, reduce dose, or discontinue ITOVEBI if severe diarrhea occurs. ( 2.4 , 5.3 ) Embryo-Fetal Toxicity : ITOVEBI can cause fetal harm. Advise patients of potential risk to a fetus and to use effective non-hormonal contraception. ( 5.4 , 8.1 , 8.3 ) Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
Contraindications
None.
Adverse Reactions
The recommended dose reduction levels of ITOVEBI for adverse reactions are listed in Table 1 . Permanently discontinue ITOVEBI if patients are unable to tolerate the second dose reduction. Table 1: Dose Reduction for Adverse Reactions Dose Level Dose and Schedule Recommended starting dose 9 mg daily First dose reduction 6 mg daily Second dose reduction 3 mg daily The recommended dosage modifications of ITOVEBI for adverse reactions are summarized in Table 2 . Table 2: Recommended Dosage Modifications for Adverse Reactions Adverse Reaction Severity Dosage Modification Hyperglycemia Before initiating treatment with ITOVEBI, test FPG or FBG, and HbA 1C levels, and optimize plasma/blood glucose levels in all patients. After initiating treatment with ITOVEBI, monitor FPG or FBG levels based on the recommended schedule, and as clinically indicated [see Warnings and Precautions (5.1) ]. [see Warnings and Precautions (5.1) ] Fasting glucose levels (FPG or FBG) > ULN to 160 mg/dL (> ULN – 8.9 mmol/L) No adjustment of ITOVEBI required. Consider dietary modifications and ensure adequate hydration. Initiate or intensify oral anti-hyperglycemic medications for patients with risk factors for hyperglycemia. Fasting glucose levels > 160 to 250 mg/dL (> 8.9 – 13.9 mmol/L) Withhold ITOVEBI until FPG or FBG ≤ 160 mg/dL (≤ 8.9 mmol/L). Initiate or intensify anti-hyperglycemic medications. Resume ITOVEBI at the same dose level. If FPG or FBG persists > 200 – 250 mg/dL (> 11.1 – 13.9 mmol/L) for 7 days under appropriate anti-hyperglycemic treatment, consider consultation with a healthcare professional experienced in the treatment of hyperglycemia. Fasting glucose levels > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) Withhold ITOVEBI. Initiate or intensify anti-hyperglycemic medications. Administer appropriate hydration if required. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L) within 7 days, resume ITOVEBI at the same dose level. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L) in ≥ 8 days, resume ITOVEBI at one lower dose level. If FPG or FBG > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) recurs within 30 days, withhold ITOVEBI until FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L). Resume ITOVEBI at one lower dose level. Fasting glucose levels > 500 mg/dL (> 27.8 mmol/L) Withhold ITOVEBI. Initiate or intensify anti-hyperglycemic medications. Assess for volume depletion and ketosis and administer appropriate hydration. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L), resume ITOVEBI at one lower dose level. If FPG or FBG > 500 mg/dL (> 27.8 mmol/L) recurs within 30 days, permanently discontinue ITOVEBI. Stomatitis [see Warnings and Precautions (5.2) ] Grade 1 Based on CTCAE version 5.0. No adjustment of ITOVEBI required. Initiate or intensify appropriate medical therapy (e.g., corticosteroid-containing mouthwash) as clinically indicated. Grade 2 Withhold ITOVEBI until recovery to Grade ≤ 1. Initiate or intensify appropriate medical therapy. Resume ITOVEBI at the same dose level. For recurrent Grade 2 stomatitis, withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Grade 3 Withhold ITOVEBI until recovery to Grade ≤ 1. Initiate or intensify appropriate medical therapy. Resume ITOVEBI at one lower dose level. Grade 4 Permanently discontinue ITOVEBI. Diarrhea [see Warnings and Precautions (5.3) ] Grade 1 No adjustment of ITOVEBI required. Initiate appropriate medical therapy and monitor as clinically indicated. Grade 2 Withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at the same dose level. Initiate or intensify appropriate medical therapy and monitor as clinically indicated. For recurrent Grade 2 diarrhea, withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Grade 3 Withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Initiate or intensify appropriate medical therapy and monitor as clinically indicated. Grade 4 Permanently discontinue ITOVEBI. Hematologic Toxicities [see Adverse Reactions (6.1) ] Grade 1, 2, or 3 No adjustment of ITOVEBI required. Monitor complete blood count and for signs or symptoms of hematologic toxicities as clinically indicated. Grade 4 Withhold ITOVEBI until recovery to Grade ≤ 2. Resume ITOVEBI at the same dose level or reduce to one lower dose level as clinically indicated. Other Adverse Reactions [see Adverse Reactions (6.1) ] Grade 1 No adjustment of ITOVEBI required. Grade 2 Consider withholding ITOVEBI, if clinically indicated, until recovery to Grade ≤ 1. Resume ITOVEBI at the same dose level. Grade 3 (first event) Withhold ITOVEBI until recovery to Grade ≤ 1. Resume ITOVEBI at the same dose level or one lower dose level based on clinical evaluation. Grade 3 (recurrent) Withhold ITOVEBI until recovery to Grade ≤ 1. Resume ITOVEBI at one lower dose level. Grade 4 Permanently discontinue ITOVEBI.
Storage and Handling
ITOVEBI is supplied in the following strengths and package configurations: Package Configuration Tablet Strength NDC Tablet Description Bottle of 28 tablets 3 mg 50242-084-08 Red and round convex-shaped with an "INA 3" debossing on one side Bottle of 28 tablets 9 mg 50242-079-08 Pink and oval-shaped with an "INA 9" debossing on one side
How Supplied
ITOVEBI is supplied in the following strengths and package configurations: Package Configuration Tablet Strength NDC Tablet Description Bottle of 28 tablets 3 mg 50242-084-08 Red and round convex-shaped with an "INA 3" debossing on one side Bottle of 28 tablets 9 mg 50242-079-08 Pink and oval-shaped with an "INA 9" debossing on one side
Medication Information
Warnings and Precautions
Hyperglycemia : ITOVEBI can cause severe or fatal hyperglycemia including ketoacidosis. Before initiating treatment with ITOVEBI, test fasting plasma glucose (FPG), HbA1c, and optimize blood glucose. Initiate or optimize anti-hyperglycemic medications as clinically indicated. Interrupt, reduce dose, or discontinue ITOVEBI if severe hyperglycemia occurs. ( 2.4 , 5.1 ) Stomatitis : ITOVEBI can cause severe stomatitis. Consider treating with a corticosteroid-containing mouthwash if stomatitis occurs. Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity. ( 2.4 , 5.2 ) Diarrhea : ITOVEBI can cause diarrhea, which may be severe, and result in dehydration and acute kidney injury. Advise patients to start anti-diarrheal treatment, increase oral fluids, and notify their healthcare provider if severe diarrhea occurs. Interrupt, reduce dose, or discontinue ITOVEBI if severe diarrhea occurs. ( 2.4 , 5.3 ) Embryo-Fetal Toxicity : ITOVEBI can cause fetal harm. Advise patients of potential risk to a fetus and to use effective non-hormonal contraception. ( 5.4 , 8.1 , 8.3 ) Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
Indications and Usage
ITOVEBI, in combination with palbociclib and fulvestrant, is indicated for the treatment of adults with endocrine-resistant, PIK3CA -mutated, hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy [see Clinical Studies (14.1) ] .
Dosage and Administration
Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimen. ( 2.1 ) Recommended dosage: 9 mg orally once daily with or without food. ( 2.3 ) See Full Prescribing Information for dosage modifications of ITOVEBI due to adverse reactions. ( 2.4 ) Reduce the starting dose in patients with moderate renal impairment. ( 2.5 )
Contraindications
None.
Adverse Reactions
The recommended dose reduction levels of ITOVEBI for adverse reactions are listed in Table 1 . Permanently discontinue ITOVEBI if patients are unable to tolerate the second dose reduction. Table 1: Dose Reduction for Adverse Reactions Dose Level Dose and Schedule Recommended starting dose 9 mg daily First dose reduction 6 mg daily Second dose reduction 3 mg daily The recommended dosage modifications of ITOVEBI for adverse reactions are summarized in Table 2 . Table 2: Recommended Dosage Modifications for Adverse Reactions Adverse Reaction Severity Dosage Modification Hyperglycemia Before initiating treatment with ITOVEBI, test FPG or FBG, and HbA 1C levels, and optimize plasma/blood glucose levels in all patients. After initiating treatment with ITOVEBI, monitor FPG or FBG levels based on the recommended schedule, and as clinically indicated [see Warnings and Precautions (5.1) ]. [see Warnings and Precautions (5.1) ] Fasting glucose levels (FPG or FBG) > ULN to 160 mg/dL (> ULN – 8.9 mmol/L) No adjustment of ITOVEBI required. Consider dietary modifications and ensure adequate hydration. Initiate or intensify oral anti-hyperglycemic medications for patients with risk factors for hyperglycemia. Fasting glucose levels > 160 to 250 mg/dL (> 8.9 – 13.9 mmol/L) Withhold ITOVEBI until FPG or FBG ≤ 160 mg/dL (≤ 8.9 mmol/L). Initiate or intensify anti-hyperglycemic medications. Resume ITOVEBI at the same dose level. If FPG or FBG persists > 200 – 250 mg/dL (> 11.1 – 13.9 mmol/L) for 7 days under appropriate anti-hyperglycemic treatment, consider consultation with a healthcare professional experienced in the treatment of hyperglycemia. Fasting glucose levels > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) Withhold ITOVEBI. Initiate or intensify anti-hyperglycemic medications. Administer appropriate hydration if required. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L) within 7 days, resume ITOVEBI at the same dose level. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L) in ≥ 8 days, resume ITOVEBI at one lower dose level. If FPG or FBG > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) recurs within 30 days, withhold ITOVEBI until FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L). Resume ITOVEBI at one lower dose level. Fasting glucose levels > 500 mg/dL (> 27.8 mmol/L) Withhold ITOVEBI. Initiate or intensify anti-hyperglycemic medications. Assess for volume depletion and ketosis and administer appropriate hydration. If FPG or FBG decreases to ≤ 160 mg/dL (≤ 8.9 mmol/L), resume ITOVEBI at one lower dose level. If FPG or FBG > 500 mg/dL (> 27.8 mmol/L) recurs within 30 days, permanently discontinue ITOVEBI. Stomatitis [see Warnings and Precautions (5.2) ] Grade 1 Based on CTCAE version 5.0. No adjustment of ITOVEBI required. Initiate or intensify appropriate medical therapy (e.g., corticosteroid-containing mouthwash) as clinically indicated. Grade 2 Withhold ITOVEBI until recovery to Grade ≤ 1. Initiate or intensify appropriate medical therapy. Resume ITOVEBI at the same dose level. For recurrent Grade 2 stomatitis, withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Grade 3 Withhold ITOVEBI until recovery to Grade ≤ 1. Initiate or intensify appropriate medical therapy. Resume ITOVEBI at one lower dose level. Grade 4 Permanently discontinue ITOVEBI. Diarrhea [see Warnings and Precautions (5.3) ] Grade 1 No adjustment of ITOVEBI required. Initiate appropriate medical therapy and monitor as clinically indicated. Grade 2 Withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at the same dose level. Initiate or intensify appropriate medical therapy and monitor as clinically indicated. For recurrent Grade 2 diarrhea, withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Grade 3 Withhold ITOVEBI until recovery to Grade ≤ 1, then resume ITOVEBI at one lower dose level. Initiate or intensify appropriate medical therapy and monitor as clinically indicated. Grade 4 Permanently discontinue ITOVEBI. Hematologic Toxicities [see Adverse Reactions (6.1) ] Grade 1, 2, or 3 No adjustment of ITOVEBI required. Monitor complete blood count and for signs or symptoms of hematologic toxicities as clinically indicated. Grade 4 Withhold ITOVEBI until recovery to Grade ≤ 2. Resume ITOVEBI at the same dose level or reduce to one lower dose level as clinically indicated. Other Adverse Reactions [see Adverse Reactions (6.1) ] Grade 1 No adjustment of ITOVEBI required. Grade 2 Consider withholding ITOVEBI, if clinically indicated, until recovery to Grade ≤ 1. Resume ITOVEBI at the same dose level. Grade 3 (first event) Withhold ITOVEBI until recovery to Grade ≤ 1. Resume ITOVEBI at the same dose level or one lower dose level based on clinical evaluation. Grade 3 (recurrent) Withhold ITOVEBI until recovery to Grade ≤ 1. Resume ITOVEBI at one lower dose level. Grade 4 Permanently discontinue ITOVEBI.
Storage and Handling
ITOVEBI is supplied in the following strengths and package configurations: Package Configuration Tablet Strength NDC Tablet Description Bottle of 28 tablets 3 mg 50242-084-08 Red and round convex-shaped with an "INA 3" debossing on one side Bottle of 28 tablets 9 mg 50242-079-08 Pink and oval-shaped with an "INA 9" debossing on one side
How Supplied
ITOVEBI is supplied in the following strengths and package configurations: Package Configuration Tablet Strength NDC Tablet Description Bottle of 28 tablets 3 mg 50242-084-08 Red and round convex-shaped with an "INA 3" debossing on one side Bottle of 28 tablets 9 mg 50242-079-08 Pink and oval-shaped with an "INA 9" debossing on one side
Description
Warnings and Precautions, Hyperglycemia ( 5.1 ) 09/2025
Section 42229-5
Locally Advanced or Metastatic Breast Cancer
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 12/2025 | |
|
PATIENT INFORMATION
ITOVEBI® (eye-TOVE-bee) (inavolisib) tablets, for oral use |
||
| Important: ITOVEBI is used with palbociclib and fulvestrant. You should also read the Patient Information that comes with palbociclib and fulvestrant. | ||
|
What is ITOVEBI?
ITOVEBI is a prescription medicine used in combination with the medicines palbociclib and fulvestrant to treat adults who have hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer that has an abnormal phosphatidylinositol-3-kinase catalytic subunit alpha (PIK3CA) gene, and has spread to nearby tissue or lymph nodes (locally advanced), or to other parts of the body (metastatic), and has come back after hormone (endocrine) therapy. Your healthcare provider will test your cancer for abnormal PIK3CA genes to make sure that ITOVEBI is right for you. It is not known if ITOVEBI is safe and effective in children. |
||
Before you take ITOVEBI, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take ITOVEBI?
|
||
|
What are the possible side effects of ITOVEBI? ITOVEBI may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
The most common side effects and abnormal blood test results of ITOVEBI when used in combination with palbociclib and fulvestrant include: |
||
|
|
|
| ITOVEBI may affect fertility in males and in females who are able to become pregnant. Talk to your healthcare provider if this is a concern for you. These are not all of the possible side effects of ITOVEBI. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ITOVEBI?
|
||
|
General information about the safe and effective use of ITOVEBI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ITOVEBI for a condition for which it was not prescribed. Do not give ITOVEBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ITOVEBI that is written for health professionals. |
||
|
What are the ingredients in ITOVEBI?
Active ingredient: inavolisib Inactive ingredients: lactose, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The tablet film-coating contains iron oxide red, and iron oxide yellow (in the 9 mg tablet only), macrogol/polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Distributed by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 ITOVEBI is a registered trademark of Genentech, Inc. For more information, go to www.ITOVEBI.com or call 1-877-436-3683. |
Section 43683-2
| Warnings and Precautions, Hyperglycemia (5.1) | 09/2025 |
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F) [see USP Controlled Room Temperature]. Store in the original container and keep the bottle tightly closed in order to protect from moisture.
5.3 Diarrhea
Severe diarrhea, including dehydration and acute kidney injury, can occur in patients treated with ITOVEBI.
Diarrhea occurred in 48% of patients treated with ITOVEBI in combination with palbociclib and fulvestrant, including Grade 3 events in 3.7% of patients. The median time to first onset was 15 days (range: 2 to 602 days).
Diarrhea led to dose interruptions in 7% of patients, and dose reductions in 1.2% of patients. Anti-diarrheal medicines were used in 28% (46/162) of patients who received ITOVEBI in combination with palbociclib and fulvestrant to manage symptoms.
Monitor patients for signs and symptoms of diarrhea. Advise patients to increase oral fluids and start anti-diarrheal treatment at the first sign of diarrhea while taking ITOVEBI. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity [see Dosage and Administration (2.4)].
11 Description
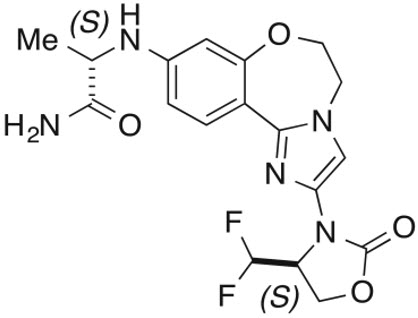
ITOVEBI contains inavolisib, a kinase inhibitor. The chemical name of inavolisib is (2S)-2-[[2-[(4S)-4-(difluoromethyl)-2-oxo-oxazolidin-3-yl]-5,6-dihydroimidazo[1,2-d][1,4]benzoxazepin-9-yl]amino]propanamide. Inavolisib is a white to off-white, greyish pink, greyish orange, or greyish yellow powder or powder with lumps. Inavolisib demonstrates pH-dependent aqueous solubility; the greatest solubility is at low pH, and solubility decreases with increasing pH. The molecular formula for inavolisib is C18H19F2N5O4 and the molecular weight is 407.37 g/mol. The chemical structure of inavolisib is shown below:
ITOVEBI film-coated tablets are supplied for oral administration with two strengths that contain 3 mg and 9 mg of inavolisib. The tablets also contain lactose, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The film-coating contains the following inactive ingredients: polyvinyl alcohol (partially hydrolyzed), titanium dioxide, macrogol/polyethylene glycol, talc, iron oxide red, and iron oxide yellow (in the 9 mg tablet only).
5.2 Stomatitis
Severe stomatitis can occur in patients treated with ITOVEBI.
Stomatitis occurred in 51% of patients treated with ITOVEBI in combination with palbociclib and fulvestrant, including Grade 3 events in 6% of patients. The median time to first onset was 13 days (range: 1 to 610 days).
Stomatitis led to dose interruption in 10%, to dose reduction in 3.7%, and to discontinuation of ITOVEBI in 0.6% of patients.
In patients who received ITOVEBI in combination with palbociclib and fulvestrant, 38% used a mouthwash containing corticosteroid for management or prophylaxis of stomatitis.
Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity [see Dosage and Administration (2.4)].
5.1 Hyperglycemia
Severe or fatal hyperglycemia, including ketoacidosis, can occur in patients treated with ITOVEBI. Ketoacidosis with a fatal outcome has occurred in the postmarketing setting.
Increased fasting glucose occurred in 85% of patients treated with ITOVEBI, including 22% of patients with Grade 2 (FPG > 160 to 250 mg/dL), 12% with Grade 3 (FPG > 250 to 500 mg/dL), and 0.6% with Grade 4 (FPG > 500 mg/dL) events.
In INAVO120, 46% (74/162) of patients who received ITOVEBI were treated with oral anti-hyperglycemic medications and 7% (11/162) were treated with insulin to manage increased fasting glucose. In patients who experienced increased fasting glucose of > 160 mg/dL, 96% (52/54) had an improvement in fasting glucose of at least one grade level with a median time to improvement of 8 days (range: 2 to 43 days).
Among patients with hyperglycemia, the median time to first onset was 7 days (range: 2 to 955 days). Hyperglycemia led to dose interruption in 28%, to dose reduction in 2.5%, and to discontinuation of ITOVEBI in 1.2% of patients.
The safety of ITOVEBI in patients with Type 1 diabetes mellitus, or Type 2 diabetes mellitus requiring ongoing anti-hyperglycemic treatment have not been studied.
Before initiating treatment with ITOVEBI, test fasting glucose levels (FPG or FBG), HbA1C levels, and optimize fasting glucose.
After initiating treatment with ITOVEBI, or in patients who experience hyperglycemia after initiating treatment with ITOVEBI, monitor or self-monitor fasting glucose levels once every 3 days for the first week (Day 1 to 7), then once every week for the next 3 weeks (Day 8 to 28), then once every 2 weeks for the next 8 weeks, then once every 4 weeks thereafter, and as clinically indicated. Monitor HbA1C every 3 months and as clinically indicated.
Manage hyperglycemia with anti-hyperglycemic medications as clinically indicated. During treatment with anti-hyperglycemic medication, continue monitoring fasting glucose levels. Patients with a history of well-controlled Type 2 diabetes mellitus may require intensified anti-hyperglycemic treatment and close monitoring of fasting glucose levels.
Consider consultation with a healthcare professional experienced in the treatment of hyperglycemia, and initiation of fasting glucose monitoring at home for patients who have risk factors for hyperglycemia or who experience hyperglycemia. Advise patients of the signs and symptoms of hyperglycemia and counsel patients on lifestyle changes.
Based on the severity of the hyperglycemia, ITOVEBI may require dose interruption, reduction, or discontinuation [see Dosage and Administration (2.4)].
8.4 Pediatric Use
The safety and efficacy of ITOVEBI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 162 patients who received ITOVEBI in INAVO120, 15% were ≥ 65 years of age, and 3% were ≥ 75 years of age.
Dosage modifications or interruptions of ITOVEBI due to adverse reactions occurred at a higher incidence for patients ≥ 65 years of age compared to younger patients (79% versus 68%, respectively).
Clinical studies of ITOVEBI did not include sufficient numbers of patients ≥ 65 years of age to determine whether they respond differently from younger patients.
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hyperglycemia [see Warnings and Precautions (5.1)]
- Stomatitis [see Warnings and Precautions (5.2)]
- Diarrhea [see Warnings and Precautions (5.3)]
8.6 Renal Impairment
Reduce the dosage in patients with moderate renal impairment (eGFR 30 to < 60 mL/min based on CKD-EPI) [see Dosage and Administration (2.5)]. No dosage modification is recommended in patients with mild renal impairment (eGFR 60 to < 90 mL/min). ITOVEBI has not been evaluated in patients with severe renal impairment (eGFR < 30 mL/min).
12.3 Pharmacokinetics
Inavolisib pharmacokinetics are presented as geometric mean (geometric coefficient of variation [geo CV]%) following administration of the approved recommended dosage unless otherwise specified. The inavolisib steady-state AUC is 1,010 h*ng/mL (25%) and Cmax is 69 ng/mL (27%). Steady-state concentrations are predicted to be attained by day 5.
Inavolisib accumulation is approximately 2-fold.
Inavolisib steady-state AUC is proportional with dose from 6 to 12 mg (0.7 to 1.3 times the approved recommended dosage).
2.1 Patient Selection
Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimens [see Clinical Studies (14.1)].
Information on FDA-approved tests for the detection of PIK3CA mutations in breast cancer is available at: http://www.fda.gov/companiondiagnostics.
2.3 Recommended Dosage
The recommended dosage of ITOVEBI is 9 mg taken orally once daily, with or without food, until disease progression or unacceptable toxicity.
Advise patients to take ITOVEBI at approximately the same time each day.
Swallow ITOVEBI tablet(s) whole. Do not chew, crush, or split prior to swallowing.
If a patient misses a dose, instruct the patient to take the missed dose as soon as possible within 9 hours. After more than 9 hours, instruct the patient to skip the dose and take the next dose at the scheduled time.
If a patient vomits a dose, instruct patients not to take an additional dose on that day and resume the usual dosing schedule the next day.
Administer ITOVEBI in combination with palbociclib and fulvestrant. The recommended dosage of palbociclib is 125 mg taken orally once daily for 21 consecutive days followed by 7 days off treatment to comprise a cycle of 28 days. Refer to the Full Prescribing Information for palbociclib and fulvestrant for dosing information.
For premenopausal and perimenopausal women, administer a luteinizing hormone-releasing hormone (LHRH) agonist in accordance with local clinical practice.
For men, consider administering an LHRH agonist in accordance with local clinical practice.
1 Indications and Usage
ITOVEBI, in combination with palbociclib and fulvestrant, is indicated for the treatment of adults with endocrine-resistant, PIK3CA-mutated, hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy [see Clinical Studies (14.1)].
12.1 Mechanism of Action
Inavolisib is an inhibitor of phosphatidylinositol 3-kinase (PI3K) with inhibitory activity predominantly against PI3Kα. In vitro, inavolisib induced the degradation of mutated PI3K catalytic alpha subunit p110α (encoded by the PIK3CA gene), inhibited phosphorylation of the downstream target AKT, reduced cellular proliferation, and induced apoptosis in PIK3CA-mutated breast cancer cell lines. In vivo, inavolisib reduced tumor growth in PIK3CA-mutated, estrogen receptor-positive, breast cancer xenograft models. The combination of inavolisib with palbociclib and fulvestrant increased tumor growth inhibition compared to each treatment alone or the doublet combinations.
5.4 Embryo Fetal Toxicity
Based on findings in animals and its mechanism of action, ITOVEBI can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. In an animal reproduction study, oral administration of inavolisib to pregnant rats during the period of organogenesis caused adverse developmental outcomes, including embryo-fetal mortality, structural abnormalities, and alterations to growth at maternal exposures approximately equivalent to the human exposure at the recommended dose of 9 mg/day based on area under the curve (AUC).
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective non-hormonal contraception during treatment with ITOVEBI and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ITOVEBI and for 1 week after the last dose [see Use in Specific Populations (8.1 and 8.3)].
ITOVEBI is used in combination with palbociclib and fulvestrant. Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
5 Warnings and Precautions
- Hyperglycemia: ITOVEBI can cause severe or fatal hyperglycemia including ketoacidosis. Before initiating treatment with ITOVEBI, test fasting plasma glucose (FPG), HbA1c, and optimize blood glucose. Initiate or optimize anti-hyperglycemic medications as clinically indicated. Interrupt, reduce dose, or discontinue ITOVEBI if severe hyperglycemia occurs. (2.4, 5.1)
- Stomatitis: ITOVEBI can cause severe stomatitis. Consider treating with a corticosteroid-containing mouthwash if stomatitis occurs. Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity. (2.4, 5.2)
- Diarrhea: ITOVEBI can cause diarrhea, which may be severe, and result in dehydration and acute kidney injury. Advise patients to start anti-diarrheal treatment, increase oral fluids, and notify their healthcare provider if severe diarrhea occurs. Interrupt, reduce dose, or discontinue ITOVEBI if severe diarrhea occurs. (2.4, 5.3)
-
Embryo-Fetal Toxicity: ITOVEBI can cause fetal harm. Advise patients of potential risk to a fetus and to use effective non-hormonal contraception. (5.4, 8.1, 8.3)
Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
2 Dosage and Administration
- Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimen. (2.1)
- Recommended dosage: 9 mg orally once daily with or without food. (2.3)
- See Full Prescribing Information for dosage modifications of ITOVEBI due to adverse reactions. (2.4)
- Reduce the starting dose in patients with moderate renal impairment. (2.5)
3 Dosage Forms and Strengths
Tablets:
- 3 mg: red and round convex-shaped with an "INA 3" debossing on one side.
- 9 mg: pink and oval-shaped with an "INA 9" debossing on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ITOVEBI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition Disorders: Ketoacidosis
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
ITOVEBI is supplied in the following strengths and package configurations:
| Package Configuration | Tablet Strength | NDC | Tablet Description |
|---|---|---|---|
| Bottle of 28 tablets | 3 mg | 50242-084-08 | Red and round convex-shaped with an "INA 3" debossing on one side |
| Bottle of 28 tablets | 9 mg | 50242-079-08 | Pink and oval-shaped with an "INA 9" debossing on one side |
13.2 Animal Toxicology And/or Pharmacology
Lens degeneration, characterized by lens fiber swelling, separation of lens fibers, and/or accumulation of subcapsular proteinaceous material, was observed in rats at an oral inavolisib dose of 10 mg/kg/day (6.3 times the human exposure at the recommended dose of 9 mg/day based on AUC). In dogs, lens fiber swelling and lens cortex vacuolation were observed at oral inavolisib doses ≥ 0.3 mg/kg/day (≥ 0.5 times the human exposure at the recommended dose based on AUC) and ≥ 1 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose based on AUC), respectively. Lens degeneration was present in rats following a 4-week recovery period but was not present in dogs following a 12-week recovery period.
2.4 Dosage Modifications for Adverse Reactions
The recommended dose reduction levels of ITOVEBI for adverse reactions are listed in Table 1. Permanently discontinue ITOVEBI if patients are unable to tolerate the second dose reduction.
| Dose Level | Dose and Schedule |
|---|---|
| Recommended starting dose | 9 mg daily |
| First dose reduction | 6 mg daily |
| Second dose reduction | 3 mg daily |
The recommended dosage modifications of ITOVEBI for adverse reactions are summarized in Table 2.
| Adverse Reaction | Severity | Dosage Modification |
|---|---|---|
|
Hyperglycemia Before initiating treatment with ITOVEBI, test FPG or FBG, and HbA1C levels, and optimize plasma/blood glucose levels in all patients. After initiating treatment with ITOVEBI, monitor FPG or FBG levels based on the recommended schedule, and as clinically indicated [see Warnings and Precautions (5.1)].
[see Warnings and Precautions (5.1)] |
Fasting glucose levels (FPG or FBG) > ULN to 160 mg/dL (> ULN – 8.9 mmol/L) |
|
| Fasting glucose levels > 160 to 250 mg/dL (> 8.9 – 13.9 mmol/L) |
|
|
| Fasting glucose levels > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) |
|
|
| Fasting glucose levels > 500 mg/dL (> 27.8 mmol/L) |
|
|
|
Stomatitis
[see Warnings and Precautions (5.2)] |
Grade 1 Based on CTCAE version 5.0.
|
|
| Grade 2 |
|
|
| Grade 3 |
|
|
| Grade 4 |
|
|
|
Diarrhea
[see Warnings and Precautions (5.3)] |
Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 |
|
|
| Grade 4 |
|
|
|
Hematologic Toxicities
[see Adverse Reactions (6.1)] |
Grade 1, 2, or 3 |
|
| Grade 4 |
|
|
|
Other Adverse Reactions
[see Adverse Reactions (6.1)] |
Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 (first event) |
|
|
| Grade 3 (recurrent) |
|
|
| Grade 4 |
|
8.3 Females and Males of Reproductive Potential
ITOVEBI is used in combination with palbociclib and fulvestrant. Refer to the Full Prescribing Information of palbociclib and fulvestrant for contraception and infertility information.
ITOVEBI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Principal Display Panel 3 Mg Tablet Bottle Carton
NDC 50242-084-08
Itovebi®
(inavolisib)
Tablets
3 mg
Swallow tablets whole.
DO NOT chew, crush,
or split tablets.
28 Tablets
Rx only
Genentech
11039843
Principal Display Panel 9 Mg Tablet Bottle Carton
NDC 50242-079-08
Itovebi®
(inavolisib)
Tablets
9 mg
Swallow tablets whole.
DO NOT chew, crush,
or split tablets.
28 Tablets
Rx only
Genentech
11039960
2.2 Recommended Evaluation Before Initiating Itovebi
Evaluate fasting plasma glucose (FPG)/blood glucose (FBG) and hemoglobin A1C (HbA1C) and optimize blood glucose prior to starting ITOVEBI and at regular intervals during treatment [see Warnings and Precautions (5.1)].
2.5 Dosage Modification for Moderate Renal Impairment
The recommended starting dosage of ITOVEBI for patients with moderate renal impairment (eGFR 30 to < 60 mL/min based on CKD-EPI) is 6 mg orally once daily [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with inavolisib have not been conducted.
Inavolisib was not mutagenic in the bacterial reverse mutation (Ames) assay. Inavolisib was clastogenic in an in vitro human lymphocyte micronucleus assay. Inavolisib was not genotoxic in an in vivo rat bone marrow micronucleus test and did not induce DNA break in a liver comet assay.
Fertility studies with inavolisib have not been conducted. In repeat-dose toxicity studies, inavolisib was administered orally once daily for up to 3 months duration in rats and dogs.
In male rats, dose-dependent atrophy of the prostate and seminal vesicle and decreased organ weights of the prostate, seminal vesicle, epididymis and testis were observed at doses ≥ 1.5 mg/kg/day (≥ 0.4 times the human exposure at the recommended dose of 9 mg/day based on AUC). In male dogs, focal inspissation of seminiferous tubule contents and multinucleated spermatids in the testis and epithelial degeneration/necrosis in the epididymis were observed following 4 weeks of dosing at ≥ 1.5 mg/kg/day (≥ 2 times the human exposure at the recommended dose of 9 mg/day based on AUC) and decreased sperm count was observed following 3 months of dosing at ≥ 1 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose of 9 mg/day based on AUC). Findings in dogs were not observed following a recovery period.
In female rats, atrophy in the uterus and vagina, decreased ovarian follicles, and findings suggestive of an interruption/alteration of the estrous cycle were observed following up to 3 months of dosing at doses ≥ 3 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose of 9 mg/day based on AUC). Findings in the uterus, vagina, and estrous cycle observed in the 4-week toxicity study were not observed following recovery. Recovery was not assessed in the 3-month study in rats.
Structured Label Content
Section 42229-5 (42229-5)
Locally Advanced or Metastatic Breast Cancer
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 12/2025 | |
|
PATIENT INFORMATION
ITOVEBI® (eye-TOVE-bee) (inavolisib) tablets, for oral use |
||
| Important: ITOVEBI is used with palbociclib and fulvestrant. You should also read the Patient Information that comes with palbociclib and fulvestrant. | ||
|
What is ITOVEBI?
ITOVEBI is a prescription medicine used in combination with the medicines palbociclib and fulvestrant to treat adults who have hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer that has an abnormal phosphatidylinositol-3-kinase catalytic subunit alpha (PIK3CA) gene, and has spread to nearby tissue or lymph nodes (locally advanced), or to other parts of the body (metastatic), and has come back after hormone (endocrine) therapy. Your healthcare provider will test your cancer for abnormal PIK3CA genes to make sure that ITOVEBI is right for you. It is not known if ITOVEBI is safe and effective in children. |
||
Before you take ITOVEBI, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take ITOVEBI?
|
||
|
What are the possible side effects of ITOVEBI? ITOVEBI may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
The most common side effects and abnormal blood test results of ITOVEBI when used in combination with palbociclib and fulvestrant include: |
||
|
|
|
| ITOVEBI may affect fertility in males and in females who are able to become pregnant. Talk to your healthcare provider if this is a concern for you. These are not all of the possible side effects of ITOVEBI. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ITOVEBI?
|
||
|
General information about the safe and effective use of ITOVEBI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ITOVEBI for a condition for which it was not prescribed. Do not give ITOVEBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ITOVEBI that is written for health professionals. |
||
|
What are the ingredients in ITOVEBI?
Active ingredient: inavolisib Inactive ingredients: lactose, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The tablet film-coating contains iron oxide red, and iron oxide yellow (in the 9 mg tablet only), macrogol/polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Distributed by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 ITOVEBI is a registered trademark of Genentech, Inc. For more information, go to www.ITOVEBI.com or call 1-877-436-3683. |
Section 43683-2 (43683-2)
| Warnings and Precautions, Hyperglycemia (5.1) | 09/2025 |
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F) [see USP Controlled Room Temperature]. Store in the original container and keep the bottle tightly closed in order to protect from moisture.
5.3 Diarrhea
Severe diarrhea, including dehydration and acute kidney injury, can occur in patients treated with ITOVEBI.
Diarrhea occurred in 48% of patients treated with ITOVEBI in combination with palbociclib and fulvestrant, including Grade 3 events in 3.7% of patients. The median time to first onset was 15 days (range: 2 to 602 days).
Diarrhea led to dose interruptions in 7% of patients, and dose reductions in 1.2% of patients. Anti-diarrheal medicines were used in 28% (46/162) of patients who received ITOVEBI in combination with palbociclib and fulvestrant to manage symptoms.
Monitor patients for signs and symptoms of diarrhea. Advise patients to increase oral fluids and start anti-diarrheal treatment at the first sign of diarrhea while taking ITOVEBI. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity [see Dosage and Administration (2.4)].
11 Description (11 DESCRIPTION)
ITOVEBI contains inavolisib, a kinase inhibitor. The chemical name of inavolisib is (2S)-2-[[2-[(4S)-4-(difluoromethyl)-2-oxo-oxazolidin-3-yl]-5,6-dihydroimidazo[1,2-d][1,4]benzoxazepin-9-yl]amino]propanamide. Inavolisib is a white to off-white, greyish pink, greyish orange, or greyish yellow powder or powder with lumps. Inavolisib demonstrates pH-dependent aqueous solubility; the greatest solubility is at low pH, and solubility decreases with increasing pH. The molecular formula for inavolisib is C18H19F2N5O4 and the molecular weight is 407.37 g/mol. The chemical structure of inavolisib is shown below:
ITOVEBI film-coated tablets are supplied for oral administration with two strengths that contain 3 mg and 9 mg of inavolisib. The tablets also contain lactose, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The film-coating contains the following inactive ingredients: polyvinyl alcohol (partially hydrolyzed), titanium dioxide, macrogol/polyethylene glycol, talc, iron oxide red, and iron oxide yellow (in the 9 mg tablet only).
5.2 Stomatitis
Severe stomatitis can occur in patients treated with ITOVEBI.
Stomatitis occurred in 51% of patients treated with ITOVEBI in combination with palbociclib and fulvestrant, including Grade 3 events in 6% of patients. The median time to first onset was 13 days (range: 1 to 610 days).
Stomatitis led to dose interruption in 10%, to dose reduction in 3.7%, and to discontinuation of ITOVEBI in 0.6% of patients.
In patients who received ITOVEBI in combination with palbociclib and fulvestrant, 38% used a mouthwash containing corticosteroid for management or prophylaxis of stomatitis.
Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity [see Dosage and Administration (2.4)].
5.1 Hyperglycemia
Severe or fatal hyperglycemia, including ketoacidosis, can occur in patients treated with ITOVEBI. Ketoacidosis with a fatal outcome has occurred in the postmarketing setting.
Increased fasting glucose occurred in 85% of patients treated with ITOVEBI, including 22% of patients with Grade 2 (FPG > 160 to 250 mg/dL), 12% with Grade 3 (FPG > 250 to 500 mg/dL), and 0.6% with Grade 4 (FPG > 500 mg/dL) events.
In INAVO120, 46% (74/162) of patients who received ITOVEBI were treated with oral anti-hyperglycemic medications and 7% (11/162) were treated with insulin to manage increased fasting glucose. In patients who experienced increased fasting glucose of > 160 mg/dL, 96% (52/54) had an improvement in fasting glucose of at least one grade level with a median time to improvement of 8 days (range: 2 to 43 days).
Among patients with hyperglycemia, the median time to first onset was 7 days (range: 2 to 955 days). Hyperglycemia led to dose interruption in 28%, to dose reduction in 2.5%, and to discontinuation of ITOVEBI in 1.2% of patients.
The safety of ITOVEBI in patients with Type 1 diabetes mellitus, or Type 2 diabetes mellitus requiring ongoing anti-hyperglycemic treatment have not been studied.
Before initiating treatment with ITOVEBI, test fasting glucose levels (FPG or FBG), HbA1C levels, and optimize fasting glucose.
After initiating treatment with ITOVEBI, or in patients who experience hyperglycemia after initiating treatment with ITOVEBI, monitor or self-monitor fasting glucose levels once every 3 days for the first week (Day 1 to 7), then once every week for the next 3 weeks (Day 8 to 28), then once every 2 weeks for the next 8 weeks, then once every 4 weeks thereafter, and as clinically indicated. Monitor HbA1C every 3 months and as clinically indicated.
Manage hyperglycemia with anti-hyperglycemic medications as clinically indicated. During treatment with anti-hyperglycemic medication, continue monitoring fasting glucose levels. Patients with a history of well-controlled Type 2 diabetes mellitus may require intensified anti-hyperglycemic treatment and close monitoring of fasting glucose levels.
Consider consultation with a healthcare professional experienced in the treatment of hyperglycemia, and initiation of fasting glucose monitoring at home for patients who have risk factors for hyperglycemia or who experience hyperglycemia. Advise patients of the signs and symptoms of hyperglycemia and counsel patients on lifestyle changes.
Based on the severity of the hyperglycemia, ITOVEBI may require dose interruption, reduction, or discontinuation [see Dosage and Administration (2.4)].
8.4 Pediatric Use
The safety and efficacy of ITOVEBI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 162 patients who received ITOVEBI in INAVO120, 15% were ≥ 65 years of age, and 3% were ≥ 75 years of age.
Dosage modifications or interruptions of ITOVEBI due to adverse reactions occurred at a higher incidence for patients ≥ 65 years of age compared to younger patients (79% versus 68%, respectively).
Clinical studies of ITOVEBI did not include sufficient numbers of patients ≥ 65 years of age to determine whether they respond differently from younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hyperglycemia [see Warnings and Precautions (5.1)]
- Stomatitis [see Warnings and Precautions (5.2)]
- Diarrhea [see Warnings and Precautions (5.3)]
8.6 Renal Impairment
Reduce the dosage in patients with moderate renal impairment (eGFR 30 to < 60 mL/min based on CKD-EPI) [see Dosage and Administration (2.5)]. No dosage modification is recommended in patients with mild renal impairment (eGFR 60 to < 90 mL/min). ITOVEBI has not been evaluated in patients with severe renal impairment (eGFR < 30 mL/min).
12.3 Pharmacokinetics
Inavolisib pharmacokinetics are presented as geometric mean (geometric coefficient of variation [geo CV]%) following administration of the approved recommended dosage unless otherwise specified. The inavolisib steady-state AUC is 1,010 h*ng/mL (25%) and Cmax is 69 ng/mL (27%). Steady-state concentrations are predicted to be attained by day 5.
Inavolisib accumulation is approximately 2-fold.
Inavolisib steady-state AUC is proportional with dose from 6 to 12 mg (0.7 to 1.3 times the approved recommended dosage).
2.1 Patient Selection
Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimens [see Clinical Studies (14.1)].
Information on FDA-approved tests for the detection of PIK3CA mutations in breast cancer is available at: http://www.fda.gov/companiondiagnostics.
2.3 Recommended Dosage
The recommended dosage of ITOVEBI is 9 mg taken orally once daily, with or without food, until disease progression or unacceptable toxicity.
Advise patients to take ITOVEBI at approximately the same time each day.
Swallow ITOVEBI tablet(s) whole. Do not chew, crush, or split prior to swallowing.
If a patient misses a dose, instruct the patient to take the missed dose as soon as possible within 9 hours. After more than 9 hours, instruct the patient to skip the dose and take the next dose at the scheduled time.
If a patient vomits a dose, instruct patients not to take an additional dose on that day and resume the usual dosing schedule the next day.
Administer ITOVEBI in combination with palbociclib and fulvestrant. The recommended dosage of palbociclib is 125 mg taken orally once daily for 21 consecutive days followed by 7 days off treatment to comprise a cycle of 28 days. Refer to the Full Prescribing Information for palbociclib and fulvestrant for dosing information.
For premenopausal and perimenopausal women, administer a luteinizing hormone-releasing hormone (LHRH) agonist in accordance with local clinical practice.
For men, consider administering an LHRH agonist in accordance with local clinical practice.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ITOVEBI, in combination with palbociclib and fulvestrant, is indicated for the treatment of adults with endocrine-resistant, PIK3CA-mutated, hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy [see Clinical Studies (14.1)].
12.1 Mechanism of Action
Inavolisib is an inhibitor of phosphatidylinositol 3-kinase (PI3K) with inhibitory activity predominantly against PI3Kα. In vitro, inavolisib induced the degradation of mutated PI3K catalytic alpha subunit p110α (encoded by the PIK3CA gene), inhibited phosphorylation of the downstream target AKT, reduced cellular proliferation, and induced apoptosis in PIK3CA-mutated breast cancer cell lines. In vivo, inavolisib reduced tumor growth in PIK3CA-mutated, estrogen receptor-positive, breast cancer xenograft models. The combination of inavolisib with palbociclib and fulvestrant increased tumor growth inhibition compared to each treatment alone or the doublet combinations.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on findings in animals and its mechanism of action, ITOVEBI can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. In an animal reproduction study, oral administration of inavolisib to pregnant rats during the period of organogenesis caused adverse developmental outcomes, including embryo-fetal mortality, structural abnormalities, and alterations to growth at maternal exposures approximately equivalent to the human exposure at the recommended dose of 9 mg/day based on area under the curve (AUC).
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective non-hormonal contraception during treatment with ITOVEBI and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ITOVEBI and for 1 week after the last dose [see Use in Specific Populations (8.1 and 8.3)].
ITOVEBI is used in combination with palbociclib and fulvestrant. Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hyperglycemia: ITOVEBI can cause severe or fatal hyperglycemia including ketoacidosis. Before initiating treatment with ITOVEBI, test fasting plasma glucose (FPG), HbA1c, and optimize blood glucose. Initiate or optimize anti-hyperglycemic medications as clinically indicated. Interrupt, reduce dose, or discontinue ITOVEBI if severe hyperglycemia occurs. (2.4, 5.1)
- Stomatitis: ITOVEBI can cause severe stomatitis. Consider treating with a corticosteroid-containing mouthwash if stomatitis occurs. Monitor patients for signs and symptoms of stomatitis. Withhold, reduce dose, or permanently discontinue ITOVEBI based on severity. (2.4, 5.2)
- Diarrhea: ITOVEBI can cause diarrhea, which may be severe, and result in dehydration and acute kidney injury. Advise patients to start anti-diarrheal treatment, increase oral fluids, and notify their healthcare provider if severe diarrhea occurs. Interrupt, reduce dose, or discontinue ITOVEBI if severe diarrhea occurs. (2.4, 5.3)
-
Embryo-Fetal Toxicity: ITOVEBI can cause fetal harm. Advise patients of potential risk to a fetus and to use effective non-hormonal contraception. (5.4, 8.1, 8.3)
Refer to the Full Prescribing Information of palbociclib and fulvestrant for pregnancy and contraception information.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Select patients for the treatment of HR-positive, HER2-negative, locally advanced or metastatic breast cancer with ITOVEBI based on the presence of one or more PIK3CA mutations in plasma specimen. (2.1)
- Recommended dosage: 9 mg orally once daily with or without food. (2.3)
- See Full Prescribing Information for dosage modifications of ITOVEBI due to adverse reactions. (2.4)
- Reduce the starting dose in patients with moderate renal impairment. (2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets:
- 3 mg: red and round convex-shaped with an "INA 3" debossing on one side.
- 9 mg: pink and oval-shaped with an "INA 9" debossing on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ITOVEBI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition Disorders: Ketoacidosis
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ITOVEBI is supplied in the following strengths and package configurations:
| Package Configuration | Tablet Strength | NDC | Tablet Description |
|---|---|---|---|
| Bottle of 28 tablets | 3 mg | 50242-084-08 | Red and round convex-shaped with an "INA 3" debossing on one side |
| Bottle of 28 tablets | 9 mg | 50242-079-08 | Pink and oval-shaped with an "INA 9" debossing on one side |
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Lens degeneration, characterized by lens fiber swelling, separation of lens fibers, and/or accumulation of subcapsular proteinaceous material, was observed in rats at an oral inavolisib dose of 10 mg/kg/day (6.3 times the human exposure at the recommended dose of 9 mg/day based on AUC). In dogs, lens fiber swelling and lens cortex vacuolation were observed at oral inavolisib doses ≥ 0.3 mg/kg/day (≥ 0.5 times the human exposure at the recommended dose based on AUC) and ≥ 1 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose based on AUC), respectively. Lens degeneration was present in rats following a 4-week recovery period but was not present in dogs following a 12-week recovery period.
2.4 Dosage Modifications for Adverse Reactions
The recommended dose reduction levels of ITOVEBI for adverse reactions are listed in Table 1. Permanently discontinue ITOVEBI if patients are unable to tolerate the second dose reduction.
| Dose Level | Dose and Schedule |
|---|---|
| Recommended starting dose | 9 mg daily |
| First dose reduction | 6 mg daily |
| Second dose reduction | 3 mg daily |
The recommended dosage modifications of ITOVEBI for adverse reactions are summarized in Table 2.
| Adverse Reaction | Severity | Dosage Modification |
|---|---|---|
|
Hyperglycemia Before initiating treatment with ITOVEBI, test FPG or FBG, and HbA1C levels, and optimize plasma/blood glucose levels in all patients. After initiating treatment with ITOVEBI, monitor FPG or FBG levels based on the recommended schedule, and as clinically indicated [see Warnings and Precautions (5.1)].
[see Warnings and Precautions (5.1)] |
Fasting glucose levels (FPG or FBG) > ULN to 160 mg/dL (> ULN – 8.9 mmol/L) |
|
| Fasting glucose levels > 160 to 250 mg/dL (> 8.9 – 13.9 mmol/L) |
|
|
| Fasting glucose levels > 250 to 500 mg/dL (> 13.9 – 27.8 mmol/L) |
|
|
| Fasting glucose levels > 500 mg/dL (> 27.8 mmol/L) |
|
|
|
Stomatitis
[see Warnings and Precautions (5.2)] |
Grade 1 Based on CTCAE version 5.0.
|
|
| Grade 2 |
|
|
| Grade 3 |
|
|
| Grade 4 |
|
|
|
Diarrhea
[see Warnings and Precautions (5.3)] |
Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 |
|
|
| Grade 4 |
|
|
|
Hematologic Toxicities
[see Adverse Reactions (6.1)] |
Grade 1, 2, or 3 |
|
| Grade 4 |
|
|
|
Other Adverse Reactions
[see Adverse Reactions (6.1)] |
Grade 1 |
|
| Grade 2 |
|
|
| Grade 3 (first event) |
|
|
| Grade 3 (recurrent) |
|
|
| Grade 4 |
|
8.3 Females and Males of Reproductive Potential
ITOVEBI is used in combination with palbociclib and fulvestrant. Refer to the Full Prescribing Information of palbociclib and fulvestrant for contraception and infertility information.
ITOVEBI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Principal Display Panel 3 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 3 mg Tablet Bottle Carton)
NDC 50242-084-08
Itovebi®
(inavolisib)
Tablets
3 mg
Swallow tablets whole.
DO NOT chew, crush,
or split tablets.
28 Tablets
Rx only
Genentech
11039843
Principal Display Panel 9 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 9 mg Tablet Bottle Carton)
NDC 50242-079-08
Itovebi®
(inavolisib)
Tablets
9 mg
Swallow tablets whole.
DO NOT chew, crush,
or split tablets.
28 Tablets
Rx only
Genentech
11039960
2.2 Recommended Evaluation Before Initiating Itovebi (2.2 Recommended Evaluation Before Initiating ITOVEBI)
Evaluate fasting plasma glucose (FPG)/blood glucose (FBG) and hemoglobin A1C (HbA1C) and optimize blood glucose prior to starting ITOVEBI and at regular intervals during treatment [see Warnings and Precautions (5.1)].
2.5 Dosage Modification for Moderate Renal Impairment
The recommended starting dosage of ITOVEBI for patients with moderate renal impairment (eGFR 30 to < 60 mL/min based on CKD-EPI) is 6 mg orally once daily [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with inavolisib have not been conducted.
Inavolisib was not mutagenic in the bacterial reverse mutation (Ames) assay. Inavolisib was clastogenic in an in vitro human lymphocyte micronucleus assay. Inavolisib was not genotoxic in an in vivo rat bone marrow micronucleus test and did not induce DNA break in a liver comet assay.
Fertility studies with inavolisib have not been conducted. In repeat-dose toxicity studies, inavolisib was administered orally once daily for up to 3 months duration in rats and dogs.
In male rats, dose-dependent atrophy of the prostate and seminal vesicle and decreased organ weights of the prostate, seminal vesicle, epididymis and testis were observed at doses ≥ 1.5 mg/kg/day (≥ 0.4 times the human exposure at the recommended dose of 9 mg/day based on AUC). In male dogs, focal inspissation of seminiferous tubule contents and multinucleated spermatids in the testis and epithelial degeneration/necrosis in the epididymis were observed following 4 weeks of dosing at ≥ 1.5 mg/kg/day (≥ 2 times the human exposure at the recommended dose of 9 mg/day based on AUC) and decreased sperm count was observed following 3 months of dosing at ≥ 1 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose of 9 mg/day based on AUC). Findings in dogs were not observed following a recovery period.
In female rats, atrophy in the uterus and vagina, decreased ovarian follicles, and findings suggestive of an interruption/alteration of the estrous cycle were observed following up to 3 months of dosing at doses ≥ 3 mg/kg/day (≥ 1.2 times the human exposure at the recommended dose of 9 mg/day based on AUC). Findings in the uterus, vagina, and estrous cycle observed in the 4-week toxicity study were not observed following recovery. Recovery was not assessed in the 3-month study in rats.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:16.094170 · Updated: 2026-03-14T22:43:58.484368