Polyethylene Glycol 3350
5d2b2570-444b-4263-b339-bd81171979d6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
MARLEX PHARMACEUTICAL S , INC ---------- Polyethylene Glycol 3350 powder, for solution Drug Facts
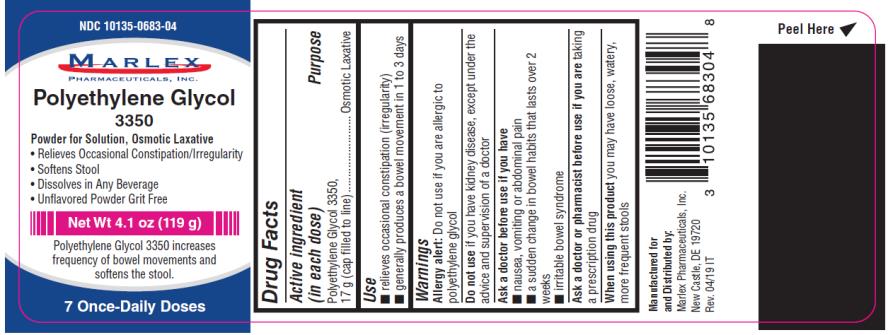
Purpose
Osmotic Laxative
Medication Information
Purpose
Osmotic Laxative
Description
MARLEX PHARMACEUTICAL S , INC ---------- Polyethylene Glycol 3350 powder, for solution Drug Facts
Use
- relieves occasional constipation (irregularity) generally
- produces a bowel movement in 1 to 3 days
Section 42229-5
MARLEX PHARMACEUTICAL S , INC
----------
Polyethylene Glycol 3350 powder, for solution
Drug Facts
Warnings
Allergy alert:
Do not use if you are allergic to polyethylene glycol
Directions
-
do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line (clear section in cap)
- adults and children 17 years of age and older:
○ fill to top of clear section in cap which is marked to indicate the correct dose (17 g)
○ stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
○ use once a day
○ use no more than 7 days
- children 16 years of age or under: ask a doctor
Do Not Use
if you have kidney disease, except under the advice and supervision of a doctor
Other Information
- store at 20° - 25°C (68° - 77°F)
- tamper- evident: do not use if foil seal under cap, printed with “SEALED for YOUR PROTECTION” is missing, open or broken
Inactive Ingredient
none
Questions Or Comments?
1-800-FDA-1088
Manufactured for & Distributed by:
Marlex Pharmaceuticals, Inc.
New Castle, DE 19720
Rev. 04/19IT
Principal Display Panel
NDC 10135-0683-04
Polyethylene Glycol
3350
Powder for Solution, Osmotic Laxative
Net Wt 4.1 oz (119 g)
7 Once-Daily Doses
When Using This Product
you may loose, watery, more frequent stools
Stop Use and Ask A Doctor If
- you have rectal bleeding or your nausea, bloating, cramping or abdominal pain gets worse. These may be signs of a serious condition.
- you get diarrhea
- you need to use a laxative for longer than 1 week
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Dose)
Polyethylene Glycol 3350, 17 g (cap filled to line)
Ask A Doctor Before Use If You Have
- nausea, vomiting or abdominal pain
- a sudden change in bowel habits that lasts over 2 weeks
- irritable bowel syndrome
Ask Doctor Or Pharmacist Before Use If You Are
taking a prescription drug
Structured Label Content
Use
- relieves occasional constipation (irregularity) generally
- produces a bowel movement in 1 to 3 days
Section 42229-5 (42229-5)
MARLEX PHARMACEUTICAL S , INC
----------
Polyethylene Glycol 3350 powder, for solution
Drug Facts
Purpose
Osmotic Laxative
Warnings
Allergy alert:
Do not use if you are allergic to polyethylene glycol
Directions
-
do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line (clear section in cap)
- adults and children 17 years of age and older:
○ fill to top of clear section in cap which is marked to indicate the correct dose (17 g)
○ stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
○ use once a day
○ use no more than 7 days
- children 16 years of age or under: ask a doctor
Do Not Use (Do not use)
if you have kidney disease, except under the advice and supervision of a doctor
Other Information (Other information)
- store at 20° - 25°C (68° - 77°F)
- tamper- evident: do not use if foil seal under cap, printed with “SEALED for YOUR PROTECTION” is missing, open or broken
Inactive Ingredient (Inactive ingredient)
none
Questions Or Comments? (Questions or comments?)
1-800-FDA-1088
Manufactured for & Distributed by:
Marlex Pharmaceuticals, Inc.
New Castle, DE 19720
Rev. 04/19IT
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 10135-0683-04
Polyethylene Glycol
3350
Powder for Solution, Osmotic Laxative
Net Wt 4.1 oz (119 g)
7 Once-Daily Doses
When Using This Product (When using this product)
you may loose, watery, more frequent stools
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you have rectal bleeding or your nausea, bloating, cramping or abdominal pain gets worse. These may be signs of a serious condition.
- you get diarrhea
- you need to use a laxative for longer than 1 week
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Dose) (Active ingredient (in each dose))
Polyethylene Glycol 3350, 17 g (cap filled to line)
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- nausea, vomiting or abdominal pain
- a sudden change in bowel habits that lasts over 2 weeks
- irritable bowel syndrome
Ask Doctor Or Pharmacist Before Use If You Are (Ask doctor or pharmacist before use if you are)
taking a prescription drug
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:48.709352 · Updated: 2026-03-14T22:55:33.121473