These Highlights Do Not Include All The Information Needed To Use Bavencio Safely And Effectively. See Full Prescribing Information For Bavencio.
5cd725a1-2fa4-408a-a651-57a7b84b2118
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for: Merkel Cell Carcinoma (MCC) Adults and pediatric patients 12 years and older with metastatic MCC. ( 1.1 , 14.1 ) Urothelial Carcinoma (UC) Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. ( 1.2 , 14.2 ) Patients with locally advanced or metastatic UC who: Have disease progression during or following platinum-containing chemotherapy. ( 1.2 , 14.2 ) Have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.2 , 14.2 ) Renal Cell Carcinoma (RCC) First-line treatment, in combination with axitinib, of patients with advanced RCC. ( 1.3 , 14.3 )
Indications and Usage
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for: Merkel Cell Carcinoma (MCC) Adults and pediatric patients 12 years and older with metastatic MCC. ( 1.1 , 14.1 ) Urothelial Carcinoma (UC) Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. ( 1.2 , 14.2 ) Patients with locally advanced or metastatic UC who: Have disease progression during or following platinum-containing chemotherapy. ( 1.2 , 14.2 ) Have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.2 , 14.2 ) Renal Cell Carcinoma (RCC) First-line treatment, in combination with axitinib, of patients with advanced RCC. ( 1.3 , 14.3 )
Dosage and Administration
Premedicate for the first 4 infusions and subsequently as needed. ( 2.1 ) Merkel Cell Carcinoma : 800 mg every 2 weeks. ( 2.2 ) Urothelial Carcinoma ; 800 mg every 2 weeks. ( 2.3 ) Renal Cell Carcinoma : 800 mg every 2 weeks in combination with axitinib 5 mg orally twice daily. ( 2.4 ) Administer BAVENCIO as an intravenous infusion over 60 minutes.
Warnings and Precautions
Immune-Mediated Adverse Reactions ( 5.1 ) Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and may result in solid organ transplant rejection. Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. Withhold or permanently discontinue based on severity and type of reaction. Infusion-related reactions : Interrupt, slow the rate of infusion, or permanently discontinue BAVENCIO based on severity of reaction. ( 5.2 ) Complications of allogeneic HSCT : Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Major adverse cardiovascular events : Optimize management of cardiovascular risk factors. Discontinue BAVENCIO in combination with axitinib for Grade 3-4 events. ( 5.4 ) Embryo-fetal toxicity : BAVENCIO can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.5 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1) ] Infusion-related reactions [see Warnings and Precautions (5.2) ] Complications of allogeneic HSCT [see Warnings and Precautions (5.3) ] Major adverse cardiovascular events [see Warnings and Precautions (5.4) ]
Storage and Handling
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
How Supplied
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
Medication Information
Warnings and Precautions
Immune-Mediated Adverse Reactions ( 5.1 ) Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and may result in solid organ transplant rejection. Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. Withhold or permanently discontinue based on severity and type of reaction. Infusion-related reactions : Interrupt, slow the rate of infusion, or permanently discontinue BAVENCIO based on severity of reaction. ( 5.2 ) Complications of allogeneic HSCT : Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Major adverse cardiovascular events : Optimize management of cardiovascular risk factors. Discontinue BAVENCIO in combination with axitinib for Grade 3-4 events. ( 5.4 ) Embryo-fetal toxicity : BAVENCIO can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.5 , 8.1 , 8.3 )
Indications and Usage
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for: Merkel Cell Carcinoma (MCC) Adults and pediatric patients 12 years and older with metastatic MCC. ( 1.1 , 14.1 ) Urothelial Carcinoma (UC) Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. ( 1.2 , 14.2 ) Patients with locally advanced or metastatic UC who: Have disease progression during or following platinum-containing chemotherapy. ( 1.2 , 14.2 ) Have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.2 , 14.2 ) Renal Cell Carcinoma (RCC) First-line treatment, in combination with axitinib, of patients with advanced RCC. ( 1.3 , 14.3 )
Dosage and Administration
Premedicate for the first 4 infusions and subsequently as needed. ( 2.1 ) Merkel Cell Carcinoma : 800 mg every 2 weeks. ( 2.2 ) Urothelial Carcinoma ; 800 mg every 2 weeks. ( 2.3 ) Renal Cell Carcinoma : 800 mg every 2 weeks in combination with axitinib 5 mg orally twice daily. ( 2.4 ) Administer BAVENCIO as an intravenous infusion over 60 minutes.
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1) ] Infusion-related reactions [see Warnings and Precautions (5.2) ] Complications of allogeneic HSCT [see Warnings and Precautions (5.3) ] Major adverse cardiovascular events [see Warnings and Precautions (5.4) ]
Storage and Handling
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
How Supplied
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
Description
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for: Merkel Cell Carcinoma (MCC) Adults and pediatric patients 12 years and older with metastatic MCC. ( 1.1 , 14.1 ) Urothelial Carcinoma (UC) Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. ( 1.2 , 14.2 ) Patients with locally advanced or metastatic UC who: Have disease progression during or following platinum-containing chemotherapy. ( 1.2 , 14.2 ) Have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.2 , 14.2 ) Renal Cell Carcinoma (RCC) First-line treatment, in combination with axitinib, of patients with advanced RCC. ( 1.3 , 14.3 )
Section 42229-5
First-Line Maintenance Treatment of Urothelial Carcinoma
BAVENCIO is indicated for the maintenance treatment of patients with locally advanced or metastatic urothelial carcinoma (UC) that has not progressed with first-line platinum-containing chemotherapy [see Clinical Studies (14.2)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 06/2025 | |||||
|
MEDICATION GUIDE
BAVENCIO® (buh-VEN-see-oh) (avelumab) injection |
||||||
|
What is the most important information I should know about BAVENCIO?
BAVENCIO is a medicine that may treat certain cancers by working with your immune system. BAVENCIO can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during treatment or even after your treatment has ended. Call or see your healthcare provider right away if you get any new or worsening signs or symptoms, including: Lung problems. |
||||||
|
|
|
||||
Intestinal problems.
|
||||||
|
|
|||||
| Hormone gland problems. | ||||||
|
|
|||||
| Kidney problems. | ||||||
|
|
|||||
| Skin problems. | ||||||
|
|
|||||
Problems can also happen in other organs and tissues. These are not all of the signs or symptoms of immune system problems that can happen with BAVENCIO. Call or see your healthcare provider right away for any new or worsening signs or symptoms, which may include:
|
||||||
| Infusion-related reactions can sometimes be severe or life-threatening. Signs and symptoms of infusion-related reactions may include: | ||||||
|
|
|||||
|
Complications, including graft-versus-host-disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with BAVENCIO. Your healthcare provider will monitor you for these complications. Heart problems. When BAVENCIO is used with the medicine axitinib, severe heart problems can happen and can lead to death. Signs and symptoms of heart problems may include: |
||||||
|
|
|||||
| Getting medical treatment right away may help keep these problems from becoming more serious. Your healthcare provider will check you for these problems during your treatment with BAVENCIO. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with BAVENCIO if you have severe side effects. | ||||||
|
What is BAVENCIO?
BAVENCIO is a prescription medicine used to treat:
|
||||||
Before you receive BAVENCIO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||
How will I receive BAVENCIO?
|
||||||
|
What are the possible side effects of BAVENCIO? BAVENCIO can cause serious side effects, including: The most common side effects of BAVENCIO in people with MCC include: |
||||||
|
|
|||||
| The most common side effects of BAVENCIO as maintenance treatment in people with UC whose cancer responded or stabilized after platinum-containing chemotherapy as first treatment include: | ||||||
|
|
|||||
| The most common side effects of BAVENCIO in people with UC after platinum-containing chemotherapy that did not work, or is no longer working, include: | ||||||
|
|
|||||
| The most common side effects of BAVENCIO when given with axitinib in people with RCC include: | ||||||
|
|
|||||
| These are not all the possible side effects of BAVENCIO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||
|
General information about the safe and effective use of BAVENCIO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about BAVENCIO that is written for health professionals. |
||||||
|
What are the ingredients in BAVENCIO?
Active ingredient: avelumab Inactive ingredients: D-mannitol, glacial acetic acid, polysorbate 20, sodium hydroxide, and Water for Injection Manufactured by: EMD Serono, Inc., Boston, MA 02210, USA, U.S. License No. 1773. Marketed by: EMD Serono, Inc., MA, USA. BAVENCIO is a trademark of Merck KGaA, Darmstadt, Germany. For more information, call toll-free 1-844-826-8371 or go to www.bavencio.com. |
||||||
Section 44425-7
Store refrigerated at 36°F to 46°F (2°C to 8°C) in original package to protect from light.
Do not freeze or shake the vial.
The vial stopper is not made with natural rubber latex.
11 Description
Avelumab is a programmed death ligand1 (PD-L1) blocking antibody. Avelumab is a human IgG1 lambda monoclonal antibody produced in Chinese hamster ovary cells and has a molecular weight of approximately 147 kDa.
BAVENCIO (avelumab) Injection for intravenous use is a sterile, preservative-free, non-pyrogenic, clear, colorless to slightly yellow solution. Each single-dose vial contains 200 mg avelumab in 10 mL (20 mg/mL). Each mL contains 20 mg avelumab, D-mannitol (51 mg), glacial acetic acid (0.6 mg), polysorbate 20 (0.5 mg), sodium hydroxide (0.3 mg), and Water for Injection. The pH range of the solution is 5.0 – 5.6.
2.1 Premedication
Premedicate patients with an antihistamine and with acetaminophen prior to the first 4 infusions of BAVENCIO. Premedication should be administered for subsequent BAVENCIO doses based upon clinical judgment and presence/severity of prior infusion reactions [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
8.4 Pediatric Use
The safety and effectiveness of BAVENCIO have been established in pediatric patients aged 12 years and older for metastatic MCC. Use of BAVENCIO in this age group is supported by evidence from adequate and well-controlled studies of BAVENCIO in adults with additional population pharmacokinetic data demonstrating that age and body weight had no clinically meaningful effect on the steady state exposure of avelumab, that drug exposure is generally similar between adults and pediatric patients age 12 years and older for monoclonal antibodies, and that the course of MCC is sufficiently similar in adult and pediatric patients to allow extrapolation of data in adults to pediatric patients. The recommended dose in pediatric patients 12 years of age or greater is the same as that in adults [see Dosage and Administration (2.2), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness of BAVENCIO have not been established in pediatric patients less than 12 years of age.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of BAVENCIO or of other avelumab products.
ADA responses following administration of BAVENCIO 10 mg/kg every 2 weeks were evaluated during the respective treatment periods in each trial. The ADA and neutralizing antibody (nAb) incidences are listed in Table 9.
| Trial Name Details of each treatment regimen are described in Section 14 [see Clinical Studies (14)].
|
Treatment Period (months) | ADA | nAb |
|---|---|---|---|
| ADA: anti-avelumab antibodies; UC: urothelial carcinoma | |||
| JAVELIN Merkel 200 Part A | 48 | 8.9% (7/79) | 71% (5/7) |
| JAVELIN Merkel 200 Part B | 35 | 8.2% (9/110) | 89% (8/9) |
| JAVELIN Bladder 100 | 37 | 19% (62/326) | 97% (60/62) |
| JAVELIN Solid Tumor, UC Cohort | 59 | 18% (41/226) | Not tested |
| JAVELIN Renal 101 | 29 | 16% (65/411) | 78% (51/65) |
In patients with advanced UC or advanced RCC, avelumab clearance was approximately 15% higher in patients who tested positive for ADA as compared to clearance in patients who tested negative for ADA; which is not considered clinically meaningful. The effect of ADA on the efficacy or safety could not be determined due to the low occurrence of ADAs.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1)]
- Infusion-related reactions [see Warnings and Precautions (5.2)]
- Complications of allogeneic HSCT [see Warnings and Precautions (5.3)]
- Major adverse cardiovascular events [see Warnings and Precautions (5.4)]
12.2 Pharmacodynamics
Avelumab exposure-response relationships and the time course of pharmacodynamic response are not fully characterized.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of avelumab as a single agent was characterized in patients who received BAVENCIO at doses ranging from 1 to 20 mg/kg every 2 weeks (0.1 to 2 times of the approved recommended dosage). The exposure of avelumab increased dose-proportionally from 10 to 20 mg/kg every 2 weeks. Steady-state concentrations of avelumab were reached after approximately 4 to 6 weeks (2 to 3 cycles) and the systemic accumulation was 1.25-fold.
2.5 Dose Modifications
No dose reduction for BAVENCIO is recommended. In general, withhold BAVENCIO for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue BAVENCIO for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating corticosteroids.
Dosage modifications for BAVENCIO for adverse reactions that require management different from these general guidelines are summarized in Table 1.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.03
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, SJS = Stevens-Johnson syndrome, TEN = toxic epidermal necrosis, DRESS = drug rash with eosinophilia and systemic symptoms | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of last dose or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating corticosteroids.
|
| Grade 3 or 4 | Permanently discontinue | |
| Colitis | Grade 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
| Hepatitis with no tumor involvement of the liver For liver enzyme elevations in patients treated with combination therapy, see Table 2 |
AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue BAVENCIO based on recommendations for hepatitis where there is no tumor involvement of the liver.
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grade 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grade 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis | Grade 2, 3 or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grade 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Infusion-related reactions [see Warnings and Precautions (5.2)] | Grade 1 or 2 | Interrupt or slow the rate of infusion |
| Grade 3 or 4 | Permanently discontinue |
Table 2 presents dosage modifications that are different from those described above in Table 1 for BAVENCIO used as monotherapy or in the Full Prescribing Information for the drug administered in combination.
| Treatment | Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.03
|
Dosage Modification |
|---|---|---|---|
| BAVENCIO in combination with axitinib | Liver enzyme elevations | ALT or AST at least 3 times ULN but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN | Withhold both BAVENCIO and axitinib until adverse reactions recover to Grades 0-1 Consider corticosteroid therapy
Consider rechallenge with BAVENCIO or axitinib or sequential rechallenge with both BAVENCIO and axitinib after recovery Dose reduction according to the axitinib Full Prescribing Information should be considered if rechallenging with axitinib.
|
| ALT or AST at least 10 times ULN or more than 3 times ULN with concurrent total bilirubin at least 2 times ULN | Permanently discontinue both BAVENCIO and axitinib |
1 Indications and Usage
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for:
Merkel Cell Carcinoma (MCC)
Urothelial Carcinoma (UC)
- Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. (1.2, 14.2)
- Patients with locally advanced or metastatic UC who:
Renal Cell Carcinoma (RCC)
12.1 Mechanism of Action
PD-L1 may be expressed on tumor cells and tumor-infiltrating immune cells and can contribute to the inhibition of the anti-tumor immune response in the tumor microenvironment. Binding of PD-L1 to the PD-1 and B7.1 receptors found on T cells and antigen presenting cells suppresses cytotoxic T-cell activity, T-cell proliferation, and cytokine production. Avelumab binds PD-L1 and blocks the interaction between PD-L1 and its receptors PD-1 and B7.1. This interaction releases the inhibitory effects of PD-L1 on the immune response resulting in the restoration of immune responses, including anti-tumor immune responses. Avelumab has also been shown to induce antibody-dependent cell-mediated cytotoxicity (ADCC) in vitro. In syngeneic mouse tumor models, blocking PD-L1 activity resulted in decreased tumor growth.
5.5 Embryo Fetal Toxicity
Based on its mechanism of action, BAVENCIO can cause fetal harm when administered to a pregnant woman. Animal studies have demonstrated that inhibition of the PD-1/PD-L1 pathway can lead to increased risk of immune-mediated rejection of the developing fetus resulting in fetal death. If this drug is used during pregnancy, or if the patient becomes pregnant while taking BAVENCIO, inform the patient of the potential risk to a fetus. Advise females of childbearing potential to use effective contraception during treatment with BAVENCIO and for at least one month after the last dose of BAVENCIO [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
-
Immune-Mediated Adverse Reactions (5.1)
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and may result in solid organ transplant rejection.
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment.
- Withhold or permanently discontinue based on severity and type of reaction.
- Infusion-related reactions: Interrupt, slow the rate of infusion, or permanently discontinue BAVENCIO based on severity of reaction. (5.2)
- Complications of allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Major adverse cardiovascular events: Optimize management of cardiovascular risk factors. Discontinue BAVENCIO in combination with axitinib for Grade 3-4 events. (5.4)
- Embryo-fetal toxicity: BAVENCIO can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.5, 8.1, 8.3)
2 Dosage and Administration
- Premedicate for the first 4 infusions and subsequently as needed. (2.1)
- Merkel Cell Carcinoma: 800 mg every 2 weeks. (2.2)
- Urothelial Carcinoma; 800 mg every 2 weeks. (2.3)
- Renal Cell Carcinoma: 800 mg every 2 weeks in combination with axitinib 5 mg orally twice daily. (2.4)
Administer BAVENCIO as an intravenous infusion over 60 minutes.
3 Dosage Forms and Strengths
Injection: 200 mg/10 mL (20 mg/mL), clear, colorless to slightly yellow solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of BAVENCIO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: neutropenia
Hepatobiliary disorders: sclerosing cholangitis
2.3 Recommended Dosage for Uc
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
2.2 Recommended Dosage for Mcc
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.
2.4 Recommended Dosage for Rcc
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks in combination with axitinib 5 mg orally taken twice daily (12 hours apart) with or without food until disease progression or unacceptable toxicity.
When axitinib is used in combination with BAVENCIO, dose escalation of axitinib above the initial 5 mg dose may be considered at intervals of two weeks or longer. Review the Full Prescribing Information for axitinib prior to initiation.
5.2 Infusion Related Reactions
BAVENCIO can cause severe or life-threatening infusion-related reactions [see Adverse Reactions (6.1)]. Premedicate with antihistamine and acetaminophen prior to the first 4 infusions. Monitor patients for signs and symptoms of infusion-related reactions including pyrexia, chills, flushing, hypotension, dyspnea, wheezing, back pain, abdominal pain, and urticaria. Interrupt or slow the rate of infusion for mild or moderate infusion-related reactions. Stop the infusion and permanently discontinue BAVENCIO for severe (Grade 3) or life-threatening (Grade 4) infusion-related reactions [see Dosage and Administration (2.5) and Adverse Reactions (6.1)].
Infusion-related reactions occurred in 26% of patients treated with BAVENCIO including 3 (0.2%) Grade 4 and 10 (0.5%) Grade 3 infusion-related reactions. Ninety-three percent of patients received premedication with antihistamine and acetaminophen. Eleven (85%) of the 13 patients with Grade ≥ 3 reactions were treated with intravenous corticosteroids. Fifteen percent of patients had infusion-related reactions that occurred after the BAVENCIO infusion was completed.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in the WARNINGS AND PRECAUTIONS section reflect exposure to BAVENCIO 10 mg/kg intravenously every 2 weeks as a single agent in 1854 patients enrolled in the JAVELIN Merkel 200 and JAVELIN Solid Tumor trials and to BAVENCIO 10 mg/kg intravenously every 2 weeks in combination with axitinib 5 mg orally twice daily in 489 patients enrolled in the JAVELIN Renal 100 and JAVELIN Renal 101 trials. In the BAVENCIO monotherapy population, 25% of patients were exposed for ≥ 6 months and 9% were exposed for ≥ 12 months. The population characteristics of BAVENCIO in combination with axitinib are shown below. When BAVENCIO was used in combination with axitinib, 70% of patients were exposed for ≥ 6 months and 31% were exposed for ≥ 12 months. The following criteria were used to classify an adverse reaction as immune-mediated: onset within 90 days after last dose of BAVENCIO, no spontaneous resolution within 7 days of onset, treatment with corticosteroids or other immunosuppressant or hormone replacement therapy, biopsy consistent with immune-mediated reaction, and no other clear etiology.
1.3 Advanced Renal Cell Carcinoma
BAVENCIO in combination with axitinib is indicated for the first-line treatment of patients with advanced renal cell carcinoma (RCC) [see Clinical Studies (14.3)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
14.3 Advanced Renal Cell Carcinoma
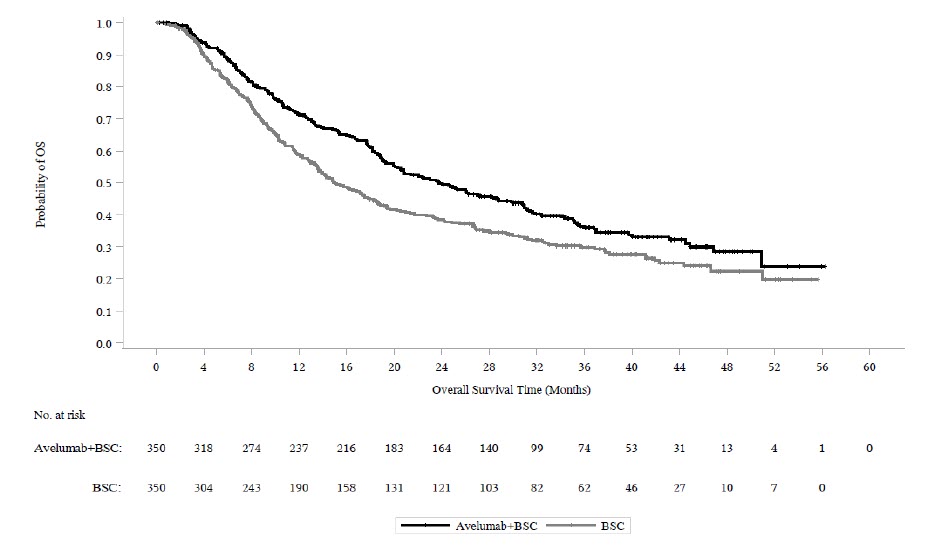
The efficacy and safety of BAVENCIO in combination with axitinib was demonstrated in the JAVELIN Renal 101 trial (NCT02684006), a randomized, multicenter, open-label, study of BAVENCIO in combination with axitinib in 886 patients with untreated advanced RCC regardless of tumor PD-L1 expression [intent-to-treat (ITT) population]. Patients with autoimmune disease or conditions requiring systemic immunosuppression were excluded.
Randomization was stratified according to Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) (0 vs. 1) and region (United States vs. Canada/Western Europe vs. the rest of the world). Patients were randomized (1:1) to one of the following treatment arms:
- BAVENCIO 10 mg/kg intravenous infusion every 2 weeks in combination with axitinib 5 mg twice daily orally (N=442). Patients who tolerated axitinib 5 mg twice daily without Grade 2 or greater axitinib-related adverse events for 2 consecutive weeks could increase to 7 mg and then subsequently to 10 mg twice daily. Axitinib could be interrupted or reduced to 3 mg twice daily and subsequently to 2 mg twice daily to manage toxicity.
- Sunitinib 50 mg once daily orally for 4 weeks followed by 2 weeks off (N=444) until radiographic or clinical progression or unacceptable toxicity.
Treatment with BAVENCIO and axitinib continued until RECIST v1.1-defined progression of disease by Blinded Independent Central Review (BICR) assessment or unacceptable toxicity. Administration BAVENCIO and axitinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed at baseline, after randomization at 6 weeks, then every 6 weeks thereafter up to 18 months after randomization, and every 12 weeks thereafter until documented confirmed disease progression by BICR.
Baseline characteristics were a median age of 61 years (range: 27 to 88); 38% of patients were 65 years or older; 75% were male; 75% were White, 15% Asian, 2% Black, 1% American Indian or Alaskan Native, 7% unknown; 4% were Hispanic or Latino; ECOG PS was 0 (63%) or 1 (37%); and 63% of patients were PD-L1 positive, 28% were PD-L1 negative, and 8% had unknown PD-L1 status. Patient distribution by International Metastatic Renal Cell Carcinoma Database (IMDC) risk groups was 21% favorable, 62% intermediate, and 16% poor.
The major efficacy outcome measures were progression-free survival (PFS), as assessed by an BICR using RECIST v1.1 and overall survival (OS) in patients with PD-L1-positive tumors using a clinical trial assay (PD-L1 expression level ≥ 1%). PFS was statistically significant in patients with PD-L1-positive tumors [HR 0.61 (95% CI: 0.48, 0.79)] and in the ITT population. The final analysis for OS was not statistically significant for either the PD-L1-positive or ITT population.
Efficacy results for the ITT population are presented in Table 14 and Figure 2.
| Efficacy Endpoints | BAVENCIO plus Axitinib (N=442) |
Sunitinib (N=444) |
|---|---|---|
| BICR: Blinded Independent Central Review; CI: Confidence interval; NE: Not estimable; NS: not statistically significant. | ||
|
Progression-Free Survival (PFS) Based on BICR assessment.
|
||
| Events (%) | 180 (41) | 216 (49) |
| Median in months (95% CI) | 13.8 (11.1, NE) | 8.4 (6.9, 11.1) |
| Hazard ratio (95% CI) | 0.69 (0.56, 0.84) | |
| p-value p-value based on 2-sided stratified log-rank.
|
0.0002 | |
| Overall Survival (OS) | ||
| Events (%) | 283 (64) | 295 (66) |
| Median in months (95% CI) | 44.8 (39.7, 51.1) | 38.9 (31.4, 45.2) |
| Hazard ratio (95% CI) | 0.88 (0.75, 1.04) | |
| p-value | NS | |
| Confirmed Objective Response Rate (ORR) | ||
| Objective Response Rate n (%) | 227 (51.4) | 114 (25.7) |
| (95% CI) | (46.6, 56.1) | (21.7, 30.0) |
| Complete Response (CR) n (%) | 15 (3.4) | 8 (1.8) |
| Partial Response (PR) n (%) | 212 (48) | 106 (24) |
Figure 2: K-M Estimates for PFS based on BICR Assessment – ITT
1.1 Metastatic Merkel Cell Carcinoma
BAVENCIO (avelumab) is indicated for the treatment of adults and pediatric patients 12 years and older with metastatic Merkel cell carcinoma (MCC) [see Clinical Studies (14.1)].
16 How Supplied/storage and Handling
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
5.3 Complications of Allogeneic Hsct
Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after being treated with a PD-1/PD-L1 blocking antibody. Transplant-related complications include hyperacute graft-versus-host-disease (GVHD), acute GVHD, chronic GVHD, hepatic veno-occlusive disease (VOD) after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between PD-1/PD-L1 blockade and allogeneic HSCT.
Follow patients closely for evidence of transplant-related complications and intervene promptly. Consider the benefit versus risks of treatment with a PD-1/PD-L1 blocking antibody prior to or after an allogeneic HSCT.
14.1 Metastatic Merkel Cell Carcinoma
The efficacy and safety of BAVENCIO was demonstrated in the JAVELIN Merkel 200 trial (NCT02155647), an open-label, single--arm, multi-center study conducted in patients with histologically confirmed metastatic MCC. This trial consisted of two parts; Part A enrolled patients with metastatic MCC whose disease had progressed on or after chemotherapy administered for distant metastatic disease, and Part B enrolled patients with metastatic MCC who were treatment-naïve. The trial excluded patients with autoimmune disease; medical conditions requiring systemic immunosuppression; prior organ or allogeneic stem cell transplantation; prior treatment with anti-PD-1, anti-PD-L1, or anti-CTLA-4 antibodies; CNS metastases; infection with HIV, hepatitis B, or hepatitis C; or ECOG performance score ≥ 2.
Patients received BAVENCIO 10 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity. Patients with radiological disease progression not associated with significant clinical deterioration, defined as no new or worsening symptoms, no change in performance status for greater than 2 weeks, and no need for salvage therapy, could continue treatment. Tumor response assessments were performed every 6 weeks.
The major efficacy outcome measures were confirmed overall response rate (ORR) according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 as assessed by a blinded independent central review committee (IRC) and IRC-assessed duration of response.
13.2 Animal Toxicology And/or Pharmacology
In animal models, inhibition of PD-L1/PD-1 signaling increased the severity of some infections and enhanced inflammatory responses. M. tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-L1 and PD-1 knockout mice and mice receiving PD-L1 blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus.
Principal Display Panel 10 Ml Vial Carton
NDC 44087-3535-1
BAVENCIO®
(avelumab)
Injection
200 mg/10 mL
(20 mg/mL)
For intravenous infusion
after dilution
Single-dose vial
Discard unused portion.
Dispense the enclosed
Medication Guide to each
patient.
1 vial
Rx only
EMD
SERONO
5.4 Major Adverse Cardiovascular Events (mace)
BAVENCIO in combination with axitinib can cause severe and fatal cardiovascular events. Consider baseline and periodic evaluations of left ventricular ejection fraction. Monitor for signs and symptoms of cardiovascular events. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Discontinue BAVENCIO and axitinib for Grade 3-4 cardiovascular events.
MACE occurred in 7% of patients with advanced RCC treated with BAVENCIO in combination with axitinib compared to 3.4% treated with sunitinib in a randomized trial, JAVELIN Renal 101. These events included death due to cardiac events (1.4%), Grade 3-4 myocardial infarction (2.8%), and Grade 3-4 congestive heart failure (1.8%). Median time to onset of MACE was 4.2 months (range: 2 days to 24.5 months).
5.1 Severe and Fatal Immune Mediated Adverse Reactions
BAVENCIO is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under Warnings and Precautions may not include all possible severe and fatal immune-mediated reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue. Immune-mediated adverse reactions can occur at any time after starting treatment with a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue BAVENCIO depending on severity [see Dosage and Administration (2.5)]. In general, if BAVENCIO requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic corticosteroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted to assess the potential of avelumab for genotoxicity or carcinogenicity.
Fertility studies have not been conducted with avelumab; however, an assessment of male and female reproductive organs was included in 3-month repeat-dose toxicity study in Cynomolgus monkeys. Weekly administration of avelumab did not result in any notable effects in the male and female reproductive organs.
Structured Label Content
Section 42229-5 (42229-5)
First-Line Maintenance Treatment of Urothelial Carcinoma
BAVENCIO is indicated for the maintenance treatment of patients with locally advanced or metastatic urothelial carcinoma (UC) that has not progressed with first-line platinum-containing chemotherapy [see Clinical Studies (14.2)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 06/2025 | |||||
|
MEDICATION GUIDE
BAVENCIO® (buh-VEN-see-oh) (avelumab) injection |
||||||
|
What is the most important information I should know about BAVENCIO?
BAVENCIO is a medicine that may treat certain cancers by working with your immune system. BAVENCIO can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during treatment or even after your treatment has ended. Call or see your healthcare provider right away if you get any new or worsening signs or symptoms, including: Lung problems. |
||||||
|
|
|
||||
Intestinal problems.
|
||||||
|
|
|||||
| Hormone gland problems. | ||||||
|
|
|||||
| Kidney problems. | ||||||
|
|
|||||
| Skin problems. | ||||||
|
|
|||||
Problems can also happen in other organs and tissues. These are not all of the signs or symptoms of immune system problems that can happen with BAVENCIO. Call or see your healthcare provider right away for any new or worsening signs or symptoms, which may include:
|
||||||
| Infusion-related reactions can sometimes be severe or life-threatening. Signs and symptoms of infusion-related reactions may include: | ||||||
|
|
|||||
|
Complications, including graft-versus-host-disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with BAVENCIO. Your healthcare provider will monitor you for these complications. Heart problems. When BAVENCIO is used with the medicine axitinib, severe heart problems can happen and can lead to death. Signs and symptoms of heart problems may include: |
||||||
|
|
|||||
| Getting medical treatment right away may help keep these problems from becoming more serious. Your healthcare provider will check you for these problems during your treatment with BAVENCIO. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with BAVENCIO if you have severe side effects. | ||||||
|
What is BAVENCIO?
BAVENCIO is a prescription medicine used to treat:
|
||||||
Before you receive BAVENCIO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||||
How will I receive BAVENCIO?
|
||||||
|
What are the possible side effects of BAVENCIO? BAVENCIO can cause serious side effects, including: The most common side effects of BAVENCIO in people with MCC include: |
||||||
|
|
|||||
| The most common side effects of BAVENCIO as maintenance treatment in people with UC whose cancer responded or stabilized after platinum-containing chemotherapy as first treatment include: | ||||||
|
|
|||||
| The most common side effects of BAVENCIO in people with UC after platinum-containing chemotherapy that did not work, or is no longer working, include: | ||||||
|
|
|||||
| The most common side effects of BAVENCIO when given with axitinib in people with RCC include: | ||||||
|
|
|||||
| These are not all the possible side effects of BAVENCIO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||
|
General information about the safe and effective use of BAVENCIO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about BAVENCIO that is written for health professionals. |
||||||
|
What are the ingredients in BAVENCIO?
Active ingredient: avelumab Inactive ingredients: D-mannitol, glacial acetic acid, polysorbate 20, sodium hydroxide, and Water for Injection Manufactured by: EMD Serono, Inc., Boston, MA 02210, USA, U.S. License No. 1773. Marketed by: EMD Serono, Inc., MA, USA. BAVENCIO is a trademark of Merck KGaA, Darmstadt, Germany. For more information, call toll-free 1-844-826-8371 or go to www.bavencio.com. |
||||||
Section 44425-7 (44425-7)
Store refrigerated at 36°F to 46°F (2°C to 8°C) in original package to protect from light.
Do not freeze or shake the vial.
The vial stopper is not made with natural rubber latex.
11 Description (11 DESCRIPTION)
Avelumab is a programmed death ligand1 (PD-L1) blocking antibody. Avelumab is a human IgG1 lambda monoclonal antibody produced in Chinese hamster ovary cells and has a molecular weight of approximately 147 kDa.
BAVENCIO (avelumab) Injection for intravenous use is a sterile, preservative-free, non-pyrogenic, clear, colorless to slightly yellow solution. Each single-dose vial contains 200 mg avelumab in 10 mL (20 mg/mL). Each mL contains 20 mg avelumab, D-mannitol (51 mg), glacial acetic acid (0.6 mg), polysorbate 20 (0.5 mg), sodium hydroxide (0.3 mg), and Water for Injection. The pH range of the solution is 5.0 – 5.6.
2.1 Premedication
Premedicate patients with an antihistamine and with acetaminophen prior to the first 4 infusions of BAVENCIO. Premedication should be administered for subsequent BAVENCIO doses based upon clinical judgment and presence/severity of prior infusion reactions [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
8.4 Pediatric Use
The safety and effectiveness of BAVENCIO have been established in pediatric patients aged 12 years and older for metastatic MCC. Use of BAVENCIO in this age group is supported by evidence from adequate and well-controlled studies of BAVENCIO in adults with additional population pharmacokinetic data demonstrating that age and body weight had no clinically meaningful effect on the steady state exposure of avelumab, that drug exposure is generally similar between adults and pediatric patients age 12 years and older for monoclonal antibodies, and that the course of MCC is sufficiently similar in adult and pediatric patients to allow extrapolation of data in adults to pediatric patients. The recommended dose in pediatric patients 12 years of age or greater is the same as that in adults [see Dosage and Administration (2.2), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness of BAVENCIO have not been established in pediatric patients less than 12 years of age.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of BAVENCIO or of other avelumab products.
ADA responses following administration of BAVENCIO 10 mg/kg every 2 weeks were evaluated during the respective treatment periods in each trial. The ADA and neutralizing antibody (nAb) incidences are listed in Table 9.
| Trial Name Details of each treatment regimen are described in Section 14 [see Clinical Studies (14)].
|
Treatment Period (months) | ADA | nAb |
|---|---|---|---|
| ADA: anti-avelumab antibodies; UC: urothelial carcinoma | |||
| JAVELIN Merkel 200 Part A | 48 | 8.9% (7/79) | 71% (5/7) |
| JAVELIN Merkel 200 Part B | 35 | 8.2% (9/110) | 89% (8/9) |
| JAVELIN Bladder 100 | 37 | 19% (62/326) | 97% (60/62) |
| JAVELIN Solid Tumor, UC Cohort | 59 | 18% (41/226) | Not tested |
| JAVELIN Renal 101 | 29 | 16% (65/411) | 78% (51/65) |
In patients with advanced UC or advanced RCC, avelumab clearance was approximately 15% higher in patients who tested positive for ADA as compared to clearance in patients who tested negative for ADA; which is not considered clinically meaningful. The effect of ADA on the efficacy or safety could not be determined due to the low occurrence of ADAs.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1)]
- Infusion-related reactions [see Warnings and Precautions (5.2)]
- Complications of allogeneic HSCT [see Warnings and Precautions (5.3)]
- Major adverse cardiovascular events [see Warnings and Precautions (5.4)]
12.2 Pharmacodynamics
Avelumab exposure-response relationships and the time course of pharmacodynamic response are not fully characterized.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of avelumab as a single agent was characterized in patients who received BAVENCIO at doses ranging from 1 to 20 mg/kg every 2 weeks (0.1 to 2 times of the approved recommended dosage). The exposure of avelumab increased dose-proportionally from 10 to 20 mg/kg every 2 weeks. Steady-state concentrations of avelumab were reached after approximately 4 to 6 weeks (2 to 3 cycles) and the systemic accumulation was 1.25-fold.
2.5 Dose Modifications
No dose reduction for BAVENCIO is recommended. In general, withhold BAVENCIO for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue BAVENCIO for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating corticosteroids.
Dosage modifications for BAVENCIO for adverse reactions that require management different from these general guidelines are summarized in Table 1.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.03
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, SJS = Stevens-Johnson syndrome, TEN = toxic epidermal necrosis, DRESS = drug rash with eosinophilia and systemic symptoms | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of last dose or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating corticosteroids.
|
| Grade 3 or 4 | Permanently discontinue | |
| Colitis | Grade 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
| Hepatitis with no tumor involvement of the liver For liver enzyme elevations in patients treated with combination therapy, see Table 2 |
AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue BAVENCIO based on recommendations for hepatitis where there is no tumor involvement of the liver.
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grade 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grade 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis | Grade 2, 3 or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grade 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Infusion-related reactions [see Warnings and Precautions (5.2)] | Grade 1 or 2 | Interrupt or slow the rate of infusion |
| Grade 3 or 4 | Permanently discontinue |
Table 2 presents dosage modifications that are different from those described above in Table 1 for BAVENCIO used as monotherapy or in the Full Prescribing Information for the drug administered in combination.
| Treatment | Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.03
|
Dosage Modification |
|---|---|---|---|
| BAVENCIO in combination with axitinib | Liver enzyme elevations | ALT or AST at least 3 times ULN but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN | Withhold both BAVENCIO and axitinib until adverse reactions recover to Grades 0-1 Consider corticosteroid therapy
Consider rechallenge with BAVENCIO or axitinib or sequential rechallenge with both BAVENCIO and axitinib after recovery Dose reduction according to the axitinib Full Prescribing Information should be considered if rechallenging with axitinib.
|
| ALT or AST at least 10 times ULN or more than 3 times ULN with concurrent total bilirubin at least 2 times ULN | Permanently discontinue both BAVENCIO and axitinib |
1 Indications and Usage (1 INDICATIONS AND USAGE)
BAVENCIO is a programmed death ligand-1 (PD-L1) blocking antibody indicated for:
Merkel Cell Carcinoma (MCC)
Urothelial Carcinoma (UC)
- Maintenance treatment of patients with locally advanced or metastatic UC that has not progressed with first-line platinum-containing chemotherapy. (1.2, 14.2)
- Patients with locally advanced or metastatic UC who:
Renal Cell Carcinoma (RCC)
12.1 Mechanism of Action
PD-L1 may be expressed on tumor cells and tumor-infiltrating immune cells and can contribute to the inhibition of the anti-tumor immune response in the tumor microenvironment. Binding of PD-L1 to the PD-1 and B7.1 receptors found on T cells and antigen presenting cells suppresses cytotoxic T-cell activity, T-cell proliferation, and cytokine production. Avelumab binds PD-L1 and blocks the interaction between PD-L1 and its receptors PD-1 and B7.1. This interaction releases the inhibitory effects of PD-L1 on the immune response resulting in the restoration of immune responses, including anti-tumor immune responses. Avelumab has also been shown to induce antibody-dependent cell-mediated cytotoxicity (ADCC) in vitro. In syngeneic mouse tumor models, blocking PD-L1 activity resulted in decreased tumor growth.
5.5 Embryo Fetal Toxicity (5.5 Embryo-Fetal Toxicity)
Based on its mechanism of action, BAVENCIO can cause fetal harm when administered to a pregnant woman. Animal studies have demonstrated that inhibition of the PD-1/PD-L1 pathway can lead to increased risk of immune-mediated rejection of the developing fetus resulting in fetal death. If this drug is used during pregnancy, or if the patient becomes pregnant while taking BAVENCIO, inform the patient of the potential risk to a fetus. Advise females of childbearing potential to use effective contraception during treatment with BAVENCIO and for at least one month after the last dose of BAVENCIO [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
Immune-Mediated Adverse Reactions (5.1)
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and may result in solid organ transplant rejection.
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment.
- Withhold or permanently discontinue based on severity and type of reaction.
- Infusion-related reactions: Interrupt, slow the rate of infusion, or permanently discontinue BAVENCIO based on severity of reaction. (5.2)
- Complications of allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Major adverse cardiovascular events: Optimize management of cardiovascular risk factors. Discontinue BAVENCIO in combination with axitinib for Grade 3-4 events. (5.4)
- Embryo-fetal toxicity: BAVENCIO can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.5, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Premedicate for the first 4 infusions and subsequently as needed. (2.1)
- Merkel Cell Carcinoma: 800 mg every 2 weeks. (2.2)
- Urothelial Carcinoma; 800 mg every 2 weeks. (2.3)
- Renal Cell Carcinoma: 800 mg every 2 weeks in combination with axitinib 5 mg orally twice daily. (2.4)
Administer BAVENCIO as an intravenous infusion over 60 minutes.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 200 mg/10 mL (20 mg/mL), clear, colorless to slightly yellow solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of BAVENCIO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: neutropenia
Hepatobiliary disorders: sclerosing cholangitis
2.3 Recommended Dosage for Uc (2.3 Recommended Dosage for UC)
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
2.2 Recommended Dosage for Mcc (2.2 Recommended Dosage for MCC)
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.
2.4 Recommended Dosage for Rcc (2.4 Recommended Dosage for RCC)
The recommended dosage of BAVENCIO is 800 mg administered as an intravenous infusion over 60 minutes every 2 weeks in combination with axitinib 5 mg orally taken twice daily (12 hours apart) with or without food until disease progression or unacceptable toxicity.
When axitinib is used in combination with BAVENCIO, dose escalation of axitinib above the initial 5 mg dose may be considered at intervals of two weeks or longer. Review the Full Prescribing Information for axitinib prior to initiation.
5.2 Infusion Related Reactions (5.2 Infusion-Related Reactions)
BAVENCIO can cause severe or life-threatening infusion-related reactions [see Adverse Reactions (6.1)]. Premedicate with antihistamine and acetaminophen prior to the first 4 infusions. Monitor patients for signs and symptoms of infusion-related reactions including pyrexia, chills, flushing, hypotension, dyspnea, wheezing, back pain, abdominal pain, and urticaria. Interrupt or slow the rate of infusion for mild or moderate infusion-related reactions. Stop the infusion and permanently discontinue BAVENCIO for severe (Grade 3) or life-threatening (Grade 4) infusion-related reactions [see Dosage and Administration (2.5) and Adverse Reactions (6.1)].
Infusion-related reactions occurred in 26% of patients treated with BAVENCIO including 3 (0.2%) Grade 4 and 10 (0.5%) Grade 3 infusion-related reactions. Ninety-three percent of patients received premedication with antihistamine and acetaminophen. Eleven (85%) of the 13 patients with Grade ≥ 3 reactions were treated with intravenous corticosteroids. Fifteen percent of patients had infusion-related reactions that occurred after the BAVENCIO infusion was completed.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in the WARNINGS AND PRECAUTIONS section reflect exposure to BAVENCIO 10 mg/kg intravenously every 2 weeks as a single agent in 1854 patients enrolled in the JAVELIN Merkel 200 and JAVELIN Solid Tumor trials and to BAVENCIO 10 mg/kg intravenously every 2 weeks in combination with axitinib 5 mg orally twice daily in 489 patients enrolled in the JAVELIN Renal 100 and JAVELIN Renal 101 trials. In the BAVENCIO monotherapy population, 25% of patients were exposed for ≥ 6 months and 9% were exposed for ≥ 12 months. The population characteristics of BAVENCIO in combination with axitinib are shown below. When BAVENCIO was used in combination with axitinib, 70% of patients were exposed for ≥ 6 months and 31% were exposed for ≥ 12 months. The following criteria were used to classify an adverse reaction as immune-mediated: onset within 90 days after last dose of BAVENCIO, no spontaneous resolution within 7 days of onset, treatment with corticosteroids or other immunosuppressant or hormone replacement therapy, biopsy consistent with immune-mediated reaction, and no other clear etiology.
1.3 Advanced Renal Cell Carcinoma
BAVENCIO in combination with axitinib is indicated for the first-line treatment of patients with advanced renal cell carcinoma (RCC) [see Clinical Studies (14.3)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
14.3 Advanced Renal Cell Carcinoma
The efficacy and safety of BAVENCIO in combination with axitinib was demonstrated in the JAVELIN Renal 101 trial (NCT02684006), a randomized, multicenter, open-label, study of BAVENCIO in combination with axitinib in 886 patients with untreated advanced RCC regardless of tumor PD-L1 expression [intent-to-treat (ITT) population]. Patients with autoimmune disease or conditions requiring systemic immunosuppression were excluded.
Randomization was stratified according to Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) (0 vs. 1) and region (United States vs. Canada/Western Europe vs. the rest of the world). Patients were randomized (1:1) to one of the following treatment arms:
- BAVENCIO 10 mg/kg intravenous infusion every 2 weeks in combination with axitinib 5 mg twice daily orally (N=442). Patients who tolerated axitinib 5 mg twice daily without Grade 2 or greater axitinib-related adverse events for 2 consecutive weeks could increase to 7 mg and then subsequently to 10 mg twice daily. Axitinib could be interrupted or reduced to 3 mg twice daily and subsequently to 2 mg twice daily to manage toxicity.
- Sunitinib 50 mg once daily orally for 4 weeks followed by 2 weeks off (N=444) until radiographic or clinical progression or unacceptable toxicity.
Treatment with BAVENCIO and axitinib continued until RECIST v1.1-defined progression of disease by Blinded Independent Central Review (BICR) assessment or unacceptable toxicity. Administration BAVENCIO and axitinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed at baseline, after randomization at 6 weeks, then every 6 weeks thereafter up to 18 months after randomization, and every 12 weeks thereafter until documented confirmed disease progression by BICR.
Baseline characteristics were a median age of 61 years (range: 27 to 88); 38% of patients were 65 years or older; 75% were male; 75% were White, 15% Asian, 2% Black, 1% American Indian or Alaskan Native, 7% unknown; 4% were Hispanic or Latino; ECOG PS was 0 (63%) or 1 (37%); and 63% of patients were PD-L1 positive, 28% were PD-L1 negative, and 8% had unknown PD-L1 status. Patient distribution by International Metastatic Renal Cell Carcinoma Database (IMDC) risk groups was 21% favorable, 62% intermediate, and 16% poor.
The major efficacy outcome measures were progression-free survival (PFS), as assessed by an BICR using RECIST v1.1 and overall survival (OS) in patients with PD-L1-positive tumors using a clinical trial assay (PD-L1 expression level ≥ 1%). PFS was statistically significant in patients with PD-L1-positive tumors [HR 0.61 (95% CI: 0.48, 0.79)] and in the ITT population. The final analysis for OS was not statistically significant for either the PD-L1-positive or ITT population.
Efficacy results for the ITT population are presented in Table 14 and Figure 2.
| Efficacy Endpoints | BAVENCIO plus Axitinib (N=442) |
Sunitinib (N=444) |
|---|---|---|
| BICR: Blinded Independent Central Review; CI: Confidence interval; NE: Not estimable; NS: not statistically significant. | ||
|
Progression-Free Survival (PFS) Based on BICR assessment.
|
||
| Events (%) | 180 (41) | 216 (49) |
| Median in months (95% CI) | 13.8 (11.1, NE) | 8.4 (6.9, 11.1) |
| Hazard ratio (95% CI) | 0.69 (0.56, 0.84) | |
| p-value p-value based on 2-sided stratified log-rank.
|
0.0002 | |
| Overall Survival (OS) | ||
| Events (%) | 283 (64) | 295 (66) |
| Median in months (95% CI) | 44.8 (39.7, 51.1) | 38.9 (31.4, 45.2) |
| Hazard ratio (95% CI) | 0.88 (0.75, 1.04) | |
| p-value | NS | |
| Confirmed Objective Response Rate (ORR) | ||
| Objective Response Rate n (%) | 227 (51.4) | 114 (25.7) |
| (95% CI) | (46.6, 56.1) | (21.7, 30.0) |
| Complete Response (CR) n (%) | 15 (3.4) | 8 (1.8) |
| Partial Response (PR) n (%) | 212 (48) | 106 (24) |
Figure 2: K-M Estimates for PFS based on BICR Assessment – ITT
1.1 Metastatic Merkel Cell Carcinoma
BAVENCIO (avelumab) is indicated for the treatment of adults and pediatric patients 12 years and older with metastatic Merkel cell carcinoma (MCC) [see Clinical Studies (14.1)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
BAVENCIO (avelumab) Injection is a sterile, preservative-free, and clear, colorless to slightly yellow solution for intravenous infusion supplied as a single-dose vial of 200 mg/10 mL (20 mg/mL), individually packed into a carton (NDC 44087-3535-1).
5.3 Complications of Allogeneic Hsct (5.3 Complications of Allogeneic HSCT)
Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after being treated with a PD-1/PD-L1 blocking antibody. Transplant-related complications include hyperacute graft-versus-host-disease (GVHD), acute GVHD, chronic GVHD, hepatic veno-occlusive disease (VOD) after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between PD-1/PD-L1 blockade and allogeneic HSCT.
Follow patients closely for evidence of transplant-related complications and intervene promptly. Consider the benefit versus risks of treatment with a PD-1/PD-L1 blocking antibody prior to or after an allogeneic HSCT.
14.1 Metastatic Merkel Cell Carcinoma
The efficacy and safety of BAVENCIO was demonstrated in the JAVELIN Merkel 200 trial (NCT02155647), an open-label, single--arm, multi-center study conducted in patients with histologically confirmed metastatic MCC. This trial consisted of two parts; Part A enrolled patients with metastatic MCC whose disease had progressed on or after chemotherapy administered for distant metastatic disease, and Part B enrolled patients with metastatic MCC who were treatment-naïve. The trial excluded patients with autoimmune disease; medical conditions requiring systemic immunosuppression; prior organ or allogeneic stem cell transplantation; prior treatment with anti-PD-1, anti-PD-L1, or anti-CTLA-4 antibodies; CNS metastases; infection with HIV, hepatitis B, or hepatitis C; or ECOG performance score ≥ 2.
Patients received BAVENCIO 10 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity. Patients with radiological disease progression not associated with significant clinical deterioration, defined as no new or worsening symptoms, no change in performance status for greater than 2 weeks, and no need for salvage therapy, could continue treatment. Tumor response assessments were performed every 6 weeks.
The major efficacy outcome measures were confirmed overall response rate (ORR) according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 as assessed by a blinded independent central review committee (IRC) and IRC-assessed duration of response.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In animal models, inhibition of PD-L1/PD-1 signaling increased the severity of some infections and enhanced inflammatory responses. M. tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-L1 and PD-1 knockout mice and mice receiving PD-L1 blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus.
Principal Display Panel 10 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton)
NDC 44087-3535-1
BAVENCIO®
(avelumab)
Injection
200 mg/10 mL
(20 mg/mL)
For intravenous infusion
after dilution
Single-dose vial
Discard unused portion.
Dispense the enclosed
Medication Guide to each
patient.
1 vial
Rx only
EMD
SERONO
5.4 Major Adverse Cardiovascular Events (mace) (5.4 Major Adverse Cardiovascular Events (MACE))
BAVENCIO in combination with axitinib can cause severe and fatal cardiovascular events. Consider baseline and periodic evaluations of left ventricular ejection fraction. Monitor for signs and symptoms of cardiovascular events. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Discontinue BAVENCIO and axitinib for Grade 3-4 cardiovascular events.
MACE occurred in 7% of patients with advanced RCC treated with BAVENCIO in combination with axitinib compared to 3.4% treated with sunitinib in a randomized trial, JAVELIN Renal 101. These events included death due to cardiac events (1.4%), Grade 3-4 myocardial infarction (2.8%), and Grade 3-4 congestive heart failure (1.8%). Median time to onset of MACE was 4.2 months (range: 2 days to 24.5 months).
5.1 Severe and Fatal Immune Mediated Adverse Reactions (5.1 Severe and Fatal Immune-Mediated Adverse Reactions)
BAVENCIO is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under Warnings and Precautions may not include all possible severe and fatal immune-mediated reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue. Immune-mediated adverse reactions can occur at any time after starting treatment with a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue BAVENCIO depending on severity [see Dosage and Administration (2.5)]. In general, if BAVENCIO requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic corticosteroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted to assess the potential of avelumab for genotoxicity or carcinogenicity.
Fertility studies have not been conducted with avelumab; however, an assessment of male and female reproductive organs was included in 3-month repeat-dose toxicity study in Cynomolgus monkeys. Weekly administration of avelumab did not result in any notable effects in the male and female reproductive organs.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:45.434493 · Updated: 2026-03-14T22:27:57.216256