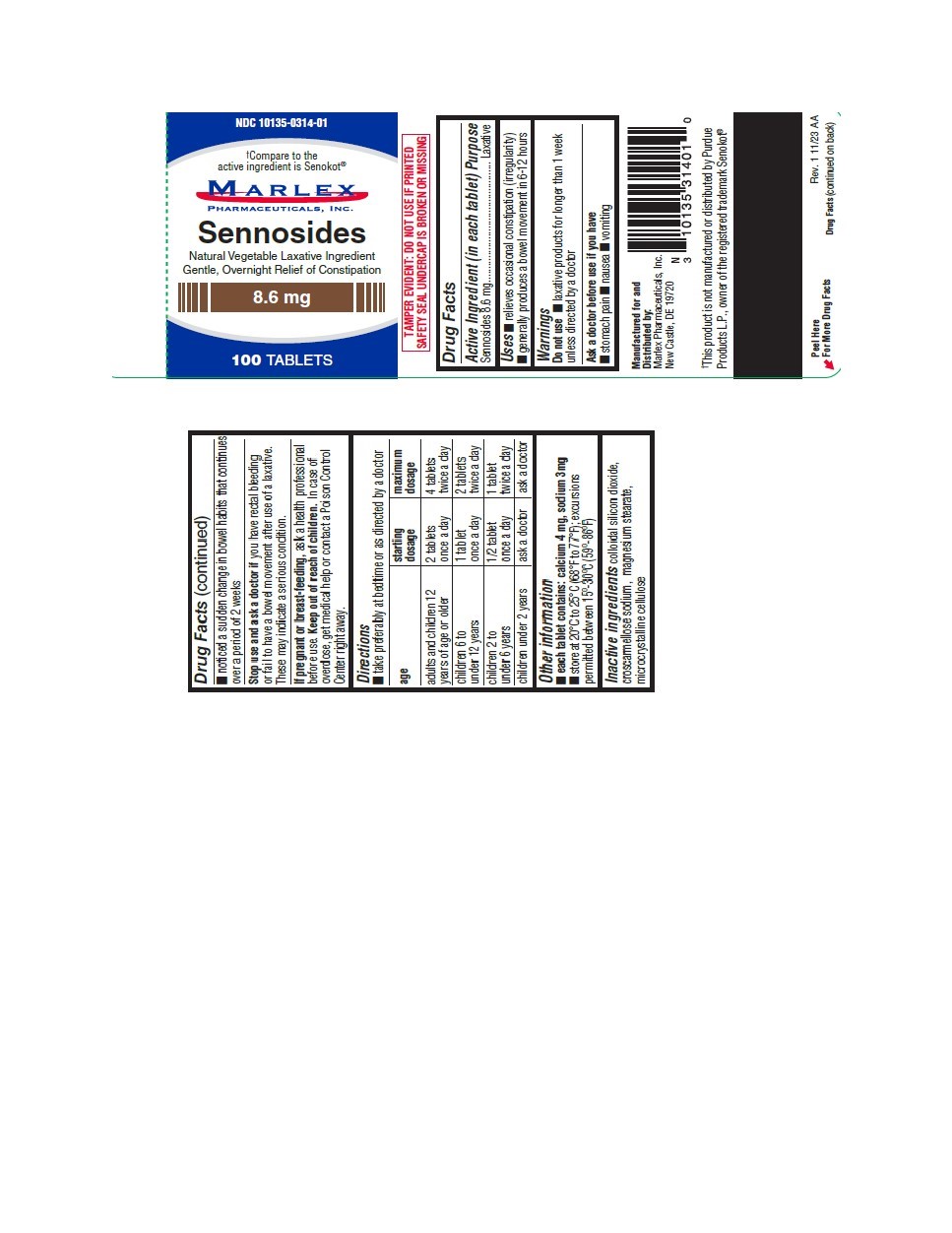
Sennosides
5ca287a3-c4f6-402d-978e-cd1328bdbc47
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SENNOSIDES 8.6mg Tablet Marlex Pharmaceuticals, Inc. Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Drug Facts
Purpose
Laxative
Medication Information
Purpose
Laxative
Description
SENNOSIDES 8.6mg Tablet Marlex Pharmaceuticals, Inc. Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Drug Facts
Uses
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 6-12 hours
Section 42229-5
SENNOSIDES 8.6mg Tablet
Marlex Pharmaceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
| adults and children 12 years of age and older | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years of age | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years of age | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Do Not Use
- laxative products for longer than 1 week unless directed by a doctor
Other Information
- Each tablet contains:
Calcium 4
mg
, Sodium 3mg
- Store at 20°C to 25°C (68°F to 77°F) excursions permitted between 15° to 30°C (59°-86°F).
Inactive Ingredients
Colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose.
*Compare to Senokot®
Product color may vary slightly due to nature of ingredients.
*Marlex Pharmaceuticals, Inc. is not affiliated with the owner of the registered trademark Senokot®.
Principal Display Panel
NDC 10135-0314-01
Sennosides
8.6 mg
100 TABLETS
Stop Use and Ask A Doctor If
you have rectal bleeding or fail to have a bowel movement after use of a laxative. These may indicate a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Sennosides 8.6 mg
Ask A Doctor Before Use If You Have
- stomach Pain
- Nausea
- Vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
Structured Label Content
Uses
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 6-12 hours
Section 42229-5 (42229-5)
SENNOSIDES 8.6mg Tablet
Marlex Pharmaceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Purpose
Laxative
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
| adults and children 12 years of age and older | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years of age | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years of age | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Do Not Use (Do not use)
- laxative products for longer than 1 week unless directed by a doctor
Other Information (Other information)
- Each tablet contains:
Calcium 4
mg
, Sodium 3mg
- Store at 20°C to 25°C (68°F to 77°F) excursions permitted between 15° to 30°C (59°-86°F).
Inactive Ingredients (Inactive ingredients)
Colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose.
*Compare to Senokot®
Product color may vary slightly due to nature of ingredients.
*Marlex Pharmaceuticals, Inc. is not affiliated with the owner of the registered trademark Senokot®.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 10135-0314-01
Sennosides
8.6 mg
100 TABLETS
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
you have rectal bleeding or fail to have a bowel movement after use of a laxative. These may indicate a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Sennosides 8.6 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- stomach Pain
- Nausea
- Vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:18.948998 · Updated: 2026-03-14T23:10:35.893664