5c0805c7-9b92-4c08-877c-64e720308430
34390-5
HUMAN OTC DRUG LABEL
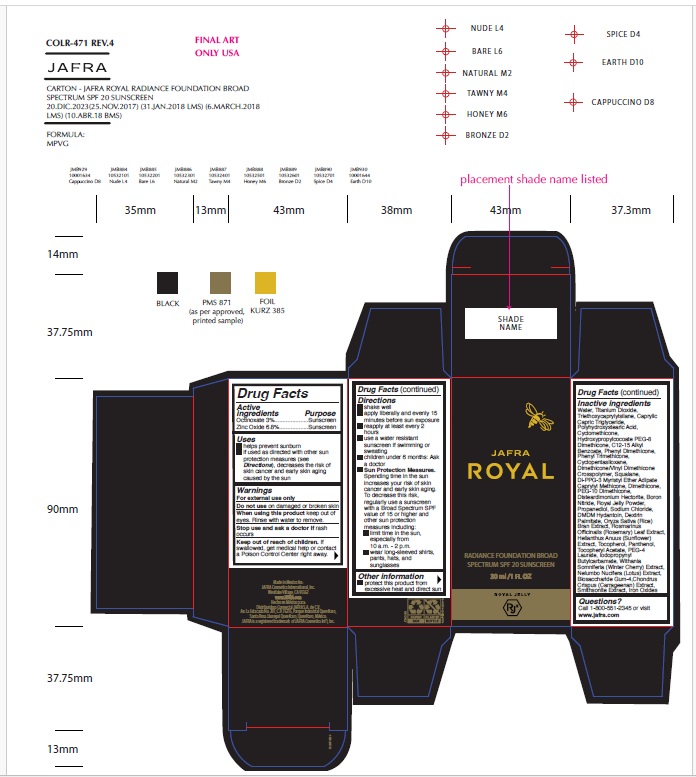
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Octinoxate 3% .................. Sunscreen Zinc Oxide 6.8% ................ Sunscreen
Purpose
Octinoxate 3% .................. Sunscreen Zinc Oxide 6.8% ................ Sunscreen
Medication Information
Purpose
Octinoxate 3% .................. Sunscreen Zinc Oxide 6.8% ................ Sunscreen
Description
Octinoxate 3% .................. Sunscreen Zinc Oxide 6.8% ................ Sunscreen
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 50565-1
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- shake well
- apply liberally and evenly 15 minutes before sun exposure
- reapply at least every 2 hours
- use a water resistant sunscreen if swimming or sweating
- children under 6 months of age: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m.-2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
Questions?
Call 1-800-551-2345 or visit www.jafra.com
Product Label
Other Information
- protect this product from excessive heat and direct sun
Inactive Ingredients
Water, Caprylic/Capric Triglyceride, C12-15 Alkyl Benzoate, Hydroxypropylcocoate PEG-8 Dimethicone, Petroleum Distillates, Phenyl Trimethicone, Phenyl Dimethicone, Di-PPG-3 Myristyl Ether Adipate, Squalane, Caprylyl Methicone, C10-11 Isoalkane, Boron Nitride, Disteardimonium Hectorite, Propanediol, Sodium Chloride, Phenoxyethanol, Dimethicone, Triethoxycaprylylsilane, Dimethicone/Vinyl Dimethicone Crosspolymer, PEG-10 Dimethicone, Propylene Carbonate, Polyhydroxystearic Acid, Oryza Sativa (Rice) Bran Extract, Tocopheryl Acetate, Panthenol, Royal Jelly Powder, Biosaccharide Gum-4, Smithsonite Extract, Ethylhexylglycerin, Withania Somnifera Extract, Laureth-3, Laureth-25, Helianthus Annuus (Sunflower) Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Tocopherol, Chondrus Crispus Extract, Nelumbo Nucifera Extract, Titanium Dioxide (CI 77891), Iron Oxides (CI 77491/CI 77492/CI 77499)
Active Ingredients purpose
Octinoxate 3% .................. Sunscreen
Zinc Oxide 6.8% ................ Sunscreen
Structured Label Content
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 50565-1 (50565-1)
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- shake well
- apply liberally and evenly 15 minutes before sun exposure
- reapply at least every 2 hours
- use a water resistant sunscreen if swimming or sweating
- children under 6 months of age: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m.-2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
Questions?
Call 1-800-551-2345 or visit www.jafra.com
Product Label (Product label)
Other Information (Other information)
- protect this product from excessive heat and direct sun
Inactive Ingredients (Inactive ingredients)
Water, Caprylic/Capric Triglyceride, C12-15 Alkyl Benzoate, Hydroxypropylcocoate PEG-8 Dimethicone, Petroleum Distillates, Phenyl Trimethicone, Phenyl Dimethicone, Di-PPG-3 Myristyl Ether Adipate, Squalane, Caprylyl Methicone, C10-11 Isoalkane, Boron Nitride, Disteardimonium Hectorite, Propanediol, Sodium Chloride, Phenoxyethanol, Dimethicone, Triethoxycaprylylsilane, Dimethicone/Vinyl Dimethicone Crosspolymer, PEG-10 Dimethicone, Propylene Carbonate, Polyhydroxystearic Acid, Oryza Sativa (Rice) Bran Extract, Tocopheryl Acetate, Panthenol, Royal Jelly Powder, Biosaccharide Gum-4, Smithsonite Extract, Ethylhexylglycerin, Withania Somnifera Extract, Laureth-3, Laureth-25, Helianthus Annuus (Sunflower) Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Tocopherol, Chondrus Crispus Extract, Nelumbo Nucifera Extract, Titanium Dioxide (CI 77891), Iron Oxides (CI 77491/CI 77492/CI 77499)
Active Ingredients purpose (Active ingredients Purpose)
Octinoxate 3% .................. Sunscreen
Zinc Oxide 6.8% ................ Sunscreen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:00.536666 · Updated: 2026-03-14T23:12:08.624404