These Highlights Do Not Include All The Information Needed To Use Inluriyo Safely And Effectively. See Full Prescribing Information For Inluriyo.
5bc172e4-e8a2-441a-be52-279a7f890196
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 ( ESR1 )-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
Indications and Usage
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 ( ESR1 )-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
Dosage and Administration
Select patients for treatment based on the presence of ESR1 mutations. ( 2.1 ) The recommended dosage is 400 mg orally once daily, on an empty stomach. ( 2.2 ) Reduce the dose in patients with moderate or severe hepatic impairment ( 2.4 , 8.6 ) Dosage modifications may be required. ( 2.3 , 2.4 , 2.5 )
Warnings and Precautions
Embryo-Fetal Toxicity: INLURIYO can cause fetal harm. Advise of the potential risk to a fetus and to use effective contraception. ( 5.1 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The recommended INLURIYO dosage modifications for adverse reactions are provided in Tables 1 and 2 . The recommended dose reduction is to 200 mg once daily. Permanently discontinue INLURIYO in patients who are unable to tolerate 200 mg once daily. Table 1: INLURIYO Dosage Modification - Adverse Reactions (except hepatotoxicity) Grade INLURIYO Dosage Modifications Persistent or recurrent Grade 2 that does not resolve with maximal supportive measures within 7 days to baseline or Grade 1 Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at the same dose level. Grade 3 or 4 (except non-hepatic asymptomatic laboratory changes) Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at next lower dose level. Table 2: INLURIYO Dosage Modification - Hepatotoxicity Abbreviation: ALT = alanine aminotransferase, AST = aspartate aminotransferase, TBL=total bilirubin, ULN = upper limit of normal. Monitor alanine aminotransferase (ALT)/aspartate aminotransferase (AST) during imlunestrant therapy as clinically indicated. Liver Transaminase INLURIYO Dosage Modifications Persistent or Recurrent: AST/ALT >3.0-5.0×ULN Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at the same dose level. If AST/ALT at baseline is within the normal range: AST/ALT >5.0-20×ULN Or If AST/ALT at baseline is above ULN: AST/ALT ≥3 × baseline (if AST/ALT≥1.5 x ULN at baseline) Or AST/ALT >8 × ULN (whichever is the lower threshold) Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at next lower dose level or discontinue if receiving 200 mg daily. AST/ALT >20.0×ULN Or ALT or AST ≥ 3 × ULN concurrent with TBL ≥ 2 × ULN (if ALT or AST < 1.5 × ULN at baseline), in the absence of cholestasis Or ALT or AST ≥ 2 × baseline concurrent with TBL ≥ 2 × ULN (if ALT or AST ≥ 1.5 × ULN at baseline), in the absence of cholestasis Discontinue INLURIYO.
Drug Interactions
Strong CYP3A Inhibitors: Avoid concomitant use with INLURIYO. If concomitant use cannot be avoided, decrease the INLURIYO dosage. ( 2.5 , 7.1 ) Strong CYP3A Inducers: Avoid concomitant use with INLURIYO. If concomitant use cannot be avoided, increase the INLURIYO dosage ( 2.5 , 7.1 ).
Medication Information
Warnings and Precautions
Embryo-Fetal Toxicity: INLURIYO can cause fetal harm. Advise of the potential risk to a fetus and to use effective contraception. ( 5.1 , 8.1 , 8.3 )
Indications and Usage
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 ( ESR1 )-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
Dosage and Administration
Select patients for treatment based on the presence of ESR1 mutations. ( 2.1 ) The recommended dosage is 400 mg orally once daily, on an empty stomach. ( 2.2 ) Reduce the dose in patients with moderate or severe hepatic impairment ( 2.4 , 8.6 ) Dosage modifications may be required. ( 2.3 , 2.4 , 2.5 )
Dosage Forms and Strengths
How Supplied
INLURIYO 200 mg tablets are white capsule-shaped tablets with “LILLY” on one side and “1717” and an elongated 4-point starburst on the other side.
INLURIYO 200 mg tablets are supplied in either a 28-count or 56-count bottle configuration.
- 28-count: NDC 0002-1717-28
- 56-count: NDC 0002-1717-56
Contraindications
None.
Adverse Reactions
The recommended INLURIYO dosage modifications for adverse reactions are provided in Tables 1 and 2 . The recommended dose reduction is to 200 mg once daily. Permanently discontinue INLURIYO in patients who are unable to tolerate 200 mg once daily. Table 1: INLURIYO Dosage Modification - Adverse Reactions (except hepatotoxicity) Grade INLURIYO Dosage Modifications Persistent or recurrent Grade 2 that does not resolve with maximal supportive measures within 7 days to baseline or Grade 1 Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at the same dose level. Grade 3 or 4 (except non-hepatic asymptomatic laboratory changes) Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at next lower dose level. Table 2: INLURIYO Dosage Modification - Hepatotoxicity Abbreviation: ALT = alanine aminotransferase, AST = aspartate aminotransferase, TBL=total bilirubin, ULN = upper limit of normal. Monitor alanine aminotransferase (ALT)/aspartate aminotransferase (AST) during imlunestrant therapy as clinically indicated. Liver Transaminase INLURIYO Dosage Modifications Persistent or Recurrent: AST/ALT >3.0-5.0×ULN Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at the same dose level. If AST/ALT at baseline is within the normal range: AST/ALT >5.0-20×ULN Or If AST/ALT at baseline is above ULN: AST/ALT ≥3 × baseline (if AST/ALT≥1.5 x ULN at baseline) Or AST/ALT >8 × ULN (whichever is the lower threshold) Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at next lower dose level or discontinue if receiving 200 mg daily. AST/ALT >20.0×ULN Or ALT or AST ≥ 3 × ULN concurrent with TBL ≥ 2 × ULN (if ALT or AST < 1.5 × ULN at baseline), in the absence of cholestasis Or ALT or AST ≥ 2 × baseline concurrent with TBL ≥ 2 × ULN (if ALT or AST ≥ 1.5 × ULN at baseline), in the absence of cholestasis Discontinue INLURIYO.
Drug Interactions
Strong CYP3A Inhibitors: Avoid concomitant use with INLURIYO. If concomitant use cannot be avoided, decrease the INLURIYO dosage. ( 2.5 , 7.1 ) Strong CYP3A Inducers: Avoid concomitant use with INLURIYO. If concomitant use cannot be avoided, increase the INLURIYO dosage ( 2.5 , 7.1 ).
Description
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 ( ESR1 )-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
Section 42229-5
Strong CYP3A Inhibitors
Avoid concomitant use with strong CYP3A inhibitors. If concomitant use cannot be avoided, decrease the INLURIYO dosage to 200 mg once daily [see Drug Interactions (7.1)].
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration |
Issued: September 2025 |
|
PATIENT INFORMATION
INLURIYOTM (en-loo-ree-yoh) imlunestrant tablets |
|
|
What is INLURIYO?
INLURIYO is a prescription medicine used to treat adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 (ESR1)-mutated advanced breast cancer or breast cancer that has spread to other parts of the body (metastatic), and whose disease has progressed after at least 1 line of endocrine therapy. Your healthcare provider will perform a test to make sure that INLURIYO is right for you. It is not known if INLURIYO is safe and effective in children. |
|
Before taking INLURIYO, tell your healthcare provider about all of your medical conditions including if you:
|
|
Females who are able to become pregnant:
|
|
Males with female partners who are able to become pregnant:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the counter medicines, vitamins, and herbal supplements. INLURIYO and other medicines may affect each other causing side effects. Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. | |
How should I take INLURIYO?
|
|
|
What are the possible side effects of INLURIYO?
The most common side effects of INLURIYO include: |
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or completely stop treatment with INLURIYO if you develop certain side effects. INLURIYO may affect fertility in males and in females who are able to become pregnant. Talk to your healthcare provider if this is a concern for you. These are not all of the possible side effects of INLURIYO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store INLURIYO?
|
|
| Keep INLURIYO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of INLURIYO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use INLURIYO for a condition for which it was not prescribed. Do not give INLURIYO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for more information about INLURIYO. |
|
|
What are the ingredients in INLURIYO?
Active ingredient: imlunestrant Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, and microcrystalline cellulose. The tablet coating contains polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. INLURIYO is a trademark of Eli Lilly and Company. All rights reserved. Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA Copyright © 2025, Eli Lilly and Company. All rights reserved. For more information, go to www.INLURIYO.com or call 1-800-545-5979 |
INL-0001-PPI-202509
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F). Excursions between 15°C to 30°C (59°F to 86°F) are permitted [see USP Controlled Room Temperature].
Section 69763-1
Disposal
Dispose unused medication via a take-back option if available. Otherwise, follow FDA instructions for disposing medication in the household trash, www.fda.gov/drugdisposal. Do NOT flush down the toilet.
11 Description
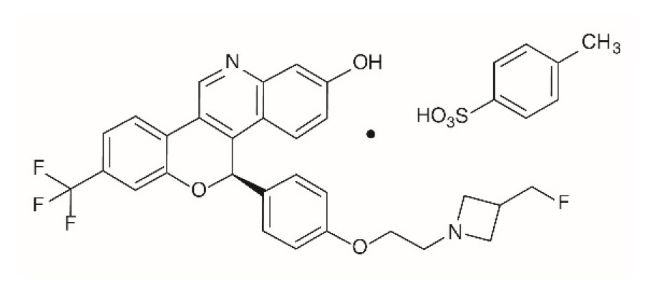
INLURIYO tablets contain imlunestrant, an estrogen receptor antagonist. The chemical name for imlunestrant tosylate is (5R)-5-(4-(2-(3-(flouromethyl)azetidin-1-yl)ethoxy)phenyl)-8-(trifluoromethyl)-5H-(1)benzopyrano(4,3-c)quinolin-2-ol, tosylate salt (1:1). Imlunestrant tosylate is a white to practically white to yellow powder with the empirical formula C29H24F4N2O3.C7H8O3S and a molecular weight 696.71 g/mol. The aqueous solubility of imlunestrant tosylate is slightly soluble at low pH, insoluble at neutral pH, and sparingly soluble at high pH. The chemical structure of imlunestrant tosylate is shown below:
INLURIYO are tablets for oral administration. Each INLURIYO tablet is available as capsule-shaped, film-coated tablet that contains 200 mg imlunestrant (equivalent to 265.66 mg imlunestrant tosylate). The tablet contains the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, and microcrystalline cellulose. The tablets are coated using a common white coating, which consists of polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of INLURIYO have not been established in pediatric patients.
8.5 Geriatric Use
Of 327 patients who received INLURIYO in the EMBER-3 study, 118 patients were ≥ 65 years of age and 37 patients were ≥ 75 years of age. No overall differences in safety or effectiveness of INLURIYO have been observed between patients 65 years of age and older and younger adult patients.
14 Clinical Studies
The efficacy of INLURIYO was evaluated in EMBER-3 (NCT04975308), a randomized, open-label, active-controlled, multicenter trial that enrolled 874 adult patients with ER+, HER2- locally advanced or metastatic breast cancer, who were previously treated with an aromatase inhibitor either alone or in combination with a CDK4/6 inhibitor. Patients were excluded if they were eligible to receive a PARP inhibitor. Patients were required to have progressed:
- Within 12 months of completing neoadjuvant or adjuvant aromatase inhibitor therapy with no systemic treatment for recurrent disease or
- Greater than 12 months after neoadjuvant or adjuvant endocrine therapy or de novo metastatic disease and had progressed on only one line of aromatase inhibitor therapy.
Patients were randomized 1:1:1 to INLURIYO 400 mg orally once daily; or investigator's choice of endocrine therapy [fulvestrant 500 mg IM on days 1, 15, 29, and once monthly thereafter (n=111) or exemestane 25 mg orally once daily (n=6)]; or an additional investigational combination regimen. Randomization was stratified by previous treatment with CDK4/6 inhibitor (yes vs no), presence of visceral metastasis (yes vs no), and region (East Asia vs North America/Western Europe vs Others). ESR1m status was determined by blood circulating tumor deoxyribonucleic acid (ctDNA) analysis using the Guardant360 CDx assay and was limited to specific ESR1 mutations in the ligand binding domain. Patients were treated until disease progression or unacceptable toxicity.
The major efficacy outcome was investigator assessed progression-free survival (PFS) according to RECIST v1.1. Other efficacy measures included overall survival (OS), blinded independent review committee (BIRC)-assessed PFS, and objective response rate (ORR).
Among the patients on the INLURIYO arm or investigator's choice of endocrine therapy who were positive for ESR1m (N=256), the median age was 61 years (range: 28-85 years); all patients were female, of which 11% were pre/perimenopausal; 61% were White, 26% Asian, 4% Black, 4% were American Indian or Alaskan Native, 4.7% missing, 0.8% multiple, 19% were Hispanic/Latino; and baseline ECOG performance status was 0 (63%) or 1 (37%). Most patients had visceral metastasis (59%) at baseline. Of the patients enrolled, 21% had received no endocrine therapy and 79% had received one line of endocrine therapy in the advanced or metastatic setting. Overall, 70% of patients were treated with a prior CDK4/6i, 2.3% treated in the adjuvant setting and 67% treated in the advanced or metastatic setting.
The efficacy results from these patients are summarized in Table 5 and Figure 1. There was a statistically significant difference in investigator-assessed PFS in the ESR1m population for INLURIYO compared to investigator's choice of endocrine therapy (fulvestrant or exemestane). PFS assessment based on a BIRC was consistent with the investigator assessment. At the time of PFS analysis, overall survival data was immature with 31% of deaths in the ESR1m population.
|
a Investigator Assessed |
||
|
b per RECIST v1.1 |
||
|
c Based on the stratified Cox proportional hazard model |
||
|
d Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.04) |
||
|
INLURIYO
N=138 |
Fulvestrant or Exemestane
N=118 |
|
| Progression-free Survival (PFS) a, b | ||
| Number of PFS Events, n (%) | 109 (79) | 102 (86) |
| Median in months (95% CI) | 5.5 (3.9, 7.4) | 3.8 (3.7, 5.5) |
| Hazard Ratio (95% CI)c | 0.62 (0.46, 0.82) | |
| p-valued | 0.0008 | |
| Objective Response Rate b | ||
| Patients with Measurable Disease | 112 | 91 |
| ORR (95% CI) | 14.3 (7.8, 20.8) | 7.7 (2.2, 13.2) |
| Complete response rate, % | 0.9 | 0 |
| Partial response rate, % | 13.4 | 7.7 |
Figure 1: Kaplan-Meier Plot of Investigator-Assessed PFS for Patients with ESR1 m, Treated with INLURIYO or Fulvestrant/Exemestane in EMBER-3
4 Contraindications
None.
6 Adverse Reactions
The most common (incidence ≥10%) adverse reactions, including laboratory abnormalities were: hemoglobin decreased, musculoskeletal pain, calcium decreased, neutrophils decreased, AST increased, fatigue, diarrhea, ALT increased, triglycerides increased, nausea, platelets decreased, constipation, cholesterol increased, and abdominal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
12.2 Pharmacodynamics
Imlunestrant exposure-response relationships and the time course of pharmacodynamics have not been fully characterized.
12.3 Pharmacokinetics
Imlunestrant pharmacokinetics were observed at steady state at the approved recommended dosage and are presented as mean (%CV) unless otherwise specified. The maximum concentration (Cmax) of imlunestrant is 141 ng/mL (45%) and the area under the concentration-time curve (AUC) is 2,400 ng*h/mL (46%). Imlunestrant Cmax and AUC increase in a dose proportional manner over a dosage range of 200 mg to 1,200 mg (0.5 to 3 times the approved recommended dosage) once daily. Steady-state is reached in approximately 6 days and the accumulation is 2.3-fold based on AUC.
2.1 Patient Selection
Select patients for treatment of ER-positive, HER2-negative advanced or metastatic breast cancer with INLURIYO based on the presence of ESR1 mutation(s) in a plasma specimen using an FDA-approved test [see Indications and Usage (1) and Clinical Studies (14.1)].
Information on FDA-approved tests for the detection of ESR1 mutations in breast cancer is available at: https://www.fda.gov/CompanionDiagnostics.
8.6 Hepatic Impairment
1 Indications and Usage
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 (ESR1)-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
12.1 Mechanism of Action
Imlunestrant is an estrogen receptor (ER) antagonist that binds to ERα. In vitro, imlunestrant induced degradation of ERα, leading to inhibition of ER-dependent gene transcription and cellular proliferation in ER+ breast cancer cells. Imlunestrant demonstrated in vitro and in vivo anti-tumor activity in ER+ breast cancer xenograft models, including models with ESR1 mutations.
5.1 Embryo Fetal Toxicity
Based on findings in animals and its mechanism of action, INLURIYO can cause fetal harm when administered to a pregnant woman. In an animal reproduction study, oral administration of imlunestrant to pregnant rats during the period of organogenesis led to embryo-fetal mortality and structural abnormalities at maternal exposures that were below the human exposure at the recommended dose based on AUC.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with INLURIYO and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with INLURIYO and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
2 Dosage and Administration
3 Dosage Forms and Strengths
INLURIYO tablets contain 200 mg imlunestrant and are white, film coated-capsule-shaped tablets, with “LILLY” on one side and “1717” and elongated 4-point starburst on the other side.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of INLURIYO was evaluated in 651 patients with ER+, HER2- locally advanced or metastatic breast cancer previously treated with endocrine therapy with or without a prior CDK4/6 inhibitor in EMBER-3 [see Clinical Studies (14.1)]. Patients received INLURIYO 400 mg orally, once daily (n=327), or standard of care (n=324) consisting of either fulvestrant (n=292) or exemestane (n=32). Among patients who were treated with INLURIYO, the median duration of exposure was 5.6 months (range: 0.2 to 28.6 months) in EMBER-3.
Serious adverse reactions occurred in 10% of patients who received INLURIYO. Serious adverse reactions in > 1% of patients included pleural effusion (1.2%). Fatal adverse reactions occurred in 1.8% of patients who received INLURIYO, including cardiac arrest, acute myocardial infarction, right ventricular failure, hypovolemic shock, and upper gastrointestinal hemorrhage (each 0.3%).
Permanent treatment discontinuation of INLURIYO due to an adverse reaction occurred in 4.6% of patients. Adverse reactions which resulted in permanent discontinuation of INLURIYO included increased alanine aminotransferase (0.9%), abdominal pain, fatigue, fractured sacrum, hepatotoxicity, neuropathy peripheral, and pyrexia (each 0.3%).
Dosage interruption of INLURIYO due to an adverse reaction occurred in 10% of patients. Adverse reactions which required dosage interruption in >0.5% were vomiting (1.5%); increased aspartate aminotransferase and COVID-19 (each 0.9%); and increased alanine aminotransferase, anemia, diarrhea, decreased neutrophil count, and pyrexia (each 0.6%).
Dose reductions of INLURIYO due to an adverse reaction occurred in 2.4% of patients. Adverse reactions which required dose reductions were increased aspartate aminotransferase (0.6%); and increased alanine aminotransferase, anemia, fatigue, interstitial lung disease, nausea, neutropenia, and vomiting (each 0.3%).
The most common (≥10%) adverse reactions, including laboratory abnormalities, were hemoglobin decreased, musculoskeletal pain, calcium decreased, neutrophils decreased, AST increased, fatigue, diarrhea, ALT increased, triglycerides increased, nausea, platelets decreased, constipation, cholesterol increased, and abdominal pain.
Table 3 summarizes the adverse reactions in EMBER-3.
|
a Adverse reactions were graded using NCI CTCAE version 5.0. |
||||
|
b Includes other related terms |
||||
| Adverse Reaction a |
INLURIYO
N=327 |
Fulvestrant or Exemestane
N=324 |
||
|
All Grades % |
Grades 3 or 4 % |
All Grades % |
Grades 3 or 4 % |
|
| Musculoskeletal Disorders | ||||
| Musculoskeletal Pain | 30 | 3.7 | 29 | 1.9 |
| General Disorders and Administration Site Conditions | ||||
| Fatigueb | 23 | 0.3 | 14 | 0.6 |
| Gastrointestinal Disorders | ||||
| Diarrhea | 22 | 0.6 | 12 | 0.0 |
| Nausea | 17 | 0.3 | 13 | 0.0 |
| Constipation | 10 | 0 | 6 | 0.3 |
| Abdominal painb | 10 | 0.3 | 6 | 0.6 |
Clinically relevant adverse reactions (<10%) in patients who received INLURIYO included: vomiting (9%), headache (9%), cough (9%), decreased appetite (8%), hot flush (7%), pruritus (3.7%), dyspepsia (2.8%), and stomatitis (2.4%).
Table 4 summarizes the laboratory abnormalities in EMBER-3.
|
a Graded according to NCI CTCAE version 5 |
||||
|
b The denominator used to calculate the rate varied from 252 to 325 based on the number of patients with a baseline value and at least one post-treatment value. |
||||
| Lab Abnormality a | INLURIYO b | Fulvestrant or Exemestane b | ||
|
All Grades % |
Grades 3 or 4 % |
All Grades % |
Grades 3 or 4 % |
|
| Hematology | ||||
| Hemoglobin decreased | 30 | 1.2 | 35 | 3.4 |
| Neutrophils decreased | 26 | 4 | 29 | 4.7 |
| Platelets decreased | 16 | 1.8 | 14 | 1.3 |
| Chemistry | ||||
| Calcium decreased | 26 | 0 | 19 | 0.6 |
| Aspartate aminotransferase increased | 25 | 1.9 | 27 | 2.3 |
| Alanine aminotransferase increased | 21 | 1.3 | 23 | 1.0 |
| Triglycerides increased | 21 | 0 | 22 | 1.2 |
| Cholesterol increased | 10 | 0 | 12 | 0 |
8 Use in Specific Populations
- Lactation: Advise not to breastfeed. (8.2)
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information).
2.2 Recommended Dosage and Administration
The recommended dosage of INLURIYO is 400 mg orally once daily until disease progression or unacceptable toxicity.
Take on an empty stomach at least 2 hours before food, or 1 hour after food [see Clinical Pharmacology (12.3)]. Take INLURIYO tablets at approximately the same time daily. Swallow the tablets whole. Do not split, crush, or chew the tablets.
Pre/perimenopausal women and men should receive a gonadotropin-releasing hormone agonist (GnRH) according to current clinical practice standards.
If patient misses a dose by 6 or more hours or vomits, instruct the patient to take the next dose the following day at its scheduled time.
13.2 Animal Toxicology And/or Pharmacology
In a 6-month repeat-dose toxicity study, oral administration of imlunestrant to rats resulted in epithelial hyperplasia in the urinary bladder, granulosa cell hyperplasia in the ovary, and hyperplasia in the mammary gland at doses ≥ 10 mg/kg/day (≥ 4 times the human AUC at the recommended dose). These effects, except urinary bladder effects, were reversible after a 3-month recovery period.
2.3 Dosage Modifications for Adverse Reactions
The recommended INLURIYO dosage modifications for adverse reactions are provided in Tables 1 and 2.
The recommended dose reduction is to 200 mg once daily.
Permanently discontinue INLURIYO in patients who are unable to tolerate 200 mg once daily.
| Grade | INLURIYO Dosage Modifications |
| Persistent or recurrent Grade 2 that does not resolve with maximal supportive measures within 7 days to baseline or Grade 1 | Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at the same dose level. |
| Grade 3 or 4 (except non-hepatic asymptomatic laboratory changes) | Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at next lower dose level. |
|
Abbreviation: ALT = alanine aminotransferase, AST = aspartate aminotransferase, TBL=total bilirubin, ULN = upper limit of normal. |
|
| Monitor alanine aminotransferase (ALT)/aspartate aminotransferase (AST) during imlunestrant therapy as clinically indicated. | |
| Liver Transaminase | INLURIYO Dosage Modifications |
|
Persistent or Recurrent:
AST/ALT >3.0-5.0×ULN |
Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at the same dose level. |
|
If AST/ALT at baseline is within the normal range:
AST/ALT >5.0-20×ULN Or If AST/ALT at baseline is above ULN: AST/ALT ≥3 × baseline (if AST/ALT≥1.5 x ULN at baseline) Or AST/ALT >8 × ULN (whichever is the lower threshold) |
Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at next lower dose level or discontinue if receiving 200 mg daily. |
| AST/ALT >20.0×ULN Or ALT or AST ≥ 3 × ULN concurrent with TBL ≥ 2 × ULN (if ALT or AST < 1.5 × ULN at baseline), in the absence of cholestasis Or ALT or AST ≥ 2 × baseline concurrent with TBL ≥ 2 × ULN (if ALT or AST ≥ 1.5 × ULN at baseline), in the absence of cholestasis |
Discontinue INLURIYO. |
2.4 Dosage in Patients With Hepatic Impairment
The recommended dosage of INLURIYO for patients with moderate (Child-Pugh B) or severe (Child-Pugh C) hepatic impairment is 200 mg once daily. Monitor for increased adverse reactions [see Clinical Pharmacology (12.3)].
8.3 Females and Males of Reproductive Potential
INLURIYO can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Package Label – Inluriyo 200 Mg 56 Count Bottle
NDC 0002-1717-56
Rx only
InluriyoTM
(imlunestrant) tablets
200 mg
56 tablets
Lilly
Structured Label Content
Dosage Forms and Strengths (34069-5)
How Supplied
INLURIYO 200 mg tablets are white capsule-shaped tablets with “LILLY” on one side and “1717” and an elongated 4-point starburst on the other side.
INLURIYO 200 mg tablets are supplied in either a 28-count or 56-count bottle configuration.
- 28-count: NDC 0002-1717-28
- 56-count: NDC 0002-1717-56
Section 42229-5 (42229-5)
Strong CYP3A Inhibitors
Avoid concomitant use with strong CYP3A inhibitors. If concomitant use cannot be avoided, decrease the INLURIYO dosage to 200 mg once daily [see Drug Interactions (7.1)].
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration |
Issued: September 2025 |
|
PATIENT INFORMATION
INLURIYOTM (en-loo-ree-yoh) imlunestrant tablets |
|
|
What is INLURIYO?
INLURIYO is a prescription medicine used to treat adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 (ESR1)-mutated advanced breast cancer or breast cancer that has spread to other parts of the body (metastatic), and whose disease has progressed after at least 1 line of endocrine therapy. Your healthcare provider will perform a test to make sure that INLURIYO is right for you. It is not known if INLURIYO is safe and effective in children. |
|
Before taking INLURIYO, tell your healthcare provider about all of your medical conditions including if you:
|
|
Females who are able to become pregnant:
|
|
Males with female partners who are able to become pregnant:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the counter medicines, vitamins, and herbal supplements. INLURIYO and other medicines may affect each other causing side effects. Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. | |
How should I take INLURIYO?
|
|
|
What are the possible side effects of INLURIYO?
The most common side effects of INLURIYO include: |
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or completely stop treatment with INLURIYO if you develop certain side effects. INLURIYO may affect fertility in males and in females who are able to become pregnant. Talk to your healthcare provider if this is a concern for you. These are not all of the possible side effects of INLURIYO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store INLURIYO?
|
|
| Keep INLURIYO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of INLURIYO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use INLURIYO for a condition for which it was not prescribed. Do not give INLURIYO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for more information about INLURIYO. |
|
|
What are the ingredients in INLURIYO?
Active ingredient: imlunestrant Inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, and microcrystalline cellulose. The tablet coating contains polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. INLURIYO is a trademark of Eli Lilly and Company. All rights reserved. Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA Copyright © 2025, Eli Lilly and Company. All rights reserved. For more information, go to www.INLURIYO.com or call 1-800-545-5979 |
INL-0001-PPI-202509
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F). Excursions between 15°C to 30°C (59°F to 86°F) are permitted [see USP Controlled Room Temperature].
Section 69763-1 (69763-1)
Disposal
Dispose unused medication via a take-back option if available. Otherwise, follow FDA instructions for disposing medication in the household trash, www.fda.gov/drugdisposal. Do NOT flush down the toilet.
11 Description (11 DESCRIPTION)
INLURIYO tablets contain imlunestrant, an estrogen receptor antagonist. The chemical name for imlunestrant tosylate is (5R)-5-(4-(2-(3-(flouromethyl)azetidin-1-yl)ethoxy)phenyl)-8-(trifluoromethyl)-5H-(1)benzopyrano(4,3-c)quinolin-2-ol, tosylate salt (1:1). Imlunestrant tosylate is a white to practically white to yellow powder with the empirical formula C29H24F4N2O3.C7H8O3S and a molecular weight 696.71 g/mol. The aqueous solubility of imlunestrant tosylate is slightly soluble at low pH, insoluble at neutral pH, and sparingly soluble at high pH. The chemical structure of imlunestrant tosylate is shown below:
INLURIYO are tablets for oral administration. Each INLURIYO tablet is available as capsule-shaped, film-coated tablet that contains 200 mg imlunestrant (equivalent to 265.66 mg imlunestrant tosylate). The tablet contains the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, and microcrystalline cellulose. The tablets are coated using a common white coating, which consists of polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of INLURIYO have not been established in pediatric patients.
8.5 Geriatric Use
Of 327 patients who received INLURIYO in the EMBER-3 study, 118 patients were ≥ 65 years of age and 37 patients were ≥ 75 years of age. No overall differences in safety or effectiveness of INLURIYO have been observed between patients 65 years of age and older and younger adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of INLURIYO was evaluated in EMBER-3 (NCT04975308), a randomized, open-label, active-controlled, multicenter trial that enrolled 874 adult patients with ER+, HER2- locally advanced or metastatic breast cancer, who were previously treated with an aromatase inhibitor either alone or in combination with a CDK4/6 inhibitor. Patients were excluded if they were eligible to receive a PARP inhibitor. Patients were required to have progressed:
- Within 12 months of completing neoadjuvant or adjuvant aromatase inhibitor therapy with no systemic treatment for recurrent disease or
- Greater than 12 months after neoadjuvant or adjuvant endocrine therapy or de novo metastatic disease and had progressed on only one line of aromatase inhibitor therapy.
Patients were randomized 1:1:1 to INLURIYO 400 mg orally once daily; or investigator's choice of endocrine therapy [fulvestrant 500 mg IM on days 1, 15, 29, and once monthly thereafter (n=111) or exemestane 25 mg orally once daily (n=6)]; or an additional investigational combination regimen. Randomization was stratified by previous treatment with CDK4/6 inhibitor (yes vs no), presence of visceral metastasis (yes vs no), and region (East Asia vs North America/Western Europe vs Others). ESR1m status was determined by blood circulating tumor deoxyribonucleic acid (ctDNA) analysis using the Guardant360 CDx assay and was limited to specific ESR1 mutations in the ligand binding domain. Patients were treated until disease progression or unacceptable toxicity.
The major efficacy outcome was investigator assessed progression-free survival (PFS) according to RECIST v1.1. Other efficacy measures included overall survival (OS), blinded independent review committee (BIRC)-assessed PFS, and objective response rate (ORR).
Among the patients on the INLURIYO arm or investigator's choice of endocrine therapy who were positive for ESR1m (N=256), the median age was 61 years (range: 28-85 years); all patients were female, of which 11% were pre/perimenopausal; 61% were White, 26% Asian, 4% Black, 4% were American Indian or Alaskan Native, 4.7% missing, 0.8% multiple, 19% were Hispanic/Latino; and baseline ECOG performance status was 0 (63%) or 1 (37%). Most patients had visceral metastasis (59%) at baseline. Of the patients enrolled, 21% had received no endocrine therapy and 79% had received one line of endocrine therapy in the advanced or metastatic setting. Overall, 70% of patients were treated with a prior CDK4/6i, 2.3% treated in the adjuvant setting and 67% treated in the advanced or metastatic setting.
The efficacy results from these patients are summarized in Table 5 and Figure 1. There was a statistically significant difference in investigator-assessed PFS in the ESR1m population for INLURIYO compared to investigator's choice of endocrine therapy (fulvestrant or exemestane). PFS assessment based on a BIRC was consistent with the investigator assessment. At the time of PFS analysis, overall survival data was immature with 31% of deaths in the ESR1m population.
|
a Investigator Assessed |
||
|
b per RECIST v1.1 |
||
|
c Based on the stratified Cox proportional hazard model |
||
|
d Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.04) |
||
|
INLURIYO
N=138 |
Fulvestrant or Exemestane
N=118 |
|
| Progression-free Survival (PFS) a, b | ||
| Number of PFS Events, n (%) | 109 (79) | 102 (86) |
| Median in months (95% CI) | 5.5 (3.9, 7.4) | 3.8 (3.7, 5.5) |
| Hazard Ratio (95% CI)c | 0.62 (0.46, 0.82) | |
| p-valued | 0.0008 | |
| Objective Response Rate b | ||
| Patients with Measurable Disease | 112 | 91 |
| ORR (95% CI) | 14.3 (7.8, 20.8) | 7.7 (2.2, 13.2) |
| Complete response rate, % | 0.9 | 0 |
| Partial response rate, % | 13.4 | 7.7 |
Figure 1: Kaplan-Meier Plot of Investigator-Assessed PFS for Patients with ESR1 m, Treated with INLURIYO or Fulvestrant/Exemestane in EMBER-3
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common (incidence ≥10%) adverse reactions, including laboratory abnormalities were: hemoglobin decreased, musculoskeletal pain, calcium decreased, neutrophils decreased, AST increased, fatigue, diarrhea, ALT increased, triglycerides increased, nausea, platelets decreased, constipation, cholesterol increased, and abdominal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
Imlunestrant exposure-response relationships and the time course of pharmacodynamics have not been fully characterized.
12.3 Pharmacokinetics
Imlunestrant pharmacokinetics were observed at steady state at the approved recommended dosage and are presented as mean (%CV) unless otherwise specified. The maximum concentration (Cmax) of imlunestrant is 141 ng/mL (45%) and the area under the concentration-time curve (AUC) is 2,400 ng*h/mL (46%). Imlunestrant Cmax and AUC increase in a dose proportional manner over a dosage range of 200 mg to 1,200 mg (0.5 to 3 times the approved recommended dosage) once daily. Steady-state is reached in approximately 6 days and the accumulation is 2.3-fold based on AUC.
2.1 Patient Selection
Select patients for treatment of ER-positive, HER2-negative advanced or metastatic breast cancer with INLURIYO based on the presence of ESR1 mutation(s) in a plasma specimen using an FDA-approved test [see Indications and Usage (1) and Clinical Studies (14.1)].
Information on FDA-approved tests for the detection of ESR1 mutations in breast cancer is available at: https://www.fda.gov/CompanionDiagnostics.
8.6 Hepatic Impairment
1 Indications and Usage (1 INDICATIONS AND USAGE)
INLURIYO is indicated for the treatment of adults with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, estrogen receptor-1 (ESR1)-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
12.1 Mechanism of Action
Imlunestrant is an estrogen receptor (ER) antagonist that binds to ERα. In vitro, imlunestrant induced degradation of ERα, leading to inhibition of ER-dependent gene transcription and cellular proliferation in ER+ breast cancer cells. Imlunestrant demonstrated in vitro and in vivo anti-tumor activity in ER+ breast cancer xenograft models, including models with ESR1 mutations.
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
Based on findings in animals and its mechanism of action, INLURIYO can cause fetal harm when administered to a pregnant woman. In an animal reproduction study, oral administration of imlunestrant to pregnant rats during the period of organogenesis led to embryo-fetal mortality and structural abnormalities at maternal exposures that were below the human exposure at the recommended dose based on AUC.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with INLURIYO and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with INLURIYO and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
INLURIYO tablets contain 200 mg imlunestrant and are white, film coated-capsule-shaped tablets, with “LILLY” on one side and “1717” and elongated 4-point starburst on the other side.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of INLURIYO was evaluated in 651 patients with ER+, HER2- locally advanced or metastatic breast cancer previously treated with endocrine therapy with or without a prior CDK4/6 inhibitor in EMBER-3 [see Clinical Studies (14.1)]. Patients received INLURIYO 400 mg orally, once daily (n=327), or standard of care (n=324) consisting of either fulvestrant (n=292) or exemestane (n=32). Among patients who were treated with INLURIYO, the median duration of exposure was 5.6 months (range: 0.2 to 28.6 months) in EMBER-3.
Serious adverse reactions occurred in 10% of patients who received INLURIYO. Serious adverse reactions in > 1% of patients included pleural effusion (1.2%). Fatal adverse reactions occurred in 1.8% of patients who received INLURIYO, including cardiac arrest, acute myocardial infarction, right ventricular failure, hypovolemic shock, and upper gastrointestinal hemorrhage (each 0.3%).
Permanent treatment discontinuation of INLURIYO due to an adverse reaction occurred in 4.6% of patients. Adverse reactions which resulted in permanent discontinuation of INLURIYO included increased alanine aminotransferase (0.9%), abdominal pain, fatigue, fractured sacrum, hepatotoxicity, neuropathy peripheral, and pyrexia (each 0.3%).
Dosage interruption of INLURIYO due to an adverse reaction occurred in 10% of patients. Adverse reactions which required dosage interruption in >0.5% were vomiting (1.5%); increased aspartate aminotransferase and COVID-19 (each 0.9%); and increased alanine aminotransferase, anemia, diarrhea, decreased neutrophil count, and pyrexia (each 0.6%).
Dose reductions of INLURIYO due to an adverse reaction occurred in 2.4% of patients. Adverse reactions which required dose reductions were increased aspartate aminotransferase (0.6%); and increased alanine aminotransferase, anemia, fatigue, interstitial lung disease, nausea, neutropenia, and vomiting (each 0.3%).
The most common (≥10%) adverse reactions, including laboratory abnormalities, were hemoglobin decreased, musculoskeletal pain, calcium decreased, neutrophils decreased, AST increased, fatigue, diarrhea, ALT increased, triglycerides increased, nausea, platelets decreased, constipation, cholesterol increased, and abdominal pain.
Table 3 summarizes the adverse reactions in EMBER-3.
|
a Adverse reactions were graded using NCI CTCAE version 5.0. |
||||
|
b Includes other related terms |
||||
| Adverse Reaction a |
INLURIYO
N=327 |
Fulvestrant or Exemestane
N=324 |
||
|
All Grades % |
Grades 3 or 4 % |
All Grades % |
Grades 3 or 4 % |
|
| Musculoskeletal Disorders | ||||
| Musculoskeletal Pain | 30 | 3.7 | 29 | 1.9 |
| General Disorders and Administration Site Conditions | ||||
| Fatigueb | 23 | 0.3 | 14 | 0.6 |
| Gastrointestinal Disorders | ||||
| Diarrhea | 22 | 0.6 | 12 | 0.0 |
| Nausea | 17 | 0.3 | 13 | 0.0 |
| Constipation | 10 | 0 | 6 | 0.3 |
| Abdominal painb | 10 | 0.3 | 6 | 0.6 |
Clinically relevant adverse reactions (<10%) in patients who received INLURIYO included: vomiting (9%), headache (9%), cough (9%), decreased appetite (8%), hot flush (7%), pruritus (3.7%), dyspepsia (2.8%), and stomatitis (2.4%).
Table 4 summarizes the laboratory abnormalities in EMBER-3.
|
a Graded according to NCI CTCAE version 5 |
||||
|
b The denominator used to calculate the rate varied from 252 to 325 based on the number of patients with a baseline value and at least one post-treatment value. |
||||
| Lab Abnormality a | INLURIYO b | Fulvestrant or Exemestane b | ||
|
All Grades % |
Grades 3 or 4 % |
All Grades % |
Grades 3 or 4 % |
|
| Hematology | ||||
| Hemoglobin decreased | 30 | 1.2 | 35 | 3.4 |
| Neutrophils decreased | 26 | 4 | 29 | 4.7 |
| Platelets decreased | 16 | 1.8 | 14 | 1.3 |
| Chemistry | ||||
| Calcium decreased | 26 | 0 | 19 | 0.6 |
| Aspartate aminotransferase increased | 25 | 1.9 | 27 | 2.3 |
| Alanine aminotransferase increased | 21 | 1.3 | 23 | 1.0 |
| Triglycerides increased | 21 | 0 | 22 | 1.2 |
| Cholesterol increased | 10 | 0 | 12 | 0 |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise not to breastfeed. (8.2)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information).
2.2 Recommended Dosage and Administration
The recommended dosage of INLURIYO is 400 mg orally once daily until disease progression or unacceptable toxicity.
Take on an empty stomach at least 2 hours before food, or 1 hour after food [see Clinical Pharmacology (12.3)]. Take INLURIYO tablets at approximately the same time daily. Swallow the tablets whole. Do not split, crush, or chew the tablets.
Pre/perimenopausal women and men should receive a gonadotropin-releasing hormone agonist (GnRH) according to current clinical practice standards.
If patient misses a dose by 6 or more hours or vomits, instruct the patient to take the next dose the following day at its scheduled time.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In a 6-month repeat-dose toxicity study, oral administration of imlunestrant to rats resulted in epithelial hyperplasia in the urinary bladder, granulosa cell hyperplasia in the ovary, and hyperplasia in the mammary gland at doses ≥ 10 mg/kg/day (≥ 4 times the human AUC at the recommended dose). These effects, except urinary bladder effects, were reversible after a 3-month recovery period.
2.3 Dosage Modifications for Adverse Reactions
The recommended INLURIYO dosage modifications for adverse reactions are provided in Tables 1 and 2.
The recommended dose reduction is to 200 mg once daily.
Permanently discontinue INLURIYO in patients who are unable to tolerate 200 mg once daily.
| Grade | INLURIYO Dosage Modifications |
| Persistent or recurrent Grade 2 that does not resolve with maximal supportive measures within 7 days to baseline or Grade 1 | Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at the same dose level. |
| Grade 3 or 4 (except non-hepatic asymptomatic laboratory changes) | Suspend until toxicity resolves to baseline or ≤Grade 1. Resume INLURIYO at next lower dose level. |
|
Abbreviation: ALT = alanine aminotransferase, AST = aspartate aminotransferase, TBL=total bilirubin, ULN = upper limit of normal. |
|
| Monitor alanine aminotransferase (ALT)/aspartate aminotransferase (AST) during imlunestrant therapy as clinically indicated. | |
| Liver Transaminase | INLURIYO Dosage Modifications |
|
Persistent or Recurrent:
AST/ALT >3.0-5.0×ULN |
Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at the same dose level. |
|
If AST/ALT at baseline is within the normal range:
AST/ALT >5.0-20×ULN Or If AST/ALT at baseline is above ULN: AST/ALT ≥3 × baseline (if AST/ALT≥1.5 x ULN at baseline) Or AST/ALT >8 × ULN (whichever is the lower threshold) |
Suspend until toxicity resolves to baseline or to >ULN-3.0×ULN. Resume INLURIYO at next lower dose level or discontinue if receiving 200 mg daily. |
| AST/ALT >20.0×ULN Or ALT or AST ≥ 3 × ULN concurrent with TBL ≥ 2 × ULN (if ALT or AST < 1.5 × ULN at baseline), in the absence of cholestasis Or ALT or AST ≥ 2 × baseline concurrent with TBL ≥ 2 × ULN (if ALT or AST ≥ 1.5 × ULN at baseline), in the absence of cholestasis |
Discontinue INLURIYO. |
2.4 Dosage in Patients With Hepatic Impairment (2.4 Dosage in Patients with Hepatic Impairment)
The recommended dosage of INLURIYO for patients with moderate (Child-Pugh B) or severe (Child-Pugh C) hepatic impairment is 200 mg once daily. Monitor for increased adverse reactions [see Clinical Pharmacology (12.3)].
8.3 Females and Males of Reproductive Potential
INLURIYO can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Package Label – Inluriyo 200 Mg 56 Count Bottle (PACKAGE LABEL – Inluriyo 200 mg 56 count bottle)
NDC 0002-1717-56
Rx only
InluriyoTM
(imlunestrant) tablets
200 mg
56 tablets
Lilly
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:30.672637 · Updated: 2026-03-14T22:50:42.037952