Drug Facts
591ffd18-fbd2-9087-e053-2991aa0ab340
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
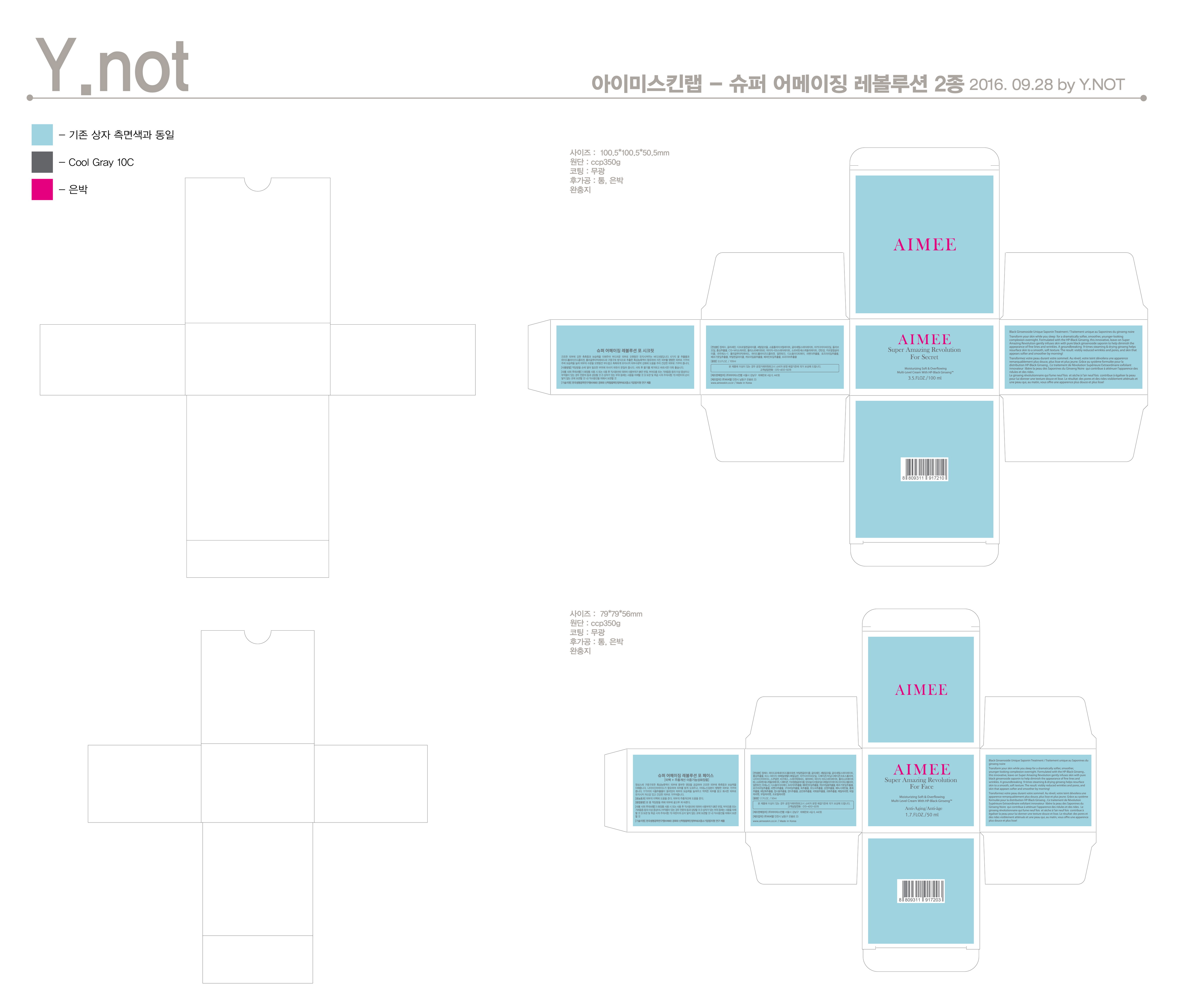
Composition & Product
Identifiers & Packaging
Description
Niacinamide, Adenosine
Medication Information
Warnings and Precautions
For external use only.
Avoid contact with eyes. Not for human consumption.
Discontinue use if irritation occurs.
If irritation persists, consult a physician.
Indications and Usage
Apply a proper amount on the skin and spread evenly.
Dosage and Administration
for external use only
Description
Niacinamide, Adenosine
Section 50565-1
keep out of reach of the children
Section 51727-6
Water, Butylene Glycol, Glycerin, Panax Ginseng Root, etc.
Section 51945-4
Section 55105-1
Skin Protectant - Anti-wrinkle & Whitening care
Section 55106-9
Niacinamide, Adenosine
Structured Label Content
Indications and Usage (34067-9)
Apply a proper amount on the skin and spread evenly.
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
For external use only.
Avoid contact with eyes. Not for human consumption.
Discontinue use if irritation occurs.
If irritation persists, consult a physician.
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
Water, Butylene Glycol, Glycerin, Panax Ginseng Root, etc.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Skin Protectant - Anti-wrinkle & Whitening care
Section 55106-9 (55106-9)
Niacinamide, Adenosine
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:24.880371 · Updated: 2026-03-14T22:54:51.534540