Reveos®
58bdabc8-283a-47bd-8abe-dd9284f3f497
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
The Reveos ® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components. Rx only.
Indications and Usage
The Reveos ® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components. Rx only.
Dosage and Administration
Follow the instructions for collecting blood with the Reveos SELECT set. ( 2 ) Follow the instructions for post-processing and red blood cell leukoreduction with the Reveos SELECT set. ( 2 )
Warnings and Precautions
Single-use product. Do not reuse. This product is intended to be single use only and is not intended to be reused or re-sterilized in any manner. Terumo Blood and Cell Technologies cannot ensure the functionality or sterility of the product if it is reused or re-sterilized. Reuse of a single-use disposable set may result in: Product performance issues due to loss of product integrity, including but not limited to the following: Fluid leaks Parts that are warped or deformed Plastics that are brittle and discolored Filters that have reduced filtration capabilities Viral infections such as hepatitis or human immunodeficiency virus (HIV) Bacterial infections Cross-contamination Any of these risks could result in serious injury or death. These risks are shared by product users, donors, patients, and recipients of the end product of the device. Inspect the packaging and the blood bag set prior to use. Do not use the set if any of the following conditions are present: There are tears or holes in the outer aluminum foil packaging or in the individual transparent packaging wrap. The tubing has severe kinks. The blood bag set is incorrectly assembled. The blood bag set is defective or damaged, or there are any leaks from the fluid-filled components of the set. Any clamps are closed. The needle cap is not in place. The solutions are cloudy or discolored or contain particulates. Using the blood bag set under these conditions may result in product contamination or poor performance during collection and/or processing. Note : It is normal to have some condensation in the outer aluminum foil pouch and individual transparent packaging wrap due to sterilization. The blood bag set is no longer sterile if any of the following conditions occur: You disconnect the sample/diversion bag before you seal the sample/diversion tubing. You remove blood samples before you seal the sample/diversion tubing. The integrity of the set is compromised for any reason. Manage the blood bag set according to your institution's SOP. Do not process whole blood less than 2 hours after collection. Processing blood too soon after collection may result in incomplete RBC leukoreduction and/or a reduced platelet yield. Residual leukocytes are a by-product of the processing procedure and contain mostly white blood cells (WBC), with some plasma, platelets, and red blood cells (RBC). Residual leukocytes are not intended for transfusion. Use aseptic technique during blood collection to ensure donor safety and product quality. Do not vent the blood bag set.
Contraindications
None.
Adverse Reactions
To report SUSPECTED ADVERSE REACTIONS, contact Terumo BCT, Inc. at 1-877-339-4228 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches. CATALOG NUMBER NDC NUMBER 6FO506A0 Carton: 82906-506-16 Aluminum foil: 82906-506-02 Primary Collect Bag: 82906-506-01
How Supplied
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches. CATALOG NUMBER NDC NUMBER 6FO506A0 Carton: 82906-506-16 Aluminum foil: 82906-506-02 Primary Collect Bag: 82906-506-01
Medication Information
Warnings and Precautions
Single-use product. Do not reuse. This product is intended to be single use only and is not intended to be reused or re-sterilized in any manner. Terumo Blood and Cell Technologies cannot ensure the functionality or sterility of the product if it is reused or re-sterilized. Reuse of a single-use disposable set may result in: Product performance issues due to loss of product integrity, including but not limited to the following: Fluid leaks Parts that are warped or deformed Plastics that are brittle and discolored Filters that have reduced filtration capabilities Viral infections such as hepatitis or human immunodeficiency virus (HIV) Bacterial infections Cross-contamination Any of these risks could result in serious injury or death. These risks are shared by product users, donors, patients, and recipients of the end product of the device. Inspect the packaging and the blood bag set prior to use. Do not use the set if any of the following conditions are present: There are tears or holes in the outer aluminum foil packaging or in the individual transparent packaging wrap. The tubing has severe kinks. The blood bag set is incorrectly assembled. The blood bag set is defective or damaged, or there are any leaks from the fluid-filled components of the set. Any clamps are closed. The needle cap is not in place. The solutions are cloudy or discolored or contain particulates. Using the blood bag set under these conditions may result in product contamination or poor performance during collection and/or processing. Note : It is normal to have some condensation in the outer aluminum foil pouch and individual transparent packaging wrap due to sterilization. The blood bag set is no longer sterile if any of the following conditions occur: You disconnect the sample/diversion bag before you seal the sample/diversion tubing. You remove blood samples before you seal the sample/diversion tubing. The integrity of the set is compromised for any reason. Manage the blood bag set according to your institution's SOP. Do not process whole blood less than 2 hours after collection. Processing blood too soon after collection may result in incomplete RBC leukoreduction and/or a reduced platelet yield. Residual leukocytes are a by-product of the processing procedure and contain mostly white blood cells (WBC), with some plasma, platelets, and red blood cells (RBC). Residual leukocytes are not intended for transfusion. Use aseptic technique during blood collection to ensure donor safety and product quality. Do not vent the blood bag set.
Indications and Usage
The Reveos ® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components. Rx only.
Dosage and Administration
Follow the instructions for collecting blood with the Reveos SELECT set. ( 2 ) Follow the instructions for post-processing and red blood cell leukoreduction with the Reveos SELECT set. ( 2 )
Contraindications
None.
Adverse Reactions
To report SUSPECTED ADVERSE REACTIONS, contact Terumo BCT, Inc. at 1-877-339-4228 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches. CATALOG NUMBER NDC NUMBER 6FO506A0 Carton: 82906-506-16 Aluminum foil: 82906-506-02 Primary Collect Bag: 82906-506-01
How Supplied
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches. CATALOG NUMBER NDC NUMBER 6FO506A0 Carton: 82906-506-16 Aluminum foil: 82906-506-02 Primary Collect Bag: 82906-506-01
Description
The Reveos ® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components. Rx only.
Section 42229-5
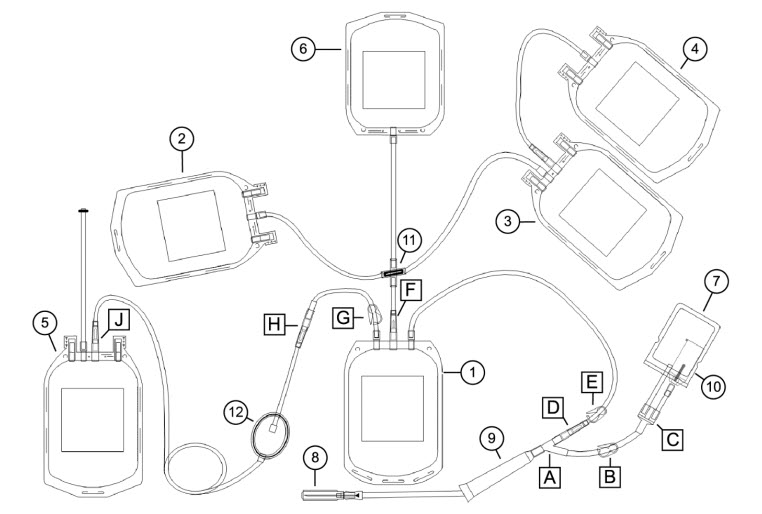
Set Diagram
| 1 Whole blood bag | 7 Sample/diversion bag | A, C: Y connector |
| 2 Interim platelet unit (IPU) bag | 8 Needle | B, G: Blue clamp |
| 3 Plasma bag | 9 Needle injury protector (NIP) | D, F, H, J: CLIKTIP frangible connector |
| 4 Plasma cryoprecipitate reduced bag | 10 Sample tube holder/luer adapter | E: White clamp |
| 5 Red blood cell (RBC) bag | 11 Cross-connector | |
| 6 Residual leukocyte bag | 12 RBC leukoreduction filter |
This document provides blood collection and post-processing instructions specific to the Reveos SELECT set. For warnings, cautions, and instructions on processing whole blood with the Reveos system, see the Reveos system operator's manual.
Note: Refer to the instructions for use provided by the manufacturer of your tubing sealer to ensure that the tubing sealer is appropriate for the tubing on the blood bag set.
Note: You must validate the process for producing, storing, and handling Cryoprecipitated Antihemophilic Factor within your institution's standard operating procedure (SOP), including procedures for using the plasma cryoprecipitate reduced bag.
Storage
- Long-term storage temperature: 1 °C to 30 °C
- Permitted temperature excursions:
- –20 °C to 1 °C for up to 2 weeks
- Up to 50 °C for up to 1 week
Use blood bag sets within 28 days after you open the outer aluminum foil pouch. To store unused blood bag sets, return them to the outer aluminum foil pouch and reclose the pouch with tape or a clip. Once you open the transparent packing wrap, you must use the blood bag set within 7 days, not exceeding 28 days from when you opened the outer aluminum foil pouch. Each outer aluminum foil pouch contains sachets that absorb oxygen. Dispose of the sachets and the outer aluminum foil pouch with normal waste.
11 Description
The Reveos SELECT set has been evaluated for use with the Reveos system.
The blood and fluid pathways of the blood bag set are steam-sterilized and are non-pyrogenic.
The blood bag set is intended for use by appropriately trained phlebotomists who collect whole blood and by blood center personnel who process whole blood using the Reveos system.
The formulas of the active ingredients are provided in Tables 1 and 2.
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Dextrose (anhydrous) | C6H12O6 | 180.16 g/mol |
| Trisodium Citrate (dihydrate) | C6H5Na3O72H2O | 294.10 g/mol |
| Citric Acid (monohydrate) | C6H8O7H2O | 210.14 g/mol |
| Sodium Dihydrogen Phosphate (dihydrate) | NaH2PO42H2O | 156.01 g/mol |
| Water for Injection | H2O | 18.02 g/mol |
Each 70 mL of CPD contains: Dextrose (anhydrous) 1.624 g, Trisodium Citrate (dihydrate) 1.841 g, Citric Acid (monohydrate) 0.229 g, Sodium Dihydrogen Phosphate (dihydrate) 0.176 g, and water for injection up to 70 mL.
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Dextrose (anhydrous) | C6H12O6 | 180.16 g/ mol |
| Sodium Chloride | NaCl | 58.44 g/mol |
| Mannitol | C6H14O6 | 182.17 g/mol |
| Adenine | C5H5N5 | 135.13 g/mol |
| Water for injection | H2O | 18.02 g/mol |
Each 111 mL of AS-5 contains: Dextrose (anhydrous) 0.908 g, Sodium Chloride 0.973 g, Mannitol 0.583 g, Adenine 0.035 g, and water for injection up to 111 mL.
The PVC plasticized with DEHP bags are not made with natural rubber latex.
The bags contain materials that have been tested to demonstrate the suitability of the containers for storing pharmaceutical solutions. The bags are nontoxic and biologically inert. The blood bag set is a closed system and is not dependent upon entry of external air during administration. The blood bag set is overwrapped to provide protection from the physical environment and to provide an additional moisture barrier when necessary.
4 Contraindications
None.
6. Adverse Reactions
To report SUSPECTED ADVERSE REACTIONS, contact Terumo BCT, Inc. at 1-877-339-4228 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Return of Used Product
If for any reason this product must be returned to Terumo BCT, Inc., a returned goods authorization (an RGA number) is required from Terumo BCT prior to shipping. Instructions for cleaning and materials, including appropriate shipping containers, proper labeling, and an RGA number, may be obtained from the Terumo BCT Quality Assurance Department. IT IS THE RESPONSIBILITY OF THE HEALTH CARE INSTITUTION TO ADEQUATELY PREPARE AND IDENTIFY THE PRODUCT FOR RETURN SHIPMENT. Please contact your local representative for information regarding returned goods and product complaints.
| Description | Primary tubing | Sample tube holder/luer adapter tubing | ||
| Item(s) | 1, 2, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 | 3 | ||
| Wall Thickness | 0.7 mm | 1.3 mm | ||
| Outer Diameter | 4.4 mm | 4.4 mm | ||
| Inner Diameter | 3.0 mm | 1.8 mm | ||
Tubing Set Composition
- Primary tubing: Polyvinyl chloride (PVC) plasticized with Di(2-ethylhexyl)phthalate (DEHP)
- Bags: PVC plasticized with DEHP
- RBC leukoreduction filter: PVC plasticized with DEHP housing, polybutylterephthalate media
- Access needle: Stainless steel (contains cobalt)
- Needle injury protector (NIP): Polypropylene (PP)
- Sample tube holder/luer adapter: PVC/polycarbonate housing, stainless steel needle
- Clamps: Polyacetal
- Cross-connector: PVC
Not made with natural rubber latex.
1 Indications and Usage
The Reveos® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components.
Rx only.
5 Warnings and Precautions
- Single-use product. Do not reuse. This product is intended to be single use only and is not intended to be reused or re-sterilized in any manner. Terumo Blood and Cell Technologies cannot ensure the functionality or sterility of the product if it is reused or re-sterilized. Reuse of a single-use disposable set may result in:
- Product performance issues due to loss of product integrity, including but not limited to the following:
- Fluid leaks
- Parts that are warped or deformed
- Plastics that are brittle and discolored
- Filters that have reduced filtration capabilities
- Viral infections such as hepatitis or human immunodeficiency virus (HIV)
- Bacterial infections
- Cross-contamination
- Product performance issues due to loss of product integrity, including but not limited to the following:
- Inspect the packaging and the blood bag set prior to use. Do not use the set if any of the following conditions are present:
- There are tears or holes in the outer aluminum foil packaging or in the individual transparent packaging wrap.
- The tubing has severe kinks.
- The blood bag set is incorrectly assembled.
- The blood bag set is defective or damaged, or there are any leaks from the fluid-filled components of the set.
- Any clamps are closed.
- The needle cap is not in place.
- The solutions are cloudy or discolored or contain particulates.
Note: It is normal to have some condensation in the outer aluminum foil pouch and individual transparent packaging wrap due to sterilization. - The blood bag set is no longer sterile if any of the following conditions occur:
- You disconnect the sample/diversion bag before you seal the sample/diversion tubing.
- You remove blood samples before you seal the sample/diversion tubing.
- The integrity of the set is compromised for any reason.
- Do not process whole blood less than 2 hours after collection. Processing blood too soon after collection may result in incomplete RBC leukoreduction and/or a reduced platelet yield.
- Residual leukocytes are a by-product of the processing procedure and contain mostly white blood cells (WBC), with some plasma, platelets, and red blood cells (RBC). Residual leukocytes are not intended for transfusion.
- Use aseptic technique during blood collection to ensure donor safety and product quality.
- Do not vent the blood bag set.
2 Dosage and Administration
3 Dosage Forms and Strengths
70 mL Citrate Phosphate Dextrose (CPD) anticoagulant is a sterile solution in a PVC plasticized with DEHP bag. Each 70 mL of CPD contains: Dextrose (anhydrous) 1.624 g, Trisodium Citrate (dihydrate) 1.841 g, Citric Acid (monohydrate) 0.229 g, Sodium Dihydrogen Phosphate (dihydrate) 0.176 g, and water for injection up to 70 mL.
111 mL Additive Solution 5 (AS-5) red cell preservative solution is a sterile solution in a PVC plasticized with DEHP bag. Each 111 mL of AS-5 contains: Dextrose (anhydrous) 0.908 g, Sodium Chloride 0.973 g, Mannitol 0.583 g, Adenine 0.035 g, and water for injection up to 111 mL.
8 Use in Specific Populations
The Reveos SELECT set has not been adequately studied in controlled clinical trials with specific populations.
16 How Supplied/storage and Handling
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches.
| CATALOG NUMBER | NDC NUMBER | |
|---|---|---|
| 6FO506A0 | Carton: | 82906-506-16 |
| Aluminum foil: | 82906-506-02 | |
| Primary Collect Bag: | 82906-506-01 |
Principal Display Panel 70 Ml Bag Pouch Case Label
TERUMO Reveos® SELECT
CPD/AS-5 RED CELL PRESERVATIVE SOLUTION FOR COLLECTION OF 500mL OF BLOOD
CAUTIONS:
For single use only. Sterile fluid path, sterilized
using steam. Non-pyrogenic fluid path.
Do not re-sterilize.
Read the instructions carefully before use.
Do not use if package is damaged.
Store between 1°C and 30°C
Manufactured by :
TERUMO BCT Vietnam Co., Ltd.
Long Duc Industrial Park, Long Duc Commune,
Long Thanh District, Dong Nai Province, Vietnam
For :
TERUMO BCT, Inc.
10811 W. Collins ave.
Lakewood, CO 80215
Rx ONLY
D0000036575-A
XXXX
LOT : LOT #
MFG DATE : Date of Mfg.
EXPIRY DATE : Exp. Date
CAT. NO. : 6FO506A0
UNITS : 016
NDC : 82906-506-16
NDC (ISBT 128)
Structured Label Content
Section 42229-5 (42229-5)
Set Diagram
| 1 Whole blood bag | 7 Sample/diversion bag | A, C: Y connector |
| 2 Interim platelet unit (IPU) bag | 8 Needle | B, G: Blue clamp |
| 3 Plasma bag | 9 Needle injury protector (NIP) | D, F, H, J: CLIKTIP frangible connector |
| 4 Plasma cryoprecipitate reduced bag | 10 Sample tube holder/luer adapter | E: White clamp |
| 5 Red blood cell (RBC) bag | 11 Cross-connector | |
| 6 Residual leukocyte bag | 12 RBC leukoreduction filter |
This document provides blood collection and post-processing instructions specific to the Reveos SELECT set. For warnings, cautions, and instructions on processing whole blood with the Reveos system, see the Reveos system operator's manual.
Note: Refer to the instructions for use provided by the manufacturer of your tubing sealer to ensure that the tubing sealer is appropriate for the tubing on the blood bag set.
Note: You must validate the process for producing, storing, and handling Cryoprecipitated Antihemophilic Factor within your institution's standard operating procedure (SOP), including procedures for using the plasma cryoprecipitate reduced bag.
Storage (STORAGE)
- Long-term storage temperature: 1 °C to 30 °C
- Permitted temperature excursions:
- –20 °C to 1 °C for up to 2 weeks
- Up to 50 °C for up to 1 week
Use blood bag sets within 28 days after you open the outer aluminum foil pouch. To store unused blood bag sets, return them to the outer aluminum foil pouch and reclose the pouch with tape or a clip. Once you open the transparent packing wrap, you must use the blood bag set within 7 days, not exceeding 28 days from when you opened the outer aluminum foil pouch. Each outer aluminum foil pouch contains sachets that absorb oxygen. Dispose of the sachets and the outer aluminum foil pouch with normal waste.
11 Description (11 DESCRIPTION)
The Reveos SELECT set has been evaluated for use with the Reveos system.
The blood and fluid pathways of the blood bag set are steam-sterilized and are non-pyrogenic.
The blood bag set is intended for use by appropriately trained phlebotomists who collect whole blood and by blood center personnel who process whole blood using the Reveos system.
The formulas of the active ingredients are provided in Tables 1 and 2.
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Dextrose (anhydrous) | C6H12O6 | 180.16 g/mol |
| Trisodium Citrate (dihydrate) | C6H5Na3O72H2O | 294.10 g/mol |
| Citric Acid (monohydrate) | C6H8O7H2O | 210.14 g/mol |
| Sodium Dihydrogen Phosphate (dihydrate) | NaH2PO42H2O | 156.01 g/mol |
| Water for Injection | H2O | 18.02 g/mol |
Each 70 mL of CPD contains: Dextrose (anhydrous) 1.624 g, Trisodium Citrate (dihydrate) 1.841 g, Citric Acid (monohydrate) 0.229 g, Sodium Dihydrogen Phosphate (dihydrate) 0.176 g, and water for injection up to 70 mL.
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Dextrose (anhydrous) | C6H12O6 | 180.16 g/ mol |
| Sodium Chloride | NaCl | 58.44 g/mol |
| Mannitol | C6H14O6 | 182.17 g/mol |
| Adenine | C5H5N5 | 135.13 g/mol |
| Water for injection | H2O | 18.02 g/mol |
Each 111 mL of AS-5 contains: Dextrose (anhydrous) 0.908 g, Sodium Chloride 0.973 g, Mannitol 0.583 g, Adenine 0.035 g, and water for injection up to 111 mL.
The PVC plasticized with DEHP bags are not made with natural rubber latex.
The bags contain materials that have been tested to demonstrate the suitability of the containers for storing pharmaceutical solutions. The bags are nontoxic and biologically inert. The blood bag set is a closed system and is not dependent upon entry of external air during administration. The blood bag set is overwrapped to provide protection from the physical environment and to provide an additional moisture barrier when necessary.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6. Adverse Reactions (6. ADVERSE REACTIONS)
To report SUSPECTED ADVERSE REACTIONS, contact Terumo BCT, Inc. at 1-877-339-4228 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Return of Used Product (RETURN OF USED PRODUCT)
If for any reason this product must be returned to Terumo BCT, Inc., a returned goods authorization (an RGA number) is required from Terumo BCT prior to shipping. Instructions for cleaning and materials, including appropriate shipping containers, proper labeling, and an RGA number, may be obtained from the Terumo BCT Quality Assurance Department. IT IS THE RESPONSIBILITY OF THE HEALTH CARE INSTITUTION TO ADEQUATELY PREPARE AND IDENTIFY THE PRODUCT FOR RETURN SHIPMENT. Please contact your local representative for information regarding returned goods and product complaints.
| Description | Primary tubing | Sample tube holder/luer adapter tubing | ||
| Item(s) | 1, 2, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 | 3 | ||
| Wall Thickness | 0.7 mm | 1.3 mm | ||
| Outer Diameter | 4.4 mm | 4.4 mm | ||
| Inner Diameter | 3.0 mm | 1.8 mm | ||
Tubing Set Composition
- Primary tubing: Polyvinyl chloride (PVC) plasticized with Di(2-ethylhexyl)phthalate (DEHP)
- Bags: PVC plasticized with DEHP
- RBC leukoreduction filter: PVC plasticized with DEHP housing, polybutylterephthalate media
- Access needle: Stainless steel (contains cobalt)
- Needle injury protector (NIP): Polypropylene (PP)
- Sample tube holder/luer adapter: PVC/polycarbonate housing, stainless steel needle
- Clamps: Polyacetal
- Cross-connector: PVC
Not made with natural rubber latex.
1 Indications and Usage (1 INDICATIONS AND USAGE)
The Reveos® SELECT set is intended to collect a unit of whole blood and to process the whole blood unit on the Reveos Automated Blood Processing System, producing blood components.
Rx only.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Single-use product. Do not reuse. This product is intended to be single use only and is not intended to be reused or re-sterilized in any manner. Terumo Blood and Cell Technologies cannot ensure the functionality or sterility of the product if it is reused or re-sterilized. Reuse of a single-use disposable set may result in:
- Product performance issues due to loss of product integrity, including but not limited to the following:
- Fluid leaks
- Parts that are warped or deformed
- Plastics that are brittle and discolored
- Filters that have reduced filtration capabilities
- Viral infections such as hepatitis or human immunodeficiency virus (HIV)
- Bacterial infections
- Cross-contamination
- Product performance issues due to loss of product integrity, including but not limited to the following:
- Inspect the packaging and the blood bag set prior to use. Do not use the set if any of the following conditions are present:
- There are tears or holes in the outer aluminum foil packaging or in the individual transparent packaging wrap.
- The tubing has severe kinks.
- The blood bag set is incorrectly assembled.
- The blood bag set is defective or damaged, or there are any leaks from the fluid-filled components of the set.
- Any clamps are closed.
- The needle cap is not in place.
- The solutions are cloudy or discolored or contain particulates.
Note: It is normal to have some condensation in the outer aluminum foil pouch and individual transparent packaging wrap due to sterilization. - The blood bag set is no longer sterile if any of the following conditions occur:
- You disconnect the sample/diversion bag before you seal the sample/diversion tubing.
- You remove blood samples before you seal the sample/diversion tubing.
- The integrity of the set is compromised for any reason.
- Do not process whole blood less than 2 hours after collection. Processing blood too soon after collection may result in incomplete RBC leukoreduction and/or a reduced platelet yield.
- Residual leukocytes are a by-product of the processing procedure and contain mostly white blood cells (WBC), with some plasma, platelets, and red blood cells (RBC). Residual leukocytes are not intended for transfusion.
- Use aseptic technique during blood collection to ensure donor safety and product quality.
- Do not vent the blood bag set.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
70 mL Citrate Phosphate Dextrose (CPD) anticoagulant is a sterile solution in a PVC plasticized with DEHP bag. Each 70 mL of CPD contains: Dextrose (anhydrous) 1.624 g, Trisodium Citrate (dihydrate) 1.841 g, Citric Acid (monohydrate) 0.229 g, Sodium Dihydrogen Phosphate (dihydrate) 0.176 g, and water for injection up to 70 mL.
111 mL Additive Solution 5 (AS-5) red cell preservative solution is a sterile solution in a PVC plasticized with DEHP bag. Each 111 mL of AS-5 contains: Dextrose (anhydrous) 0.908 g, Sodium Chloride 0.973 g, Mannitol 0.583 g, Adenine 0.035 g, and water for injection up to 111 mL.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
The Reveos SELECT set has not been adequately studied in controlled clinical trials with specific populations.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
The blood bag sets are packaged in outer aluminum foil pouches. Each outer aluminum foil pouch contains 2 blood bag sets. Each case contains 8 outer aluminum foil pouches.
| CATALOG NUMBER | NDC NUMBER | |
|---|---|---|
| 6FO506A0 | Carton: | 82906-506-16 |
| Aluminum foil: | 82906-506-02 | |
| Primary Collect Bag: | 82906-506-01 |
Principal Display Panel 70 Ml Bag Pouch Case Label (PRINCIPAL DISPLAY PANEL - 70 mL Bag Pouch Case Label)
TERUMO Reveos® SELECT
CPD/AS-5 RED CELL PRESERVATIVE SOLUTION FOR COLLECTION OF 500mL OF BLOOD
CAUTIONS:
For single use only. Sterile fluid path, sterilized
using steam. Non-pyrogenic fluid path.
Do not re-sterilize.
Read the instructions carefully before use.
Do not use if package is damaged.
Store between 1°C and 30°C
Manufactured by :
TERUMO BCT Vietnam Co., Ltd.
Long Duc Industrial Park, Long Duc Commune,
Long Thanh District, Dong Nai Province, Vietnam
For :
TERUMO BCT, Inc.
10811 W. Collins ave.
Lakewood, CO 80215
Rx ONLY
D0000036575-A
XXXX
LOT : LOT #
MFG DATE : Date of Mfg.
EXPIRY DATE : Exp. Date
CAT. NO. : 6FO506A0
UNITS : 016
NDC : 82906-506-16
NDC (ISBT 128)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:18.931162 · Updated: 2026-03-14T22:22:13.322156