Uline 279.003/279ad
57c7bf8f-c17a-41b5-9b66-e4d16d6650a7
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium chloride 0.13%
Purpose
Antibacterial
Medication Information
Purpose
Antibacterial
Description
Benzalkonium chloride 0.13%
Use
for handwashing to decrease bacteria on the skin
Warnings
For external use only: hands only
Directions
- wet hands
- apply palmful to hands
- scrub thoroughly
- rinse thoroughly
Adverse Reaction
Distributed by: ULINE, 12575 Uline Drive
Pleasant Prairie, WI 53158
1-800-295-5510
uline.com
Active Ingredient
Benzalkonium chloride 0.13%
Inactive Ingredients
water, lauramine oxide, cocamidopropyl betaine, lauramidopropylamine oxide, sodium chloride, myristamidopropylamine oxide, glycerin, fragrance, disteareth-75 IPDI, PEG-150 distearate, citric acid, tetrasodium EDTA, benzophenone-4, sodium benzoate, red 33, red 40, yellow 5
When Using This Product
avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
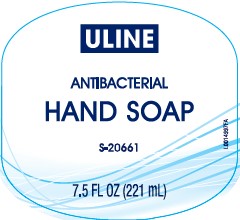
Principal Display Panel
ULINE
ANTIBACTERIAL
HAND SOAP
S-20661
7.5 FL OZ (221 mL)
Stop Use and Ask A Doctor If
- irritation and redness develop
- condition persists for more than 72 hours
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
for handwashing to decrease bacteria on the skin
Purpose
Antibacterial
Warnings
For external use only: hands only
Directions
- wet hands
- apply palmful to hands
- scrub thoroughly
- rinse thoroughly
Adverse Reaction (ADVERSE REACTION)
Distributed by: ULINE, 12575 Uline Drive
Pleasant Prairie, WI 53158
1-800-295-5510
uline.com
Active Ingredient (Active ingredient)
Benzalkonium chloride 0.13%
Inactive Ingredients (Inactive ingredients)
water, lauramine oxide, cocamidopropyl betaine, lauramidopropylamine oxide, sodium chloride, myristamidopropylamine oxide, glycerin, fragrance, disteareth-75 IPDI, PEG-150 distearate, citric acid, tetrasodium EDTA, benzophenone-4, sodium benzoate, red 33, red 40, yellow 5
When Using This Product (When using this product)
avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Principal Display Panel (principal display panel)
ULINE
ANTIBACTERIAL
HAND SOAP
S-20661
7.5 FL OZ (221 mL)
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- irritation and redness develop
- condition persists for more than 72 hours
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:25.619705 · Updated: 2026-03-14T23:13:46.303627