Paclitaxel Injection, Usp
5721b9bf-c3c1-4b73-b96c-5fbe3147b6a5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
(Patient Information Included) SAGENT ® Rx only
Indications and Usage
Paclitaxel Injection, USP is indicated as subsequent therapy for the treatment of advanced carcinoma of the ovary. As first-line therapy, paclitaxel is indicated in combination with cisplatin. Paclitaxel is indicated for the adjuvant treatment of node-positive breast cancer administered sequentially to standard doxorubicin-containing combination chemotherapy. In the clinical trial, there was an overall favorable effect on disease-free and overall survival in the total population of patients with receptor-positive and receptor-negative tumors, but the benefit has been specifically demonstrated by available data (median follow-up 30 months) only in the patients with estrogen and progesterone receptor-negative tumors. (See CLINICAL STUDIES: Breast Carcinoma . ) Paclitaxel Injection, USP is indicated for the treatment of breast cancer after failure of combination chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy. Prior therapy should have included an anthracycline unless clinically contraindicated. Paclitaxel, in combination with cisplatin, is indicated for the first-line treatment of nonsmall cell lung cancer in patients who are not candidates for potentially curative surgery and/or radiation therapy. Paclitaxel is indicated for the second-line treatment of AIDS-related Kaposi's sarcoma.
Dosage and Administration
NOTE: Contact of the undiluted concentrate with plasticized PVC equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP [di-(2-ethylhexyl)phthalate], which may be leached from PVC infusion bags or sets, diluted paclitaxel solutions should be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets. All patients should be premedicated prior to paclitaxel administration in order to prevent severe hypersensitivity reactions. Such premedication may consist of dexamethasone 20 mg PO administered approximately 12 and 6 hours before paclitaxel, diphenhydramine (or its equivalent) 50 mg I.V. 30 to 60 minutes prior to paclitaxel, and cimetidine (300 mg) or ranitidine (50 mg) I.V. 30 to 60 minutes before paclitaxel. For patients with carcinoma of the ovary the following regimen is recommended: (see CLINICAL STUDIES: Ovarian Carcinoma ): 1) For previously untreated patients with carcinoma of the ovary, one of the following recommended regimens may be given every 3 weeks. In selecting the appropriate regimen, differences in toxicities should be considered (see Table 11 in ADVERSE REACTIONS: Disease-Specific Adverse Event Experiences ). Paclitaxel administered intravenously over 3 hours at a dose of 175 mg/m 2 followed by cisplatin at a dose of 75 mg/m 2 ; or Paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m 2 followed by cisplatin at a dose of 75 mg/m 2 . 2) In patients previously treated with chemotherapy for carcinoma of the ovary, paclitaxel has been used at several doses and schedules; however, the optimal regimen is not yet clear. (See CLINICAL STUDIES: Ovarian Carcinoma section). The recommended regimen is paclitaxel 135 mg/m 2 or 175 mg/m 2 administered intravenously over 3 hours every 3 weeks. For patients with carcinoma of the breast, the following is recommended (see CLINICAL STUDIES: Breast Carcinoma section): 1) For the adjuvant treatment of node-positive breast cancer, the recommended regimen is paclitaxel, at a dose of 175 mg/m 2 intravenously over 3 hours every 3 weeks for 4 courses administered sequentially to doxorubicin-containing combination chemotherapy. The clinical trial used 4 courses of doxorubicin and cyclophosphamide (see CLINICAL STUDIES: Breast Carcinoma ). 2) After failure of initial chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy, paclitaxel at a dose of 175 mg/m 2 administered intravenously over 3 hours every 3 weeks has been shown to be effective. For patients with non-small cell lung carcinoma, the recommended regimen, given every 3 weeks, is paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m 2 followed by cisplatin, 75 mg/m 2 . For patients with AIDS-related Kaposi's sarcoma, paclitaxel administered at a dose of 135 mg/m 2 given intravenously over 3 hours every 3 weeks or at a dose of 100 mg/m 2 given intravenously over 3 hours every 2 weeks is recommended (dose intensity 45 to 50 mg/m 2 /week). In the 2 clinical trials evaluating these schedules (see CLINICAL STUDIES: AIDS-Related Kaposi's Sarcoma ), the former schedule (135 mg/m 2 every 3 weeks) was more toxic than the latter. In addition, all patients with low performance status were treated with the latter schedule (100 mg/m 2 every 2 weeks). Based upon the immunosuppression in patients with advanced HIV disease, the following modifications are recommended in these patients: 1) Reduce the dose of dexamethasone as 1 of the 3 premedication drugs to 10 mg PO (instead of 20 mg PO); 2) Initiate or repeat treatment with paclitaxel only if the neutrophil count is at least 1,000 cells/mm 3 ; 3) Reduce the dose of subsequent courses of paclitaxel by 20% for patients who experience severe neutropenia (neutrophil <500 cells/mm 3 for a week or longer); and 4) Initiate concomitant hematopoietic growth factor (G-CSF) as clinically indicated. For the therapy of patients with solid tumors (ovary, breast and NSCLC), courses of paclitaxel should not be repeated until the neutrophil count is at least 1,500 cells/mm 3 and the platelet count is at least 100,000 cells/mm 3 . Paclitaxel should not be given to patients with AIDS-related Kaposi's sarcoma if the baseline or subsequent neutrophil count is less than 1,000 cells/mm 3 . Patients who experience severe neutropenia (neutrophil <500 cells/mm 3 for a week or longer) or severe peripheral neuropathy during Paclitaxel Injection, USP therapy should have dosage reduced by 20% for subsequent courses of paclitaxel. The incidence of neurotoxicity and the severity of neutropenia increase with dose.
Contraindications
Paclitaxel Injection, USP is contraindicated in patients who have a history of hypersensitivity reactions to paclitaxel or other drugs formulated in Polyoxyl 35 Castor Oil, NF. Paclitaxel Injection, USP should not be used in patients with solid tumors who have baseline neutrophil counts of <1,500 cells/mm 3 or in patients with AIDS-related Kaposi's sarcoma with baseline neutrophil counts of <1,000 cells/mm 3 .
Adverse Reactions
Pooled Analysis of Adverse Event Experiences from Single-Agent Studies: Data in the following table are based on the experience of 812 patients (493 with ovarian carcinoma and 319 with breast carcinoma) enrolled in 10 studies who received single-agent Paclitaxel Injection, USP. Two hundred and seventy-five patients were treated in eight Phase 2 studies with paclitaxel doses ranging from 135 to 300 mg/m 2 administered over 24 hours (in four of these studies, G-CSF was administered as hematopoietic support). Three hundred and one patients were treated in the randomized Phase 3 ovarian carcinoma study which compared two doses (135 or 175 mg/m 2 ) and two schedules (3 or 24 hours) of paclitaxel. Two hundred and thirty-six patients with breast carcinoma received paclitaxel (135 or 175 mg/m 2 ) administered over 3 hours in a controlled study. Table 10. Summary a of Adverse Events in Patients with Solid Tumors Receiving Single-Agent Paclitaxel Percent of Patients (n=812) a Based on worst course analysis. b All patients received premedication. c During the first 3 hours of infusion. † Severe events are defined as at least Grade III toxicity. • Bone Marrow - Neutropenia <2,000/mm 3 90 <500/mm 3 52 - Leukopenia <4,000/mm 3 90 <1,000/mm 3 17 - Thrombocytopenia <100,000/mm 3 20 <50,000/mm 3 7 - Anemia <11 g/dL 78 <8 g/dL 16 - Infections 30 - Bleeding 14 - Red Cell Transfusions 25 - Platelet Transfusions 2 • Hypersensitivity Reaction b - All - Severe † 41 2 • Cardiovascular - Vital Sign Changes c - Bradycardia (n=537) - Hypotension (n=532) - Significant Cardiovascular Events 3 12 1 • Abnormal ECG - All Pts - Pts with normal baseline (n=559) 23 14 • Peripheral Neuropathy - Any symptoms - Severe symptoms † 60 3 • Myalgia/Arthralgia - Any symptoms - Severe symptoms † 60 8 • Gastrointestinal - Nausea and vomiting - Diarrhea - Mucositis 52 38 31 • Alopecia 87 • Hepatic (Pts with normal baseline and on study data) - Bilirubin elevations (n=765) - Alkaline phosphatase elevations (n=575) - AST (SGOT) elevations (n=591) 7 22 19 • Injection Site Reaction 13 None of the observed toxicities were clearly influenced by age.
How Supplied
Paclitaxel Injection, USP is supplied as follows: NDC Paclitaxel Injection, USP (6 mg per mL) Package Factor 25021-255-05 30 mg per 5 mL Multi-Dose Vial 1 vial per carton 25021-255-17 100 mg per 16.7 mL Multi-Dose Vial 1 vial per carton 25021-255-50 300 mg per 50 mL Multi-Dose Vial 1 vial per carton
Medication Information
Indications and Usage
Paclitaxel Injection, USP is indicated as subsequent therapy for the treatment of advanced carcinoma of the ovary. As first-line therapy, paclitaxel is indicated in combination with cisplatin.
Paclitaxel is indicated for the adjuvant treatment of node-positive breast cancer administered sequentially to standard doxorubicin-containing combination chemotherapy. In the clinical trial, there was an overall favorable effect on disease-free and overall survival in the total population of patients with receptor-positive and receptor-negative tumors, but the benefit has been specifically demonstrated by available data (median follow-up 30 months) only in the patients with estrogen and progesterone receptor-negative tumors. (See CLINICAL STUDIES: Breast Carcinoma.)
Paclitaxel Injection, USP is indicated for the treatment of breast cancer after failure of combination chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy. Prior therapy should have included an anthracycline unless clinically contraindicated.
Paclitaxel, in combination with cisplatin, is indicated for the first-line treatment of nonsmall cell lung cancer in patients who are not candidates for potentially curative surgery and/or radiation therapy.
Paclitaxel is indicated for the second-line treatment of AIDS-related Kaposi's sarcoma.
Dosage and Administration
NOTE: Contact of the undiluted concentrate with plasticized PVC equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP [di-(2-ethylhexyl)phthalate], which may be leached from PVC infusion bags or sets, diluted paclitaxel solutions should be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets.
All patients should be premedicated prior to paclitaxel administration in order to prevent severe hypersensitivity reactions. Such premedication may consist of dexamethasone 20 mg PO administered approximately 12 and 6 hours before paclitaxel, diphenhydramine (or its equivalent) 50 mg I.V. 30 to 60 minutes prior to paclitaxel, and cimetidine (300 mg) or ranitidine (50 mg) I.V. 30 to 60 minutes before paclitaxel.
For patients with carcinoma of the ovary the following regimen is recommended: (see CLINICAL STUDIES: Ovarian Carcinoma ):
-
1)For previously untreated patients with carcinoma of the ovary, one of the following recommended regimens may be given every 3 weeks. In selecting the appropriate regimen, differences in toxicities should be considered (see Table 11 in ADVERSE REACTIONS: Disease-Specific Adverse Event Experiences ).
- Paclitaxel administered intravenously over 3 hours at a dose of 175 mg/m2 followed by cisplatin at a dose of 75 mg/m2; or
- Paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m2 followed by cisplatin at a dose of 75 mg/m2.
-
2)In patients previously treated with chemotherapy for carcinoma of the ovary, paclitaxel has been used at several doses and schedules; however, the optimal regimen is not yet clear. (See CLINICAL STUDIES: Ovarian Carcinoma section). The recommended regimen is paclitaxel 135 mg/m2 or 175 mg/m2 administered intravenously over 3 hours every 3 weeks.
For patients with carcinoma of the breast, the following is recommended (see CLINICAL STUDIES: Breast Carcinoma section):
-
1)For the adjuvant treatment of node-positive breast cancer, the recommended regimen is paclitaxel, at a dose of 175 mg/m2 intravenously over 3 hours every 3 weeks for 4 courses administered sequentially to doxorubicin-containing combination chemotherapy. The clinical trial used 4 courses of doxorubicin and cyclophosphamide (see CLINICAL STUDIES: Breast Carcinoma ).
-
2)After failure of initial chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy, paclitaxel at a dose of 175 mg/m2 administered intravenously over 3 hours every 3 weeks has been shown to be effective.
For patients with non-small cell lung carcinoma, the recommended regimen, given every 3 weeks, is paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m2 followed by cisplatin, 75 mg/m2.
For patients with AIDS-related Kaposi's sarcoma, paclitaxel administered at a dose of 135 mg/m2 given intravenously over 3 hours every 3 weeks or at a dose of 100 mg/m2 given intravenously over 3 hours every 2 weeks is recommended (dose intensity 45 to 50 mg/m2/week). In the 2 clinical trials evaluating these schedules (see CLINICAL STUDIES: AIDS-Related Kaposi's Sarcoma ), the former schedule (135 mg/m2 every 3 weeks) was more toxic than the latter. In addition, all patients with low performance status were treated with the latter schedule (100 mg/m2 every 2 weeks).
Based upon the immunosuppression in patients with advanced HIV disease, the following modifications are recommended in these patients:
-
1)Reduce the dose of dexamethasone as 1 of the 3 premedication drugs to 10 mg PO (instead of 20 mg PO);
-
2)Initiate or repeat treatment with paclitaxel only if the neutrophil count is at least 1,000 cells/mm3;
-
3)Reduce the dose of subsequent courses of paclitaxel by 20% for patients who experience severe neutropenia (neutrophil <500 cells/mm3 for a week or longer); and
-
4)Initiate concomitant hematopoietic growth factor (G-CSF) as clinically indicated.
For the therapy of patients with solid tumors (ovary, breast and NSCLC), courses of paclitaxel should not be repeated until the neutrophil count is at least 1,500 cells/mm3 and the platelet count is at least 100,000 cells/mm3. Paclitaxel should not be given to patients with AIDS-related Kaposi's sarcoma if the baseline or subsequent neutrophil count is less than 1,000 cells/mm3. Patients who experience severe neutropenia (neutrophil <500 cells/mm3 for a week or longer) or severe peripheral neuropathy during Paclitaxel Injection, USP therapy should have dosage reduced by 20% for subsequent courses of paclitaxel. The incidence of neurotoxicity and the severity of neutropenia increase with dose.
Contraindications
Paclitaxel Injection, USP is contraindicated in patients who have a history of hypersensitivity reactions to paclitaxel or other drugs formulated in Polyoxyl 35 Castor Oil, NF.
Paclitaxel Injection, USP should not be used in patients with solid tumors who have baseline neutrophil counts of <1,500 cells/mm3 or in patients with AIDS-related Kaposi's sarcoma with baseline neutrophil counts of <1,000 cells/mm3.
Adverse Reactions
Pooled Analysis of Adverse Event Experiences from Single-Agent Studies: Data in the following table are based on the experience of 812 patients (493 with ovarian carcinoma and 319 with breast carcinoma) enrolled in 10 studies who received single-agent Paclitaxel Injection, USP. Two hundred and seventy-five patients were treated in eight Phase 2 studies with paclitaxel doses ranging from 135 to 300 mg/m2 administered over 24 hours (in four of these studies, G-CSF was administered as hematopoietic support). Three hundred and one patients were treated in the randomized Phase 3 ovarian carcinoma study which compared two doses (135 or 175 mg/m2) and two schedules (3 or 24 hours) of paclitaxel. Two hundred and thirty-six patients with breast carcinoma received paclitaxel (135 or 175 mg/m2) administered over 3 hours in a controlled study.
|
Percent of Patients
(n=812) |
||
|---|---|---|
|
a Based on worst course analysis. |
||
|
b All patients received premedication. |
||
|
c During the first 3 hours of infusion. |
||
|
† Severe events are defined as at least Grade III toxicity. |
||
| • Bone Marrow |
|
|
| - Neutropenia | <2,000/mm3 | 90 |
| <500/mm3
|
52 |
|
| - Leukopenia | <4,000/mm3 | 90 |
| <1,000/mm3 | 17 |
|
| - Thrombocytopenia | <100,000/mm3 | 20 |
| <50,000/mm3 | 7 |
|
| - Anemia | <11 g/dL | 78 |
| <8 g/dL | 16 |
|
| - Infections |
30 |
|
| - Bleeding |
14 |
|
| - Red Cell Transfusions |
25 |
|
| - Platelet Transfusions | 2 | |
| • Hypersensitivity Reaction
b
- All - Severe† |
41 2 |
|
|
• Cardiovascular
- Vital Sign Changesc - Bradycardia (n=537) - Hypotension (n=532) - Significant Cardiovascular Events |
3 12 1 |
|
|
• Abnormal ECG
- All Pts - Pts with normal baseline (n=559) |
23 14 |
|
|
• Peripheral Neuropathy
- Any symptoms - Severe symptoms† |
60 3 |
|
|
• Myalgia/Arthralgia
- Any symptoms - Severe symptoms† |
60 8 |
|
|
• Gastrointestinal
- Nausea and vomiting - Diarrhea - Mucositis |
52 38 31 |
|
| • Alopecia | 87 | |
|
• Hepatic (Pts with normal baseline and on study data)
- Bilirubin elevations (n=765) - Alkaline phosphatase elevations (n=575) - AST (SGOT) elevations (n=591) |
7 22 19 |
|
| • Injection Site Reaction | 13 |
None of the observed toxicities were clearly influenced by age.
How Supplied
Paclitaxel Injection, USP is supplied as follows:
| NDC | Paclitaxel Injection, USP (6 mg per mL) | Package Factor |
| 25021-255-05 | 30 mg per 5 mL Multi-Dose Vial | 1 vial per carton |
| 25021-255-17 | 100 mg per 16.7 mL Multi-Dose Vial | 1 vial per carton |
| 25021-255-50 | 300 mg per 50 mL Multi-Dose Vial | 1 vial per carton |
Description
Paclitaxel Injection, USP is a clear colorless to slightly yellow viscous solution. It is supplied as a nonaqueous solution intended for dilution with a suitable parenteral fluid prior to intravenous infusion. Paclitaxel is available in 30 mg (5 mL), 100 mg (16.7 mL), and 300 mg (50 mL) multi-dose vials. Each mL of sterile nonpyrogenic solution contains 6 mg paclitaxel, 527 mg of Polyoxyl 35 Castor Oil, NF, 49.7% (v/v) Dehydrated Alcohol, USP and 2 mg Citric Acid, USP.
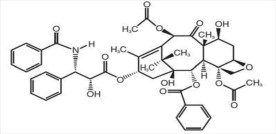
Paclitaxel is a natural product with antitumor activity. Paclitaxel is obtained via an extraction process from Taxus X media ‘Hicksii’. The chemical name for paclitaxel is (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,-12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca [3,4] benz [1,2-b] oxet-5-one 6,12b-diacetate, 12-benzoate, 9-ester with (2R,3S)-N-benzoyl-3-phenylisoserine.
Paclitaxel has the following structural formula:
Paclitaxel is a white to off-white crystalline powder with the empirical formula C47H51NO14 and a molecular weight of 853.9. It is highly lipophilic, insoluble in water, and melts at around 216 to 217°C.
Section 34073-7
Drug Interactions: In a Phase I trial using escalating doses of paclitaxel (110 to 200 mg/m2) and cisplatin (50 or 75 mg/m2) given as sequential infusions, myelosuppression was more profound when paclitaxel was given after cisplatin than with the alternate sequence (i.e., paclitaxel before cisplatin). Pharmacokinetic data from these patients demonstrated a decrease in paclitaxel clearance of approximately 33% when paclitaxel was administered following cisplatin.
The metabolism of paclitaxel is catalyzed by cytochrome P450 isoenzymes CYP2C8 and CYP3A4. In the absence of formal clinical drug interaction studies, caution should be exercised when administering paclitaxel concomitantly with known substrates or inhibitors of the cytochrome P450 isoenzymes CYP2C8 and CYP3A4. Caution should be exercised when paclitaxel is concomitantly administered with known substrates (e.g., midazolam, buspirone, felodipine, lovastatin, eletriptan, sildenafil, simvastatin, and triazolam), inhibitors (e.g., atazanavir, clarithromycin, indinavir, itraconazole, ketoconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin), and inducers (e.g., rifampin and carbamazepine) of CYP3A4. (See CLINICAL PHARMACOLOGY section.)
Caution should also be exercised when paclitaxel is concomitantly administered with known substrates (e.g., repaglinide and rosiglitazone), inhibitors (e.g., gemfibrozil), and inducers (e.g., rifampin) of CYP2C8. (See CLINICAL PHARMACOLOGY.)
Potential interactions between paclitaxel, a substrate of CYP3A4, and protease inhibitors (ritonavir, saquinavir, indinavir, and nelfinavir), which are substrates and/or inhibitors of CYP3A4, have not been evaluated in clinical trials.
Reports in the literature suggest that plasma levels of doxorubicin (and its active metabolite doxorubicinol) may be increased when paclitaxel and doxorubicin are used in combination.
Section 34076-0
Information for Patients: (See Patient Information Leaflet.)
Section 34080-2
Nursing Mothers: It is not known whether the drug is excreted in human milk. Following intravenous administration of carbon-14 labeled paclitaxel to rats on days 9 to 10 postpartum, concentrations of radioactivity in milk were higher than in plasma and declined in parallel with the plasma concentrations. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants, it is recommended that nursing be discontinued when receiving paclitaxel therapy.
Section 34081-0
Pediatric Use: The safety and effectiveness of paclitaxel in pediatric patients have not been established.
There have been reports of central nervous system (CNS) toxicity (rarely associated with death) in a clinical trial in pediatric patients in which paclitaxel was infused intravenously over 3 hours at doses ranging from 350 mg/m2 to 420 mg/m2. The toxicity is most likely attributable to the high dose of the ethanol component of the Paclitaxel Injection, USP vehicle given over a short infusion time. The use of concomitant antihistamines may intensify this effect. Although a direct effect of the paclitaxel itself cannot be discounted, the high doses used in this study (over twice the recommended adult dosage) must be considered in assessing the safety of paclitaxel for use in this population.
Section 34082-8
Geriatric Use: Of 2228 patients who received paclitaxel in 8 clinical studies evaluating its safety and effectiveness in the treatment of advanced ovarian cancer, breast carcinoma, or NSCLC, and 1570 patients who were randomized to receive paclitaxel in the adjuvant breast cancer study, 649 patients (17%) were 65 years or older and 49 patients (1%) were 75 years or older. In most studies, severe myelosuppression was more frequent in elderly patients; in some studies, severe neuropathy was more common in elderly patients. In 2 clinical studies in NSCLC, the elderly patients treated with paclitaxel had a higher incidence of cardiovascular events. Estimates of efficacy appeared similar in elderly patients and in younger patients; however, comparative efficacy cannot be determined with confidence due to the small number of elderly patients studied. In a study of first-line treatment of ovarian cancer, elderly patients had a lower median survival than younger patients, but no other efficacy parameters favored the younger group. Table 9 presents the incidences of Grade IV neutropenia and severe neuropathy in clinical studies according to age.
|
INDICATION (Study/Regimen) |
Patients (n/total [%]) | |||
|---|---|---|---|---|
|
Neutropenia
(Grade IV) |
Peripheral Neuropathy
(Grades III/IV) |
|||
| Age (y) | Age (y) | |||
| ≥65 | <65 | ≥65 | <65 | |
|
* p<0.05 |
||||
|
a Paclitaxel dose in mg/m2/infusion duration in hours; cisplatin doses in mg/m2. |
||||
|
b Peripheral neuropathy was included within the neurotoxicity category in the Intergroup First-Line Ovarian Cancer study (see Table 11). |
||||
|
c Paclitaxel dose in mg/m2/infusion duration in hours. |
||||
|
d Paclitaxel (T) following 4 courses of doxorubicin and cyclophosphamide (AC) at a dose of 175 mg/m2/3 hours every 3 weeks for 4 courses. |
||||
|
e Peripheral neuropathy reported as neurosensory toxicity in the Intergroup Adjuvant Breast Cancer study (see Table 13). |
||||
|
f Peripheral neuropathy reported as neurosensory toxicity in the ECOG NSCLC study (see Table 15). |
||||
|
• OVARIAN Cancer
(Intergroup First-Line/T175/3 c75a) (GOG-111 First-Line/T135/24 c75a) (Phase 3 Second-Line/T175/3c) (Phase 3 Second-Line/T175/24c) (Phase 3 Second-Line/T135/3c) (Phase 3 Second-Line/T135/24c) (Phase 3 Second-Line Pooled) |
34/83 (41) 48/61 (79) 5/19 (26) 21/25 (84) 4/16 (25) 17/22 (77) 47/82 (57)* |
78/252 (31) 106/129 (82) 21/76 (28) 57/79 (72) 10/81 (12) 53/83 (64) 141/319 (44) |
24/84 (29)*b 3/62 (5) 1/19 (5) 0/25 (0) 0/17 (0) 0/22 (0) 1/83 (1) |
46/255 (18)b 2/134 (1) 0/76 (0) 2/80 (3) 0/81 (0) 0/83 (0) 2/320 (1) |
|
• Adjuvant BREAST Cancer
(Intergroup/AC followed by Td) |
56/102 (55) |
734/1468 (50) |
5/102 (5)e |
46/1468 (3)e |
|
• BREAST Cancer after Failure of Initial Therapy
(Phase 3/T175/3c) (Phase 3/T135/3c) |
7/24 (29) 7/20 (35) |
56/200 (28) 37/207 (18) |
3/25 (12) 0/20 (0) |
12/204 (6) 6/209 (3) |
| • Non-Small Cell LUNG Cancer
(ECOG/T135/24 c75a) (Phase 3/T175/3 c80a) |
58/71 (82) 37/89 (42)* |
86/124 (69) 56/267 (21) |
9/71 (13)f 11/91 (12)* |
16/124 (13)f 11/271 (4) |
Section 34083-6
Carcinogenesis, Mutagenesis, Impairment of Fertility: The carcinogenic potential of paclitaxel has not been studied.
Paclitaxel has been shown to be clastogenic in vitro (chromosome aberrations in human lymphocytes) and in vivo (micronucleus test in mice). Paclitaxel was not mutagenic in the Ames test of the CHO/HGPRT gene mutation assay.
Administration of paclitaxel prior to and during mating produced impairment of fertility in male and female rats at doses equal to or greater than 1 mg/kg/day (about 0.04 the daily maximum recommended human dose on a mg/m2 basis). At this dose, paclitaxel caused reduced fertility and reproductive indices, and increased embryo- and fetotoxicity. (See WARNINGS section.)
Section 42228-7
Pregnancy: Pregnancy Category D. (See WARNINGS section.)
Section 42229-5
(Patient Information Included)
SAGENT®
Rx only
Section 42230-3
PATIENT INFORMATION
Paclitaxel Injection, USP
Read this patient information leaflet before you start taking paclitaxel. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is the most important information I should know about paclitaxel?
Paclitaxel can cause serious side effects including death.
Serious allergic reactions (anaphylaxis) can happen in people who receive paclitaxel.
Anaphylaxis is a serious medical emergency that can lead to death and must be treated right away.
Tell your healthcare provider right away if you have any of these signs of an allergic reaction:
- trouble breathing
- sudden swelling of your face, lips, tongue, throat, or trouble swallowing
- hives (raised bumps) or rash
Your healthcare provider will give you medicines to lessen your chance of having an allergic reaction.
What is paclitaxel?
Paclitaxel is a prescription medicine used to treat some forms of:
- ovarian cancer
- breast cancer
- lung cancer
- Kaposi's sarcoma
It is not known if paclitaxel is safe or effective in children.
Who should not receive paclitaxel?
Do not receive paclitaxel if:
- you are allergic to any of the ingredients in paclitaxel. See the end of this leaflet for a complete list of ingredients in paclitaxel.
- are allergic to medicines containing polyoxyethylated castor oil.
- you have low white blood cell counts.
What should I tell my healthcare provider before receiving paclitaxel?
Before receiving paclitaxel, tell your healthcare provider about all your medical conditions, including if you:
- have liver problems
- have heart problems
- are pregnant or plan to become pregnant. paclitaxel can harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- are breast-feeding or plan to breast-feed. It is not known if paclitaxel passes into your breast milk. You and your healthcare provider should decide if you will receive paclitaxel or breast-feed.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How will I receive paclitaxel?
- Paclitaxel is injected into a vein (intravenous [IV] infusion) by your healthcare provider.
Your healthcare provider will do certain tests while you receive paclitaxel.
What are the possible side effects of paclitaxel?
Tell your healthcare provider right away if you have:
- severe stomach pain
- severe diarrhea
The most common side effects of paclitaxel include:
- low red blood cell count (anemia) feeling weak or tired
- hair loss
- numbness, tingling, or burning in your hands or feet (neuropathy)
- joint and muscle pain
- nausea and vomiting
- hypersensitivity reaction - trouble breathing; sudden swelling of your face, lips, tongue, throat, or trouble swallowing; hives (raised bumps) or rash
- diarrhea
- mouth or lip sores (mucositis)
- infections - if you have a fever (temperature above 100.4°F) or other sign of infection, tell your healthcare provider right away
- swelling of your hands, face, or feet
- bleeding events
- irritation at the injection site
- low blood pressure (hypotension)
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of paclitaxel. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
General information about the safe and effective use of paclitaxel.
Medicines are sometimes prescribed for purposes other than those listed in a patient information leaflet. Do not use paclitaxel for a condition for which it was not prescribed.
Do not give paclitaxel to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about paclitaxel. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about paclitaxel that is written for health professionals.
What are the ingredients in paclitaxel?
Active ingredient: paclitaxel.
Inactive ingredients include: Polyoxyl 35 castor oil, NF and dehydrated alcohol, USP and Citric Acid, USP.
What is cancer?
Under normal conditions, the cells in your body divide and grow in an orderly, controlled way. Cell division and growth are necessary for the human body to perform its functions and to repair itself, when necessary. Cancer cells are different from normal cells because they are not able to control their own growth. The reasons for this abnormal growth are not yet fully understood. A tumor is a mass of unhealthy cells that are dividing and growing fast and in an uncontrolled way. When a tumor invades surrounding healthy body tissue, it is known as a malignant tumor. A malignant tumor can spread (metastasize) from its original site to other parts of the body if not found and treated early.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
November 2023
SAGENT Pharmaceuticals ®
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-255-05
Rx only
Paclitaxel Injection, USP
30 mg per 5 mL
(6 mg per mL)
Caution: Dilution required prior to Intravenous Infusion
CAUTION: Cytotoxic Agent
Sterile Multi-Dose Vial
Warning
Paclitaxel Injection, USP should be administered under the supervision of a physician experienced in the use of cancer chemotherapeutic agents. Appropriate management of complications is possible only when adequate diagnostic and treatment facilities are readily available.
Anaphylaxis and severe hypersensitivity reactions characterized by dyspnea and hypotension requiring treatment, angioedema, and generalized urticaria have occurred in 2% to 4% of patients receiving paclitaxel in clinical trials. Fatal reactions have occurred in patients despite premedication. All patients should be pretreated with corticosteroids, diphenhydramine, and H2 antagonists. (See DOSAGE AND ADMINISTRATION section.) Patients who experience severe hypersensitivity reactions to paclitaxel should not be rechallenged with the drug.
Paclitaxel therapy should not be given to patients with solid tumors who have baseline neutrophil counts of less than 1,500 cells/mm3 and should not be given to patients with AIDS-related Kaposi's sarcoma if the baseline neutrophil count is less than 1,000 cells/mm3. In order to monitor the occurrence of bone marrow suppression, primarily neutropenia, which may be severe and result in infection, it is recommended that frequent peripheral blood cell counts be performed on all patients receiving paclitaxel.
Warnings
Anaphylaxis and severe hypersensitivity reactions characterized by dyspnea and hypotension requiring treatment, angioedema, and generalized urticaria have occurred in 2% to 4% of patients receiving paclitaxel in clinical trials. Fatal reactions have occurred in patients despite premedication. All patients should be pretreated with corticosteroids, diphenhydramine, and H2 antagonists. (See DOSAGE AND ADMINISTRATION section.) Patients who experience severe hypersensitivity reactions to paclitaxel should not be rechallenged with the drug.
Bone marrow suppression (primarily neutropenia) is dose-dependent and is the dose-limiting toxicity. Neutrophil nadirs occurred at a median of 11 days. Paclitaxel should not be administered to patients with baseline neutrophil counts of less than 1,500 cells/mm3 (<1,000 cells/mm3 for patients with KS). Frequent monitoring of blood counts should be instituted during paclitaxel treatment. Patients should not be re-treated with subsequent cycles of paclitaxel until neutrophils recover to a level >1,500 cells/mm3 (>1,000 cells/mm3 for patients with KS) and platelets recover to a level >100,000 cells/mm3.
Severe conduction abnormalities have been documented in <1% of patients during paclitaxel therapy and in some cases requiring pacemaker placement. If patients develop significant conduction abnormalities during paclitaxel infusion, appropriate therapy should be administered and continuous cardiac monitoring should be performed during subsequent therapy with paclitaxel.
Overdosage
There is no known antidote for paclitaxel overdosage. The primary anticipated complications of overdosage would consist of bone marrow suppression, peripheral neurotoxicity, and mucositis. Overdoses in pediatric patients may be associated with acute ethanol toxicity (see PRECAUTIONS: Pediatric Use section).
References
- NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004165.
- OSHA Technical Manual, TED 1-0.15A, Section VI: Chapter 2. Controlling occupational exposure to hazardous drugs. OSHA, 1999. http://www.osha.gov/dts/osta/otm/otm_vi/otm_vi_2.html.
- American Society of Health-System Pharmacists. ASHP guidelines on handling hazardous drugs. Am J Health-Syst Pharm. 2006; 63:1172-1193.
- Polovich M, White JM, Kelleher LO, eds. 2005. Chemotherapy and biotherapy guidelines and recommendations for practice. 2nd ed. Pittsburgh, PA: Oncology Nursing Society.
Brands listed are the trademarks of their respective owners.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
November 2023
SAGENT Pharmaceuticals ®
Precautions
Contact of the undiluted concentrate with plasticized polyvinyl chloride (PVC) equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP [di-(2-ethylhexyl) phthalate], which may be leached from PVC infusion bags or sets, diluted Paclitaxel Injection, USP solutions should preferably be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets.
Paclitaxel should be administered through an in-line filter with a microporous membrane not greater than 0.22 microns. Use of filter devices such as IVEX-2® filters which incorporate short inlet and outlet PVC-coated tubing has not resulted in significant leaching of DEHP.
Adjuvant Therapy
A Phase 3 Intergroup study (Cancer and Leukemia Group B [CALGB], Eastern Cooperative Oncology Group [ECOG], North Central Cancer Treatment Group [NCCTG], and Southwest Oncology Group [SWOG]) randomized 3170 patients with node-positive breast carcinoma to adjuvant therapy with paclitaxel or to no further chemotherapy following 4 courses of doxorubicin and cyclophosphamide (AC). This multicenter trial was conducted in women with histologically positive lymph nodes following either a mastectomy or segmental mastectomy and nodal dissections. The 3 x 2 factorial study was designed to assess the efficacy and safety of 3 different dose levels of doxorubicin (A) and to evaluate the effect of the addition of paclitaxel administered following the completion of AC therapy. After stratification for the number of positive lymph nodes (1 to 3, 4 to 9, or 10+), patients were randomized to receive cyclophosphamide at a dose of 600 mg/m2 and doxorubicin at doses of either 60 mg/m2 (on day 1), 75 mg/m2 (in 2 divided doses on days 1 and 2), or 90 mg/m2 (in 2 divided doses on days 1 and 2 with prophylactic G-CSF support and ciprofloxacin) every 3 weeks for 4 courses and either paclitaxel 175 mg/m2 as a 3-hour infusion every 3 weeks for 4 additional courses or no additional chemotherapy. Patients whose tumors were positive were to receive subsequent tamoxifen treatment (20 mg daily for 5 years); patients who received segmental mastectomies prior to study were to receive breast irradiation after recovery from treatment-related toxicities.
At the time of the current analysis, median follow-up was 30.1 months. Of the 2066 patients who were hormone receptor positive, 93% received tamoxifen. The primary analyses of disease-free survival and overall survival used multivariate Cox models, which included paclitaxel administration, doxorubicin dose, number of positive lymph nodes, tumor size, menopausal status, and estrogen receptor status as factors. Based on the model for disease-free survival, patients receiving AC followed by paclitaxel had a 22% reduction in the risk of disease recurrence compared to patients randomized to AC alone (Hazard Ratio [HR]=0.78, 95% CI, 0.67 to 0.91, p=0.0022). They also had a 26% reduction in the risk of death (HR=0.74, 95% CI, 0.60 to 0.92, p=0.0065). For disease-free survival and overall survival, p-values were not adjusted for interim analyses. Kaplan-Meier curves are shown in Figures 3 and 4. Increasing the dose of doxorubicin higher than 60 mg/m2 had no effect on either disease-free survival or overall survival.
Figure 3. Disease-Free Survival: AC Versus AC+T
Figure 4. Survival: AC Versus AC+T
Subset Analyses: Subsets defined by variables of known prognostic importance in adjuvant breast carcinoma were examined, including number of positive lymph nodes, tumor size, hormone receptor status, and menopausal status. Such analyses must be interpreted with care, as the most secure finding is the overall study result. In general, a reduction in hazard similar to the overall reduction was seen with paclitaxel for both disease-free and overall survival in all of the larger subsets with one exception; patients with receptor-positive tumors had a smaller reduction in hazard (HR=0.92) for disease-free survival with paclitaxel than other groups. Results of subset analyses are shown in Table 4.
|
a Positive for either estrogen or progesterone receptors. |
|||||
|
b Negative or missing for both estrogen and progesterone receptors (both missing: n=15). |
|||||
|
Patient Subset |
Disease-Free Survival | Overall Survival | |||
|
No. of
Patients |
No. of
Recurrences |
Hazard Ratio
(95% CI) |
No. of
Deaths |
Hazard Ratio
(95% CI) |
|
| • No. of Positive Nodes
1 to 3 4 to 9 10+ |
1449 1310 360 |
221 274 129 |
0.72 (0.55 to 0.94) 0.78 (0.61 to 0.99) 0.93 (0.66 to 1.31) |
107 148 87 |
0.76 (0.52 to 1.12) 0.66 (0.47 to 0.91) 0.90 (0.59 to 1.36) |
| • Tumor Size (cm)
≤2 >2 and ≤5 >5 |
1096 1611 397 |
153 358 111 |
0.79 (0.57 to 1.08) 0.79 (0.64 to 0.97) 0.75 (0.51 to 1.08) |
67 201 72 |
0.73 (0.45 to 1.18) 0.74 (0.56 to 0.98) 0.73 (0.46 to 1.16) |
| • Menopausal Status
Pre Post |
1929 1183 |
374 250 |
0.83 (0.67 to 1.01) 0.73 (0.57 to 0.93) |
187 155 |
0.72 (0.54 to 0.97) 0.77 (0.56 to 1.06) |
| • Receptor Status
Positivea Negative/Unknownb |
2066 1055 |
293 331 |
0.92 (0.73 to 1.16) 0.68 (0.55 to 0.85) |
126 216 |
0.83 (0.59 to 1.18) 0.71 (0.54 to 0.93) |
These retrospective subgroup analyses suggest that the beneficial effect of paclitaxel is clearly established in the receptor-negative subgroup, but the benefit in receptor-positive patients is not yet clear. With respect to menopausal status, the benefit of paclitaxel is consistent (see Table 4 and Figures 5 to 8).
Figure 5. Disease-Free Survival: Receptor Status Negative/Unknown AC Versus AC+T
Figure 6. Disease-Free Survival: Receptor Status Positive AC Versus AC+T
Figure 7. Disease-Free Survival: Premenopausal AC Versus AC+T
Figure 8. Disease-Free Survival: Postmenopausal AC Versus AC+T
The adverse event profile for the patients who received paclitaxel subsequent to AC was consistent with that seen in the pooled analysis of data from 812 patients (Table 10) treated with single-agent paclitaxel in 10 clinical studies. These adverse events are described in the ADVERSE REACTIONS section in tabular (Tables 10 and 13) and narrative form.
Storage Conditions
Store the vials in original cartons between 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Retain in carton until time of use.
Handling and Disposal: See DOSAGE AND ADMINISTRATION: Preparation and Administration Precautions.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Clinical Pharmacology
Paclitaxel is a novel antimicrotubule agent that promotes the assembly of microtubules from tubulin dimers and stabilizes microtubules by preventing depolymerization. This stability results in the inhibition of the normal dynamic reorganization of the microtubule network that is essential for vital interphase and mitotic cellular functions. In addition, paclitaxel induces abnormal arrays or “bundles” of microtubules throughout the cell cycle and multiple asters of microtubules during mitosis.
Following intravenous administration of paclitaxel, paclitaxel plasma concentrations declined in a biphasic manner. The initial rapid decline represents distribution to the peripheral compartment and elimination of the drug. The later phase is due, in part, to a relatively slow efflux of paclitaxel from the peripheral compartment.
Pharmacokinetic parameters of paclitaxel following 3- and 24-hour infusions of paclitaxel at dose levels of 135 and 175 mg/m2 were determined in a Phase 3 randomized study in ovarian cancer patients and are summarized in the following table:
|
Cmax = Maximum plasma concentration |
||||||
|
AUC (0-∞) = Area under the plasma concentration-time curve from time 0 to infinity |
||||||
|
CLT = Total body clearance |
||||||
|
Dose
(mg/m 2 ) |
Infusion Duration (h) |
N
(patients) |
C
max
(ng/mL) |
AUC (0-∞)
(ng•h/mL) |
T-HALF (h) |
CL
T
(L/h/m 2 ) |
| 135 | 24 | 2 | 195 | 6300 | 52.7 | 21.7 |
| 175 | 24 | 4 | 365 | 7993 | 15.7 | 23.8 |
| 135 | 3 | 7 | 2170 | 7952 | 13.1 | 17.7 |
| 175 | 3 | 5 | 3650 | 15007 | 20.2 | 12.2 |
It appeared that with the 24-hour infusion of paclitaxel, a 30% increase in dose (135 mg/m2 versus 175 mg/m2) increased the Cmax by 87%, whereas the AUC (0-∞) remained proportional. However, with a 3-hour infusion, for a 30% increase in dose, the Cmax and AUC (0-∞) were increased by 68% and 89%, respectively. The mean apparent volume of distribution at steady state, with the 24-hour infusion of paclitaxel, ranged from 227 to 688 L/m2 indicating extensive extravascular distribution and/or tissue binding of paclitaxel.
The pharmacokinetics of paclitaxel were also evaluated in adult cancer patients who received single doses of 15 to 135 mg/m2 given by 1-hour infusions (n=15), 30 to 275 mg/m2 given by 6-hour infusions (n=36), and 200 to 275 mg/m2 given by 24-hour infusions (n=54) in Phase 1 & 2 studies. Values for CLT and volume of distribution were consistent with the findings in the Phase 3 study. The pharmacokinetics of paclitaxel in patients with AIDS-related Kaposi's sarcoma have not been studied.
In vitro studies of binding to human serum proteins, using paclitaxel concentrations ranging from 0.1 to 50 mcg/mL, indicate that between 89% to 98% of drug is bound; the presence of cimetidine, ranitidine, dexamethasone, or diphenhydramine did not affect protein binding of paclitaxel.
After intravenous administration of 15 to 275 mg/m2 doses of Paclitaxel Injection, USP as 1-, 6-, or 24-hour infusions, mean values for cumulative urinary recovery of unchanged drug ranged from 1.3% to 12.6% of the dose, indicating extensive non-renal clearance. In five patients administered a 225 or 250 mg/m2 dose of radiolabeled paclitaxel as a 3-hour infusion, a mean of 71% of the radioactivity was excreted in the feces in 120 hours, and 14% was recovered in the urine. Total recovery of radioactivity ranged from 56% to 101% of the dose. Paclitaxel represented a mean of 5% of the administered radioactivity recovered in the feces, while metabolites, primarily 6α-hydroxypaclitaxel, accounted for the balance. In vitro studies with human liver microsomes and tissue slices showed that paclitaxel was metabolized primarily to 6α-hydroxypaclitaxel by the cytochrome P450 isozyme CYP2C8; and to two minor metabolites, 3'-p-hydroxypaclitaxel and 6α, 3'-p-dihydroxy-paclitaxel, by CYP3A4. In vitro, the metabolism of paclitaxel to 6α-hydroxypaclitaxel was inhibited by a number of agents (ketoconazole, verapamil, diazepam, quinidine, dexamethasone, cyclosporin, teniposide, etoposide, and vincristine), but the concentrations used exceeded those found in vivo following normal therapeutic doses. Testosterone, 17α-ethinyl estradiol, retinoic acid, and quercetin, a specific inhibitor of CYP2C8, also inhibited the formation of 6α-hydroxypaclitaxel in vitro. The pharmacokinetics of paclitaxel may also be altered in vivo as a result of interactions with compounds that are substrates, inducers, or inhibitors of CYP2C8 and/or CYP3A4. (See PRECAUTIONS: Drug Interactions section.)
The disposition and toxicity of paclitaxel 3-hour infusion were evaluated in 35 patients with varying degrees of hepatic function. Relative to patients with normal bilirubin, plasma paclitaxel exposure in patients with abnormal serum bilirubin ≤2 times upper limit of normal (ULN) administered 175 mg/m2 was increased, but with no apparent increase in the frequency or severity of toxicity. In 5 patients with serum total bilirubin >2 times ULN, there was a statistically nonsignificant higher incidence of severe myelosuppression, even at a reduced dose (110 mg/m2), but no observed increase in plasma exposure. (See PRECAUTIONS: Hepatic and DOSAGE AND ADMINISTRATION.) The effect of renal or hepatic dysfunction on the disposition of paclitaxel has not been investigated.
Possible interactions of paclitaxel with concomitantly administered medications have not been formally investigated.
Aids Related Kaposi's Sarcoma
Data from 2, Phase 2 open-label studies support the use of paclitaxel as second-line therapy in patients with AIDS-related Kaposi's sarcoma. Fifty-nine of the 85 patients enrolled in these studies had previously received systemic therapy, including interferon alpha (32%), DaunoXome® (31%), DOXIL® (2%), and doxorubicin containing chemotherapy (42%), with 64% having received prior anthracyclines. Eighty-five percent of the pretreated patients had progressed on, or could not tolerate, prior systemic therapy.1
In Study CA139-174, patients received paclitaxel at 135 mg/m2 as a 3-hour infusion every 3 weeks (intended dose intensity 45 mg/m2/week). If no dose-limiting toxicity was observed, patients were to receive 155 mg/m2 and 175 mg/m2 in subsequent courses. Hematopoietic growth factors were not to be used initially. In Study CA139-281, patients received paclitaxel at 100 mg/m2 as a 3-hour infusion every 2 weeks (intended dose intensity 50 mg/m2/week). In this study patients could be receiving hematopoietic growth factors before the start of paclitaxel therapy, or this support was to be initiated as indicated; the dose of paclitaxel was not increased. The dose intensity of paclitaxel used in this patient population was lower than the dose intensity recommended for other solid tumors.
All patients had widespread and poor-risk disease. Applying the ACTG staging criteria to patients with prior systemic therapy, 93% were poor risk for extent of disease (T1), 88% had a CD4 count <200 cells/mm3 (I1), and 97% had poor risk considering their systemic illness (S1).
All patients in Study CA139-174 had a Karnofsky performance status of 80 or 90 at baseline; in Study CA139-281, there were 26 (46%) patients with a Karnofsky performance status of 70 or worse at baseline.
|
Prior Systemic Therapy
(n=59) |
|
| Visceral ± edema ± oral ± cutaneous | 42 |
| Edema or lymph nodes ± oral ± cutaneous | 41 |
| Oral ± cutaneous | 10 |
| Cutaneous only | 7 |
Although the planned dose intensity in the 2 studies was slightly different (45 mg/m2/week in Study CA139-174 and 50 mg/m2/week in Study CA139-281), delivered dose intensity was 38 to 39 mg/m2/week in both studies, with a similar range (20 to 24 to 51 to 61).
Non Small Cell Lung Carcinoma (nsclc)
In a Phase 3 open-label randomized study conducted by the ECOG, 599 patients were randomized to either paclitaxel (T) 135 mg/m2 as a 24-hour infusion in combination with cisplatin (c) 75 mg/m2, paclitaxel (T) 250 mg/m2 as a 24-hour infusion in combination with cisplatin (c) 75 mg/m2 with G-CSF support, or cisplatin (c) 75 mg/m2 on day 1, followed by etoposide (VP) 100 mg/m2 on days 1, 2, and 3 (control). Response rates, median time to progression, median survival, and 1-year survival rates are given in the following table. The reported p-values have not been adjusted for multiple comparisons. There were statistically significant differences favoring each of the paclitaxel plus cisplatin arms for response rate and time to tumor progression. There was no statistically significant difference in survival between either paclitaxel plus cisplatin arm and the cisplatin plus etoposide arm.
|
a Etoposide (VP) 100 mg/m2 was administered IV on days 1, 2, and 3. |
|||
|
b Compared to cisplatin/etoposide. |
|||
|
T135/24
c75 (n=198) |
T250/24
c75 (n=201) |
VP100
a
c75 (n=200) |
|
| • Response
- rate (percent) - p-valueb |
25 0.001 |
23 <0.001 |
12 |
|
• Time to Progression
- median (months) - p-valueb |
4.3 0.05 |
4.9 0.004 |
2.7 |
|
• Survival
- median (months) - p-valueb |
9.3 0.12 |
10.0 0.08 |
7.4 |
|
• 1-Year Survival
- percent of patients |
36 |
40 |
32 |
In the ECOG study, the Functional Assessment of Cancer Therapy-Lung (FACT-L) questionnaire had 7 subscales that measured subjective assessment of treatment. Of the 7, the Lung Cancer Specific Symptoms subscale favored the paclitaxel 135 mg/m2/24 hour plus cisplatin arm compared to the cisplatin/etoposide arm. For all other factors, there was no difference in the treatment groups.
The adverse event profile for patients who received paclitaxel in combination with cisplatin in this study was generally consistent with that seen for the pooled analysis of data from 812 patients treated with single-agent paclitaxel in 10 clinical studies. These adverse events and adverse events from the Phase 3 first-line NSCLC study are described in the ADVERSE REACTIONS section in tabular (Tables 10 and 15) and narrative form.
Adverse Event Experiences By Body System
The following discussion refers to the overall safety database of 812 patients with solid tumors treated with single-agent paclitaxel in clinical studies. Toxicities that occurred with greater severity or frequency in previously untreated patients with ovarian carcinoma or NSCLC who received paclitaxel in combination with cisplatin or in patients with breast cancer who received paclitaxel after doxorubicin/cyclophosphamide in the adjuvant setting and that occurred with a difference that was clinically significant in these populations are also described.
The frequency and severity of important adverse events for the Phase 3 ovarian carcinoma, breast carcinoma, NSCLC, and the Phase 2 Kaposi's sarcoma carcinoma studies are presented above in tabular form by treatment arm. In addition, rare events have been reported from postmarketing experience or from other clinical studies. The frequency and severity of adverse events have been generally similar for patients receiving paclitaxel for the treatment of ovarian, breast, or lung carcinoma or Kaposi's sarcoma, but patients with AIDS-related Kaposi's sarcoma may have more frequent and severe hematologic toxicity, infections (including opportunistic infections, see Table 16), and febrile neutropenia. These patients require a lower dose intensity and supportive care. (See CLINICAL STUDIES: AIDS-Related Kaposi's Sarcoma .) Toxicities that were observed only in or were noted to have occurred with greater severity in the population with Kaposi's sarcoma and that occurred with a difference that was clinically significant in this population are described. Elevated liver function tests and renal toxicity have a higher incidence in KS patients as compared to patients with solid tumors.
Structured Label Content
Section 34073-7 (34073-7)
Drug Interactions: In a Phase I trial using escalating doses of paclitaxel (110 to 200 mg/m2) and cisplatin (50 or 75 mg/m2) given as sequential infusions, myelosuppression was more profound when paclitaxel was given after cisplatin than with the alternate sequence (i.e., paclitaxel before cisplatin). Pharmacokinetic data from these patients demonstrated a decrease in paclitaxel clearance of approximately 33% when paclitaxel was administered following cisplatin.
The metabolism of paclitaxel is catalyzed by cytochrome P450 isoenzymes CYP2C8 and CYP3A4. In the absence of formal clinical drug interaction studies, caution should be exercised when administering paclitaxel concomitantly with known substrates or inhibitors of the cytochrome P450 isoenzymes CYP2C8 and CYP3A4. Caution should be exercised when paclitaxel is concomitantly administered with known substrates (e.g., midazolam, buspirone, felodipine, lovastatin, eletriptan, sildenafil, simvastatin, and triazolam), inhibitors (e.g., atazanavir, clarithromycin, indinavir, itraconazole, ketoconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin), and inducers (e.g., rifampin and carbamazepine) of CYP3A4. (See CLINICAL PHARMACOLOGY section.)
Caution should also be exercised when paclitaxel is concomitantly administered with known substrates (e.g., repaglinide and rosiglitazone), inhibitors (e.g., gemfibrozil), and inducers (e.g., rifampin) of CYP2C8. (See CLINICAL PHARMACOLOGY.)
Potential interactions between paclitaxel, a substrate of CYP3A4, and protease inhibitors (ritonavir, saquinavir, indinavir, and nelfinavir), which are substrates and/or inhibitors of CYP3A4, have not been evaluated in clinical trials.
Reports in the literature suggest that plasma levels of doxorubicin (and its active metabolite doxorubicinol) may be increased when paclitaxel and doxorubicin are used in combination.
Section 34076-0 (34076-0)
Information for Patients: (See Patient Information Leaflet.)
Section 34080-2 (34080-2)
Nursing Mothers: It is not known whether the drug is excreted in human milk. Following intravenous administration of carbon-14 labeled paclitaxel to rats on days 9 to 10 postpartum, concentrations of radioactivity in milk were higher than in plasma and declined in parallel with the plasma concentrations. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants, it is recommended that nursing be discontinued when receiving paclitaxel therapy.
Section 34081-0 (34081-0)
Pediatric Use: The safety and effectiveness of paclitaxel in pediatric patients have not been established.
There have been reports of central nervous system (CNS) toxicity (rarely associated with death) in a clinical trial in pediatric patients in which paclitaxel was infused intravenously over 3 hours at doses ranging from 350 mg/m2 to 420 mg/m2. The toxicity is most likely attributable to the high dose of the ethanol component of the Paclitaxel Injection, USP vehicle given over a short infusion time. The use of concomitant antihistamines may intensify this effect. Although a direct effect of the paclitaxel itself cannot be discounted, the high doses used in this study (over twice the recommended adult dosage) must be considered in assessing the safety of paclitaxel for use in this population.
Section 34082-8 (34082-8)
Geriatric Use: Of 2228 patients who received paclitaxel in 8 clinical studies evaluating its safety and effectiveness in the treatment of advanced ovarian cancer, breast carcinoma, or NSCLC, and 1570 patients who were randomized to receive paclitaxel in the adjuvant breast cancer study, 649 patients (17%) were 65 years or older and 49 patients (1%) were 75 years or older. In most studies, severe myelosuppression was more frequent in elderly patients; in some studies, severe neuropathy was more common in elderly patients. In 2 clinical studies in NSCLC, the elderly patients treated with paclitaxel had a higher incidence of cardiovascular events. Estimates of efficacy appeared similar in elderly patients and in younger patients; however, comparative efficacy cannot be determined with confidence due to the small number of elderly patients studied. In a study of first-line treatment of ovarian cancer, elderly patients had a lower median survival than younger patients, but no other efficacy parameters favored the younger group. Table 9 presents the incidences of Grade IV neutropenia and severe neuropathy in clinical studies according to age.
|
INDICATION (Study/Regimen) |
Patients (n/total [%]) | |||
|---|---|---|---|---|
|
Neutropenia
(Grade IV) |
Peripheral Neuropathy
(Grades III/IV) |
|||
| Age (y) | Age (y) | |||
| ≥65 | <65 | ≥65 | <65 | |
|
* p<0.05 |
||||
|
a Paclitaxel dose in mg/m2/infusion duration in hours; cisplatin doses in mg/m2. |
||||
|
b Peripheral neuropathy was included within the neurotoxicity category in the Intergroup First-Line Ovarian Cancer study (see Table 11). |
||||
|
c Paclitaxel dose in mg/m2/infusion duration in hours. |
||||
|
d Paclitaxel (T) following 4 courses of doxorubicin and cyclophosphamide (AC) at a dose of 175 mg/m2/3 hours every 3 weeks for 4 courses. |
||||
|
e Peripheral neuropathy reported as neurosensory toxicity in the Intergroup Adjuvant Breast Cancer study (see Table 13). |
||||
|
f Peripheral neuropathy reported as neurosensory toxicity in the ECOG NSCLC study (see Table 15). |
||||
|
• OVARIAN Cancer
(Intergroup First-Line/T175/3 c75a) (GOG-111 First-Line/T135/24 c75a) (Phase 3 Second-Line/T175/3c) (Phase 3 Second-Line/T175/24c) (Phase 3 Second-Line/T135/3c) (Phase 3 Second-Line/T135/24c) (Phase 3 Second-Line Pooled) |
34/83 (41) 48/61 (79) 5/19 (26) 21/25 (84) 4/16 (25) 17/22 (77) 47/82 (57)* |
78/252 (31) 106/129 (82) 21/76 (28) 57/79 (72) 10/81 (12) 53/83 (64) 141/319 (44) |
24/84 (29)*b 3/62 (5) 1/19 (5) 0/25 (0) 0/17 (0) 0/22 (0) 1/83 (1) |
46/255 (18)b 2/134 (1) 0/76 (0) 2/80 (3) 0/81 (0) 0/83 (0) 2/320 (1) |
|
• Adjuvant BREAST Cancer
(Intergroup/AC followed by Td) |
56/102 (55) |
734/1468 (50) |
5/102 (5)e |
46/1468 (3)e |
|
• BREAST Cancer after Failure of Initial Therapy
(Phase 3/T175/3c) (Phase 3/T135/3c) |
7/24 (29) 7/20 (35) |
56/200 (28) 37/207 (18) |
3/25 (12) 0/20 (0) |
12/204 (6) 6/209 (3) |
| • Non-Small Cell LUNG Cancer
(ECOG/T135/24 c75a) (Phase 3/T175/3 c80a) |
58/71 (82) 37/89 (42)* |
86/124 (69) 56/267 (21) |
9/71 (13)f 11/91 (12)* |
16/124 (13)f 11/271 (4) |
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, Impairment of Fertility: The carcinogenic potential of paclitaxel has not been studied.
Paclitaxel has been shown to be clastogenic in vitro (chromosome aberrations in human lymphocytes) and in vivo (micronucleus test in mice). Paclitaxel was not mutagenic in the Ames test of the CHO/HGPRT gene mutation assay.
Administration of paclitaxel prior to and during mating produced impairment of fertility in male and female rats at doses equal to or greater than 1 mg/kg/day (about 0.04 the daily maximum recommended human dose on a mg/m2 basis). At this dose, paclitaxel caused reduced fertility and reproductive indices, and increased embryo- and fetotoxicity. (See WARNINGS section.)
Section 42228-7 (42228-7)
Pregnancy: Pregnancy Category D. (See WARNINGS section.)
Section 42229-5 (42229-5)
(Patient Information Included)
SAGENT®
Rx only
Section 42230-3 (42230-3)
PATIENT INFORMATION
Paclitaxel Injection, USP
Read this patient information leaflet before you start taking paclitaxel. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is the most important information I should know about paclitaxel?
Paclitaxel can cause serious side effects including death.
Serious allergic reactions (anaphylaxis) can happen in people who receive paclitaxel.
Anaphylaxis is a serious medical emergency that can lead to death and must be treated right away.
Tell your healthcare provider right away if you have any of these signs of an allergic reaction:
- trouble breathing
- sudden swelling of your face, lips, tongue, throat, or trouble swallowing
- hives (raised bumps) or rash
Your healthcare provider will give you medicines to lessen your chance of having an allergic reaction.
What is paclitaxel?
Paclitaxel is a prescription medicine used to treat some forms of:
- ovarian cancer
- breast cancer
- lung cancer
- Kaposi's sarcoma
It is not known if paclitaxel is safe or effective in children.
Who should not receive paclitaxel?
Do not receive paclitaxel if:
- you are allergic to any of the ingredients in paclitaxel. See the end of this leaflet for a complete list of ingredients in paclitaxel.
- are allergic to medicines containing polyoxyethylated castor oil.
- you have low white blood cell counts.
What should I tell my healthcare provider before receiving paclitaxel?
Before receiving paclitaxel, tell your healthcare provider about all your medical conditions, including if you:
- have liver problems
- have heart problems
- are pregnant or plan to become pregnant. paclitaxel can harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- are breast-feeding or plan to breast-feed. It is not known if paclitaxel passes into your breast milk. You and your healthcare provider should decide if you will receive paclitaxel or breast-feed.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How will I receive paclitaxel?
- Paclitaxel is injected into a vein (intravenous [IV] infusion) by your healthcare provider.
Your healthcare provider will do certain tests while you receive paclitaxel.
What are the possible side effects of paclitaxel?
Tell your healthcare provider right away if you have:
- severe stomach pain
- severe diarrhea
The most common side effects of paclitaxel include:
- low red blood cell count (anemia) feeling weak or tired
- hair loss
- numbness, tingling, or burning in your hands or feet (neuropathy)
- joint and muscle pain
- nausea and vomiting
- hypersensitivity reaction - trouble breathing; sudden swelling of your face, lips, tongue, throat, or trouble swallowing; hives (raised bumps) or rash
- diarrhea
- mouth or lip sores (mucositis)
- infections - if you have a fever (temperature above 100.4°F) or other sign of infection, tell your healthcare provider right away
- swelling of your hands, face, or feet
- bleeding events
- irritation at the injection site
- low blood pressure (hypotension)
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of paclitaxel. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
General information about the safe and effective use of paclitaxel.
Medicines are sometimes prescribed for purposes other than those listed in a patient information leaflet. Do not use paclitaxel for a condition for which it was not prescribed.
Do not give paclitaxel to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about paclitaxel. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about paclitaxel that is written for health professionals.
What are the ingredients in paclitaxel?
Active ingredient: paclitaxel.
Inactive ingredients include: Polyoxyl 35 castor oil, NF and dehydrated alcohol, USP and Citric Acid, USP.
What is cancer?
Under normal conditions, the cells in your body divide and grow in an orderly, controlled way. Cell division and growth are necessary for the human body to perform its functions and to repair itself, when necessary. Cancer cells are different from normal cells because they are not able to control their own growth. The reasons for this abnormal growth are not yet fully understood. A tumor is a mass of unhealthy cells that are dividing and growing fast and in an uncontrolled way. When a tumor invades surrounding healthy body tissue, it is known as a malignant tumor. A malignant tumor can spread (metastasize) from its original site to other parts of the body if not found and treated early.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
November 2023
SAGENT Pharmaceuticals ®
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-255-05
Rx only
Paclitaxel Injection, USP
30 mg per 5 mL
(6 mg per mL)
Caution: Dilution required prior to Intravenous Infusion
CAUTION: Cytotoxic Agent
Sterile Multi-Dose Vial
Warning (WARNING)
Paclitaxel Injection, USP should be administered under the supervision of a physician experienced in the use of cancer chemotherapeutic agents. Appropriate management of complications is possible only when adequate diagnostic and treatment facilities are readily available.
Anaphylaxis and severe hypersensitivity reactions characterized by dyspnea and hypotension requiring treatment, angioedema, and generalized urticaria have occurred in 2% to 4% of patients receiving paclitaxel in clinical trials. Fatal reactions have occurred in patients despite premedication. All patients should be pretreated with corticosteroids, diphenhydramine, and H2 antagonists. (See DOSAGE AND ADMINISTRATION section.) Patients who experience severe hypersensitivity reactions to paclitaxel should not be rechallenged with the drug.
Paclitaxel therapy should not be given to patients with solid tumors who have baseline neutrophil counts of less than 1,500 cells/mm3 and should not be given to patients with AIDS-related Kaposi's sarcoma if the baseline neutrophil count is less than 1,000 cells/mm3. In order to monitor the occurrence of bone marrow suppression, primarily neutropenia, which may be severe and result in infection, it is recommended that frequent peripheral blood cell counts be performed on all patients receiving paclitaxel.
Warnings (WARNINGS)
Anaphylaxis and severe hypersensitivity reactions characterized by dyspnea and hypotension requiring treatment, angioedema, and generalized urticaria have occurred in 2% to 4% of patients receiving paclitaxel in clinical trials. Fatal reactions have occurred in patients despite premedication. All patients should be pretreated with corticosteroids, diphenhydramine, and H2 antagonists. (See DOSAGE AND ADMINISTRATION section.) Patients who experience severe hypersensitivity reactions to paclitaxel should not be rechallenged with the drug.
Bone marrow suppression (primarily neutropenia) is dose-dependent and is the dose-limiting toxicity. Neutrophil nadirs occurred at a median of 11 days. Paclitaxel should not be administered to patients with baseline neutrophil counts of less than 1,500 cells/mm3 (<1,000 cells/mm3 for patients with KS). Frequent monitoring of blood counts should be instituted during paclitaxel treatment. Patients should not be re-treated with subsequent cycles of paclitaxel until neutrophils recover to a level >1,500 cells/mm3 (>1,000 cells/mm3 for patients with KS) and platelets recover to a level >100,000 cells/mm3.
Severe conduction abnormalities have been documented in <1% of patients during paclitaxel therapy and in some cases requiring pacemaker placement. If patients develop significant conduction abnormalities during paclitaxel infusion, appropriate therapy should be administered and continuous cardiac monitoring should be performed during subsequent therapy with paclitaxel.
Overdosage (OVERDOSAGE)
There is no known antidote for paclitaxel overdosage. The primary anticipated complications of overdosage would consist of bone marrow suppression, peripheral neurotoxicity, and mucositis. Overdoses in pediatric patients may be associated with acute ethanol toxicity (see PRECAUTIONS: Pediatric Use section).
References (REFERENCES)
- NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004165.
- OSHA Technical Manual, TED 1-0.15A, Section VI: Chapter 2. Controlling occupational exposure to hazardous drugs. OSHA, 1999. http://www.osha.gov/dts/osta/otm/otm_vi/otm_vi_2.html.
- American Society of Health-System Pharmacists. ASHP guidelines on handling hazardous drugs. Am J Health-Syst Pharm. 2006; 63:1172-1193.
- Polovich M, White JM, Kelleher LO, eds. 2005. Chemotherapy and biotherapy guidelines and recommendations for practice. 2nd ed. Pittsburgh, PA: Oncology Nursing Society.
Brands listed are the trademarks of their respective owners.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2023 Sagent Pharmaceuticals
November 2023
SAGENT Pharmaceuticals ®
Description (DESCRIPTION)
Paclitaxel Injection, USP is a clear colorless to slightly yellow viscous solution. It is supplied as a nonaqueous solution intended for dilution with a suitable parenteral fluid prior to intravenous infusion. Paclitaxel is available in 30 mg (5 mL), 100 mg (16.7 mL), and 300 mg (50 mL) multi-dose vials. Each mL of sterile nonpyrogenic solution contains 6 mg paclitaxel, 527 mg of Polyoxyl 35 Castor Oil, NF, 49.7% (v/v) Dehydrated Alcohol, USP and 2 mg Citric Acid, USP.
Paclitaxel is a natural product with antitumor activity. Paclitaxel is obtained via an extraction process from Taxus X media ‘Hicksii’. The chemical name for paclitaxel is (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,-12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca [3,4] benz [1,2-b] oxet-5-one 6,12b-diacetate, 12-benzoate, 9-ester with (2R,3S)-N-benzoyl-3-phenylisoserine.
Paclitaxel has the following structural formula:
Paclitaxel is a white to off-white crystalline powder with the empirical formula C47H51NO14 and a molecular weight of 853.9. It is highly lipophilic, insoluble in water, and melts at around 216 to 217°C.
Precautions (PRECAUTIONS)
Contact of the undiluted concentrate with plasticized polyvinyl chloride (PVC) equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP [di-(2-ethylhexyl) phthalate], which may be leached from PVC infusion bags or sets, diluted Paclitaxel Injection, USP solutions should preferably be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets.
Paclitaxel should be administered through an in-line filter with a microporous membrane not greater than 0.22 microns. Use of filter devices such as IVEX-2® filters which incorporate short inlet and outlet PVC-coated tubing has not resulted in significant leaching of DEHP.
How Supplied (HOW SUPPLIED)
Paclitaxel Injection, USP is supplied as follows:
| NDC | Paclitaxel Injection, USP (6 mg per mL) | Package Factor |
| 25021-255-05 | 30 mg per 5 mL Multi-Dose Vial | 1 vial per carton |
| 25021-255-17 | 100 mg per 16.7 mL Multi-Dose Vial | 1 vial per carton |
| 25021-255-50 | 300 mg per 50 mL Multi-Dose Vial | 1 vial per carton |
Adjuvant Therapy
A Phase 3 Intergroup study (Cancer and Leukemia Group B [CALGB], Eastern Cooperative Oncology Group [ECOG], North Central Cancer Treatment Group [NCCTG], and Southwest Oncology Group [SWOG]) randomized 3170 patients with node-positive breast carcinoma to adjuvant therapy with paclitaxel or to no further chemotherapy following 4 courses of doxorubicin and cyclophosphamide (AC). This multicenter trial was conducted in women with histologically positive lymph nodes following either a mastectomy or segmental mastectomy and nodal dissections. The 3 x 2 factorial study was designed to assess the efficacy and safety of 3 different dose levels of doxorubicin (A) and to evaluate the effect of the addition of paclitaxel administered following the completion of AC therapy. After stratification for the number of positive lymph nodes (1 to 3, 4 to 9, or 10+), patients were randomized to receive cyclophosphamide at a dose of 600 mg/m2 and doxorubicin at doses of either 60 mg/m2 (on day 1), 75 mg/m2 (in 2 divided doses on days 1 and 2), or 90 mg/m2 (in 2 divided doses on days 1 and 2 with prophylactic G-CSF support and ciprofloxacin) every 3 weeks for 4 courses and either paclitaxel 175 mg/m2 as a 3-hour infusion every 3 weeks for 4 additional courses or no additional chemotherapy. Patients whose tumors were positive were to receive subsequent tamoxifen treatment (20 mg daily for 5 years); patients who received segmental mastectomies prior to study were to receive breast irradiation after recovery from treatment-related toxicities.
At the time of the current analysis, median follow-up was 30.1 months. Of the 2066 patients who were hormone receptor positive, 93% received tamoxifen. The primary analyses of disease-free survival and overall survival used multivariate Cox models, which included paclitaxel administration, doxorubicin dose, number of positive lymph nodes, tumor size, menopausal status, and estrogen receptor status as factors. Based on the model for disease-free survival, patients receiving AC followed by paclitaxel had a 22% reduction in the risk of disease recurrence compared to patients randomized to AC alone (Hazard Ratio [HR]=0.78, 95% CI, 0.67 to 0.91, p=0.0022). They also had a 26% reduction in the risk of death (HR=0.74, 95% CI, 0.60 to 0.92, p=0.0065). For disease-free survival and overall survival, p-values were not adjusted for interim analyses. Kaplan-Meier curves are shown in Figures 3 and 4. Increasing the dose of doxorubicin higher than 60 mg/m2 had no effect on either disease-free survival or overall survival.
Figure 3. Disease-Free Survival: AC Versus AC+T
Figure 4. Survival: AC Versus AC+T
Subset Analyses: Subsets defined by variables of known prognostic importance in adjuvant breast carcinoma were examined, including number of positive lymph nodes, tumor size, hormone receptor status, and menopausal status. Such analyses must be interpreted with care, as the most secure finding is the overall study result. In general, a reduction in hazard similar to the overall reduction was seen with paclitaxel for both disease-free and overall survival in all of the larger subsets with one exception; patients with receptor-positive tumors had a smaller reduction in hazard (HR=0.92) for disease-free survival with paclitaxel than other groups. Results of subset analyses are shown in Table 4.
|
a Positive for either estrogen or progesterone receptors. |
|||||
|
b Negative or missing for both estrogen and progesterone receptors (both missing: n=15). |
|||||
|
Patient Subset |
Disease-Free Survival | Overall Survival | |||
|
No. of
Patients |
No. of
Recurrences |
Hazard Ratio
(95% CI) |
No. of
Deaths |
Hazard Ratio
(95% CI) |
|
| • No. of Positive Nodes
1 to 3 4 to 9 10+ |
1449 1310 360 |
221 274 129 |
0.72 (0.55 to 0.94) 0.78 (0.61 to 0.99) 0.93 (0.66 to 1.31) |
107 148 87 |
0.76 (0.52 to 1.12) 0.66 (0.47 to 0.91) 0.90 (0.59 to 1.36) |
| • Tumor Size (cm)
≤2 >2 and ≤5 >5 |
1096 1611 397 |
153 358 111 |
0.79 (0.57 to 1.08) 0.79 (0.64 to 0.97) 0.75 (0.51 to 1.08) |
67 201 72 |
0.73 (0.45 to 1.18) 0.74 (0.56 to 0.98) 0.73 (0.46 to 1.16) |
| • Menopausal Status
Pre Post |
1929 1183 |
374 250 |
0.83 (0.67 to 1.01) 0.73 (0.57 to 0.93) |
187 155 |
0.72 (0.54 to 0.97) 0.77 (0.56 to 1.06) |
| • Receptor Status
Positivea Negative/Unknownb |
2066 1055 |
293 331 |
0.92 (0.73 to 1.16) 0.68 (0.55 to 0.85) |
126 216 |
0.83 (0.59 to 1.18) 0.71 (0.54 to 0.93) |
These retrospective subgroup analyses suggest that the beneficial effect of paclitaxel is clearly established in the receptor-negative subgroup, but the benefit in receptor-positive patients is not yet clear. With respect to menopausal status, the benefit of paclitaxel is consistent (see Table 4 and Figures 5 to 8).
Figure 5. Disease-Free Survival: Receptor Status Negative/Unknown AC Versus AC+T
Figure 6. Disease-Free Survival: Receptor Status Positive AC Versus AC+T
Figure 7. Disease-Free Survival: Premenopausal AC Versus AC+T
Figure 8. Disease-Free Survival: Postmenopausal AC Versus AC+T
The adverse event profile for the patients who received paclitaxel subsequent to AC was consistent with that seen in the pooled analysis of data from 812 patients (Table 10) treated with single-agent paclitaxel in 10 clinical studies. These adverse events are described in the ADVERSE REACTIONS section in tabular (Tables 10 and 13) and narrative form.
Adverse Reactions (ADVERSE REACTIONS)
Pooled Analysis of Adverse Event Experiences from Single-Agent Studies: Data in the following table are based on the experience of 812 patients (493 with ovarian carcinoma and 319 with breast carcinoma) enrolled in 10 studies who received single-agent Paclitaxel Injection, USP. Two hundred and seventy-five patients were treated in eight Phase 2 studies with paclitaxel doses ranging from 135 to 300 mg/m2 administered over 24 hours (in four of these studies, G-CSF was administered as hematopoietic support). Three hundred and one patients were treated in the randomized Phase 3 ovarian carcinoma study which compared two doses (135 or 175 mg/m2) and two schedules (3 or 24 hours) of paclitaxel. Two hundred and thirty-six patients with breast carcinoma received paclitaxel (135 or 175 mg/m2) administered over 3 hours in a controlled study.
|
Percent of Patients
(n=812) |
||
|---|---|---|
|
a Based on worst course analysis. |
||
|
b All patients received premedication. |
||
|
c During the first 3 hours of infusion. |
||
|
† Severe events are defined as at least Grade III toxicity. |
||
| • Bone Marrow |
|
|
| - Neutropenia | <2,000/mm3 | 90 |
| <500/mm3
|
52 |
|
| - Leukopenia | <4,000/mm3 | 90 |
| <1,000/mm3 | 17 |
|
| - Thrombocytopenia | <100,000/mm3 | 20 |
| <50,000/mm3 | 7 |
|
| - Anemia | <11 g/dL | 78 |
| <8 g/dL | 16 |
|
| - Infections |
30 |
|
| - Bleeding |
14 |
|
| - Red Cell Transfusions |
25 |
|
| - Platelet Transfusions | 2 | |
| • Hypersensitivity Reaction
b
- All - Severe† |
41 2 |
|
|
• Cardiovascular
- Vital Sign Changesc - Bradycardia (n=537) - Hypotension (n=532) - Significant Cardiovascular Events |
3 12 1 |
|
|
• Abnormal ECG
- All Pts - Pts with normal baseline (n=559) |
23 14 |
|
|
• Peripheral Neuropathy
- Any symptoms - Severe symptoms† |
60 3 |
|
|
• Myalgia/Arthralgia
- Any symptoms - Severe symptoms† |
60 8 |
|
|
• Gastrointestinal
- Nausea and vomiting - Diarrhea - Mucositis |
52 38 31 |
|
| • Alopecia | 87 | |
|
• Hepatic (Pts with normal baseline and on study data)
- Bilirubin elevations (n=765) - Alkaline phosphatase elevations (n=575) - AST (SGOT) elevations (n=591) |
7 22 19 |
|
| • Injection Site Reaction | 13 |
None of the observed toxicities were clearly influenced by age.
Contraindications (CONTRAINDICATIONS)
Paclitaxel Injection, USP is contraindicated in patients who have a history of hypersensitivity reactions to paclitaxel or other drugs formulated in Polyoxyl 35 Castor Oil, NF.
Paclitaxel Injection, USP should not be used in patients with solid tumors who have baseline neutrophil counts of <1,500 cells/mm3 or in patients with AIDS-related Kaposi's sarcoma with baseline neutrophil counts of <1,000 cells/mm3.
Storage Conditions
Store the vials in original cartons between 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Retain in carton until time of use.
Handling and Disposal: See DOSAGE AND ADMINISTRATION: Preparation and Administration Precautions.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Paclitaxel is a novel antimicrotubule agent that promotes the assembly of microtubules from tubulin dimers and stabilizes microtubules by preventing depolymerization. This stability results in the inhibition of the normal dynamic reorganization of the microtubule network that is essential for vital interphase and mitotic cellular functions. In addition, paclitaxel induces abnormal arrays or “bundles” of microtubules throughout the cell cycle and multiple asters of microtubules during mitosis.
Following intravenous administration of paclitaxel, paclitaxel plasma concentrations declined in a biphasic manner. The initial rapid decline represents distribution to the peripheral compartment and elimination of the drug. The later phase is due, in part, to a relatively slow efflux of paclitaxel from the peripheral compartment.
Pharmacokinetic parameters of paclitaxel following 3- and 24-hour infusions of paclitaxel at dose levels of 135 and 175 mg/m2 were determined in a Phase 3 randomized study in ovarian cancer patients and are summarized in the following table:
|
Cmax = Maximum plasma concentration |
||||||
|
AUC (0-∞) = Area under the plasma concentration-time curve from time 0 to infinity |
||||||
|
CLT = Total body clearance |
||||||
|
Dose
(mg/m 2 ) |
Infusion Duration (h) |
N
(patients) |
C
max
(ng/mL) |
AUC (0-∞)
(ng•h/mL) |
T-HALF (h) |
CL
T
(L/h/m 2 ) |
| 135 | 24 | 2 | 195 | 6300 | 52.7 | 21.7 |
| 175 | 24 | 4 | 365 | 7993 | 15.7 | 23.8 |
| 135 | 3 | 7 | 2170 | 7952 | 13.1 | 17.7 |
| 175 | 3 | 5 | 3650 | 15007 | 20.2 | 12.2 |
It appeared that with the 24-hour infusion of paclitaxel, a 30% increase in dose (135 mg/m2 versus 175 mg/m2) increased the Cmax by 87%, whereas the AUC (0-∞) remained proportional. However, with a 3-hour infusion, for a 30% increase in dose, the Cmax and AUC (0-∞) were increased by 68% and 89%, respectively. The mean apparent volume of distribution at steady state, with the 24-hour infusion of paclitaxel, ranged from 227 to 688 L/m2 indicating extensive extravascular distribution and/or tissue binding of paclitaxel.
The pharmacokinetics of paclitaxel were also evaluated in adult cancer patients who received single doses of 15 to 135 mg/m2 given by 1-hour infusions (n=15), 30 to 275 mg/m2 given by 6-hour infusions (n=36), and 200 to 275 mg/m2 given by 24-hour infusions (n=54) in Phase 1 & 2 studies. Values for CLT and volume of distribution were consistent with the findings in the Phase 3 study. The pharmacokinetics of paclitaxel in patients with AIDS-related Kaposi's sarcoma have not been studied.
In vitro studies of binding to human serum proteins, using paclitaxel concentrations ranging from 0.1 to 50 mcg/mL, indicate that between 89% to 98% of drug is bound; the presence of cimetidine, ranitidine, dexamethasone, or diphenhydramine did not affect protein binding of paclitaxel.
After intravenous administration of 15 to 275 mg/m2 doses of Paclitaxel Injection, USP as 1-, 6-, or 24-hour infusions, mean values for cumulative urinary recovery of unchanged drug ranged from 1.3% to 12.6% of the dose, indicating extensive non-renal clearance. In five patients administered a 225 or 250 mg/m2 dose of radiolabeled paclitaxel as a 3-hour infusion, a mean of 71% of the radioactivity was excreted in the feces in 120 hours, and 14% was recovered in the urine. Total recovery of radioactivity ranged from 56% to 101% of the dose. Paclitaxel represented a mean of 5% of the administered radioactivity recovered in the feces, while metabolites, primarily 6α-hydroxypaclitaxel, accounted for the balance. In vitro studies with human liver microsomes and tissue slices showed that paclitaxel was metabolized primarily to 6α-hydroxypaclitaxel by the cytochrome P450 isozyme CYP2C8; and to two minor metabolites, 3'-p-hydroxypaclitaxel and 6α, 3'-p-dihydroxy-paclitaxel, by CYP3A4. In vitro, the metabolism of paclitaxel to 6α-hydroxypaclitaxel was inhibited by a number of agents (ketoconazole, verapamil, diazepam, quinidine, dexamethasone, cyclosporin, teniposide, etoposide, and vincristine), but the concentrations used exceeded those found in vivo following normal therapeutic doses. Testosterone, 17α-ethinyl estradiol, retinoic acid, and quercetin, a specific inhibitor of CYP2C8, also inhibited the formation of 6α-hydroxypaclitaxel in vitro. The pharmacokinetics of paclitaxel may also be altered in vivo as a result of interactions with compounds that are substrates, inducers, or inhibitors of CYP2C8 and/or CYP3A4. (See PRECAUTIONS: Drug Interactions section.)
The disposition and toxicity of paclitaxel 3-hour infusion were evaluated in 35 patients with varying degrees of hepatic function. Relative to patients with normal bilirubin, plasma paclitaxel exposure in patients with abnormal serum bilirubin ≤2 times upper limit of normal (ULN) administered 175 mg/m2 was increased, but with no apparent increase in the frequency or severity of toxicity. In 5 patients with serum total bilirubin >2 times ULN, there was a statistically nonsignificant higher incidence of severe myelosuppression, even at a reduced dose (110 mg/m2), but no observed increase in plasma exposure. (See PRECAUTIONS: Hepatic and DOSAGE AND ADMINISTRATION.) The effect of renal or hepatic dysfunction on the disposition of paclitaxel has not been investigated.
Possible interactions of paclitaxel with concomitantly administered medications have not been formally investigated.
Indications and Usage (INDICATIONS AND USAGE)
Paclitaxel Injection, USP is indicated as subsequent therapy for the treatment of advanced carcinoma of the ovary. As first-line therapy, paclitaxel is indicated in combination with cisplatin.
Paclitaxel is indicated for the adjuvant treatment of node-positive breast cancer administered sequentially to standard doxorubicin-containing combination chemotherapy. In the clinical trial, there was an overall favorable effect on disease-free and overall survival in the total population of patients with receptor-positive and receptor-negative tumors, but the benefit has been specifically demonstrated by available data (median follow-up 30 months) only in the patients with estrogen and progesterone receptor-negative tumors. (See CLINICAL STUDIES: Breast Carcinoma.)
Paclitaxel Injection, USP is indicated for the treatment of breast cancer after failure of combination chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy. Prior therapy should have included an anthracycline unless clinically contraindicated.
Paclitaxel, in combination with cisplatin, is indicated for the first-line treatment of nonsmall cell lung cancer in patients who are not candidates for potentially curative surgery and/or radiation therapy.
Paclitaxel is indicated for the second-line treatment of AIDS-related Kaposi's sarcoma.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
NOTE: Contact of the undiluted concentrate with plasticized PVC equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP [di-(2-ethylhexyl)phthalate], which may be leached from PVC infusion bags or sets, diluted paclitaxel solutions should be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets.
All patients should be premedicated prior to paclitaxel administration in order to prevent severe hypersensitivity reactions. Such premedication may consist of dexamethasone 20 mg PO administered approximately 12 and 6 hours before paclitaxel, diphenhydramine (or its equivalent) 50 mg I.V. 30 to 60 minutes prior to paclitaxel, and cimetidine (300 mg) or ranitidine (50 mg) I.V. 30 to 60 minutes before paclitaxel.
For patients with carcinoma of the ovary the following regimen is recommended: (see CLINICAL STUDIES: Ovarian Carcinoma ):
-
1)For previously untreated patients with carcinoma of the ovary, one of the following recommended regimens may be given every 3 weeks. In selecting the appropriate regimen, differences in toxicities should be considered (see Table 11 in ADVERSE REACTIONS: Disease-Specific Adverse Event Experiences ).
- Paclitaxel administered intravenously over 3 hours at a dose of 175 mg/m2 followed by cisplatin at a dose of 75 mg/m2; or
- Paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m2 followed by cisplatin at a dose of 75 mg/m2.
-
2)In patients previously treated with chemotherapy for carcinoma of the ovary, paclitaxel has been used at several doses and schedules; however, the optimal regimen is not yet clear. (See CLINICAL STUDIES: Ovarian Carcinoma section). The recommended regimen is paclitaxel 135 mg/m2 or 175 mg/m2 administered intravenously over 3 hours every 3 weeks.
For patients with carcinoma of the breast, the following is recommended (see CLINICAL STUDIES: Breast Carcinoma section):
-
1)For the adjuvant treatment of node-positive breast cancer, the recommended regimen is paclitaxel, at a dose of 175 mg/m2 intravenously over 3 hours every 3 weeks for 4 courses administered sequentially to doxorubicin-containing combination chemotherapy. The clinical trial used 4 courses of doxorubicin and cyclophosphamide (see CLINICAL STUDIES: Breast Carcinoma ).
-
2)After failure of initial chemotherapy for metastatic disease or relapse within 6 months of adjuvant chemotherapy, paclitaxel at a dose of 175 mg/m2 administered intravenously over 3 hours every 3 weeks has been shown to be effective.
For patients with non-small cell lung carcinoma, the recommended regimen, given every 3 weeks, is paclitaxel administered intravenously over 24 hours at a dose of 135 mg/m2 followed by cisplatin, 75 mg/m2.
For patients with AIDS-related Kaposi's sarcoma, paclitaxel administered at a dose of 135 mg/m2 given intravenously over 3 hours every 3 weeks or at a dose of 100 mg/m2 given intravenously over 3 hours every 2 weeks is recommended (dose intensity 45 to 50 mg/m2/week). In the 2 clinical trials evaluating these schedules (see CLINICAL STUDIES: AIDS-Related Kaposi's Sarcoma ), the former schedule (135 mg/m2 every 3 weeks) was more toxic than the latter. In addition, all patients with low performance status were treated with the latter schedule (100 mg/m2 every 2 weeks).
Based upon the immunosuppression in patients with advanced HIV disease, the following modifications are recommended in these patients:
-
1)Reduce the dose of dexamethasone as 1 of the 3 premedication drugs to 10 mg PO (instead of 20 mg PO);
-
2)Initiate or repeat treatment with paclitaxel only if the neutrophil count is at least 1,000 cells/mm3;
-
3)Reduce the dose of subsequent courses of paclitaxel by 20% for patients who experience severe neutropenia (neutrophil <500 cells/mm3 for a week or longer); and
-
4)Initiate concomitant hematopoietic growth factor (G-CSF) as clinically indicated.
For the therapy of patients with solid tumors (ovary, breast and NSCLC), courses of paclitaxel should not be repeated until the neutrophil count is at least 1,500 cells/mm3 and the platelet count is at least 100,000 cells/mm3. Paclitaxel should not be given to patients with AIDS-related Kaposi's sarcoma if the baseline or subsequent neutrophil count is less than 1,000 cells/mm3. Patients who experience severe neutropenia (neutrophil <500 cells/mm3 for a week or longer) or severe peripheral neuropathy during Paclitaxel Injection, USP therapy should have dosage reduced by 20% for subsequent courses of paclitaxel. The incidence of neurotoxicity and the severity of neutropenia increase with dose.
Aids Related Kaposi's Sarcoma (AIDS-Related Kaposi's Sarcoma)
Data from 2, Phase 2 open-label studies support the use of paclitaxel as second-line therapy in patients with AIDS-related Kaposi's sarcoma. Fifty-nine of the 85 patients enrolled in these studies had previously received systemic therapy, including interferon alpha (32%), DaunoXome® (31%), DOXIL® (2%), and doxorubicin containing chemotherapy (42%), with 64% having received prior anthracyclines. Eighty-five percent of the pretreated patients had progressed on, or could not tolerate, prior systemic therapy.1
In Study CA139-174, patients received paclitaxel at 135 mg/m2 as a 3-hour infusion every 3 weeks (intended dose intensity 45 mg/m2/week). If no dose-limiting toxicity was observed, patients were to receive 155 mg/m2 and 175 mg/m2 in subsequent courses. Hematopoietic growth factors were not to be used initially. In Study CA139-281, patients received paclitaxel at 100 mg/m2 as a 3-hour infusion every 2 weeks (intended dose intensity 50 mg/m2/week). In this study patients could be receiving hematopoietic growth factors before the start of paclitaxel therapy, or this support was to be initiated as indicated; the dose of paclitaxel was not increased. The dose intensity of paclitaxel used in this patient population was lower than the dose intensity recommended for other solid tumors.
All patients had widespread and poor-risk disease. Applying the ACTG staging criteria to patients with prior systemic therapy, 93% were poor risk for extent of disease (T1), 88% had a CD4 count <200 cells/mm3 (I1), and 97% had poor risk considering their systemic illness (S1).
All patients in Study CA139-174 had a Karnofsky performance status of 80 or 90 at baseline; in Study CA139-281, there were 26 (46%) patients with a Karnofsky performance status of 70 or worse at baseline.
|
Prior Systemic Therapy
(n=59) |
|
| Visceral ± edema ± oral ± cutaneous | 42 |
| Edema or lymph nodes ± oral ± cutaneous | 41 |
| Oral ± cutaneous | 10 |
| Cutaneous only | 7 |
Although the planned dose intensity in the 2 studies was slightly different (45 mg/m2/week in Study CA139-174 and 50 mg/m2/week in Study CA139-281), delivered dose intensity was 38 to 39 mg/m2/week in both studies, with a similar range (20 to 24 to 51 to 61).
Non Small Cell Lung Carcinoma (nsclc) (Non-Small Cell Lung Carcinoma (NSCLC))
In a Phase 3 open-label randomized study conducted by the ECOG, 599 patients were randomized to either paclitaxel (T) 135 mg/m2 as a 24-hour infusion in combination with cisplatin (c) 75 mg/m2, paclitaxel (T) 250 mg/m2 as a 24-hour infusion in combination with cisplatin (c) 75 mg/m2 with G-CSF support, or cisplatin (c) 75 mg/m2 on day 1, followed by etoposide (VP) 100 mg/m2 on days 1, 2, and 3 (control). Response rates, median time to progression, median survival, and 1-year survival rates are given in the following table. The reported p-values have not been adjusted for multiple comparisons. There were statistically significant differences favoring each of the paclitaxel plus cisplatin arms for response rate and time to tumor progression. There was no statistically significant difference in survival between either paclitaxel plus cisplatin arm and the cisplatin plus etoposide arm.
|
a Etoposide (VP) 100 mg/m2 was administered IV on days 1, 2, and 3. |
|||
|
b Compared to cisplatin/etoposide. |
|||
|
T135/24
c75 (n=198) |
T250/24
c75 (n=201) |
VP100
a
c75 (n=200) |
|
| • Response
- rate (percent) - p-valueb |
25 0.001 |
23 <0.001 |
12 |
|
• Time to Progression
- median (months) - p-valueb |
4.3 0.05 |
4.9 0.004 |
2.7 |
|
• Survival
- median (months) - p-valueb |
9.3 0.12 |
10.0 0.08 |
7.4 |
|
• 1-Year Survival
- percent of patients |
36 |
40 |
32 |
In the ECOG study, the Functional Assessment of Cancer Therapy-Lung (FACT-L) questionnaire had 7 subscales that measured subjective assessment of treatment. Of the 7, the Lung Cancer Specific Symptoms subscale favored the paclitaxel 135 mg/m2/24 hour plus cisplatin arm compared to the cisplatin/etoposide arm. For all other factors, there was no difference in the treatment groups.
The adverse event profile for patients who received paclitaxel in combination with cisplatin in this study was generally consistent with that seen for the pooled analysis of data from 812 patients treated with single-agent paclitaxel in 10 clinical studies. These adverse events and adverse events from the Phase 3 first-line NSCLC study are described in the ADVERSE REACTIONS section in tabular (Tables 10 and 15) and narrative form.
Adverse Event Experiences By Body System (Adverse Event Experiences by Body System)
The following discussion refers to the overall safety database of 812 patients with solid tumors treated with single-agent paclitaxel in clinical studies. Toxicities that occurred with greater severity or frequency in previously untreated patients with ovarian carcinoma or NSCLC who received paclitaxel in combination with cisplatin or in patients with breast cancer who received paclitaxel after doxorubicin/cyclophosphamide in the adjuvant setting and that occurred with a difference that was clinically significant in these populations are also described.
The frequency and severity of important adverse events for the Phase 3 ovarian carcinoma, breast carcinoma, NSCLC, and the Phase 2 Kaposi's sarcoma carcinoma studies are presented above in tabular form by treatment arm. In addition, rare events have been reported from postmarketing experience or from other clinical studies. The frequency and severity of adverse events have been generally similar for patients receiving paclitaxel for the treatment of ovarian, breast, or lung carcinoma or Kaposi's sarcoma, but patients with AIDS-related Kaposi's sarcoma may have more frequent and severe hematologic toxicity, infections (including opportunistic infections, see Table 16), and febrile neutropenia. These patients require a lower dose intensity and supportive care. (See CLINICAL STUDIES: AIDS-Related Kaposi's Sarcoma .) Toxicities that were observed only in or were noted to have occurred with greater severity in the population with Kaposi's sarcoma and that occurred with a difference that was clinically significant in this population are described. Elevated liver function tests and renal toxicity have a higher incidence in KS patients as compared to patients with solid tumors.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:09.687962 · Updated: 2026-03-14T22:00:59.848985