Drug Facts
570ee9fb-a495-4a99-9215-0cd1bb2e0290
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
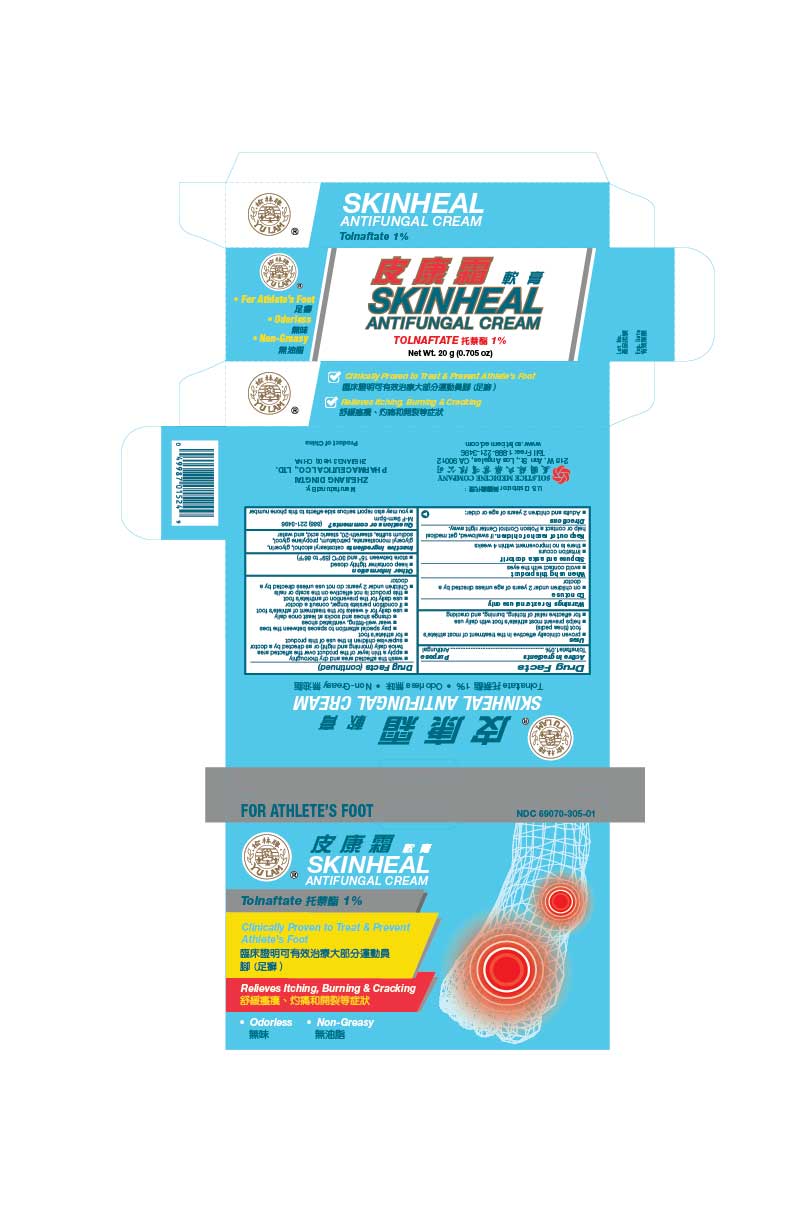
Active ingredient Tolnaftate 1.0%
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses
proven clinically effective in the treatment of most athlete’s foot (tinea pedis) and ringworm (tinea corporis)
helps prevent most athlete’s foot with daily use
for effective relief of itching, burning, and cracking
Dosage and Administration
Directions
Adults and children 2 years of age or older:
wash the affected area and dry thoroughly
apply a thin layer of the product over the affected area twice daily (morning and night) or as directed by a doctor
supervise children in the use of this product
for athlete’s foot
pay special attention to spaces between the toes
wear well-fitting, ventilated shoes
change shoes and socks at least once daily
use daily for 4 weeks for the treatment of athlete’s foot and ringworm
if condition persists longer, consult a doctor
use daily for the prevention of athlete’s foot
this product is not effective on the scalp or nails
Children under 2 years: do not use unless directed by a doctor
Description
Active ingredient Tolnaftate 1.0%
Section 44425-7
Other information
keep container tightly closed
store between 15 and 30 C (59 to 86 F)
Section 50565-1
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
irritation occurs
there is no improvement within 4 weeks
Section 50567-7
When using this product
avoid contact with the eyes
Section 50570-1
Do not use
on children under 2 years of age unless directed by a doctor
Section 51727-6
Inactive ingredients cetostearyl alcohol, glycerin, glyceryl monostearate, petrolatum, propylene glycol, sodium sulfite, steareth- 20, stearic acid, and water
Section 51945-4
SKINHEAL ANTIFUNGAL CREAM
Tolnaftate 1%
Net Wt. 10 g (1/3 oz)
NDC 69070-305-01
Section 53413-1
Questions or comments?
(888) 221-3496 M-F 9am-5pm
you may also report serious side effects to this phone number
Section 55105-1
Purpose
Antifungal
Section 55106-9
Active ingredient
Tolnaftate 1.0%
Structured Label Content
Indications and Usage (34067-9)
Uses
proven clinically effective in the treatment of most athlete’s foot (tinea pedis) and ringworm (tinea corporis)
helps prevent most athlete’s foot with daily use
for effective relief of itching, burning, and cracking
Dosage and Administration (34068-7)
Directions
Adults and children 2 years of age or older:
wash the affected area and dry thoroughly
apply a thin layer of the product over the affected area twice daily (morning and night) or as directed by a doctor
supervise children in the use of this product
for athlete’s foot
pay special attention to spaces between the toes
wear well-fitting, ventilated shoes
change shoes and socks at least once daily
use daily for 4 weeks for the treatment of athlete’s foot and ringworm
if condition persists longer, consult a doctor
use daily for the prevention of athlete’s foot
this product is not effective on the scalp or nails
Children under 2 years: do not use unless directed by a doctor
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 44425-7 (44425-7)
Other information
keep container tightly closed
store between 15 and 30 C (59 to 86 F)
Section 50565-1 (50565-1)
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
irritation occurs
there is no improvement within 4 weeks
Section 50567-7 (50567-7)
When using this product
avoid contact with the eyes
Section 50570-1 (50570-1)
Do not use
on children under 2 years of age unless directed by a doctor
Section 51727-6 (51727-6)
Inactive ingredients cetostearyl alcohol, glycerin, glyceryl monostearate, petrolatum, propylene glycol, sodium sulfite, steareth- 20, stearic acid, and water
Section 51945-4 (51945-4)
SKINHEAL ANTIFUNGAL CREAM
Tolnaftate 1%
Net Wt. 10 g (1/3 oz)
NDC 69070-305-01
Section 53413-1 (53413-1)
Questions or comments?
(888) 221-3496 M-F 9am-5pm
you may also report serious side effects to this phone number
Section 55105-1 (55105-1)
Purpose
Antifungal
Section 55106-9 (55106-9)
Active ingredient
Tolnaftate 1.0%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:32.378022 · Updated: 2026-03-14T23:13:32.363247